- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Genetic Hearing Loss
Lalsa Shilpa Perepa
How to cite this article: Lalsa Shilpa P. Genetic Hearing Loss. Glob J Oto 2017; 5(3): 555662. DOI: 10.19080/GJO.2017.05.555662
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Introduction
- One of the major cause of hearing loss is gene mutations.
- Inherited from parents to the offspring
- Transferred to future generations
- Can be syndromic or non syndromic
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Need for knowing
a) New era in audiology
b) Knowledge about etiology of HL
To meet the requirements
- Deeper knowledge of pathophysiological changes
- Need for developing new tools.
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Genetic testing
- 20,000 - 25,000 genes present in humans.
- Biochemical tests - indentifies the gene for genetic disorder.
- Identifies changes from chromosomes, genes or proteins.
- Testing is used to find changes that are associated with inherited disorders.
- Confirm or rule out a suspected genetic condition or help determine a person's chance developing or passing on a genetic disorder.
- Several hundred genetic tests are currently in use, and more are being developed.
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Types of Genetic Testing
- Newborn screening
- Diagnostic testing
- Carrier testing
- Prenatal testing
- Predictive and presymptamatic testing
- Forensic testing
- Research testing
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Procedure
- Genetic tests are performed on a sample of blood, hair, skin, amniotic fluid (the fluid that surrounds a fetus during pregnancy), or other tissue.
- For example, a medical procedure called a buckle smear uses a small brush or cotton swab to collect a sample of cells from the inside surface of the cheek.
- The sample is sent to a laboratory where technicians look for specific changes in chromosomes, DNA, or proteins, depending on the suspected disorder.
- The laboratory reports the test results in writing to a person's doctor or genetic counselor.
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Results
- Not straight forward.
- Should consider family, medical histories and type of genetic test.
- Positive - chromosome or gene with abnormality identified.
- Do not say the course of severity.
- Test results negative - no copy of dangerous gene
- Does not rule out other gene causing same phenotype.
- Ambiguous and inconclusive
- Other test required to confirm
- Polymorphism - natural gene variation occurring in normal people.
- Difficulty in distinguishing from disordered gene from polymorphism
- Further testing for the family.
Cost and time
- Cost ranges from 3000 Rs to 50000 Rs.
- Increases if more testing done
- Can take several days to months for results
- Prenatal testing results - available quickly
Risks and limitations
- Physical risk - very small
- Miscarriage - prenatal testing
- Emotional problems with test results.
- Can say the possibilities in other members in the family.
- Genetic information nondiscrimination act - amended on may 21,2008 in US
Why to perform genetic testing in hearing impairment?
- Accurate genetic diagnosis based on clinical features alone is not possible.
- DNA analysis makes it possible.
- Syndromal conditions with typical clinical features not seen - genetic testing can identify.
- In future the critical decision to go for genetic testing will be taken by the audiologist.
- So it is necessary to know the available molecular testing that are available and also the various conditions that occur due to genes.
- Similar to new born hearing screening genetic testing can also become a routine clinical evaluation and audiologist should know the possible outcomes.
- Genetic testing is minimally invasive.
- . Since genetic hearing loss is more widely accepted and testing is available, genetic - phenotype correlations can be made more reliable.
- The physical effect of individual mutation or combination of mutation hearing loss can be predicted with greater confidence.
- An evaluation of the pedigree to assess which other family members may potentially benefit from the same test.
- In some cases, the DNA diagnosis may affect the type and timing of treatment, with potentially major benefits to the patient.
- Knowing the precise cause of the hearing loss will help to determine the most effective management and therapeutic options.
- . For example usher syndrome - management differs. Sign language may not be best option.
The benefits of genetic testing include the following:
- The providing an accurate diagnosis of the etiology of the hearing impairment.
- Avoiding the need for more expensive and invasive testing
- Providing the basis for prognostic information about future hearing
- Giving direction and improving genetic counseling
- Defining new guidelines for treatment (Figure 1).
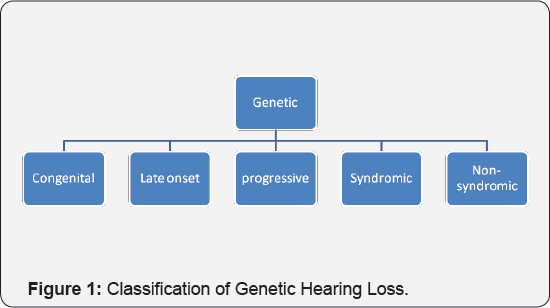
Congenital Hearing Loss
- Hearing loss present by birth.
- Genetically, it can be acquired due to various factors.
Causes
a) Consanguinity
b) Paternal / maternal deafness
c) Ear anomalies (may occur with or without syndromes)
d) Single-Gene Disorders
Consanguinity
- Common feature across many parts of the globe
- Marriage within relations
- Legal system in many cultures
- Known to be a significant cause of genetic hearing loss( Turan & Apaydin,2002)
- Causes Congenital SNHL
- Disruption in planar cell polarity pathway (PCP). PCP- helps formation of polarized structure
- Regulates embryonic development. Genetic disturbances causes disruption in PCP
- Genetic mutation of GJB2
- Siblings have higher incidence of autosomal recessive hearing impairment.
