Histological Assessment of The Regenerative Potential of Platelet-Rich Plasma for Severe Vocal Fold Scar in A Rat Model
Hiroki Matsushita, Shigeyuki Mukudai*, Keiko Hashimoto, Shota Kinoshita, Mami Kaneko, Yoichiro Sugiyama and Shigeru Hirano
Department of Otolaryngology-Head and Neck Surgery, Kyoto Prefectural University of Medicine, Japan
Submission: June 19, 2023; Published: June 30, 2023
*Corresponding author: Shigeyuki Mukudai, MD, PhD, Department of Otolaryngology-Head and Neck Surgery, Kyoto Prefectural University of Medicine, Kajii-cho, Kamigyo-ku, Kyoto 602-8566, Japan
How to cite this article: Hiroki M, Shigeyuki M, Keiko Hashimoto, Shota K, Mami K, et al. Histological Assessment of The Regenerative Potential of Platelet-Rich Plasma for Severe Vocal Fold Scar in A Rat Model. Glob J Oto, 2023; 26 (1): 556180. DOI: 10.19080/GJO.2023.26.556180
Abstract
Objectives: Platelet-rich plasma (PRP) is enriched in growth factors and has been expected for tissue regeneration in several organs. Some investigators suggest regenerative potentials of PRP for human vocal fold scar, although there has been lack of animal studies on histology. We aimed to histologically reveal the possible regenerative effects of PRP for severe vocal fold scar model of rats.
Methods: Four samples of PRP were obtained from 18-week-old male Sprague-Dawley rats. The concentration of platelet-derived growth factor-BB (PDGF-BB), hepatocyte growth factor (HGF), transforming growth factor-beta (TGF-β), and basic fibroblast growth factor (bFGF) in PRP was determined using ELISA. The vocal folds of 10-week-old male SD rats were unilaterally injured, and PRP or saline (sham) was injected into the scarred vocal folds on post-injury day 56. The degree of vocal fold fibrosis was assessed on day 140 after the injection using histology.
Results: ELISA showed variation of growth factors contained in each sample of PRP. The animal study for the treatment by one of the PRP lots indicated significantly increased hyaluronic acid deposition (p = 0.026) and reduced collagen deposition (p = 0.037) in the lamina propria of the vocal fold compared with the sham group.
Conclusion: The current study suggested that intracordal injection of PRP may have the regenerative potential for severe vocal fold scar at least on histological aspect. However, it should be kept in mind that the constitution of PRP always differs, which warrants the needs to prepare PRP which contains uniform composition of growth factors.
Keywords: Platelet-rich plasma; Vocal fold; Scar; Rats; Growth factors
Abbreviations: PRP: Platelet-Rich-Plasma; PDGF-BB: Platelet-Derived Growth Factor-BB; HGF: Hepatocyte Growth Factor; TGF-β: Transforming Growth Factor-Beta; bFGF: Basic Fibroblast Growth Factor; LP: Lamina Propria; HA: Hyaluronic Acid; GMP: Good Manufacturing Practice; TA: Thyroarytenoid; EGFR: Epithelial Growth Factor Receptor; FGFR: Fibroblast Growth Factor Receptor
Introduction
Vocal fold scarring stiffens the mucosa and leads to severe disturbances in the vibratory properties. Several factors contribute to vocal fold scarring, including inflammation, irritation, iatrogenic injury, and radiation. Vocal fold scars are characterized by decrease or loss of hyaluronic acid (HA) and over-deposition of collagen in the lamina propria (LP), resulting in intractable dysphonia [1-3]. Although several therapeutic strategies have been developed, including fat injection [4], steroids [5], HA [6], hepatocyte growth factor (HGF) [7], and basic fibroblast growth factor (bFGF) [8,9], among others, there is still no optimal standard treatment for vocal fold scar. One obstacle is the lack of a readily available HGF or bFGF drug for humans, although these growth factors have shown remarkable effects at least for mild to moderate scar in human patients [10,11]. There is a commercially available bFGF drug (Fiblast R, Kaken Pharmaceutical Co.) made in Japan and has only been approved therein. The drug is not approved for vocal fold scar and can be used only off-label. No HGF drugs are available worldwide, and a good manufacturing practice (GMP)-compatible drug is available only for clinical trials in Japan [11].
