Abstract
Background: Stereochemistry is a key determinant in drug development, as it defines the three-dimensional structure of molecules and their interactions within biological systems. Because most biological targets are inherently chiral, even small differences in molecular orientation can lead to significant changes in drug response.
Objective: This review aims to examine the role of stereochemistry in drug development, with particular emphasis on its influence on
pharmacological activity, pharmacokinetics, and overall therapeutic performance.
Methods: An extensive review of published literature was undertaken to outline essential stereochemical concepts, including chirality,
enantiomers, diastereomers, and absolute configuration, and to relate these concepts to current approaches in drug design and optimization.
Results: Although stereoisomers share the same chemical composition, they often differ in their biological behavior. One isomer may provide
the intended therapeutic effect, while another may be less active or associated with adverse effects. Recent advances in stereoselective synthesis
and analytical methods have enabled the production of single-enantiomer drugs, which offer improved selectivity and safety. In addition, the
concept of chiral switching has emerged as an effective strategy to enhance the clinical value of existing drugs. Various examples from clinical
practice further highlight the importance of stereochemistry in determining drug action.
Conclusion: A clear understanding of stereochemistry is essential for the rational design of safe and effective drugs. Integrating
stereochemical considerations into drug development can improve therapeutic outcomes and reduce unwanted effects, thereby supporting the
advancement of modern pharmacotherapy.
Keywords:Stereochemistry; Chirality; Enantiomers; Chiral Switch; Drug Development; Pharmacotherapy
Introduction
Background
Stereochemistry is a branch of chemistry that studies how atoms are arranged in relation to one another within molecules. Since the prefix “stereo-” denotes “three-dimensionality,” stereochemistry is often referred to as 3D chemistry.[1] Stereoisomers are molecules whose atoms are joined in the same sequence but differ in how they are arranged in space; stereochemistry takes these spatial characteristics of molecules into account.[2] Stereoselectivity is shown in drug deposition, especially in activities that rely on contact with chiral biological macromolecules, such as drug metabolism, binding to plasma proteins, and active transport pathways.[3] Stereoisomers are molecules with identical chemical formulae that only differ in their spatial arrangement.[4] The study of chiral compounds is the most significant area in stereochemistry. The majority of chiral medicines were given as racemates in the early 1980s. [5] When chiral centers, often referred to as stereo genic or asymmetric centers, occur inside substances, stereoisomerism results. [6] As shown in Table 1, numerous types of chemical structures are included in stereochemistry. Stereochemistry relies heavily on chirality [7] (Figure 1) (Table 1).
Chirality
A geometric characteristic associated with three-dimensional objects’ lack of symmetry is called chirality [8] (Table 2). Chirality is the ability of a molecule to exist in two asymmetric forms that are mirror images of one another but cannot be superimposed, without altering the bond ordering, atom-to-atom interactions, or atomic composition [9]. Drug chirality has emerged as a key subject in the creation, discovery, development, patenting, and promotion of novel pharmaceuticals [10,11]. Chiral compounds are those that have one or more chiral centers [12]. Chirality also defines a drug’s pharmacology by affecting its binding affinity and interactions with its target. (Figure 2) [13] The enantiomers of chiral compounds, such as the right and left hands, are not overlaid on one another [14] (Figure 3).



Enantiomer
Enantiomers are stereoisomers that exist as non-super imposable mirror images [15]. Enantiomers can also be characterized by their absolute configuration, which is determined by the sequence in which the constituents are arranged around the chiral center [16]. While one enantiomer may be active, the other could be inactive and may cause side effects and/or show toxicity [17,18]. An established instance of enantiomer-related toxicity involves the (R)- and (S) enantiomers of thalidomide [17,19]. R-(-)-thalidomide is a non-harmful substance with calming effects, whereas S-(+) thalidomide is teratogenic and causes deformities in embryos when given to expectant mothers [20] (Figure 4). Enantiomers share the same physical and chemical characteristics, including molecular weight, solubility, and melting point [21] (Table 2) (Figure 5).




Diastereomer
Stereoisomers which cannot be superimposed and are not mirror images of each other [22]. Diastereomer are distinguished by variations in their physical properties, as well as some differences in their chemical behavior towards both achiral and chiral reagents [23]. Two molecules (or substances) are classified as Diastereomer if they are stereoisomerically related but not enantiomerically related [7].