- Increased risk of polygenic inheritance (Northern et al.)
- Also, more common in near relations 1st cousins- greater risk. Distant cousins- lesser risk.
Paternal or maternal deafness
- Either of the parent has hearing loss
- SNHL common occurrence
- Deletion of genetic material on the long arm of chromosome 15
- Loss of multiple genes in that region
- SRTC on Chromosome 15 - cause of HL
Single gene disorders
- Single mutated gene- cause
- Passes to generations in several ways
- Genetic protein -non functional
- Defect on autosome of X chromosome - affects one member of the gene pair or both carrying the same gene pair
- Pattern of inheritance depends on the phenotype.
Ear anomalies
Late onset genetic hearing loss
- Occurs later in life
- 9 different types of genes localized
- DFNA10 on chromosome 6 - most common
- Connexin 6 also has impact
- Mostly Age related hearing impairment
- Can be triggered by environmental factors
- No exact gene identified till date.
Late onset Progressive genetic hearing loss
a) Autosomal dominant
b) Occurs at any age
c) It may be due to some defective genes that might cause deafness, but expressed later in life.
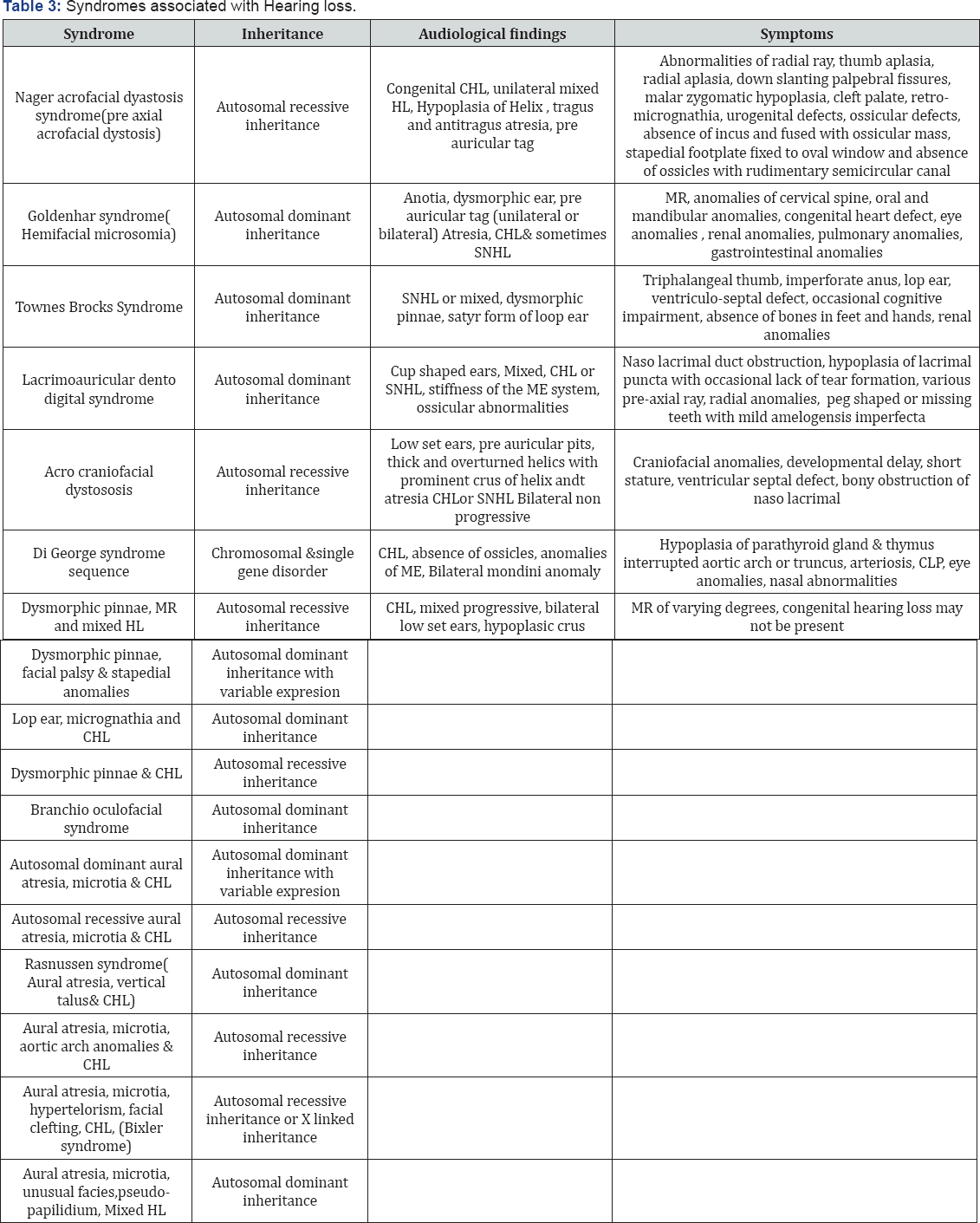
Syndromic hearing loss
A particular set of congenital signs repeatedly occurring in a generally consistent pattern is known as "syndrome".