Moreover, growth factors have shown marked improvement of voice in cases of mild to moderate scars; however, these have proven difficult in treating severe scars [7,8]. One possible strategy to overcome the severe scarring may be via the combination of various growth factors. Platelets play an important role in elementary hemostasis and wound healing [10,11]. Platelet-rich plasma (PRP) is plasma with concentrated platelets isolated from blood, enriched in several growth factors, including platelet-derived growth factor-BB (PDGF-BB, an isoform of PDGF family) [12], transforming growth factor-beta (TGF-β), HGF, and bFGF [13]. PRP has previously shown beneficial effects on wound healing with few side effects and is clinically used in dentistry, plastic surgery, and orthopedic surgery [14-19]. Additionally, PRP has shown beneficial effects on wound healing in an acute vocal fold injury model of rats [20].
More recently, some investigators applied PRP for human patients with vocal fold scar and reported improvements of acoustic parameters or voice handicap index as preliminary results [21,22]. Moreover, fortunately, PRP can be harvested easily from the patient and can be rather implemented worldwide. However, the critical issue resides in the lack of studies that clearly show the regenerative effects of PRP for vocal fold scars in animal models. Only preliminary human outcome studies have been reported with mixed results, and thus, reports confirming the histological improvement of scarred vocal folds using PRP treatment in animals are warranted. This study aimed to investigate the possible anti-fibrotic and regenerative effects of PRP for severe vocal fold scar in rats.
Materials and Methods
Experimental animals
All experiments were approved by the Institutional Animal Care and Use Committee of the Kyoto Prefectural University of Medicine. Male Sprague-Dawley rats (Shimizu Laboratory Supplies Co, Kyoto, Japan) were housed with standard bedding and had free access to food and water. The temperature was maintained at 22°C with a 12-h light/dark cycle.
PRP preparation
PRP was prepared as previously described [23]. Briefly, blood (4-8mL) from 18-week-old male rats was drawn using a 10mL syringe through cardiac puncture (1mL; 0.8% citric acid, 2.0% citric acid Na, and 2.45% glucose), and 2mL of blood was transferred to another tube, followed by PRP preparation. Blood was centrifuged at 2,000rpm (440 × g) for 10min, followed by separation into the plasma (upper layer), buffy coat (middle, white layer), and red blood cell layers (bottom). Subsequently, the upper and middle layers were collected, frozen at -80°C, and centrifuged at 3,000rpm (1,000 × g) for 15min. The precipitated platelets at the bottom of the centrifuge tube were collected with 300µL of supernatant to yield PRP. The following amounts of PRP were obtained from each lot (#1: 1,200µL, #2: 900µL, #3: 600µL, #4: 600µL).
Growth factor quantification using ELISA
Four samples of PRP containing growth factors (PDGF-BB, TGF-β, HGF, and bFGF) were assayed using diagnostic kits (Bio-Techno, Minneapolis, MI, USA). Assays were performed using the sandwich enzyme immunoassay technique. The PDGF-BB assay was performed in microplates coated with a monoclonal antibody against PDGF-BB. The samples and standards were prepared and diluted according to the manufacturer’s standard protocol. Thereafter, 50µL sample duplicates were applied to the microplate, and the plate was covered and incubated at room temperature (RT) for 2h.
The wells were then washed, an enzyme-conjugated polyclonal antibody against PDGF-BB was added, and the plate was incubated at RT for 2h. The plates were re-washed, and the substrate was added, followed by incubation for 30min at RT. Finally, 50µL of stop solution was added, and a fluorescence microplate reader (Glomax, Promega, Tokyo, Japan) was used to measure the absorbance at 450nm (OD450). A standard curve was generated, and PDGF-BB levels (pg/mL) of each sample were determined. TGF-β, HGF, and bFGF levels were determined using a similar enzyme immunoassay technique, according to the manufacturer’s instructions.
In vivo surgical procedure
Surgery was conducted as described previously on 10 ten-week-old male rats [24-28]. The right side of the rat vocal folds was entirely stripped using a 22-gauge needle, and the lamina propria was removed using micro-forceps until the thyroarytenoid (TA) muscle was visible, which has been confirmed to create severe scar of the vocal fold at 2 months after the stripping. Stripping was performed under deep anesthesia with a mixture of medetomidine hydrochloride (0.3mg/kg), midazolam hydrochloride (4mg/kg), and butorphanol tartrate (5mg/kg), along with atropine (0.005mg/kg) to reduce saliva and sputum secretion and topical 4% lidocaine to reduce vocal fold movement [29].