Chiral Switch
Chiral switching is a phrase used to characterize the creation of single enantiomers from outdated racemate medications [24] (Figure 6). By increasing selectivity and potency towards receptors, lowering side effects, lowering inter-individual variability of the therapeutic response, lowering administered doses, improving the pharmacokinetic profile, and lowering drugdrug interactions, the chiral switch strategy may have therapeutic benefits linked to an improved therapeutic index [25] (Table 2).

Isomers
A molecule having a distinct chemical structure but the same chemical formula is called an isomer [26] (Figure 6).
Absolute Configuration
Using the stereochemical descriptors R and S, the absolute configuration (AC) of an entity, such as an organic molecule, specifies the spatial arrangement of its atoms [27]. R, which comes from the Latin rectus, indicates clockwise or to the right, while S, which comes from the Latin sinister, means counterclockwise or to the left. A certain chiral center’s R or S configuration can be ascertained using exact rules based on mass and atomic number [28]. The R, S system, commonly known as the Cahn-Ingold- Prelog rules of chiral chemical naming, was introduced by three chemists: R. S. Cahn, C. K. Ingold, and V. Prelog [2].
Importance
In order to accurately describe chemical reactions,
stereochemistry takes into account both static (configuration)
and dynamic (conformation) temperature-dependent conditions
[29]. Drug stereochemistry has gained attention recently due to
developments in the techniques for stereoselective synthesis and
stereospecific analysis of chiral drug molecules, as well as growing
awareness of the possible importance of the distinct biological
characteristics of stereoisomers [30]. Stereochemistry plays a
crucial role in therapeutic pharmacology as pure enantiomers
of chiral substances have the same physical properties, but their
properties in biochemical processes can be radically different.
The following scenarios are possible:
a) One or both enantiomers may have the desired effect,
but only one may cause undesired side effects.
b) The inactive enantiomer might function as an antagonist
that competes.
c) The actions of the enantiomers may differ or be opposing.
d) Only one enantiomer may result in undesirable side
effects, while one or both enantiomers may produce the intended
effect.
e) The adverse effects of the active enantiomer may be
counteracted by the inactive enantiomer [20].
Chiral compounds make up more than half of the medications now on the market, and many of them are sold as racemates, which are equimolar mixtures of two enantiomers. Unlike many chiral man-made substances, chiral natural molecules only occur in one enantiomeric form (natural sugars are (+)-isomers, while amino acids are (-)-isomers) [31]. Enantiomer separation can be accomplished using chiral chromatographic techniques as well as kinetic and thermodynamic resolution [9].
Differences in a single stereocenter can have a significant impact on biological activity in both natural products and FDAapproved medications. For instance, 1,4-dihydropyrans are L-type calcium channel ligands that show stereoselectivity for the activity type. More specifically, the R-enantiomer inhibits the channel whereas the S-enantiomer activates it. Chirality frequently affects how a chemical smells, with enantiomers frequently smelling entirely different. For instance, carvone is what gives spearmint and caraway their respective scents (Figures 2 & 7) [32] (Table 3).


Pharmacology
Stereoisomers have different pharmacokinetic and pharmacodynamic characteristics [19]. As a remarkable chiral selector, the body’s many homochiral molecules will interact with each racemic medication in a unique way and metabolize each enantiomer via a distinct pathway to produce distinct pharmacological activity. Consequently, one isomer may exhibit the intended therapeutic effects, whilst the other may be inert or, in the worst situations, exhibit toxic or undesirable side effects [4,33]. Stereoisomerism can lead to pharmacokinetic differences such as L-methotrexate being better absorbed than D-methotrexate, Esomeprazole being more bioavailable than racemic omeprazole, and S-warfarin having a lower volume of distribution because it is more extensively bound to albumin than R-warfarin [19].
Enantiomers with equal qualitative and quantitative properties, enantiomers with different quantitative properties, and enantiomers with other qualitative attributes are the three main categories of chiral medications based on the pharmacological features of the enantiomers [34].
Enantiomers with equal qualitative and quantitative properties
Propafenone (antiarrhythmic), flecainide (antiarrhythmic), cyclophosphamide (antineoplastic), and fluoxetine (antidepressant) are among the few racemic medications that fall into this category [4].
Enantiomers with different quantitative properties
The pharmacological activity of this kind of chiral medication is attributed to one enantiomer, whereas the other enantiomer exhibits minimal or no pharmacological activity [4].
Enantiomers with other qualitative attributes
These enantiomers exhibit distinct pharmacological actions,
which encompass several circumstances:
1. enantiomers that share a biological target yet exhibit
opposing actions;
2. enantiomers containing different biological objectives
3. Among the enantiomers that may have adverse effects
4. an inactive enantiomer capable of counteracting the
adverse effects of the active enantiomer [4], (Figure 8).

