Abstract
Background: Adenoid cystic carcinoma (ACC) affecting the sinonasal and orbitofacial compartments is rare and usually diagnosed at an
advanced stage because of nonspecific early symptoms. ACC is classically neurotropic, with perineural invasion (PNI) being a major adverse
prognostic factor. We report a rare presentation of a massive cribriform-predominant ACC of the right naso-ethmoido-orbital region with skullbase
and intracranial extension but without histologic perineural or vascular invasion.
Case presentation: a 27-year-old man from Senegal and builder by occupation, developed a progressive right palpebral swelling in 2015 and
underwent an undocumented exenteration in 2017. Recurrence manifested as painful right orbital swelling with bilateral nasal obstruction and
anosmia. An endonasal biopsy in August 2024 confirmed ACC. He was referred to our center in Rabat in January 2025. Clinical exam revealed
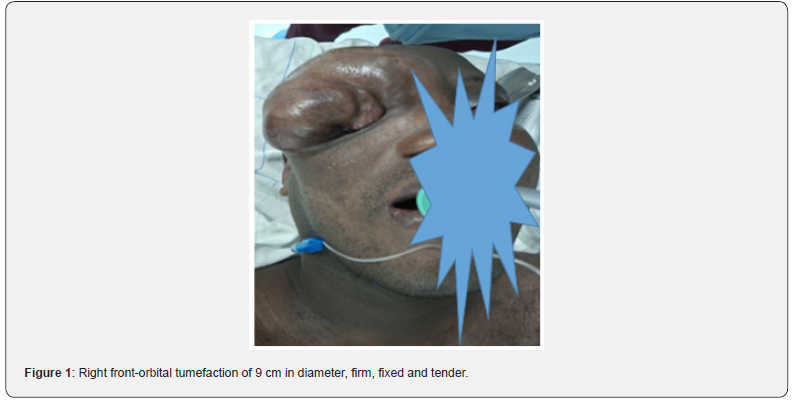
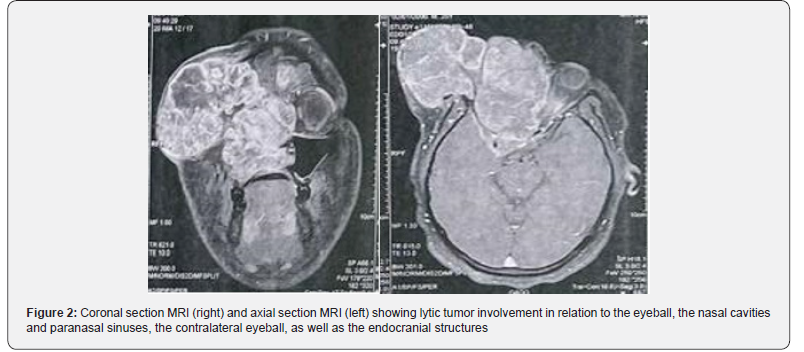
a 9-cm right fronto-orbital mass with purulent discharge and a right nasal cavity occluded by an ulcerated polypoid tumor. MRI showed a 103
× 111 × 91mm heterogeneous mass filling the ethmoid/maxillary/frontal complex with bone lysis, pachymeningeal enhancement, orbital
obliteration, cavernous sinus and cavernous ICA involvement, and extension to the nasopharynx. He underwent subtotal excision with skullbase
repair (multilayer fat graft + bio glue) and packing; directed healing followed by skin grafting. Final pathology: cribriform-predominant
ACC, greatest axis ≈10 cm, no perineural invasion, no vascular emboli. Adjuvant concurrent radio chemotherapy was delivered.
Conclusion: This case illustrates the biological heterogeneity of ACC: exceptional tumor bulk and multi-compartment invasion can coexist
with absence of PNI. Multidisciplinary management combining maximal safe resection, skull-base repair and adjuvant radiotherapy/
chemoradiotherapy is essential. Long-term surveillance is mandatory given ACC’s propensity for late local recurrence and distant metastases.
Molecular profiling may further refine prognosis and guide future targeted therapies.