Rats were placed on a custom operating table to prepare for laryngeal deployment. The larynx was visualized using a 30°C direct rigid endoscope inserted trans-orally and connected to a video system. On postoperative day 56, 50µL of PRP or saline was gently injected into the right side of the vocal folds via a 27-gauge needle connected to a 1mL syringe. Animals were divided into either sham (saline-injected) or PRP groups (n = 5 each). The PRP, as indicated by lot #1, with the most adequate quantities was chosen for the subsequent vocal fold injections. The injection infiltrated the LP as well as the TA muscle due to the technical difficulty of injecting specifically into the LP of the small vocal folds.
Histological examination
On postoperative day 140, all animals were euthanized by intraperitoneal administration of sodium pentobarbital. Each larynx was then harvested and fixed in 4% paraformaldehyde for 4h, followed by immersion in 30% sucrose for 24h at 4°C. The larynx was placed horizontally with the dorsal side at the bottom, soaked in Tissue-Tek® O.C.T. Compound (Sakura Finetek Japan, Tokyo, Japan), and frozen in liquid nitrogen. The block was sectioned coronally into 10µm sections (CM 1850 Cryostat; Leica Instruments, Nussoch, Germany), and the sections were mounted on glass slides, air-dried, and stored at -80°C until use. The sections were stained with Alcian blue pH 2.5 (Abcam, Cambridge, UK) and Elastica van Gieson (EVG; Merck KGaA, Darmstadt, Germany).
The stained area in the middle portion of the LP was measured using a BZ-X700 analyzer (Keyence, Osaka, Japan). Alcian blue staining was performed with hyaluronidase digestion to identify HA and resulted in blue staining. EVG staining was used to identify collagen that was stained pink. The BZ-X700 analyzer software was used for automated measurements of specified colors in each section, and the percentage of stained areas for each color in the LP was calculated as the density. Normalized ratios were obtained by dividing the percentage of each area on the injured side by that on the uninjured side. Two examiners performed the measurements in a blind fashion so that they could not distinguish the slides.
Statistical analysis
Data are expressed as mean ± standard error of the mean (SEM). Samples from each group were assayed in duplicate using ELISA. In vivo, impaired t-test was used to analyze the histological data. The analysis was conducted using R (Saitama Medical Center, Jichi Medical University, Saitama, Japan) and a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). Statistical significance was set at p<0.05.
Results
Growth factor levels in PRP measured using ELISA
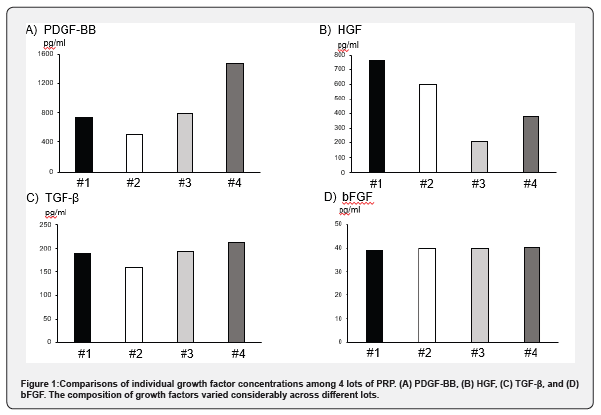
ELISA showed wide variation on the concentration of growth factors contained in each sample of PRP, particularly in PDGF-BB (872.06 ± 178.26pg/mL) and HGF (611.25 ± 141.16pg/mL) (Figure 1), while there were few differences in the concentrations of TGF-β (187.99 ± 9.50pg/mL) and bFGF (187.99 ± 9.50pg/mL) (Figure 1). The PRP of lot #1 was used for the following PRP injection study.