Pharmacologically Active Enantiomers: Configuration & Clinical examples
Avapritinib
When it comes to mutant KIT and the KIT-D816V mutation of platelet-derived growth factor receptor alpha (PDGFRA), Avapritinib, a selective orally accessible tyrosine kinase inhibitor, exhibits strong action [35]. In 2020, the FDA authorized it for the treatment of gastrointestinal stromal tumors (GISTs) with PDGFRA exon 18 mutations, and patients have shown that using it has a positive therapeutic impact. Additionally, avapritinib is being tested in clinical settings to treat several conditions, including indolent systemic Masto cytosis.
In terms of structure, avapritinib is a flexible, linear molecule with a chiral center on its rotational chain. When compared to its (R)-(+)-counterpart, (S)-(−)-avapritinib demonstrated higher c-KIT D816V inhibitory action [36].
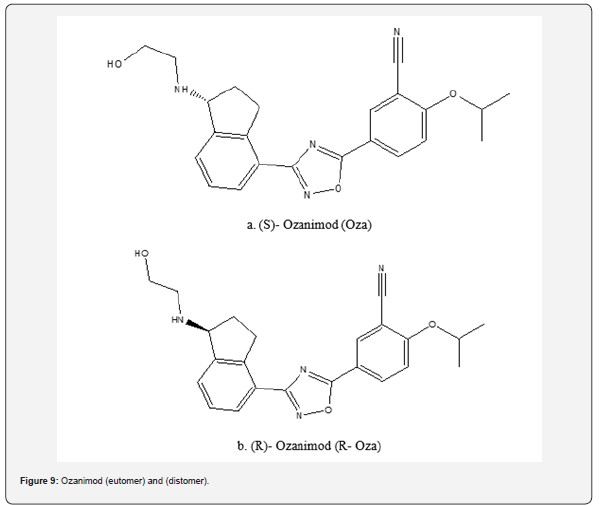
Ozanimod
Ozanimod’s unique capacity to modify immunological response and its more patient-friendly oral delivery method make it a promising option for treating multiple sclerosis in the clinical setting [37]. On March 25, 2020, Ozanimod received approval in the United States for the treatment of relapse multiple sclerosis in adults, including clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive illness [38]. With a selectivity of 27 times for S1P1 over S1P5 receptors and more than 10,000 times for S1P1 over S1P2,3,4 receptors, ozanimod is a powerful agonist of both S1P1 and S1P5 receptors [39].
S-ozanimod, or simply ozanimod, and R-ozanimod are the two enantiomeric forms of ozanimod that are implied by its single chiral carbon atom [40]. The S-enantiomer, which is more effective, is used in medicinal applications. As a result, regulatory bodies require the R antipode in an ozanimod sample to be determined as part of the required evaluations [25] (Figure 9).
Lonafarnib
Under license from Merck & Co., Eiger Biopharmaceuticals developed lonafarnib (ZokinvyTM), an oral active FTase inhibitor, to treat progeria and progeroid laminopathies as well as hepatitis D virus (HDV) infections [41]. Lonafarnib was first approved in the USA on November 20, 2020 to treat processing-deficient progeroid laminopathies (with either homozygous or compound heterozygous ZMPSTE24 mutations or heterozygous LMNA mutation with progerin-like protein accumulation) in patients ≥ 12 months of age with a body surface area (BSA) of ≥ 0.39 m2 [42] (Figure 10).

Thalidomide
Initially introduced as a sedative in the late 1950s, thalidomide was used to relieve pregnant women’s nausea. About 10,000 babies were born with phocomelia within a few years of thalidomide being widely used in Europe, Australia, and Japan [43]. Nevertheless, thalidomide has an impact on a wide range of other tissues, such as the spine, gastrointestinal tract, reproductive system, eyes, ears, legs, face, and cardiovascular system. Unfortunately, it has been estimated that up to 40% of babies die in their first year of life due to internal organ developmental variations, such as atresia of the colon and heart problems [44]. Asa result, thalidomide was outlawed in the majority of nations in 1961 [43]. Pregnancy-related drug usage has been a major source of concern since the thalidomide disaster in the 1960s because of the possibility of undesirable effects on the fetus, particularly severe congenital malformations (MCM) [45,46] (Figure 4).
