ECAP & Auditory Nerve Stimulation in Cochleostomy Versus Round Window Insertion Approaches for Cochlear Implantation
Sahar Madkhali, Rawan Mandura* and Khalid Alnoury
Otolaryngology Head and Neck Surgery Department, King Abdulaziz University, Saudi Arabia
Submission:May 16, 2022;Published:May 24, 2022
*Corresponding author: Rawan Mandura MD, Otolaryngology Department, King Abdulaziz University, Jeddah, Saudi Arabia
How to cite this article: Sahar M, Rawan M, Khalid A. ECAP & Auditory Nerve Stimulation in Cochleostomy Versus Round Window Insertion Approaches for Cochlear Implantation. Glob J Oto, 2022; 25 (2): 556158. DOI: 10.19080/GJO.2022.25.556158
Abstract
Background: Cochlear implants convert sound signal into electrical stimulation to stimulate distal auditory nerve fibers in order to produce hearing, an important factor in this process is the variation on the current levels needed to elicit hearing for each individual and stimulation channel. The two major approaches used for electrode insertion, cochleostomy and round window insertion, have different insertion angle and different radial distance (electrode-modiolar interface) which may affect the stimulation current levels. Previous studies have been investigating the ramifications of these variances upon hearing and speech perception outcomes with several means. ECAP (Electrically evoked compound action potential) is an objective measurement of how the auditory nerve responds to electrical stimulation; it can be used to explore the auditory nerve response at each electrode contact on the cochlear implant array and allows simple verification of electrode-nerve interface.
Objective: The aim of our study was to examine whether cochlear implantation using the round window approach versus cochleostomy achieves comparable action potential and auditory stimulation in apical, middle and basal electrodes of cochlea by comparing ECAP measurements in these two techniques.
Methodology: Retrospective analysis included Twenty- one Cochlear implant patients, twelve patients implanted through cochleostomy and nine patients through round window insertion, the mean age were 4.28 years ranging between (1-15 years). To avoid the influence of inter- corporation software differences, all the patients selected were implanted by devices from the cochlear company, the multichannel cochlear implants have 22 electrodes, Electrodes were split into 3 groups, from 1-7 grouped as basal electrodes, 8-15 grouped as middle, 16-22 as apical electrodes. The intraoperative ECAP measurements were collected for 2 electrodes from each group for all patients.
Results: No statistically significant differences were found between the two groups when assessed for current stimulation of high-frequency sounds [basal1 p= 0.95, basal2 p= 0.47], mid-frequency sounds [mid1 p= 0.64, mid2 p= 0.35] and low- frequency sounds [apical1 p= 0.687, apical2 p= 0.57].
Conclusion: There is no difference in the action potential of the distal portion of the auditory nerve in cochlear implant patients operated with cochleostomy approach or with round window insertion, both techniques provide comparable stimulation of the cochlear nerve for high, mid and low frequency sounds.
Keywords: Cochlear implantation; Round window approach; Cochleostomy; ECAP; Auditory nerve stimulation
Abbreviations: CI: Cochlear implantation; FDA: Food and Drug Administration; ECAP: Evoked Compound Action Potential; MCL: Maximum Comfort Level; SPSS: Statistical Package For Social Science; SD: Standard Deviation; RW: Round Window; ESRT: Electrically Evoked Stapedius Reflex Thresholds
Introduction
Cochlear implantation (CI) is a sound restoration surgery that has been utilized in cases of sever to profound deafness for both adults and children. The cochlear implants (CIs) replace the function of hair cells by converting sound signals into electrical impulses that lead to auditory nerve stimulation. Worldwide, it considered to be a common surgery as there are more than 3,24,000 people with CIs currently. Furthermore, it is estimated that approximately 50,000 CI procedures occurring annually [1,2].
In 1985, the United States Food and Drug Administration (FDA) approved multichannel CIs for adults with profound hearing loss. Subsequently, in 1990, implantation was approved for children. Afterwards, CI surgery has become the standard of care for patients with severe-to-profound sensorineural hearing loss. Multiple factors have been implicated in the hearing outcome, including both extrinsic and intrinsic factors.
