Oxidative Stress, Sensorineural Deafness Linked to Advancing Age - Presbycusis: Systematic Review
Sokolo Gedikondele Jérôme1, Longo Mbenza Benjamin1,2,3,4, Lepira Bompeka François1, Mvitu Muaka Moise1, Mambueni Thamba Christophe1, Nkodila Natuhoyila Aliocha4* and Tshibambe Tshiondo Richard5,6
1Faculty of Médecine, University of Kinshasa, Democratic Republic of Congo
2Faculty of Sciences, Walter Sisulu University, MTHATHA, South Africa
3Faculty of Médecine, University of President Joseph Kasavubu, Democratic Republic of Congo
4Faculty of Public Health, Lomo University of Research, Democratic Republic of Congo
5National Pilot Institute for Health Sciences Education of Kinshasa, Democratic Republic of Congo
6Higher Institute of Medical Techniques of Kinshasa, Democratic Republic of Congo
Submission:September 22, 2020; Published:October 13, 2020
*Corresponding author:Aliocha Nkodila Natuhoyila, Faculty of Public Health, Lomo University of Research, Democratic Republic of Congo
How to cite this article: Sokolo G J, Longo MB, Lepira B F, Mvitu M M, Mambueni T C, et al. Oxidative Stress, Sensorineural Deafness Linked to Advancing Age - Presbycusis: Systematic Review. Glob J Oto, 2020; 23 (4): 556118 DOI: 10.19080/GJO.2020.23.556118
Abstract
Background: Oxidative stress in the elderly is a serious problem due to the alteration of the otological system and leads to presbycusis. The aims of this study were to reduce information bias and disseminate the European consensus around oxidative stress in the face of certain controversies around this stress in presbycusis.
Methods: a systematic review using the google scholar, Medline, Home Pubmed, Home PMC and DMS / CDC / ONU / STDA engines, provided scientific and information on oxidative stress, deafness and presbycusis.
Results: This systematic review showed that deafness linked to sound trauma to muscular sound exposure or other loud noises such as gunfire or explosions and to a multifactorial etiology. Aging leads to progressive hearing loss (presbycusis) which is linked to the damage produced by free radicals during oxidative stress.
Conclusion: The complexity of the effects of senescence, senility, genetic factors during fetal life, ecotoxic effects and mutations are involved in presbycusis.
Keywords: Oxidative stress; Advancement in age; Presbycusis; Free radicals
Introduction
Sensorineural deafness (SNS) linked to advancing age, or presbycusis, is currently the subject of concern of scientific researchers [1]. Indeed, presbycusis is a hearing disorder that develops by a gradual and constant decrease with advancing age or senescence if not natural physiological appearance of the auditory system after the age of 60 years [2-5]. The decrease in average hearing in the very high frequencies from the age of 25 varies according to individuals from 0.5dB per year and accentuated from 65 years, 1dB from 75 years and 2dB from 85 years old [2]. Biological plausibility has also accumulated information relating to oxidative (oxidative) stress in general (1) and oxidative stress in deafness associated with advancing age better defined as presbycusis in particular [3-8]. Understanding the typical etiology and physiopathological mechanisms allows a holistic approach to oxidative stress in seniors [9]. Thus, the objective of this systematic review was to reduce information bias and disseminate the European consensus around oxidative stress in the face of certain controversies around this stress in presbycusis [1].
Methods
Engines including google scholar, Medline, Home Pubmed, Home PMC and DMS / CDC / ONU / STDA, were used to organize scientific and factual information (evidence = Factorial medicine / Evidence based medicine) around the following keywords: oxidative stress, deafness and presbycusis [10-12].
The information has been rigorously presented according to the following sections:
A. describe the main reactive radicular or non-radicular pro-oxidative species:
a. oxidative stress and mechanisms.
b. etiopathogenic form of deafness.
c. physiopathology of presbycusis in chronobiology / Illikibiology.
B. highlight the gene candidate for presbycusis, oxidative stress and mitochondrial dysfunction:
a. mitochondrial DNA deletion and mutation and presbycusis.
b. animal model of the role of oxidative stress and mitochondrial dysfunction in presbycusis.
c. preventive approach and action for the late onset of presbycusis: supplementation / diet and / or calorie restriction.
Results
This review focused on types of sensorineural hearing loss including sensorineural presbycusis [4,13].
Therefore, the results were organized as follows:
a. epidemiology and type of presbycusis.
b. free radicals and oxidative stress.
c. oxidative stress around general concepts.
d. physiopathological mechanisms of oxidative stress.
e. oxidative stress and sensorineural deafness related to age.
f. antioxidants.
g. clinical, therapeutic implications and public health perspectives around presbycusis.