Conclusion
Stereochemistry plays a crucial role in determining how drugs interact with biological systems and ultimately affects their safety and effectiveness. Differences in the spatial arrangement of molecules can lead to significant variations in therapeutic outcomes, even when the chemical composition remains the same. As discussed in this review, the use of single-enantiomer drugs and advances in stereoselective techniques have improved drug specificity while reducing unwanted effects. Approaches such as chiral switching have further strengthened the clinical value of many existing drugs. However, challenges related to synthesis, cost, and regulatory requirements still need to be addressed. A better understanding of stereochemical behavior will continue to support the development of safer and more efficient medicines in the future.
References
- Singh K, Shakya P, Kumar A, Alok S, Kamal M, et al. (2014) Stereochemistry and Its Role in Drug Design. Int J Pharm Sci Res [Internet] 5(11): 4644-4659.
- Kumar R (2021) Effects of Stereoisomers on Drug Activity. Am J Biomed Sci Res 13(3): 220-222.
- Ananthi N (2018) Role of Chirality in Drugs. Org Med Chem Int J 5(3).
- Nguyen LA, He H, Pham HC (2006) Chiral drugs: an overview. Int J Biomed Sci 2(2): 85-100.
- Huang Q, He R, Kozikowski A (2011) Stereochemistry at the Forefront in the Design and Discovery of Novel Anti-tuberculosis Agents. Curr Top Med Chem 11(7):810-818.
- Caldwell J (1992) The importance of stereochemistry in drug action and disposition. J Clin Pharmacol 32(10):925-929.
- Gal J (2020) Proposal for a New Stereochemically Informative Nonproprietary Drug Naming System. Pharmaceut Med 34(2): 113-126.
- Coelho MM, Fernandes C, Remião F, Tiritan ME (2021) Enantioselectivity in Drug Pharmacokinetics and Toxicity: Pharmacological Relevance and Analytical Methods. Molecules 26(11): 3113.
- Brooks HW, Guida CW, Daniel GK (2011) The Significance of Chirality in Drug Design and Development. Curr Top Med Chem 11(7): 760-770.
- Agranat I, Wainschtein SR, Zusman EZ (2012) The predicated demise of racemic new molecular entities is an exaggeration. Nat Rev Drug Discov 11(12): 972-973.
- Caner H, Groner E, Levy L, Agranat I (2004) Trends in the development of chiral drugs. Drug Discov Today 9(3): 105-110.
- Alum BN (2024) Exploring the Role of Stereochemistry in Organic Synthesis: Strategies, Challenges, and Applications 3(3): 69-74.
- Senkuttuvan N, Komarasamy B, Krishnamoorthy R, Sarkar S, Dhanasekaran S, et al. (2024) The significance of chirality in contemporary drug discovery-a mini review. RSC Adv 14(45): 33429-33448.
- Bialer M, Perucca E (2025) From barbiturates to ganaxolone: The importance of chirality in drug development and in understanding the actions of old and new antiseizure medications. Pharmacol Ther 268: 108808.
- Smith SW (2009) Chiral Toxicology: It’s the Same Thing…Only Different. Toxicol Sci 110(1): 4-30.
- Baker GB, Prior TI (2002) Stereochemistry and drug efficacy and development: Relevance of chirality to antidepressant and antipsychotic drugs. Ann Med 34(7-8): 537-543.
- Mwamwitwa KW, Kaibere RM, Fimbo AM, Sabitii W, Ntinginya NE, et al. (2020) A retrospective cross-sectional study to determine chirality status of registered medicines in Tanzania. Sci Rep 10(1): 1-11.
- Čižmáriková R, Habala L, Valentová J, Markuliak M (2019) Survey of pharmacological activity and pharmacokinetics of selected β-adrenergic blockers in regard to their stereochemistry. Appl Sci 9(4).
- Chhabra N, Aseri M, Padmanabhan D (2013) A review of drug isomerism and its significance. Int J Appl Basic Med Res 3(1): 16-18.
- Elder FCT, Feil EJ, Snape JN, Gaze WH, Kasprzyk HBA (2020) The role of stereochemistry of antibiotic agents in the development of antibiotic resistance in the environment. Environ Int 139: 105681.
- Sushil S, Sharma AK, Gupta AK, Navdeep D, Brashier DBS (2014) Review Article Chiral Switch- an Emerging Strategy in Therapeutics 4(2): 135-139.
- Oh H. A molecule will be achiral if it has a plane or symmetry or a center of symmetry. If a molecule is achiral, it can be superimposed on its mirror image and thus does not have an enantiomer. 1-7.
- Agranat I, Caner H, Caldwell J (2002) Putting chirality to work: The strategy of chiral switches. Nat Rev Drug Discov 1(10): 753-768.
- Somogyi A, Bochner F, Foster D (2004) Inside the isomers: The tale of chiral switches. Aust Prescr 27(2): 2-5.