Histological examination
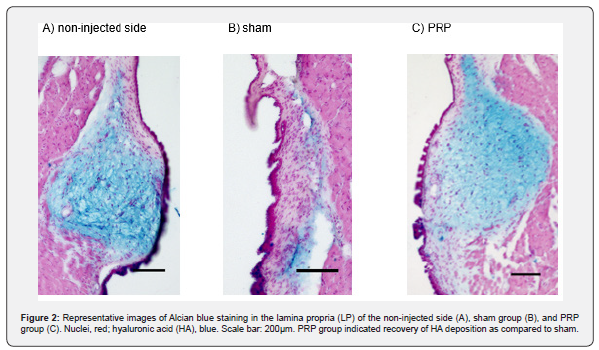
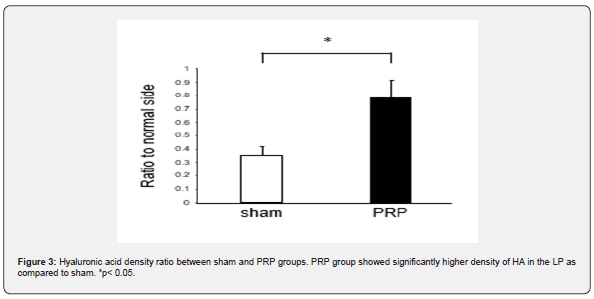
The histological examination on post-operation day 140 indicated that the injected side in the sham group had a reduced HA staining area compared to that of the non-injected side (Figure 2). In contrast, the injected side in the PRP treatment group showed a significantly restored HA staining ratio compared to that of the sham group (p = 0.026), (Figure 2 and 3). EVG staining revealed increased collagen deposition area in the injected side of the sham group compared with that of the non-injected side (Figure 4). In contrast, the collagen deposition ratio was significantly lower in the PRP treatment group than in the sham group (p = 0.037), (Figure 4 and 5).
Discussion
Vocal fold scarring is one of the most difficult pathologies in laryngology. Although growth factor therapy using HGF or bFGF has revealed significant effects for mild to moderate scars of the vocal fold in clinical trials, the effects of administration of single growth factor proved inefficacious for severe scars. The combination of growth factors may have more powerful regenerative effects. Indeed, the combination of HGF and bFGF showed enhanced regeneration of the skin after incisional wound in rats as compared to each alone [30]. PRP is regarded as the “cocktail of growth factors” and is expected to have regenerative potentials for several organs [13,31]. Some investigators reported the effects of PRP for acute wound healing of the vocal fold using animal models.
It was reported that PRP injection into acute vocal fold injury in rats considerably increased the expression of epithelial growth factor receptor (EGFR) and fibroblast growth factor receptor (FGFR) with allogenic transplant [20], similar to the results of current study. This study indicates the possible positive biological activity for better wound healing, although histological data of the treated vocal folds were lacking. Woo et al. injected PRP to injured vocal folds of rabbits autologously and indicated decreased granulation and collagen deposition in the LP treated by PRP, although the overall fibrosis did not differ between PRP-treated and sham groups [32]. These previous studies have demonstrated the positive effects of PRP on wound healing of the vocal fold; however, its anti-fibrotic effects remain unexplored.
The present study attempted to reveal the anti-fibrotic effects of PRP for chronic severe scarring of the vocal folds, and the results showed that PRP remarkably reduced fibrosis with a marked increase of HA and a decrease in collagen deposition in the LP. Despite being a preliminary study, this is the first report to confirm the anti-fibrotic effects of PRP for vocal fold scars through histology. The mechanism of the anti-fibrotic activity of PRP remains unknown. The present results indicate that PRP contains various growth factors, including PDGF, HGF, bFGF, and TGF-β, possibly among others. Unveiling every interaction among growth factors is rather challenging. However, our current study highlights the effectiveness of the current PRP lot in treating severe vocal fold scars.
Nonetheless, it remains uncertain whether other lots will yield similar results. Since the content of growth factors in PRP varies across individuals, it is crucial to consider this aspect when interpreting the results. Previous research has shown that differences in growth factors in human PRP lead to varying behavior of mesenchymal stem cells [33]. Additionally, PRP preparation methods differ across studies [34], suggesting that efforts to develop more consistent and superior quality PRP are necessary. While our study was small, the histological evidence demonstrates PRP's positive impact in treating vocal fold scars. Further research is warranted to investigate the interaction of growth factors within PRP and identify the most effective PRP preparation for treating vocal fold scars.