The intrinsic factors, however, are often difficult to modify or overcome. There are many significant predictive factors have been described in the literature [3-5]. These include, but are not limited to, pre/postlingual status [6], intracochlear trauma, and distance from modiolus [7], duration of deafness [8] and level of preimplant speech recognition [9]. Of note, Recipient age does not appear to have a significant impact on hearing outcomes in elderly candidates [10,11]. Various objective measures have been widely used for auditory nerve stimulation assessment. In addition, different techniques have been advocated, including the use of thresholds of the Electrically Evoked Compound Action Potential (ECAP) [12] And the stapedial reflex (ESR) [13]. The ECAP represents a synchronized response generated by a group of electrically activated auditory nerve fibers.
Consequently, analyzing this physiological response to the electrical stimulation transmitted by the implant, information can be obtained regarding the expected and actual function of the peripheral nerve. Therefore, this response is particularly advantageous because it allows the clinician to directly measure auditory nerve fibre potentials on implanted patients. The clinical utility of the ECAP measures have been investigated in the literature, and it has many useful applications, including objective verification of auditory nerve function in response to electrical stimulation, objective verification of electrode/device function, assistance in programming the speech processor for individuals who cannot provide reliable behavioral responses, and verification or confirmation of the accuracy of questionable behavioral responses [14]. This response can be used both during and after CI surgery. Intraoperatively, it is beneficial for intracochlear electrode placement adjustment, and also for technical functional testing. On the other hand, the ECAP recordings postoperatively can be used to measure the neuronal potentials elicited by electrode stimulation along the basilar membrane.
Additionally, these measurements are helpful in programming and fitting as they can determine the upper and lower limits of the stimulation current [the hearing threshold (THR) and the maximum comfort level (MCL)] [14-18] or to evaluate the stimulation current field along the cochlea and the interaction of individual electrodes [13-15]. The two major approaches used for electrode insertion during CI surgery, including cochleostomy and round window insertion, have different insertion angle [16] and different radial distance (electrode-modiolar interface) [17], which may affect the stimulation current levels. Previous studies have been investigating the ramifications of these variances upon hearing and speech perception outcomes with several means. The aim of our study is to examine whether cochlear implantation using the round window approach versus cochleostomy achieves comparable action potential and auditory stimulation in apical, middle and basal electrodes of cochlear by comparing ECAP measurements in these two techniques.
Methodology
Design and Setting
A retrospective controlled analysis was carried out at Department of Otolaryngology, head and neck surgery, king Abdul-Aziz University Hospital, Jeddah, Saudi Arabia. The study protocol was approved by the national biomedical ethics research committee of King Abdul-Aziz University (reference No. 133-19).
Subjects
The study included pediatric patients who had cochlear implant via cochleostomy (Group 1) or round window insertion (Group 2), from March 2019 to March 2020. All included patients had profound sensory-neural hearing loss of unknown etiology, with a normal cochlear anatomy as evidenced both in the preoperative radiological investigations (computed tomography) and during the operative procedure. Patients with congenital anomalies of the inner ear, those with other implanted devices and those with failure to obtain ECAP from either basal, middle or apical electrodes were excluded. Additionally, patients with intraoperative complications were excluded.
Procedure
To prevent inter-surgeon variance, all cochlear implantations were performed by the same surgeon, following the same standard steps of postauricular incision, simple mastoidectomy, posterior tympanotomy with full insertion of the electrodes through either round window or cochleostomy, anterior and inferior to round window. Further, to avoid any device or software programming bias, all implants were from the same manufacturer, Cochlear company.
Data Collection
With the contribution of an experienced audiologist, ECAP data were collected from the cochlear programming system. The multichannel cochlear implants comprise 22 electrodes, which were classified according to their position into 3 sections: basal (electrodes 1-7), middle (electrodes 8-15), and apical (electrodes 16-22). In each section, two intraoperative ECAP measures were conducted in two different electrodes. This resulted into 6 measurements for each patient, 2 basal, 2 middle and 2 apical, randomly selected for each group.
Statistical Analysis
Data was analyzed using the statistical package for social science (SPSS) version 20. Independent t-test was used to analyze the comparisons of mean ECAP measurements between the two groups within each of the selected electrodes; values are presented as mean and standard deviation (SD), with the t-statistics, the degree of freedom and the significance level, which was fixed as p-value<0.050.
Result
A total 21 patients were eligible, 12 implanted through cochleostomy (Group 1) and 9 through round window insertion (Group 2). The mean age of the total population was 4.28, range =1-15 years. Intraoperative ECAP measurements showed no statistically significant difference between the two surgical techniques in any of the 6 electrodes (P>0.05). However, we noted that patients who underwent round window insertion had relatively lower ECAP values in the middle electrodes (mean ECAP=180.66 versus 186.58 and 172.44 versus 183.83 mV) and relatively higher values in the apical electrodes (mean=170.88 versus 165.58 and 174.55 versus 167.16 mV), compared to cochleostomy respectively (Table 1), (Chart 1).