Associated with malformation of the ear and other organs with medical problems involving other organs.
I. Around 400 genetic syndromes that cause hearing loss identified
II. One of the contributing factors for pre lingual deafness A syndrome may be caused by
- a chromosomal problem,
- a biochemical defect,
- a Mendelian genetic defect,
- or an environmental agent.
Pattern of Inheritance
- Autosomal dominant inheritance
- Autosomal recessive inheritance
- X-linked inheritance
- Mitochondrial inheritance
Autosomal dominant inheritance
- The pattern of inheritance is Autosomal dominant if individuals in each generation are affected;
- Both males and females are equally likely to be affected (Figure 2).

iii. Affected individuals are usually heterozygote's with one normal and one deleterious copy of the gene for the disorder, and each offspring of an affected individual has a 50% chance of inheriting the deleterious allele.
iv. There may be variable expression among affected individuals, and some who must have the deleterious allele may show no phenotypic signs.
I. Autosomal recessive inheritance
A. The mode of inheritance of a disorder is Autosomal recessive if the abnormal phenotype is expressed only in individuals who have two copies of the deleterious allele.
B. Heterozygotes are not affected and are often called carriers.
C. The probability that a child of carrier parents will be affected by one quarter;
D. The probability that an unaffected sibling is a carrier is two third.
E. If a hearing impaired parents has an affected sibling or the parents are related, the etiology is most likely to be deleterious gene with the mode of inheritance being Autosomal recessive.
F. Recessive inheritance probably explains most case of profound sensorineural hearing loss when family history is negative, and there is no known environment factor that could be responsible.
II. X - Linked inheritance
i. If a disorder has an X- linked dominant pattern of inheritance with complete penetrance, then all daughters of affected fathers are affected.
ii. In the case of an X- linked recessive trait, the majority of affected individuals are male because they have only one chromosome (Figure 3).
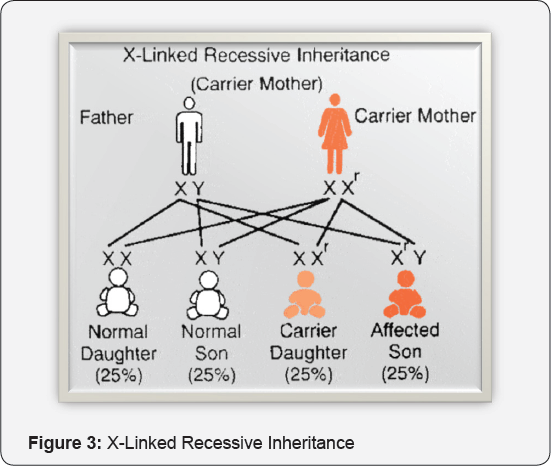
iii. Female have two X chromosomes and may be carriers, but are unlikely to be affected.
iv. Sons of carrier mother - 100% chance
v. Daughters - 50% and remaining will be 50% carriers.
vi. Note that, although females have two X chromosomes in a cell, in general one of the two is randomly activated early in embryonic development.
vii. Thus, females are mosaic, with each cell having one or other X chromosome active.
III. Mitochondrial Conditions
a) Mitochondria are structures in the cell that produce the energy that cells need to survive.
b) Neither the mitochondria nor the cell can exist without the other.
c) Changes in the mitochondrial genes can also result in syndromes involving hearing loss.
d) Mutation in the mitochondrial genome can affect energy production through adenosine triphosphase synthesis and oxidative phosphorylation. Tissues that require high levels of energy are particularly affected.
e) Typically, mitochondrial diseases involve progressive neuromuscular degeneration with ataxia, ophthalmoplegia, and progressive hearing loss [11-20].
IV. Mitochondrial mutation and syndromic hearing loss
a) Systemic neuromuscular syndromes such as Kearns Sayre Syndrome, mitochondrial encephalomyopathy, lactic acidosis and stroke like episodes etc frequently have hearing loss as one of their clinical signs. It is due to heteroplasmic mutation.
b) [Homoplasmic mutation- most healthy individuals appear to have only a single mitochondrial DNA gene type which is homophasic but in many mitochondrial diseases, the mitochondrial DNA population is mixed which is the heteroplasmic condition].
c) SNHL caused by Diabetes Mellitus is inherited by A3243G in the gene for tRNA.
d) Late onset HL develops after onset of diabetes
e) In addition to diabetes mellitus, diabetic insipidis, otic atrophy and hearing loss have been well described as the Wolfram syndrome usually an autosomal recessive condition.
V. A3243G
a) Gene defect on mitochondrial DNA (mt DNA).
b) Associated with deletion in the mitochondrial genome.
c) A & G stand for - Adenine & Guanine.
d) 3243 stands for - position of transposition.
e) The convention adopted for describing transposition defects is to show the correct neucleotide (A), then show position of transposition (3243) and then incorrect nucleotide (G).