Presbycusis
Presbycusis has been characterized by the following main types:
a. neurosensory presbycusis in hearing loss involving only the high frequencies.
b. nervous presbycusis leading to a disturbance of the thresholds on the acute frequencies and the distinction of alterations in blood circulation for all frequencies.
c. Mechanical presbycusis disturbs acute frequencies [13]. Epidemiology of deafness
The World Health Organization (WHO) considers hearing impairment as concepts of severity-progression: less severe disabling hearing loss and deafness, a profound progression from disabling hearing loss [9,14]. Disabling hearing loss forms deafness according to the advancement of age from childhood through adulthood to senescence / senility in the world: disabling hearing loss in children (hearing loss ≥30 dB at tonal audiometry [9,14,15] with 60% of cases where 34 million among the 466 million people in the world including adults (hearing loss ≥40 dB in the better ear) in the world [9,15].
The risk factors for deafness vary according to the clinical forms and the chronobiology at birth have been clearly defined and updated by Joint Committe or Infant Hearing [16,17] congenital infections (Rubella, syphilis, CMV, herpes simplex, toxoplasm), chickenpox zoster, HIV, weight<1000g, premature, perinatal hypoxia, hyperbilirubinemia, taking ototoxic drugs by the pregnant mother, bacterial meningitis, the most common mutation (conferring high risk of congenital deafness and perinatal deafness) is that of connexin 26, a protein involved in the ionic exchanges of the organ of corti [18-20].
a) hearing loss related to sound trauma is linked to the professional use of noisy devices, to muscular sound exposure or to other loud noises such as gunfire or blasts and to a multifactorial etiology.
b) aging leads to progressive hearing loss (presbycusis). This presbycusis is the main cause of deafness [21-23].
Origin of presbycusis
Presbycusis is a gradual and insidious hearing loss affecting both ears symmetrically, particularly at high frequencies, the effects of which are accentuated in a noisy environment [9,22-27]. Presbycusis affects more than a third of people aged between 60 and 70 in their old age, more than 80% of people over 80 [22- 27]. It causes cognitive, social, and physical disorders. Presbycusis is an umbrella term for deafness associated with aging [22-27]. The origin of presbycusis is multifactorial, and results from the physiological aging of the cochlea; In addition, there are additional effects of extrinsic (exposure to noise, ototoxic drugs) and intrinsic (systemic diseases and genetic disposition) factors [24,28]. The etiology of presbycusis is complex, and several factors are involved [4]. It is the result of an age-related physiological degeneration of cochlear structures combined with the cumulative effects of individual environmental, medical, and genetic factors [4].
Types of presbycusis
Physiologically presbycusis has several types, including the following:
a) sensory presbycusis with impairment in high frequencies secondary to the loss of external cells at the base of the cochlea.
b) metabolic or strial presbycusis with plateau deafness at all frequencies and which is the consequence of degenerative damage to the vascular structure (atrophy) with alteration of the endocochlear potential.
c) neural presbycusis, secondary to the loss of spiral ganglion cells and which partly explains the disorders of verbal discrimination.
Additive exogenous factors
Regular exposure to sounds over 85dB which increases the risk of hearing loss [4, 29-31].
a. Ototoxic agents, in particular antibiotics, aminoglycosides and cisplatin which damage these same cells.
b. Industrial chemical agents (toluene, trichlorethylene) and tobacco.
c. Metabolic disorder and immune diseases by progressive destruction of vasa nervorum.
Nevertheless, the impact of presbycusis would be attributable to genetic factors which would be more important for strial presbycusis (35 to 55%), mitochondrial genes associated with antioxidant agents protecting against oxidative stress to reduce the severity of deafness age-related [4]. In addition, there are other genetic variations in the control of certain enzymes that can lead to deafness [3,32]. Lin et al have shown that the black phenotype is better protected against age-related deafness through protection of the vascular streak by melanin [33-36].
Oxidative stress
Oxygen paradox
The European cooperation in science and technology (COST) adopts a multidisciplinary approach around free radicals and oxidative stress. Free radicals are very reactive molecules which have one or more unpaired electron (s): OH. Oxygen (O2) is a biradical that has two unpaired electrons [37]. Free radicals, highly reactive molecules, produce harmful effects both at the cellular and extracellular level [38].
Sources of free radicals
There are several cellular sources of free radicals [39]. These are endogenous sources:
a. exogenous sources,
b. enzymatic life,
c. non-enzymatic life,
The main sources of free radical formation can be either endogenous or exogenous; they can be physical, chemical, and biological in nature. According to enzymes and cofactors: Xanthine oxidase and hemoglobin in the endoplasmic reticulum, cytochrome P450 and b5 in the nucleus (DNA) and endoplasmic reticulum.