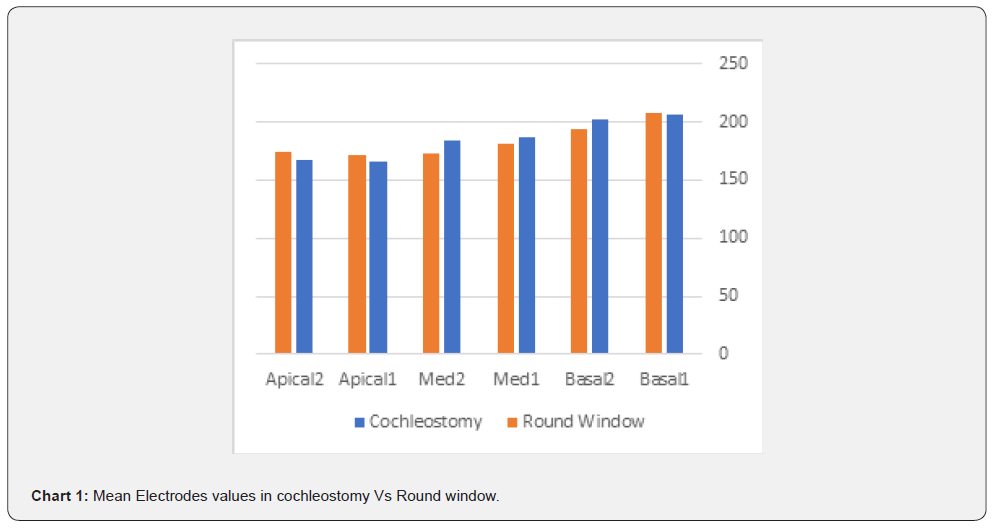
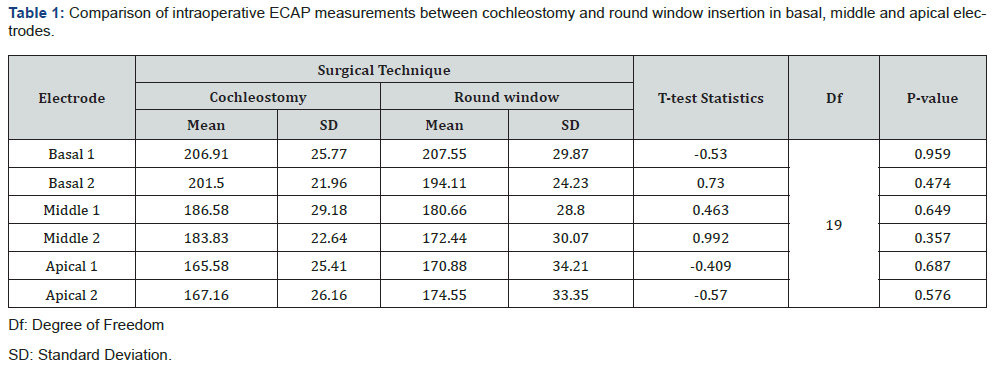
Discussion
The auditory nerve is stimulated by the electrical impulses delivered by CI, before the higher auditory neural structures receive these impulses and the decoded information produced. Theoretically, the ability of the auditory nerve to faithfully encode and process electrical stimuli should be important for CI outcomes. Studies have proved that the physiological status (i.e., number and responsiveness of neurons) of the auditory nerve may be important for CI outcomes [18,19]. The ECAP is a direct measurement of neural responses generated by auditory nerve fibers, which makes it feasible to exclusively evaluate the physiological status of the auditory nerve. It represents a synchronous response from electrically stimulated auditory nerve fibers, providing information regarding the status of the auditory nerve.
As the ECAP measured intraoperatively is one of the first markers of auditory nerve function after cochlear implant surgery, many studies have focused on evaluating the feasibility of using the ECAP to determine stimulus levels for individual electrodes in CI patients [9,6]. The intraoperative uses of the ECAP can confirm auditory nerve, device integrity, and electrode functionality, and the postoperative ECAP measurements allow the audiologist in speech processor programming as they have been previously associated with speech perception scores. In addition, the ECAP measurements have been correlated with both detection thresholds (T-levels) and maximum comfortable loudness (C-levels) [9,6], and the ECAP maximum amplitude has been linked to speech perception scores after CI [20,21].