Keywords:Cribriform; Predominant adenoid cystic carcinoma; Naso ethmoidal; Perineural invasion; Orbital swelling; Bilateral nasal obstruction
Abbreviations:ACC: Adenoid Cystic Carcinoma; PNI: Perineural Invasion
Introduction
Adenoid cystic carcinoma (ACC) is an uncommon malignant neoplasm of secretory gland origin. While ACC most often arises in major and minor salivary glands, sinonasal and orbitofacial localizations represent a minority of cases and present unique diagnostic and therapeutic challenges because of proximity to the orbit, cranial base and neurovascular structures [1-4]. ACCs are classically slow-growing but relentlessly infiltrative: perineural invasion (PNI), skull-base erosion, local recurrence and late distant metastases (commonly pulmonary) are hallmarks of the disease [2,5-7]. Histologically ACC shows cribriform, tubular and solid patterns; the cribriform and tubular variants are generally associated with more indolent behavior compared with the solid pattern, whereas PNI and solid architecture predict worse outcomes [6,8-10]. Treatment typically relies on maximal safe surgical resection when feasible, followed by adjuvant radiotherapy; the role of chemotherapy is limited and mainly used in the setting of advanced or metastatic disease [3,11-13]. We report a striking case of a very large cribriform-predominant ACC of the naso-ethmoido-orbital region with extensive skull-base and intracranial extension in a young adult patient - and, notably, absence of histologic perineural or vascular invasion [14-17]. We discuss diagnostic features, therapeutic choices, prognosis and implications for future management, with reference to recent literature (2015-2025).
Case Presentation
Patient and history
A 27-year-old man originally from Senegal and working as a builder presented with a long history of right palpebral swelling. He initially noticed a small eyelid swelling in 2015, which progressively increased. In 2017 he underwent an undocumented orbital exenteration in Senegal. Thereafter, the mass recurred, became painful and progressively larger; the patient developed bilateral nasal obstruction and anosmia but denied epistaxis, headaches or vertigo. Limited financial resources delayed further care; an endonasal biopsy performed in Dakar in August 2024 revealed adenoid cystic carcinoma. The patient sought further treatment in Morocco and was admitted to our department in Rabat in January 2025. Medical history included tobacco and alcohol use. No significant family history or prior head-and-neck radiation was reported.
Clinical examination
On presentation the face showed a right fronto-orbital tumefaction approx. 9 cm in diameter, firm, fixed and tender, with purulent discharge at the inner canthus. Endoscopic rhinologic exam demonstrated marked left septal deviation secondary to mass effect and complete filling of the right nasal cavity by an ulcerated polypoid tumor preventing passage of the endoscope. The left nasal cavity contained purulent secretions. Neurologic exam was otherwise unremarkable.
Imaging
Gadolinium-enhanced MRI of the brain and orbits (02/01/2025) demonstrated a heterogeneous tissue mass measuring 103 × 111 × 91 mm occupying the right naso-ethmoidoorbital complex. Imaging features included T1 hypo intensity, moderate T2/FLAIR hyperintensity, and very heterogeneous contrast enhancement. The lesion filled and distended the ethmoid cells with lysis of ethmoid roof, extension to skull base with pachymeningeal enhancement lifting the right frontal lobe (no intraparenchymal abnormal signal). It invaded frontal soft tissues and frontal sinus with lysis of sinus walls and subcutaneous frontal extension. Posteriorly it involved the right cavernous sinus with encasement and caliber reduction of the cavernous internal carotid artery. Laterally it obliterated the orbit, eroded the lateral orbital rim and extended to the ipsilateral superficial temporal fossa. Medially it eroded the left planum with infiltration of left medial extraconal fat. Inferiorly it filled the nasal fossae with lysis of turbinates and septum, invaded the right maxillary sinus medial wall (small retention fluid). Posterior extension continued through the choanae to the rhino pharynx. These features were consistent with an advanced, multi-compartment sinonasal malignancy involving orbit and skull base [9,17].