Endogenous sources are provided by:
a. mitochondria (Biologies),
b. phagocyte (Biology),
c. Xanthine oxidase,
d. Transition metals,
e. Paroxysms (Biology),
f. Inflammation (Biology) (Figure 1) Exogenous sources are provided by:
a. the cigarettes,
b. ionizing radicals (physical),
c. various pollution (chemical),
d. UV radiation (physical),
e. chemicals and drugs (chemical),
f. ozone (chemical).
Two production pathways for reactive oxygen species or free radicals are possible:
A. enzymatic route: activation of enzymes responsible for the oxidation of O2. These are the following enzymes:
a) NADPH - oxidase or NOX enzymes,
b) Xanthine oxidase (XO),
c) Cytochrome P450 (CytP450),
d) Myelo peroxidase.
B. non-enzymatic route:
a) heavy metals (Iron ++). Reaction of FENTON.
At the level of endogenous sources: According to enzymes and cofactors, xanthine oxidase and hemoglobin in the endoplasmic reticulum, cytochrome P450 and b5 in the DNA nucleus and endoplasmic reticulum. Oxygen brerst / NASPH oxidase, Myeloperoxidase at the level of lysosomes, the respiratory chain at the level of mitochondria, Oxidases, Flavoproteins at the level of peroxisomes. Lipo-oxygen and peroxidase in lipid membranes and lipid peroxidation and transition metals (iron) in mitochondria [41-43]. The information has been presented rigorously according to the following sections: Free radicals relate to very reactive molecules which present one or more unpaired electrons, on the other hand the reactive species (RE) can be radicular or not, they are highly reactive molecules. , corresponding respectively to O, N and Cl.
Primary reactive oxygen species (ROS):
a. Free radicals (ion): Superoxide ion (O2-), Radial hydroxyl (OH−), Nitric oxide (NO−) and Hypochlorite (OCl−)
b. Molecules: Peroxide (H2O2) Secondary or organic reactive oxygen species: lipids, carbohydrates, proteins or nucleic acids and peroxide radical (ROO−) (Table 1)
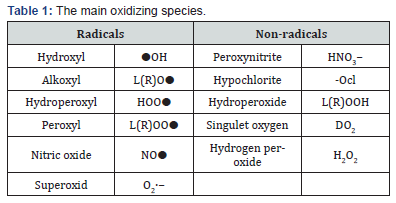
Role of free radicals
The attack or oxidation of the constituents of our body due to an excess of particularly harmful molecules called free radicals which come from the inspired oxygen. Thus lipids, proteins, sugars, and DNA are denatured by the oxidation of cells; this is a form of cell aggression. In addition, these various attacks on cells are one of the causes of cellular aging in humans. Due to their unstable or oxidative nature, active oxygen species (EOR) interact with a whole series of biological substances existing in the environment. The cellular damage produced by EOA is applied to the DNA level of protein and lipid mutations. We observe mutations in DNA, proteins and lipids or observe protein inactivation, lipid peroxidation in polyunsaturated fatty acids of the cell membrane or lipoproteins [24]. The very delicate balance between the production of energy and that of the highly oxidizing molecules generated by the mitochondria [44] ensures the normal functioning of the cell despite the formation of EOA [44].
Physio pathological mechanisms of oxidative stress Under physiological conditions, there is a balance that is established between the pro-oxidant and antioxidant system not harmful to the cellular components (lupid, carbohydrate, proteins and DNA). Under pathological conditions, an imbalance of the balance is created in favor of the pro-oxidant system. Hence oxidative stress. Which leads to cellular toxicity. Thus, oxidative stress can be defined as an attack or oxidation of the constituents of the body due to an excess of particularly harmful molecules called free radicals which come from the oxygen in the air that we breathe to live. This oxygen denatures proteins, fats, carbohydrates and even DNA, and thereby cell membranes and cells. Cells and their finest constituents “Rust” much like a piece of metal left in the open. This cellular aggression or oxidation of cells is one of the essential causes of cellular aging. Oxidation of cells remains a major cause of aging in our cells. Due to their unstable or oxidizing nature, active oxygen species will interact with a whole series of biological substances present in the environment where they are produced [24]. There are 3 physiopathological mechanisms of oxidative stress:
a) Overproduction of free radicals exceeding the antioxidant system
b) Normal production of free radicals and decreased capacity of the antioxidant system
c) A combination or association of two previous mechanisms. (cos of HIV / AIDS infections).
Biological consequences of oxidative stress These consequences are at the cellular level as well as the organs and systems.
At the cellular level
At this stage, we observe an oxidation of the essential components of the cells leading to mitochondrial lesions, an energy deficit stupefying to cell death.