The ECAP threshold and amplitude differ in accordance to region of cochlea that has been stimulated. In other word, there is significant increase in ECAP amplitude towards the apical region of the cochlea and significant decrease in ECAP threshold towards the apex. Brill et al [22] reported these findings and had explained them as following: Firstly, the narrowed distance between the recording electrode and the stimulated neural tissue (because of reduced diameter of the cochlear turns towards the apex) and secondly, the greater density or survival rate of neuronal tissue adjacent to the electrode in the apical region explain the significant increase in ECAP amplitude towards the apical region. These two factors could also lead to a reduction in the stimulation amplitude required to trigger an action potential explain the significant decrease in ECAP threshold towards the apex.
Differences in impedances of the electrode-tissue interfaces between cochlear regions cannot explain this effect because the implant uses current sources and high-impedance recording circuitry and is therefore not affected by different electrode-tissue interface impedances. Although CI procedures were originally intended only for patients with total deafness because the procedure could damage and destroy any residual hearing, different modifications have recently been implemented with the aim of minimizing intracochlear trauma and also optimizing placement of electrode contacts with respect to spiral ganglion neurons of the cochlea that lead to preservation of residual hearing post CI surgery, including soft surgical approaches and techniques such as blood and bony dust entry, steroid use, surgical site of insertion, perilymph leakage and suctioning, and depth of insertion [23]. In addition to that using electrode design and hybrid electric–acoustic processors.
This new trend of preservation of residual acoustic hearing in the cochlear apex during CI surgery is supported by growing evidence, indicating that combined electric and acoustic stimulation lends to improved speech recognition in complex listening environments, sound localization, music appreciation, and decreased listening effort [24]. Despite these advances, the degree of hearing preservation varies significantly across individuals [25,26]. An important factor in CIs performance is the modiolar electrode distance. As the tissue impedance plays a significant role in electrical current transmission, which impedes these current from getting delivered to the modiolus [27,28], a shorter distance between the current stimulus and the spiral ganglion may improve stimulus transmission while also reducing broad current spread [29], thereby improving spectral resolution. The Modiolar hugging electrode arrays, new modification of the electrode arrays, designed to reduce the distance between the stimulus and the target neural substrate, thereby increasing the likelihood of efficiently transmitting the electrical current.
Several studies have reported that these type electrodes are associated with an increased probability of inducing intracochlear damage and destroying residual hearing [16,30]. The round window insertion (RWI) and cochleostomy approaches are the two most common surgical techniques used in CI. The ideal approach for electrode array insertion is debated and there is no consensus in literature. Previous reports had been in favor of cochleostomy approaches, anterior and inferior to the round window (RW). The RW proximity to the osseous spiral lamina and membranes, intracochlear fluid dynamics made the cochleostomy approach more favorable as it reduces disruption to intracochlear fluid dynamics and the cochlear aqueduct [31] and increase electrode contact distance to the osseous spiral lamina and membranes.
Several studies, thereafter, has supported the RWI approach and there has been a growing trend towards round window insertion [32], with reports of less traumatic insertions in cadaveric dissection studies [33,16]. It has been previously proposed that cochleostomy approaches, anterior and inferior to the round window (RW), is more favorable because of RW proximity to the osseous spiral lamina and membranes, intracochlear fluid dynamics, therefore, cochleostomy approach may reduce disruption to intracochlear fluid dynamics and the cochlear aqueduct and increase electrode contact distance to the osseous spiral lamina and membranes. More recently, cadaveric dissections and clinical outcome studies have suggested that RWIs might be advantageous because this surgical technique results in less traumatic insertions than the traditional cochleostomy approach [33,16].
Jiam et al [34] reported that the RWI technique is associated with an increased likelihood of perimodiolar placement, which was confirmed radiologically, and place electrodes closer to cochlear neural substrates and minimize current spread. In this study we examined weather certain surgical approach, RWI versus cochleostomy, has a superior auditory nerve stimulation by comparing the intraoperative ECAP measurements in the in apical, middle and basal electrodes of cochlea in these two techniques. The analysis concluded that there are no significant difference in the ECAP results between the two approaches and both techniques achieved a comparable auditory nerve stimulation.