- Hancu G, Modroiu A (2022) Chiral Switch: Between Therapeutical Benefit and Marketing Strategy. Pharmaceuticals 15(2): 240.
- Baregama C (2018) Stereochemistry- racemic modification, resolution, and its importance with recently used optically active drugs. Asian J Pharm Clin Res 11(1): 3-12.
- Saito F, Schreiner PR (2020) Determination of the Absolute Configurations of Chiral Alkanes – An Analysis of the Available Tools. European J Org Chem 2020(40): 6328-6339.
- McConathy J, Owens MJ (2003) Stereochemistry in Drug Action. Prim Care Companion CNS Disord 5(2): 70-73.
- Villalobos F, Mendieta MJI, Lobo CJ, Morcillo SP, Martínez JI, et al. (2025) Diastereomeric Configuration Drives an On-Surface Specific Rearrangement into Low Bandgap Non-Benzenoid Graphene Nanoribbons. J Am Chem Soc 147(9): 7245-7254.
- Hutt AJ (2002) The development of single-isomer molecules: Why and how. CNS Spectr 7(Suppl 1): 14-22.
- Kasprzyk HB (2010) Pharmacologically active compounds in the environment and their chirality. Chem Soc Rev 39(11): 4466-4503.
- Scott KA, Ropek N, Melillo B, Schreiber SL, Cravatt BF, et al. (2022) Stereochemical diversity as a source of discovery in chemical biology. Curr Res Chem Biol 2: 100028.
- Landoni F, Soraci AL (2014) Pharmacology of Chiral Compounds 2-Arylpropionic Acid Derivatives. Curr Drug Metab 2(1): 37-51.
- Peng Sj, Zhu Yy, Luo Cy, Zhang P, Wang Fy, et al. (2024) Lab Med Discovery Chiral drugs: Sources, absolute configuration identification, pharmacological applications, and future research trends. Lab Med Discov 1(1): 100008.
- Gotlib J, Reiter A, Radia DH, Deininger MW, George TI, et al. (2021) Efficacy and safety of avapritinib in advanced systemic mastocytosis: interim analysis of the phase 2 PATHFINDER trial. Nature medicine 27: 2192-2199.
- Ya DY, Chen Z, Liang PL, Yi XL, Bei BY, et al. (2025) Absolute Configuration and Chiroptical Properties of Flexible Drug Avapritinib. Pharmaceuticals (Basel) 18(6): 833.
- Fronza M, Lorefice L, Frau J, Cocco E (2021) An Overview of the Efficacy and Safety of Ozanimod for the Treatment of Relapsing Multiple Sclerosis. Drug Des Devel Ther 15:1993-2004.
- Lamb YN (2020) Ozanimod: First Approval. Drugs 80(8): 841-848.
- Scott FL, Clemons B, Brooks J, Brahmachary E, Powell R, et al. (2016) Ozanimod (RPC1063) is a potent sphingosine-1-phosphate receptor-1 (S1P1) and receptor-5 (S1P5) agonist with autoimmune disease-modifying activity. Br J Pharmacol 173(11): 1778-1792.
- Ferencz E, Szabó ZI, Zöldhegyi A, Dombi G, Molnár G, et al. (2024) Possibilities and limitations of computer assisted chiral HPLC method development for ozanimod on polysaccharide based chiral stationary phases. Sci Rep 14(1): 26757.
- Dhillon S (2021) Lonafarnib: First Approval. Drugs 81(2): 283-289.
- (2020) ZOKINVYTM (lonafarnib) capsules, for oral use Initial U.S. Approval.
- Kim JH, Scialli AR (2011) Thalidomide: The Tragedy of Birth Defects and the Effective Treatment of Disease. Toxicological Sciences 122(1): 1-6.
- Vargesson N, Stephens T (2021) Expert Opinion on Drug Safety Thalidomide: history, withdrawal, renaissance, and safety concerns. Expert Opin Drug Saf 20(12): 1455-145
- Gils C, Pottegård A, Ennis ZN, Damkier P (2016) Perception of drug teratogenicity among general practitioners and specialists in obstetrics / gynecology: a regional and national questionnaire-based survey. BMC Pregnancy Childbirth 16: 1-7.
- Broe A, Pottegård A, Petersen I, Damkier P (2025) Prescription Drugs in Pregnancy and Congenital Malformations: A Population- Based Safety Screening Study. Pharmacoepidemiol Drug Saf 34(9): 70211.






