At the level of organs or systems
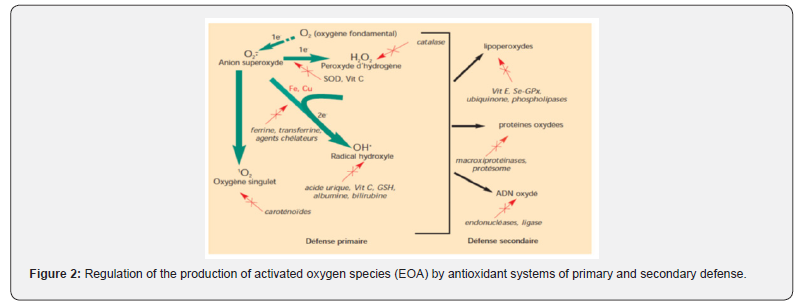
Mono-organic or multi-organic dysfunction is observed with structural damage (cells and tissues) leading to overcoming defense, adaptation, and repair mechanisms. Hence the dysfunction of organs and systems. The cell continues to evolve, thanks to the very delicate balance between the production of energy and that of the highly oxidizing molecules generated by the mitochondria [14]. This balance is ensured by several antioxidant defense systems that regulate the production of EOA [44]. These means of protection consist of enzymes.
Cellular protection against free radicals
This cellular protection corresponds to the catabolism or neutralization of reactive oxygen species or free radicals [45]. There are primary and organic antioxidants grouped into 2 types, 2 types of antioxidants: enzymatic antioxidants and nonenzymatic antioxidants. Regarding enzymatic antioxidants we have:
A. superoxide dismutase (SOD),
B. catalase (CAT)
On the other hand, non-enzymatic antioxidants come from the oxygenated blood, i.e. in the diet including vitamin C, vitamin C, β carotene, micronutrients (Se, Zn, Cu, Mn / antioxidant enzyme cofactors). Others come from endogenous sources including glutathione, albumin, uric acid, bilirubin and sometimes from plant sources including flavonoids, polyphenols.
A secondary defense system composed of proteolytic enzymes aims to prevent the accumulation in the cell of oxidized proteins, lipids, and DNA and to degrade their toxic fragments.
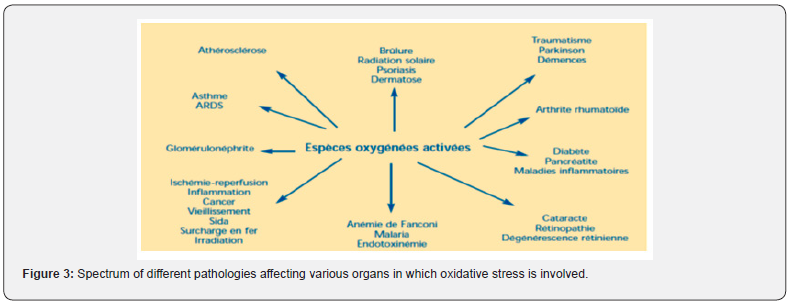
This constitutes the means of protection against EOA (Figure 2) [24,46,47]. The notion of regulation and not of total inhibition is important because the advent of molecular biology and genomics has made it possible to show that EOA, like antioxidants, have a key role in the regulation of apoptosis (programmed suicide of cells progressing to a cancerous state), in the expression of certain transcription factors or as modulators of the expression of structural genes, including antioxidant enzymes [24,46,47]. In the body, but under the action of environmental elements, several biochemical mechanisms can be activated (Table 1) [24] by producing excessively EOA which will therefore very quickly overwhelm all our antioxidant defenses (Table 2).

There is therefore the appearance of oxidative stress which is defined as a profound imbalance in the balance between prooxidants (EAO) and antioxidants in favor of the former, which leads to cell damage which is often irreversible (Figure 3) [24]. Oxidative stress is involved in the development of more than 200 pathologies (cardiovascular diseases, degenerations and inflammatories, cancer, diabetes, AIDS) [24,48,49]. This oxidative stress is a state of imbalance between the production of reactive species and the body’s defenses [49]. A state of oxidative stress exists when at least one of the following three conditions is present (Figure 1) [24,40]:
a) the excess of reactive species of O2, N2 or cl2,
b) insufficient defenses (endogenous, exogenous),
c) insufficient repair mechanisms.
Oxidative stress and sensorineural deafness related to age

Age-related sensorineural deafness considered to be presbycusis remains a phenomenon in which oxidative stress is involved. An expert group has carried out several works in the redox regulation of neurological and psychiatric diseases with particular emphasis on two relatively unexplored areas: the inner ear and schizophrenia. Of all the NOX isoforms, NOX3 is the most elusive, least studied, and even less well understood, primarily due to its restricted topological expression in the inner ear. One of the laboratories involved in this COST action shed light on the function of NOX3 in the regulation of balance and hearing function by developing sophisticated mouse models that lend themselves to investigation of this problem. The situation of hearing loss, of genetic origin or acquired after environmental exposure to noise or aging, has recently been given an important place in Redox regulation.