Surgical management
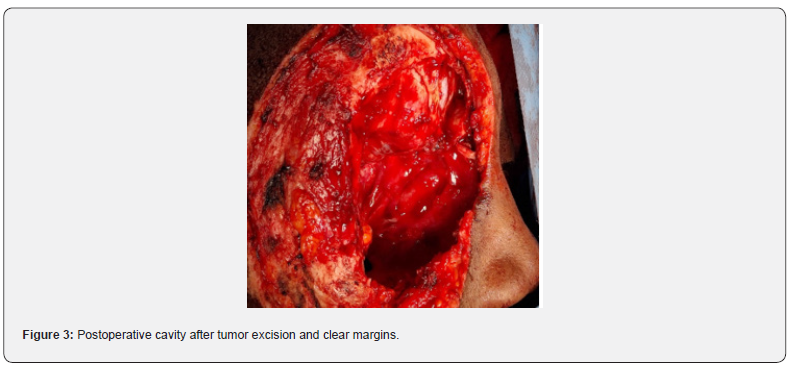
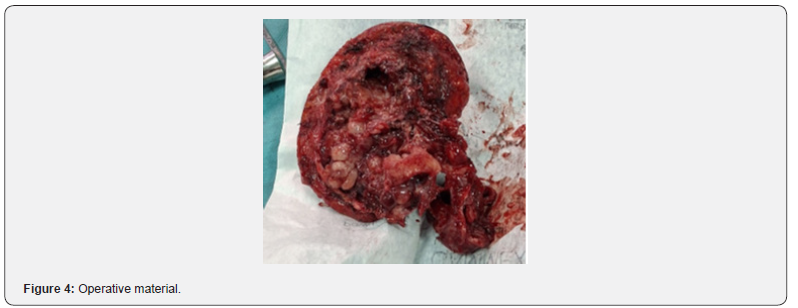
The patient underwent a wide excision of the orbital component through a circumferential incision with attempted 1-cm margins where anatomy permitted. Subcutaneous and muscular plans were dissected and the right superficial temporal artery ligated. Intraoperative findings confirmed invasion of the right orbit, frontal process of the maxilla, nasolacrimal bone, lateral and alar nasal cartilages (destroyed), ethmoid, sphenoid and frontal sinuses; the contralateral frontal sinus also appeared involved and was addressed. Fragments of suspect frontal, temporal and maxillary bone were debrided with hand instruments. A large skull-base dehiscence with meningeal prolapse was observed; the meninges were cleaned and a cerebrospinal fluid leak was repaired by multilayer technique using autologous fat graft and bioglue. Hemostasis was achieved and the cavity was packed with Gelfoam and topical agents. Directed healing was performed for several weeks until granulation allowed skin grafting.
Pathology
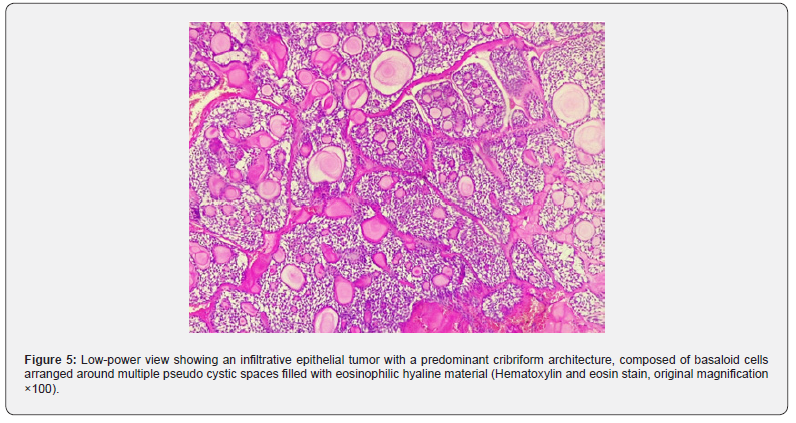
Definitive histopathology showed a cribriform-predominant adenoid cystic carcinoma, greatest dimension ≈10 cm. Importantly, extensive sampling did not demonstrate perineural invasion or vascular emboli.
Adjuvant therapy and early outcome
Given subtotal resection and extensive local disease, the patient received adjuvant concurrent radiochemotherapy (definitive radiotherapy with concurrent platinum-based radiosensitization). At early follow-up (3 months) there was no obvious radiographic gross residual tumor; the patient remains under long-term surveillance.
Discussion
Rarity and diagnostic delay
Sinonasal and orbitofacial ACCs are uncommon; because early symptoms (nasal obstruction, anosmia, mild facial swelling) are nonspecific, diagnosis is frequently delayed and tumors are often advanced at presentation [1-4,9]. This case exemplifies how financial and access barriers may allow tumors to reach extraordinary size and multi-compartment extension before definitive treatment, complicating surgical management [18-22].
Histologic pattern and prognostic implications
ACC histologic subtypes (cribriform, tubular, solid) convey prognostic information: cribriform and tubular patterns usually relate to more indolent behavior while the solid pattern and presence of PNI associate with worse local control and survival [6,8-10,22]. PNI is especially important: systematic reviews have shown PNI correlates strongly with local recurrence and reduced disease-specific survival in head and neck ACC [22,23]. The absence of PNI in a tumor of this magnitude is unusual and may indicate a different neurotropic biology; this observation supports the concept of molecular heterogeneity in ACC, in line with recent molecular studies identifying distinct fusion events (MYB/MYBL1::NFIB) and NOTCH pathway alterations that influence behavior [12,13].