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Mitochondrial Mutation and Ototoxicity
- Aminoglycoside ototoxicity is a common cause of acquired hearing loss. (Prezant et al 1993)?
- They analysed 3 chinese families where individuals developed HL after the use of aminoglycosides.
- Found that mitochondrial ribosomes and mitochondrially encoded 12S rRNA gene are the locus of such predisposition to toxicity.
- 1555DELG - The hearing loss is sensorineural and may be progressive.
- Those who have this condition may have sudden hearing loss when exposed to aminoglycoside antibiotics (e.g., neomycin, gentamycin, streptomycin, kanamycin, tobramycin, or amikacin).
- NS SNHL is almost genetically heterogeneous, with the number of involved genes estimated to be over one hundred.
- At present, pedigree analysis and audiometric morphology are still the most popular means of attempting a sub-categorization of the isolated types of genetic hearing loss.
- Genes of non-syndromic disorder are designated as DFN,
a) DFNA: Nonsyndromic deafness, autosomal dominant
b) DFNB: Nonsyndromic deafness, autosomal recessive [21-25].
c) DFNX: Nonsyndromic deafness, X-linked
d) Several recessive and dominant loci have been mapped to the same chromosome regions
Genes related to nonsyndromic deafness
ACTG1, CDH23, CLDN14, COCH, COL11A2, DFNA5, ESPN, EYA4, GJB2, GJB6, KCNQ4, MYO15A, MYO6, MYO7A, OTOF, PCDH15, POU3F4, SLC26A4, STRC, TECTA, TMC1,TMIE, TMPRSS3, USH1C, and WFS1 genes cause non syndromic deafness, with weaker evidence currently implicating genes GJB3, and MYO1A.
- Most of the autosomal recessive loci cause pre-lingual severe to profound hg loss.
- An exception is DFNB8, in which the hearing impairment is post lingual and rapidly progressive.
- Most of the autosomal dominant loci cause post lingual hearing impairment.
- Some exceptions are DFNA3, DFNA8 etc.
- X linked non syndromic hearing loss can be either pre or post lingual.
- Disorder due to DFN3 has mixed hearing loss
- DFNA1 (medical condition): Dominantly inherited hearing loss that occurs without any other symptoms - i.e. is not associated with any other condition.
- Type 1 involves a defect of the Drosophila diaphanous gene on chromosome 5q31.
- Regulation of acting polymerization in haircells of the inner ear.
- The non syndromic hearing loss can again inherited by Autosomal dominant or recessive or X - linked or even mitochondrial inheritance [26-30].
- There are many genes responsible for the complete development of cochlea and other auditory structures.
- Any mutation in these genes can cause malformation of inner ear.
- Arrest in normal development or aberrant development of inner ear structures may result in hearing impairment.
- Computerized temporal bone imaging techniques reveal that about 20% of children with congenital sensorineural hearing loss have subtle or severe abnormalities of the inner ear.
- About 65% of such abnormalities are bilateral; 35% are unilateral.
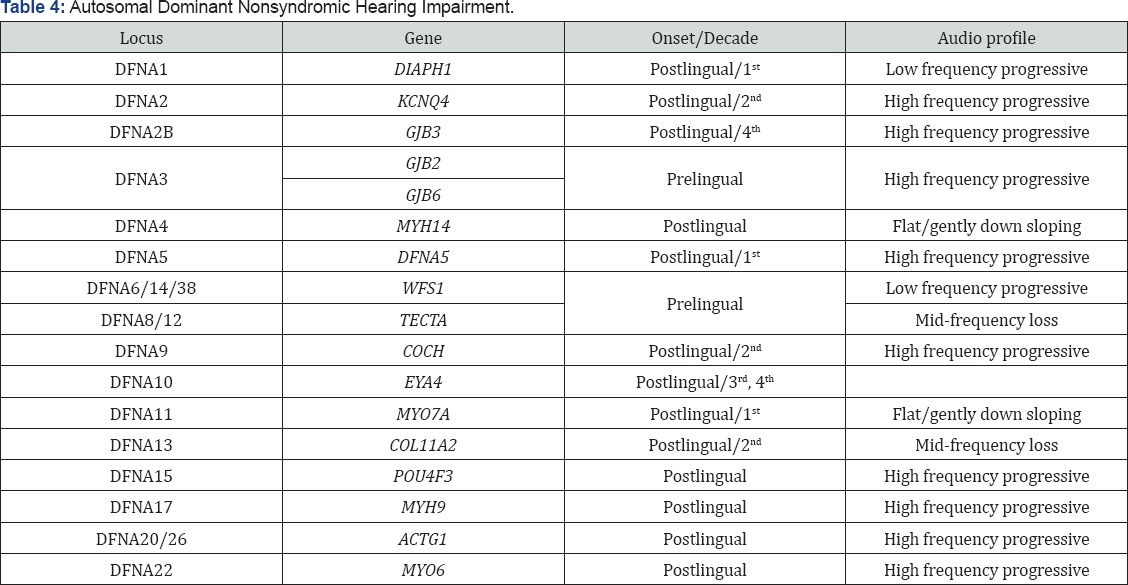
Autosomal dominant non- syndromic HL
a) Dominant progressive hearing loss is a type of non- syndromic
b) Non-congenital sensorineural hearing loss
c) Variable in age of onset and rate of progression.