Indeed, a very compelling report has described that pejvakin, a protein linked to peroxisomal function and proliferation, is the basis of a form of hereditary sensorineural deafness and that its absence or dysfunction induced by oxidative stress makes inner ear extremely vulnerable to noise, damage [50]. NOX3 is expressed in the cochlea even though mice lacking it do not have an apparent phenotype related to hearing loss. The peroxisoma generation of ROS is related to the NOX3 function is an issue that remains to be investigated. Thus, much evidence has been established on the role of oxidative stress in neurodegenerative diseases [51]. In addition, the role played by oxidative stress in metabolic syndrome and in type 2 diabetes mellitus [52]. The levels of O2 • - and • NO including their interaction significantly determine the alteration of physiological signaling by ROS / RNS is implicated in the etiopathology of diabetes [53]. The O2 • - / • NO ratio is regulated and affects the synthesis and degradation of insulin. In addition, oxidative stress contributes to the loss of pancreatic β cells, which impairs insulin secretion [54] (Figure 4).
This section covers a wide range of disorders on the topic of redox signaling and nitroxidative stress as a common modulator of disturbances related to tumor cell migration, fibrogenesis, hearing loss, neuropsychiatric disorders, metabolic syndrome. , drug metabolism, renal hemodynamics and pulmonary hypoxia. In the slide, the participation of the relevant enzymatic pathways (NOX, SOD, ALDH-2) is indicated. BMI: body mass index. The other abbreviations are defined in the text. Therefore, therapeutic strategies to prevent β cell dysfunction require better knowledge of how to protect them against the direct or indirect effects of free radicals and lipid peroxidation.
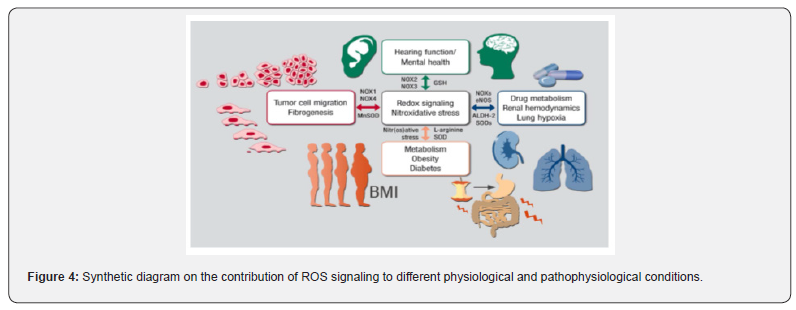
Insulin sensitivity and metabolic homeostasis depend on the ability of adipose tissue to absorb and use excess glucose and fatty acids. This buffering capacity depends on the physiological levels of • NO, the function of which can be impaired by excessive O2 formation [55]. It should be noted that redox signaling and oxidative stress can contribute to the remodeling of adipose tissue. The COST action has also addressed the toxicity and tolerance issues arising from the clinical use of organic nitrites. Recently, insight has been gained on the molecular mechanisms linked to the side effects of nitroglycerin and other nitrates in mitochondrial dysfunction and disruption of redox homeostasis. Disturbed vascular redox balance underlies the pathophysiology of important disease conditions in other organs such as the lungs and kidneys. In the lungs, chronic hypoxic pulmonary hypertension causes vasoconstriction and vascular remodeling, two phenomena that perpetuate the vicious cycle. In the kidney, animal studies using superoxide dismutase (SOD) mimetics indicate an important role of redox dysfunction in the increased vascular resistance associated with chronic kidney disease and open the possibility of therapeutic intervention.
Role of NOX3 in Pathologies of the Inner Ear
Recent research has demonstrated the major role of ROS in hearing impairment, including overexposure to noise, ototoxic drugs (eg cisplatin) and age-related hearing loss [56]. Although the sources of ROS are complex and partially understood as at the level of mitochondria. Activation of NOX enzymes, especially NOX3, plays a key role in hearing loss. An elevated level and specific expression in the inner ear identify NOX3 as a prime drug target for combating hearing loss [57]. NOX3 is a multi-subunit NADPH oxidase, functionally and structurally closely related to NOX1 and NOX2. NOX3 is active as a multiprotein complex comprising membrane bound NOX3 and p22phox and cytosolic subunits NOXO1 and NOXA1. NOX3 is crucial for the formation of otoconia, small crystals of protein carbonate involved in equilibrium [58].