Imaging and extent assessment
MRI is the modality of choice for mapping soft tissue extent, perineural spread and intracranial involvement; CT complements bone evaluation [9,17]. Our patient’s MRI documented classic features of advanced sinonasal ACC - extensive bone lysis, pachymeningeal enhancement, orbital obliteration and cavernous sinus/ICA involvement - information that dictated a maximal safe (debulking/subtotal) surgical approach and skull-base repair [24- 28].
Surgical strategy and skull-base repair
En bloc resection with negative margins is the goal for local control, but is frequently impossible in sinonasal lesions abutting skull base, orbit or cavernous sinus [11,19]. When radical resection would cause unacceptable morbidity, subtotal or functionpreserving resection to relieve mass effect and allow adjuvant therapy is an accepted approach. The CSF leak encountered was appropriately repaired by multilayer closure using autologous fat and sealant - standard practice in skull-base surgery [19]. Our approach prioritized maximal safe resection, reconstruction, and subsequent adjuvant therapy.
Role of adjuvant radiotherapy and chemoradiation
Adjuvant radiotherapy is widely recommended for highstage or incompletely resected ACCs and for PNI or positive/close margins; particle therapy (protons/carbon ions) may allow dose escalation with better normal-tissue sparing in skull-base tumors [7,14,24]. The benefit of concurrent chemotherapy is not firmly established; platinum-based radiosensitizers are commonly used in high-risk or unresectable cases, although randomized data are lacking and some series report limited long-term survival benefit [20,25]. Given our patient’s subtotal excision and extensive disease, adjuvant concurrent chemoradiotherapy represented a reasonable, evidence-supported choice.
Molecular features and future therapies
Molecular profiling (MYB/MYBL1 fusions, NOTCH alterations) refines ACC classification and suggests avenues for targeted therapy and trials. MYB::NFIB fusion is a recurrent event in ACC and has been explored as a diagnostic marker and potential therapeutic target; NOTCH pathway dysregulation is associated with aggressive behavior and is being evaluated in clinical trials [9,12,13,26,28]. Integration of routine molecular testing would add prognostic and therapeutic value in future cases.
Prognosis and follow-up
Prognosis in snACC depends on stage, histologic pattern, margin status and PNI; reported 5-year OS varies widely but declines with advanced stage and PNI [2,7,22]. ACC often metastasizes late (lungs common) and recurrences can occur many years after treatment; lifelong surveillance with interval MRI of the head and chest imaging is recommended [5,29]. The absence of PNI in our case may be a favorable histologic feature, but the large size, skull-base involvement and marginal resection imply high recurrence risk; thus, strict long-term follow-up is mandatory.
Lessons and Implications
This case highlights the importance of early referral and imaging for sinonasal masses, the need for multidisciplinary skull-base teams in complex cases, and the potential benefit of integrating molecular profiling into prognostic assessment and clinical trial selection. In low-resource settings, improving access to timely care is crucial to prevent extreme tumor progression and to enable more definitive surgery.
Limitations
As a single case, generalizability is limited. Molecular testing (MYB/MYBL1 fusion, NOTCH sequencing) was not available and would have enriched the analysis. Long-term oncologic outcomes (local control, distant metastasis, survival) remain immature.
Conclusion
We report an exceptional case of a very large cribriformpredominant adenoid cystic carcinoma centered in the nasoethmoido- orbital region with intracranial extension but lacking histologic perineural or vascular invasion. This unusual combination underscores biological heterogeneity in ACC and highlights challenges in managing advanced sinonasal tumors. Multidisciplinary care - combining maximal safe resection, skullbase repair and adjuvant radiotherapy/chemoradiotherapy - and lifelong surveillance remain essential. Expanding access to molecular profiling may soon allow better risk stratification and targeted therapeutic options for patients with ACC.
References
- Lee TH, Jin H, Park J (2024) Clinical outcomes in adenoid cystic carcinoma of the nasal cavity and paranasal sinuses: a multicenter analysis. Cancers (Basel) 16(6): 1235.