d) It is inherited in an autosomal dominant pattern.
e) Age of onset can vary from early childhood in some families to early adulthood in others.
f) Presymptomatic gene carriers may demonstrate elevated thresholds for stapedial reflexes and positive signs for recruitment.
g) Eventually the disease progresses to the level of severe to profound hearing loss.
h) More than 12 genes causing dominant progressive hearing loss have been localized.
i) Konigsmark and Gorlin defined four types of dominant progressive hearing loss:
i early onset
ii high frequency
iii midfrequency
iv low frequency
Heterogeneity has been documented for each subtype as exemplified by four types of high frequency dominant progressive hearing loss identified by audiogram configuration within family groups.
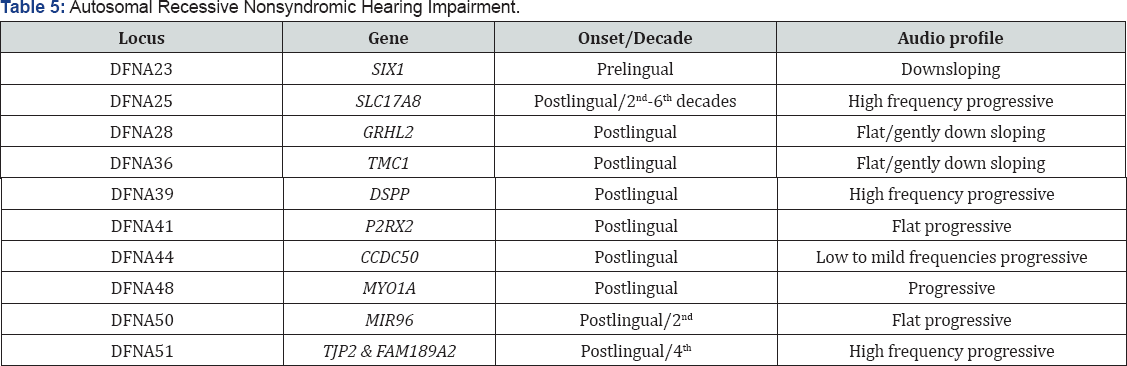
Autosomal recessive non syndromic HL
Konigsmark and Gorlin divided nonsyndromic recessive sensorineural hearing loss into three subtypes.
These are
a. congenital severe-to-profound,
b. congenital moderate,
c. and early onset
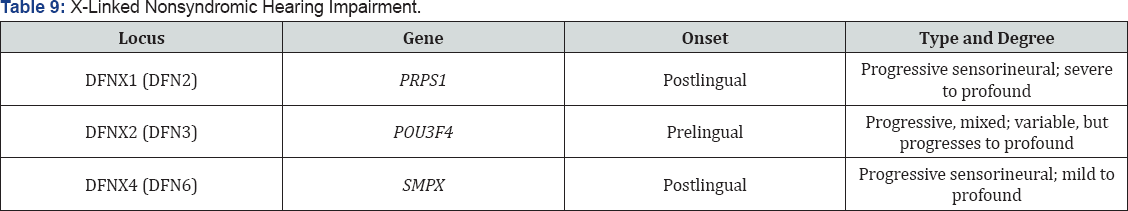
X - linked non - syndromic hearing loss
i. At least 6 loci on the X-chromosome for nonsyndromic hearing loss are known.
ii. Two types of nonsyndromic
iii. They are
iv. early onset rapidly progressive type
v. and a moderate slowly progressive type
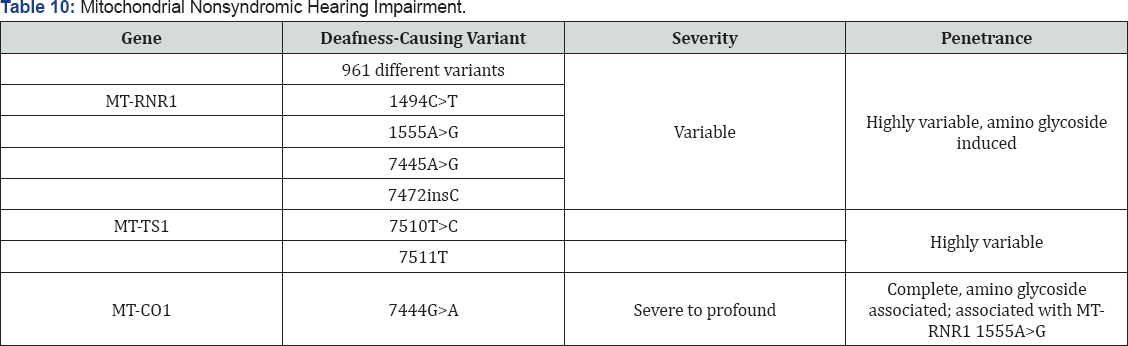
Mitochondrial Inheritance of non syndromic hearing loss
i. Majority of mutations are a cause of maternally inherited multisystem disorder.
ii. Mitochondrial inheritance is by MTRNR1 and MTTS1.
iii. May lead to severe to profound SNHL
iv. MTRNR1 causes mitochondrial inheritance which further induces HL by amino glycoside
I. Mitochondrial Determined Hearing Impairment
A. The mitochondrial DNA molecule encodes 13mRNA and 2 rRNA and 22 tRNA, that are required for assembling a functional mitochondrial protein synthesizing system.