The experiment carried out in mice carrying a loss-of-function mutation in the NOX3- complex including the common NOX1 to NOX4 subunit p22phox, or the cytosolic NOXO1- subunit have a vestibular phenotype similar to mutant NOX3 mice (called tilted head mouse). In addition, the role of NOXA1 is still poorly defined, especially in vitro. On the other hand, the physiological relevance of the expression of NOX3 in the cochlea remains uncertain because no auditory phenotype has been described for the loss of NOX3 in mutant animals. The role of NOX3 in cisplatin-induced hearing loss is in fact the best documented. In vitro the potential role of NOX3 has been demonstrated in cochlear damage on NADPH oxidase activity [59]. And therefore, an increase in the level of NOX3 mRNA has been described in the cochlea of rats treated with cisplatin, while siRNAs against NOX3 could prevent hearing loss by cisplatin [60].
Currently, NOX3 is a major source of ROS in the cochlea. However, the role of NOX3 may not be limited to ototoxicity induced by cisplatin. Likewise, oxidative stress is also a major element in other hearing pathologies such as presbycusis, Meniere’s disease or noise-induced hearing loss. The role of NOX3 in hearing loss based on the loss of functional mutant mice will be shown to be experimental in the field. Three mouse models of hearing loss have been successfully developed in one of the laboratories of this COST- action namely overexposure to noise, cisplatin and agerelated hearing loss models and an investigation of the role of NOX3 is currently ongoing through NOX3 and p22phox mutant mice. It is therefore difficult to predict whether classical pharmacological approaches, i.e., small molecule NOX3 inhibitors, therapies based on molecular biology, i.e. siRNA-mediated knock-down will be the ultimate tools for targeted NOX3 therapies, specific approaches allowing the inhibition of pathological NOX3 activity.
Clinical and Therapeutic Implications and Public Health Perspectives around Presbycusis
Information relating to the role of NOX3 in the pathologies of the inner ear demonstrated over the last decades, associating ENT specialists, professionals (speech therapists), physiologists and practicing physicians to understand overexposure to noise, the effects of ototoxic drugs well demonstrated in the genesis of age-related hearing loss [61]. Indeed, NOX3 will become a drug target of choice to fight deafness [62].
Conclusion
This review article highlighted the complexity of the effects of senescence, senility, genetic factors during fetal life, ecotoxic effects and mutations involved in presbycusis. Further studies are needed to develop prevention programs and drug molecules to delay the pejorative effects of oxidative stress associated with presbycusis.
Acknowledgment
We thank all who participated in the study
References
- Javier Egeaa, Isabel Fabregatb, Yves M Frapartc,PietroGhezzid, AgnesGörlach, et al. (2017) European contribution to the study of ROS: A summary of the findings and prospects for the future from the COST action BM1203 (EU-ROS). Redox Biology 13: 94-162.
- Jing Wang, Jean Luc Puel (2020) Presbycusis: An Update on Cochlear Mechanisms and Therapies. J Clin Med 9(1): 218.
- Anne Claude Guinchard, Philippe Estoppey, Raphaël Maire (2017) Presbyacousie: baisse de l’ouïe chez la personneâgé Forum Med Suisse 17(10): 230-235.
- Huang Q, Tang J (2010) Age-related hearing loss or presbycusis. Eur Arch Otorhinolaryngol 267(8): 1179-1191.
- Samina Salim (2017) Oxidative Stress and the Central Nervous System. J Pharmacol Exp Ther 360(1): 201-205.
- Yamasoba T, Lin F R, Someya S, AkinoriKashio, Takashi Sakamoto, et al. (2013) Current concepts in age-related hearing loss: Epidemiology and mechanistic pathways. Hear Res 303: 30-38.
- Moon I J, Byun H, Woo S, GeumYounGwak, Sung Hwa Hong, et al. (2015) Factors Associated with Age-related Hearing Impairment: A Retrospective Cohort Study. Medicine (Baltimore) 94(43): e1846.
- Organisationmondiale de la santé(OMS) (2020)Surdité et déficience auditive. Genève, mars.
- Anthony Bared, Xiaomei Ouyang, Simon Angeli, Li Lin Du, Kimberly Hoang, et al. (2010) Antioxidant enzymes, presbycusis, and ethnic variability. Otolaryngol Head Neck Surg 143(2): 263-268.
- Tavanai E, Mohammadkhani G (2017) Role of antioxidants in prevention of age-related hearing loss: a review of literature. Eur Arch Otorhinolaryngol 274(4): 1821-1834.
- Klinger Vagner Teixeira da Costa, Kelly Cristina Lira de Andrade, Maria Eduarda di Cavalcanti, Ana Claudia FigueiredoFrizzo, Aline TenórioLinsCarnaúba, et al. (2018) Hearing Loss at High Frequencies and Oxidative Stress: A New Paradigm for Different Etiologies, An Excursus into Hearing Loss, Stavros Hatzopoulos and Andrea Ciorba, IntechOpen.