Conclusion
Our study findings suggest that intracordal injection of PRP has potential anti-fibrotic and regenerative effects on chronic severe vocal fold scar. However, it is crucial to note that the composition of growth factors within PRP varies between individuals, which may influence its therapeutic efficacy in treating vocal fold scars. Thus, efforts should be made to optimize the PRP preparation method to achieve consistent levels of growth factors across individual rats. Furthermore, future research should focus on uncovering the interaction between each growth factor contained in PRP and identifying the most suitable PRP for treating vocal fold scars. These investigations are necessary to improve our understanding of PRP's therapeutic potential and further advance its clinical application.
Acknowledgment
This study was supported by a Grant-in-Aid for Scientific Research (B; Grant Number 19H03805 and C; Grant Number 22K09671) and a Grand-in-Aid for Early-Career Scientists (Grant Number 22K16902).
References
- Hirano S, Minamiguchi S, Yamashita M, Ohno T, Kanemaru S, et al. (2009) Histologic characterization of human scarred vocal folds. J Voice 23(4): 399-407.
- Rousseau B, Hirano S, Scheidt TD, Welham NV, Thibeault SL, et al. (2003) Characterization of vocal fold scarring in a canine model. Laryngoscope 113(4): 620-627.
- Rousseau B, Hirano S, Chan RW, Welham NV, Thibeault SL, et al. (2004) Characterization of chronic vocal fold scarring in a rabbit model. J Voice 18(1): 116-124.
- Sataloff RT, Spiegel JR, Hawkshaw M, Rosen DC, Heuer RJ (1997) Autologous fat implantation for vocal fold scar: a preliminary report. J Voice 11(2): 238-246.
- Govil N, Rafii BY, Paul BC, Ruiz R, Amin MR, et al. (2014) Glucocorticoids for vocal fold disease: a survey of otolaryngologists. J Voice 28(1): 82-87.
- Thibeault SL, Rousseau B, Welham NV, Hirano S, Bless DM (2004) Hyaluronan levels in acute vocal fold scar. Laryngoscope 114(4): 760-764.
- Ohno T, Yoo MJ, Swanson ER, Hirano S, Ossoff RH, et al. (2009) Regeneration of aged rat vocal folds using hepatocyte growth factor therapy. Laryngoscope 119(7): 1424-1430.
- Suzuki R, Kawai Y, Tsuji T, Hiwatashi N, Kishimoto Y, et al. (2017) Prevention of vocal fold scarring by local application of basic fibroblast growth factor in a rat vocal fold injury model. Laryngoscope 127(2): E67-E74.
- Kumai Y (2019) Pathophysiology of Fibrosis in the Vocal Fold: Current Research, Future Treatment Strategies, and Obstacles to Restoring Vocal Fold Pliability. Int J Mol Sci 20(10): 2551.
- Holinstat M (2017) Normal platelet function. Cancer Metastasis Rev 36(2): 195-198.
- Nurden AT, Nurden P, Sanchez M, Andia I, Anitua E (2008) Platelets and wound healing. Front Biosci 13: 3532-3548.
- Fredriksson L, Li H, Eriksson U (2004) The PDGF family: four gene products form five dimeric isoforms. Cytokine Growth Factor Rev 15(4): 197-204.
- Eppley BL, Woodell JE, Higgins J (2004) Platelet quantification and growth factor analysis from platelet-rich plasma: implications for wound healing. Plast Reconstr Surg 114(6): 1502-1508.
- Plachokova AS, Nikolidakis D, Mulder J, Jansen JA, Creugers NH (2008) Effect of platelet-rich plasma on bone regeneration in dentistry: a systematic review. Clin Oral Implants Res 19(6): 539-545.
- Tozum TF, Demiralp B (2003) Platelet-rich plasma: a promising innovation in dentistry. J Can Dent Assoc 69(10): 664.
- Eppley BL, Pietrzak WS, Blanton M (2006) Platelet-rich plasma: a review of biology and applications in plastic surgery. Plast Reconstr Surg 118(6): 147e-159e.
- Sommeling CE, Heyneman A, Hoeksema H, Verbelen J, Stillaert FB, et al. (2013) The use of platelet-rich plasma in plastic surgery: a systematic review. J Plast Reconstr Aesthet Surg 66(3): 301-311.