Interestingly, the same insignificant results had been reported by Hamerschmidt et al [35] which indicate that both surgical approaches produced almost the same actions potential to elicit the auditory nerve stimulation and they have concluded that the choice of either technique is primary depends the surgeon’s preference. On the contrarily, Karatas et al [36] reported that the RWI approach provided a better auditory nerve stimulation than cochleostomy approach when the electrically evoked stapedius reflex thresholds (ESRT) used for comparing the two surgical techniques.
Conclusion
Our study indicates that there is no difference in the ECAP measurement of the distal portion of the auditory nerve in cochlear implant patients operated with cochleostomy approach or with round window insertion, both techniques provide comparable stimulation of the cochlear nerve for high, mid and low frequency sounds. Although the two techniques have different insertion angle and different radial distance (electrode-modiolar interface), they do not have an effect on the ECAP and auditory nerve stimulation. Therefore, the surgical approach depends on surgeon preference.
References
- Hochmair I (2013) Cochlear implants: facts.
- (2013) National Institute on Deafness and Other Communication Disorders. NIDCD Fact Sheet: Cochlear Implants. Bethesda, MD: National Institutes of Health.
- Summerfield AQ, Marshall DH (1995) Preoperative predictors of outcomes from cochlear implantation in adults: performance and quality of life. Ann Otol Rhinol Laryngol Suppl 166: 105-8.
- Tong YC, Busby PA, Clark GM (1988) Perceptual studies on cochlear implant patients with early onset of profound hearing impairment prior to normal development of auditory, speech, and language skills. J Acoust Soc Am 84(3): 951-962.
- Adunka O, Kiefer J (2006) Impact of electrode insertion depth on intracochlear trauma. Otolaryngol Head Neck Surg 135(3): 374-382.
- Hughes ML, Brown CJ, Abbas PJ, Wolaver AA, Gervais JP (2000) Comparison of EAP thresholds with MAP levels in the nucleus 24 cochlear implant: data from children. Ear Hear 21(2): 164-174.
- Jerger J, Oliver TA, Chmiel RA (1988) Prediction of dynamic range from stapedius reflex in cochlear implant patients. Ear Hear 9(1): 4-8.
- Brown CJ, Abbas PJ, Gantz BJ (1998) Preliminary experience with neural response telemetry in the nucleus CI24 M cochlear implant. Am J Otol 19(3): 320-327.
- Brown CJ, Hughes ML, Luk B, Abbas PJ, Wolaver A, et al. (2000) The relationship between EAP and EABR thresholds and levels used to program the nucleus 24 speech processor: data from adults. Ear Hear 21(2): 151-163.
- Franck KH (2002) A model of a nucleus 24 cochlear implant fitting protocol based on the electrically evoked whole nerve action potential. Ear Hear 23(1 Suppl): 67S-71S.
- Franck KH, Norton SJ (2001) Estimation of psychophysical levels using the electrically evoked compound action potential meas- ured with the neural response telemetry capabilities of Cochlear Corporation's CI24 M device. Ear Hear 22(4): 289-299.
- Hughes ML, Vander WKR, Brown C, Abbas P, Kelsay D, et al. (2001) A longitudinal study of electrode impedance, the electrically evoked compound action potential, and behavioral measures in nucleus 24 cochlear implant users. Ear & Hear 22(6): 471-486.
- Abbas PJ, Hughes ML, Brown CJ, Miller CA (2004) Channel interaction in cochlear implant users evaluated using the electrically evoked compound action potential. Audiol Neurootol 9(4): 203-213.
- Cohen LT, Richardson LM, Saunders E, Cowan RSC (2003) Spatial spread of neural excitation in cochlear implant recipients: compari- son of improved ECAP method and psychophysical forward masking. Hear Res 179(1): 72-87.
- Cohen LT, Saunders E, Richardson LM (2004) Spatial spread of neural excitation: comparison of compound action potential and forward-masking data in cochlear implant recipients. Int J Audiol 43(6): 346-355.
- Wanna GB, Noble JH, Carlson ML, Gifford RH, Dietrich MS, et al. (2014) Impact of electrode design and surgical approach on scalar location and cochlear implant outcomes. Laryngoscope 124 Suppl 6(0 6): S1-S7.
- Saunders E, Cohen L, Aschendorff A, Shapiro W, Knight M, et al. (2002) Threshold, Comfortable Level and Impedance Changes as a Function of Electrode-Modiolar Distance. Ear Hear 23(1 Suppl): 28S-40S.