- Presbycusis in seniors: definition, symptoms and treatments.
- Cruickshanks KJ, Tweed TS, Wiley TL, Barbara E K Klein, Ronald Klein, et al. (2003) The 5-year incidence and progression of hearing loss: the epidemiology of hearing loss study. Arch Otolaryngol Head Neck Surg 129(10): 1041-1046.
- Mackenzie I, Smith A (2009) Deafness-the neglected and hidden disability. Ann Trop Med Parasitol 103(7): 565-571.
- American Academy of Pediatrics, Joint Committee on Infant Hearing. Year 2007 position statement: Principles and guidelines for early hearing detection and intervention programs. Pediatrics120(4):898-921.
- Joint Committee on Infant Hearing 1994 Position Statement. American Academy of Pediatrics Joint Committee on Infant Hearing. Pediatrics95(1):152-156.
- Petersen MB, Willems PJ (2006) Non-syndromic, autosomal-recessive deafness. Clin Genet 69(5): 371-392.
- Nickel R, Forge A (2008) Gap junctions and connexins in the inner ear: their roles in homeostasis and deafness. CurrOpinOtolaryngol Head Neck Surg16(5): 452-457.
- Kikuchi T, Kimura RS, Paul DL,T Takasaka, J C Adams, et al. (2000) Gap junction systems in the mammalian cochlea. Brain Res Brain Res Rev 32(1):163-166.
- Adrian C Davis, Howard J Hoffman (2019) Hearing loss: rising prevalence and impact. Bullet WHO97:646-646A.
- Van Eyken E, Van Camp G, Van Laer L (2007) The complexity of age-related hearing impairment: contributing environmental and genetic factors. AudiolNeurootol12(6):345-358.
- Liu XZ, Yan D (2007) Ageing and hearing loss. J Pathol 211(2):188-197.
- J Pincemail, C Heusele, F Bonté,R Limet, JO Defraigne(2001) Stress oxydant, antioxydantsnutritionnels et vieillissement. Act Med Int4:18-23.
- Yueh B, Shapiro N, MacLean CH, Shekelle PG (2003) Screening and management of adult hearing loss in primary care: scientific review. JAMA 289(15):1976-8519.
- Mazurek B1, Stöver T, Haupt H, J Gross, A Szczepek(2008) Pathogenesis and treatment of presbyacusis. Current status and future perspectives. HNO56(4):429-32.
- AnneLise ML Poirrier (2010)Mécanismes et Thérapies des Surdité Université de Liège.
- Maire R (2009) La presbyacousie: Les facteursexogènes et géné Geriatrie Pratique/Praxis (2): N°1.
- Gates GA, Mills JH (2005) Presbycusis. Lancet 366(9491):1111-1120.
- Sprinzl GM1, Riechelmann H (2010) Current trends in treating hearing loss in elderly people: a review of the technology and treatment options-a mini-review. Gerontology56(3):351-358.
- Yang CH, Schrepfer T, Schacht J (2015) Age-related hearing impairment and the triad of acquired hearing loss. Front. Front Cell Neurosci 9: 276.
- Lin FR, Thorpe R, GordonSalant S, Ferrucci L (2011) Hearing loss prevalence and risk factors among older adults in the United States. J GerontolA Biol Sci Med Sci66(5):582-590.
- Hong Miao Ren, Jihao Ren, Wei Liu (2013) Recognition and Control of the Progression of Age-Related Hearing Loss. Rejuvenation Res16(6): 475-486.
- Cruickshanks KJ, Wiley TL, Tweed TS, et al. (1998) Prevalence of hearing loss in older adults in Beaver Dam, Wisconsin. The Epidemiology of Hearing Loss Study. Am J Epidemiol148(9):879-886.
- Helzner EP, Cauley JA, Pratt SR, Joseph M Zmuda, Evelyn O Talbott, et al. (2005) Race and sex differences in age-related hearing loss: the Health, Aging and Body Composition Study. J Am Geriatr Soc53(12):2119-2127.
- Borden W T, Hoffmann R, Stuyver T, Chen B (2017) Dioxygen: What Makes This Triplet Diradical Kinetically Persistent? J Am Chem Soc139(26): 9010–9018.
- Phaniendra A, Jestadi D B, Periyasamy L (2014) Free Radicals: Properties, Sources, Targets, and Their Implication in Various Diseases. Indian J Clin Biochem 30(1): 11-26.
- Machlin L J,Bendich A (1987) Free radical tissue damage: protective role of antioxidant nutrients. FASEB 1(6): 441-445.
- A R L Lausanne (2010) Le Stress Oxydatif.Unilabs Mars.