- Mauthe T, Holzmann D, Soyka MB (2024) Outcome-oriented clinicopathological reappraisal of sinonasal adenoid cystic carcinoma: a multicenter analysis. Sci Rep 14(1): 18655.
- Fang Y, Zhang Y, Wu J (2022) Current opinions on diagnosis and treatment of adenoid cystic carcinoma. Lancet Oncol (review) 130: 105945.
- Al Ghulaiga FM, Alkhiary H, AlKhalidi H, Alkatan HM (2022) Adenoid cystic carcinoma of the orbit with bilateral cavernous sinus extension: a case report. Int J Surg Case Rep 99: 107623.
- Zupancic M (2024) Adenoid cystic carcinoma - clinical presentation and current management. Anticancer Res 44(4): 1325-1337.
- Nagao T (2018) Histologic grading and prognosis in adenoid cystic carcinoma. Am J Surg Pathol 42(3): 324-334.
- Garden AS (2019) Adjuvant radiotherapy and particle therapy for skull base adenoid cystic carcinoma: outcomes and recommendations. Int J Radiat Oncol Biol Phys 104(4): 866-875.
- Carlson ML, Teknos TN (2021) Advanced adenoid cystic carcinoma of the skull base: outcomes and challenges. Neurosurg Rev 44(5): 2885-2894.
- Sasaki T, Tanaka S, Takahashi H (2021) MRI characteristics of sinonasal ACC and skull base involvement. J Neuroradiol 48(3): 193-200.
- Hsieh CE, Huang HY (2020) Prognostic significance of histologic pattern in adenoid cystic carcinoma. Head Neck 42(6): 1143–1150.
- Amit M (2023) Endoscopic resection of sinonasal adenoid cystic carcinoma: long-term results. Laryngoscope 133(4): 884-891.
- Wagner VP (2022) MYB-NFIB fusion transcript in adenoid cystic carcinoma: current state of knowledge and future directions. Hum Pathol 176: 103745.
- Bedell M (2024) A novel scoring system for MYB RNA in situ hybridization as a diagnostic adjunct in ACC. Mod Pathol 18(1): 51.
- Rohit A (2025) Particle-based radiotherapy for sinonasal ACC: systematic review. Radiother Oncol.
- Nakagawa T, Nakahara S (2020) Adenoid cystic carcinoma of external auditory canal: outcomes and review. Otology & Neurotology 41(4): e475-e482.
- Li J (2025) Advances in treatment of adenoid cystic carcinoma: a 2025 update. Cancer Treat Rev.
- Turner A (2026) MRI and CT features of advanced sinonasal ACC with intracranial extension: diagnostic pearls. Neuroradiology 62(9): 1139-1148.
- Lorini L (2021) Prognosis and management of recurrent and/or metastatic ACC: a systematic review. Eur J Cancer 145.
- Mendenhall WM (2020) ACC of head and neck: long-term outcomes and treatment paradigms. Am J Clin Oncol 43(9): 646-652.
- Song X (2025) Long-term outcomes of platinum-based chemotherapy for T4 sinonasal ACC: multicenter study. Front Oncol.
- Dillon PM (2019) Long-term surveillance strategies in ACC: implications for practice. J Clin Oncol 37(20): 1740-1749.
- Ju J (2020) The role of perineural invasion in head and neck ACC prognosis: systematic review and meta-analysis. Eur J Cancer 137: 154-166.
- Lorini L (2021) Recurrent/metastatic adenoid cystic carcinoma: systemic therapy and outcomes. Cancer Treat Rev 99: 102246.
- Garden AS (2019) Adjuvant proton therapy for sinonasal ACC: experience and outcomes. Int J Radiat Oncol Biol Phys 104(4): 866-875.
- Fang Y (2022) Current opinions on diagnosis and treatment of ACC: review. Oral Oncol.
- Wagner VP (2023) Clinical trials targeting MYB/NOTCH in ACC: progress and limitations. Cancer 129(8): 1450-1466.
- Al Ghulaiga FM, AlKhalidi H, Alkhiary H (2022) Adenoid cystic carcinoma of the orbit with bilateral cavernous sinus extension: case and review. Int J Surg Case Rep 99: 107623.
- De Almeida LO (2025) Molecular advances in adenoid cystic carcinoma: implications for therapy. Mol Cancer.
- Coca-Pelaz A (2023) Management and outcomes in sinonasal ACC: consensus and recommendations. Head Neck 45(5):1237-1249.





