- Kim JR, Abbas PJ, Brown CJ, Etler CP, O Brien S, et al. (2010) The relationship between electrically evoked compound action potential and speech perception: a study in cochlear implant users with short electrode array. Otol Neurotol 31(7): 1041-1048.
- Kirby AE, Middlebrooks, JC (2010) Auditory temporal acuity probed with cochlear implant stimulation and cortical recording. J Neurophysiol 103(1): 531-542.
- Kim JR, Tejani VD, Abbas PJ, Brown CJ (2017) Intracochlear recording of acoustically and electrically evoked potentials in nucleus hybrid L24 cochlear implant users and their relationship to speech perception. Front Neurosci 11: 216.
- Schvartz-Leyzac KC, Pfingst BE (2018) Assessing the relationship between the electrically evoked compound action potential and speech recognition abilities in bilateral cochlear implant recipients. Ear Hear 39(2): 344-358.
- Brill S, Muller J, Hagen R, Alexander M, Steffi-Johanna B, et al. (2009) Site of cochlear stimulation and its effect on electrically evoked compound action potentials using the MED-EL standard electrode array. BioMed Eng Online 8: 40.
- Lehnhardt E (1993) Intracochlear placement of cochlear implant electrodes in soft surgery technique [in German]. HNO 41(7): 356-359.
- Turner C, Gantz BJ, Reiss L (2008) Integration of acoustic and electrical hearing. J Rehabil Res Dev 45(5): 769-778.
- Gantz BJ, Dunn C, Oleson J, Hansen M, Parkinson A, et al. (2016) Multicenter clinical trial of the Nucleus Hybrid S8 cochlear implant: final outcomes. Laryngoscope 126(4): 962-973.
- Helbig S, Adel Y, Rader T, Stover T, Baumann U (2016) Long-term hearing preservation outcomes after cochlear implantation for electric-acoustic stimulation. Otol Neurotol 37(9): e353-e359.
- Spelman FA, Clopton BM, Pfingst BE (1982) Tissue impedance and current flow in the implanted ear: implications for the cochlear prosthesis. Ann Otol Rhinol Laryngol Suppl 98: 3-8.
- Suesserman MF, Spelman FA (1993) Quantitative in vivo measurements of inner ear tissue resistivities. I. in vitro characterization. IEEE Trans Biomed Eng 40(10): 1032-1047.
- Davis TJ, Zhang D, Gifford RH, Dawant BM, Labadie RF, et al. (2016) Relationship between electrode-to-modiolus distance and current levels for adults with cochlear implants. Otol Neurotol 37(1): 31-37.
- Boyer E, Karkas A, Attye A, Lefournier V, Escude B, et al. (2015) Scalar localization by cone-beam computed tomography of cochlear implant carriers: a comparative study between straight and periomodiolar precurved electrode arrays. Otol Neurotol 36(3): 422-429.
- Lehnhardt E (1993) Intracochlear placement of cochlear implant electrodes in soft surgery technique. HNO 41(7): 356-359.
- Richard C, Fayad JN, Doherty J, Linthicum FH (2012) Round window versus cochleostomy techniques in cochlear implantation: histological findings. Otol Neurotol 33(7): 1181-1187.
- Adunka O, Unkelbach MH, Mack M, Hambek M, Gstoettner W, et al. (2004) Cochlear implantation via the round window mem- brane minimizes trauma to cochlear structures: a histologically controlled insertion study. Acta Otolaryngol 124(7): 807-812.
- Jiam NT, Jiradejvong P, Pearl MS, Limb CJ (2016) The Effect of Round Window vs Cochleostomy Surgical Approaches on Cochlear Implant Electrode Position: A Flat-Panel Computed Tomography Study. JAMA Otolaryngol Head Neck Surg 142(9): 873-880.
- Rogerio H, Luiz HS, Rodrigo KR, Gislaine RMW, Adriana KPO, et al. (2012) A comparison between neural response telemetry via cochleostomy or the round window approach in cochlear implantation. Braz J Otorhinolaryngol 78(4): 71-75.
- Karatas E, Aud MD, Baglam T, Durucu C, Baysal E, et al. (2011) Intraoperative electrically evoked stapedius reflex thresholds in children undergone cochlear implantation: round window and cochleostomy approaches. Int J Pediatr Otorhinolaryngol 75(9): 1123-1126.





