- Wallace DC (1992) Diseases of the mitochondrial DNA (review). Annu Rev Biochem 61:1175-1212.
- Giugliano D, Ceriello A, Paolisso G (1996) Oxidative stress and diabetic vascular complications. Diabetes Care 19: 257-267.
- Sawa A, Khan AA, Hester LD, Snyder SH (1997) Glyceraldehyde-3-phosphate dehydrogenase: nuclear translocation participates in neuronal and non-neuronal cell death.Proc Natl Acad Sci USA94: 11669-11674.
- Knight JA (2001) The biochemistry of aging. Advances in Clinical Chemistry35:1-62.
- Elodie Gauche, Christophe Hausswirth (2006) Stress oxydant, complémentationnutritionnelleenantioxydants et exercice. Moveme Sport Scien2(58):43-66.
- Domenico Fusco, Giuseppe Colloca, Maria Rita Lo Monaco, Matteo Cesari (2007) Effects of antioxidant supplementation on the aging process. Clin Interv Aging 2(3): 377–387.
- B Poljsak, I Milisav (2013) Aging, Oxidative Stress and Antioxidants, Oxidative Stress and Chronic Degenerative Diseases - A Role for Antioxidants, José A MoralesGonzález, IntechOpen.
- Ilaria Liguori, Gennaro Russo, Francesco Curcio, Giulia Bulli, Luisa Aran, et al. (2018) Oxidative stress, aging, and diseases. Clin Interv Aging 13:757-772.
- Li Zio, Evan R Prater, MykolaStevskiv et al. (2019) Inflammaging and Oxidative Stress in Human Diseases: From Molecular Mechanisms to Novel Treatments. Int J Mol Sci20(18):4472.
- Delmaghani S, Defourny J, Aghaie A, Beurg et al. (2015) Hypervulnerability to Sound Exposure through Impaired Adaptive Proliferation of Peroxisomes Cell163(4):894-906.
- JeanClaude Desport, Philippe Couratier (2002) Stress oxydant et maladies neurodégéné NutritCliniqMétabo16(4):253-259.
- Harani H, Koceïr E A, Zenati A,Ouadahi N (2014) P115: Stress oxydant et Diabète de type 2 : intérêt du manganèse et du chrome dans le contrôleglycémique chez le patient diabétique. NutritCliniqMétabo28(1):S128.
- L Rochette, M Zeller, Y Cottin, C Vergely (2014) Diabetes, oxidative stress and therapeutic strategies. BiochimBiophys Acta1840(9):2709-2729.
- J Wang, X Yang, J Zhang (2016) Bridges between mitochondrial oxidative stress, ER stress and mTOR signaling in pancreatic β cells. Cell Signal28(8):1099-1104.
- Jankovic A, Korac A, Buzadzic B, Stancic A, et al. (2016) Targeting the NO/superoxide ratio in adipose tissue: relevance to obesity and diabetes management. Br J Pharmacol174(12):1570-1590.
- Kopke R, Allen K A, Henderson D, Hoffer M,Frenz D, et al. (1999) A Radical Demise: Toxins and Trauma Share Common Pathways in Hair Cell Death. Anna New York AcadScie884(1): 171-191.
- Francis Rousset, Stephanie Carnesecchi, Pascal Senn, KarlHeinz Krause (2015) NOX3-Targeted Therapies for Inner Ear Pathologies. Curr Pharm Des21(41):5977-5987.
- Rainer Paffenholz, Rebecca ABergstrom, Francesca Pasutto,Philipp Wabnitz, Robert J Munroe, et al. (2004) Vestibular efects in head-tilt mice result from mutations in Nox3, encoding an NADPH oxidase. Genes Dev18(5):486-491.
- Banfi B, Malgrange B, Knisz J, Steger K,Karl Heinz Krause, et al. (2004) NOX3, a superoxide-generating NADPH oxidase of the inner ear. J Biol Chem. 279(44):46065-46072.
- Mukherjea D, Jajoo S, Kaur T, Sheehan K E, Ramkumar V, et al. (2010) Transtympanic Administration of Short Interfering (si)RNA for the NOX3 Isoform of NADPH Oxidase Protects Against Cisplatin-Induced Hearing Loss in the Rat. Antioxidants & Redox Signaling13(5):589-598.
- Florine Fournet, MélanieGuindéoRoccia (2014) La perception auditive dans le bruit chez le jeune enfant normo-entendant et implantécochlé Université Paris VI Pierre et Marie Curie.
- AnneLise ML Poirrier (2010)Mécanismes et Thérapies des SurditésNeurosensorielles. Université de Liège, Septembr





