B. The 13mRNAs are translated on mitochondrian specific genetic code into 13 proteins which are required to form the five enzyme complexes required for oxi d ativg
F. The genes for 20 different connexin proteins are present in the human genome.
G. There are two types of connexins, alpha and beta, named GJA or GJ
B followed by a number
H. The connexins of one cell align symmetrically with those of its neighbor to create continuous aqueous pores that functionally couple the adjacent cells.
I. Connexins aggregate in the plane of the plasma membrane to form a gap junction plaque.
J. Connexin genes involved in deafness are GJBJ (Cx32), which is also responsible for X- linked Charcot Marie Tooth disease , GJB3 (Cx31) involved in both deafness and a skin disease.
K. Several connexin genes (GJB1, GJB2, GJB3, GJB6 and GJA1) have been found mutated in patients with non-syndromic and/or syndromic deafness indicating an important role of these proteins.
L. As development proceeds, expression of these two genes was found in various subtypes of fibrocytes, either within the spiral limbus or along the spiral ligament, as well as in the basilar membrane cells, in the Reissner’s membrane cells, and in subsets of the cellular elements of the cochlear ganglion.
M. The genes for 20 different connexin proteins are present in the human genome.
N. There are two types of connexins, alpha and beta, named GJA or GJB followed by a number
O. Gjb3 and Gjb1 expression was spatiotemporally modulated within the sensory hair cells and the various supporting cells that compose the developing organ of Corti.
P. A transitory expression of Gjb1 was found in the basal and intermediate cells of the stria vascularis of auditory system.
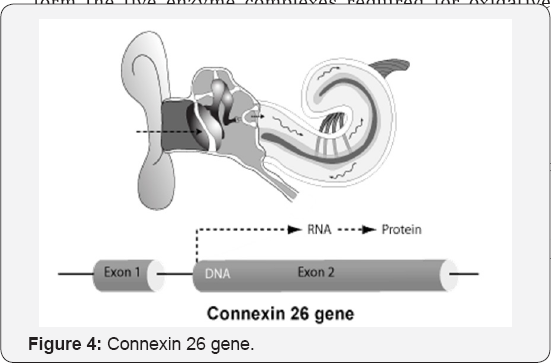
Q. Everyone has two copies of this gene, but if each parent has a flawed, recessive copy of the GJB2/ Connexin 26 gene, the baby may be born with hearing loss.
R. This is because the mutation is suspected of disrupting potassium flow in the inner ear.
S. Approximately 50% of childhood non syndromic recessive hearing loss is caused by mutations in the connexin 26 (Cx26 gene (GJB2/DFNB)
T. The most common mutation that is found in the connexin 26 gene is 35delG, which means that a G is deleted at position 35.
U. This is because the mutation is suspected of disrupting potassium flow in the inner ear.
V. Approximately 50% of childhood non syndromic recessive hearing loss is caused by mutations in the connexin 26 (Cx26 gene (GJB2/DFNB) W. The most common mutation that is found in the connexin 26 gene is 35delG, which means that a G is deleted at position 35.
X. More than 90 different mutations have been found in the coding sequences of connexin 26.
Y. Most are rare, but a few are relatively common in particular populations (e.g. 267delT and 235 delC in Ashkenazi Jewish and Asian populations, respectively).
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Multifactorial Genetic Disorders
- Some disorders appear to result from a combination of genetic factors interacting with environmental influences.
- Examples of this type of inheritance associated with hearing loss include clefting syndromes, involving conductive hearing loss, and the microtia/hemifacial microsomia/Goldenhar spectrum.
- Goldenhar's syndrome has been described as autosomal dominant in some families, although this may simply represent clustering.
- Findings in this syndrome include preauricular tags/pits, vertebral anomalies such as hypoplastic or hemivertebrae in the cervical region, epibulbar dermoids, and coloboma of the upper lid. Other conditions believed to represent multifactorial inheritance are increased susceptibility to hearing loss and hyperlipidemia.
Autosomal Chromosomal Syndromes
a) Middle ear and mastoid disease are often observe in Down syndrome children, but sensorineural hearing loss may also be present.
b) Trisomy 13, which is often lethal in the newborn period, can have significant sensorineural hearing loss in the survivors. Turner's syndrome, monosomic for all or part of one X chromosome, presents generally in female as gonadal dysgenesis, short stature, and often webbed neck or shield chest.
c) They will also have sensorineural, conductive, or mixed hearing loss, which can be progressive and may be the first evidence of the syndrome in prepubertal females.
d) SYNDROMES (Figure 5).