- Hall MP, Band PA, Meislin RJ, Jazrawi LM, Cardone DA (2009) Platelet-rich plasma: current concepts and application in sports medicine. J Am Acad Orthop Surg 17(10): 602-608.
- Alsousou J, Thompson M, Hulley P, Noble A, Willett K (2009) The biology of platelet-rich plasma and its application in trauma and orthopaedic surgery: a review of the literature. J Bone Joint Surg Br 91(8): 987-996.
- Cobden SB, Ozturk K, Duman S, Esen H, Aktan TM, et al. (2016) Treatment of Acute Vocal Fold Injury with Platelet-Rich Plasma. J Voice 30(6): 731-735.
- Woo P, Murry T (2021) Short-Term Voice Improvement after Repeated Office-Based Platelet-Rich Plasma PRP Injection in Patients with Vocal Fold Scar, Sulcus, and Atrophy. J Voice.
- van der Woerd B, O'Dell K, Castellanos CX, Bhatt N, Benssousan Y, et al. (2023) Safety of Platelet-Rich Plasma Subepithelial Infusion for Vocal Fold Scar, Sulcus, and Atrophy. Laryngoscope 133(3): 647-653.
- Sakata M, Tonomura H, Itsuji T, Ishibashi H, Takatori R, et al. (2018) Bone Regeneration of Osteoporotic Vertebral Body Defects Using Platelet-Rich Plasma and Gelatin β-Tricalcium Phosphate Sponges. Tissue Eng Part A 24(11-12): 1001-1010.
- Tateya T, Tateya I, Sohn JH, Bless DM (2005) Histologic characterization of rat vocal fold scarring. Ann Otol Rhinol Laryngol 114(3): 183-191.
- Ozawa S, Mukudai S, Kaneko M, Kinoshita S, Hashimoto K, et al. (2021) Anti-inflammatory and Antioxidant Effects of Japanese Herbal Medicine Kyoseihatekigan on Vocal Fold Wound Healing. J Voice.
- Ogawa M, Mukudai S, Sugiyama Y, Matsushita H, Kinoshita S, et al. (2022) The Effects of Amniotic Membrane Transplantation on Vocal Fold Regeneration. Laryngoscope 132(10): 2017-2025.
- Hashimoto K, Kaneko M, Kinoshita S, Ozawa S, Mukudai S, et al. (2021) Effects of Repeated Intracordal Glucocorticoid Injection on the Histology and Gene Expression of Rat Vocal Folds. J Voice.
- Tateya I, Tateya T, Sohn JH, Bless DM (2016) Histological Effect of Basic Fibroblast Growth Factor on Chronic Vocal Fold Scarring in a Rat Model. Clin Exp Otorhinolaryngol 9(1): 56-61.
- Kawai S, Takagi Y, Kaneko S, Kurosawa T (2011) Effect of three types of mixed anesthetic agents alternate to ketamine in mice. Exp Anim 60(5): 481-487.
- Ono I, Yamashita T, Hida T, Jin HY, Ito Y, et al. (2004) Combined administration of basic fibroblast growth factor protein and the hepatocyte growth factor gene enhances the regeneration of dermis in acute incisional wounds. Wound Repair Regen 12(1): 67-79.
- Miron RJ, Fujioka-Kobayashi M, Bishara M, Zhang Y, Hernandez M, et al. (2017) Platelet-Rich Fibrin and Soft Tissue Wound Healing: A Systematic Review. Tissue Eng Part B Rev 23(1): 83-99.
- Woo SH, Jeong HS, Kim JP, Koh EH, Lee SU, et al. (2014) Favorable vocal fold wound healing induced by platelet-rich plasma injection. Clin Exp Otorhinolaryngol 7(1): 47-52.
- Cho HS, Song IH, Park SY, Sung MC, Ahn MW, et al. (2011) Individual variation in growth factor concentrations in platelet-rich plasma and its influence on human mesenchymal stem cells. Korean J Lab Med 31(3): 212-218.
- Tey RV, Haldankar P, Joshi VR, Raj R, Maradi R (2022) Variability in Platelet-Rich Plasma Preparations Used in Regenerative Medicine: A Comparative Analysis. Stem Cells Int 2022: 3852898.





