Autosomal dominant
I. Locus name- DFNA
Some cause pre-lingual deafness, progressive and affects all frequencies and sometimes downward sloping type hearing loss. Mostly post-lingual deafness affecting all frequencies and begins in any decade of life.
Autosomal recessive
A. Locus name- DFNB
Some cause post-lingual deafness, can be stable or progressive and causes moderate to profound hearing loss. Most of them cause pre lingual, can be stable or progressive and causes moderate to profound hearing loss.(Tables 1 & 2).


Apert syndrome
i. AKA= Acrocephalosyndactyly
Single gene autosomal dominant caused by the mutation of FGFR2 gene, located on the long arm of chromosome 10 at 10q26.
Audiological findings
- CHL-mild to moderate
- chronic middle ear disease
- ossicular anomalies
Symptoms
- CNS growth effected
- craniofacial & limb abnormalities
- mitten hand
- low set ears posteriorly rotated
- synostosis of one or more ossicles
- Fixation of footplate of stapes
- Reduced ME space
- ET dysfunction
- Syndactyly of fingers and toes
- Brachymelia1
- MR
- Hydrocephaly
- Cognitive imparment
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Waardenburg Syndrome
- It is the most common form of inheritable congenital deafness.
- Genetic Analysis Distal end of band 2q35gene responsible is: PAX3
- There is a significant amount of variability of expression in this syndrome.
- There may be unilateral or bilateral sensorineural hearing loss in patients and the phenotypic expressions may include pigmentary anomalies and craniofacial features.
Symptoms
a) Marked facial asymmetry
b) Lagophthalmos
c) A drooping right corner of the mouth.
d) Absence of naso-frontal angle
e) Eyebrow hypertrichosis
f) Upturned nasal tip
g) Shortened upper lip
h) Pronounced cupid's bow.
A. Type I -Waardenburg syndrome is characterized by evidence of dystopia canthorum and the full symptomatology of the disease.
I. Narrow nose
II. Marked hypoplasia of the nasal bone,
III. Short philtrum
IV. Short and retro positioned maxilla.
V. Convergent strabismus (blepharophimosis)
VI. Reduced visibility of the medial sclera
VII. The head circumference, clivus length, and facial depth are smaller in affected individuals with this syndrome.
B. Type II -Waardenburg syndrome are a heterogeneous group with normally located canthi (without dystopia canthorum).
I. Sensorineural hearing loss (77%)
II. Heterochromia iridium (47%) are the 2 most important diagnostic indicators for this type.
C. Type III- Waardenburg syndrome (Klein-Waardenburg syndrome) is similar to type I but is also characterized by musculoskeletal abnormalities
I. Aplasia of the first 2 ribs
II. Lack of differentiation of the small carpal bones
III. cystic formation of the sacrum1
IV. Abnormalities of the arms1
V. Amyoplasia and stiffness of the joints
VI. Bilateral cutaneous syndactyly
VII. mental retardation
VIII. Microcephaly
IX. severe skeletal anomalies.
D. Type IV Waardenburg syndrome (Shah-Waardenburg syndrome) is the association of Waardenburg syndrome with congenital aganglionic megacolon (Hirschsprung disease).
a) Dystopia canthorum is found in 41.2-99% of persons with Waardenburg syndrome.
i. The distance between the inner angles of the eyelids is accompanied by increased distance between the inferior lacrimal points.
ii. Hageman and Delleman divided Waardenburg syndrome into 2 variants: with dystopia canthorum and without.
b) Congenital deafmutism occurs in 9-62.5% of persons with Waardenburg syndrome.
c) Different combinations of hearing loss occur: unilateral or bilateral, severe or moderate, total or moderate. Fisch separated Waardenburg syndrome into the following distinct types according to audiogram results.
d) Patients with total deafness and little residual hearing at the lower frequency
e) Patients with a moderate degree of deafness with uniform hearing loss in the lower and middle frequency with improvement in the higher frequency
f) Pigmentary disturbances of hairs in Waardenburg syndrome include 2 types of alterations: white forelock and premature graying of scalp hair, eyebrows, cilia, or body hair.
i. The white forelock is observed in 17-58.4% of persons with Waardenburg syndrome and involves the forehead (and both medial eyebrows), the vertex, or another part of scalp.
ii. The white forelock may be evident at birth or soon afterward, or it may develop later.
iii. Poliosis may persist throughout life or may disappear in the first years of life and reappear later.
iv. Patients with Waardenburg syndrome become prematurely gray in 7% of cases.
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Stickler Syndrome
a) Cleft palate and severe myopia are its characteristics features.
b) Significant sensorineural hearing loss or mixed hearing loss is present in about 15% of cases, whereas hearing loss of lesser severity may be present in up to 80% of cases.
c) Autosomal dominant- COLI1A1, COLI1A2,COL2A1, COL2A2 and several others
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Norrie Syndrome
Norrie syndrome is a sex-linked disorder that includes congenital or rapidly progressive blindness.
Alport Syndrome
I. Alport syndrome involves hearing loss associated with renal impairment of varying severity.
II. When a genetic mutation occurs, connecting structures in both the inner ear and kidney become increasingly fragile, resulting in progressive hearing impairment and kidney disease
III. Eustation tube dysfunction occurs secondarily to the cleft palate and results in conductive hearing loss.
IV. Ossicular abnormalities may also be present.
Branchio-oto-renal Syndrome
A. Branchio-oto-renal syndrome is estimated to occur in 2% of children with congenital hearing impairment.
B. Seventy-five percent of patients with branchio-oto-renal syndrome have significant hearing loss.
Treacher Collins Syndrome
I. Conductive hearing loss is present 30% of the time, but sensorineural hearing loss and vestibular dysfunction can also be present.
II. Ossicular malformations are common in these patients.
III. The syndrome is transmitted autosomal dominant with high penetrance gene located on 5q-32-q33.1, TREACLE.
Neurofibromatosis
i. Mental retardation, blindness, and sensorineural hearing loss can result from central nervous system tumors.
ii. Neurofibromatosis is classified as types 1 and 2.
iii. Autosomal dominant 22q12.2.
iv. NF2 is a tumor supressor gene
v. Acoustic neuromas are usually unilateral and occur in only 5% of affected patients.
vi. Neurofibromatosis type 2, which is a genetically distinct disorder, is characterized by bilateral acoustic neuromas.
vii. Bilateral acoustic neuromas are present in 95% of affected patients and are usually asymptomatic until early adulthood.
Ushers syndrome
a) Usher's syndrome has a prevalence of 3.5 per 100,000 populations)
b) Sensorineural hearing loss and retinitis pigmentosa characterize the syndrome.
c) Usher type 1 patients have congenital bilateral profound hearing loss and absent vestibular function
d) type 2 patients have moderate losses and normal vestibular function.
e) type 3 demonstrate progressive hearing loss and variable vestibular dysfunction and are found primarily in the Norwegian population.
Pendred Syndrome
a. Pendred’s syndrome includes thyroid goiter and profound sensorineural hearing loss.
b. Hearing loss is progressive in about 15% of patients.
c. The majority of patient present with bilateral moderate to severe sensorineural hearing impairment, with some residual hearing in the low frequencies.
Wildervaank Syndrome
I. Wildervaank's syndrome is comprised of the Klippel- Feil sign involving fused cervical vertebrae, sensorineural hearing or mixed hearing impairment, and cranial nerve 6 paralysis causing retraction of the eye on lateral gaze.
II. This syndrome is seen most commonly in female because of the high mortality associated with the X-linked dominant form in males. Isolated Klippel-Feil sequence includes hearing impairment in about one third of cases.
III. The hearing impairment is related to bony malformations of the inner ear.
Jervell and Lange-Neilsen Syndrome
Jervell and Lange-Neilsen syndrome is a rare syndrome that consists of profound sensorineural hearing loss and syncopal episodes resulting from a cardiac conduction defect. Genetic studies attribute one form of Jervell and Lange-Neilsen syndrome to homozygosity for mutations affecting a potassium channel gene (KVLQT 1) on chromosome 11p15.5, which are thought to result in delayed myocellular repolarization in the heart. The gene KCNE1 has also been shown to be responsible for the disorder.
I. Hereditary Meniere’s syndrome
a) Recurrent severe unilateral headaches
b) variable results of vestibular function tests
c) Vertigo and vomiting
d) Audiological finings- fluctuating SNHL
e) Unilateral or bilateral
f) Assymetrical and progressively deteriorates
g) Mild to severe HL
h) Autosomal recessive transmission
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Charge
- Coloboma, Heart anomalies, Atresia, Choanae, Retarded growth and development, Genital hypoplasia and ear anomalies
- Autosomal dominant inheritance
- CHL, SNHL or mixed, trough shaped configuration and vestibular anomalies
Symptoms
- CNS impairment
- Brain malformation
- Facial paresis
- Facial assymetry
- Microcephaly
- Low set posteriorly rotated ears
- Micrognathia
- Nasal root
- Semicircular canal
- Hypoplasia
- Heart anomalies
- Tracheoesophageal fistula
- Genital anomalies
- Scoliosis
- Short neck
Crouzon syndrome (Craniofacial dyostosis)
- Mutation of single gene FGFR2 located on the long arm of chromosome 10 at 10 q 26
- Autosomal dominant inheritance with variable expression
- CHL common. Mild to moderate stenosis of EAC, chronic middle ear disorder and ossicular anomalies
Symptoms
- Maxillary hypoplasia
- Malocclusion
- Soft tissue hypertrophy of hard palate
- Cognitive impairment
- Increased intracranial pressure
- Bulging eyes
- Dental crowding
- Abnormality of cranial and facial bones
- Cleft palate
- synostosis
Hunter syndrome
- X- linked recessive inheritance. Gene mapped to Xq 28 and is labelled as IDS
- CHL, Chronic middle ear disorder, glue ear, progressive HL
I Symptoms
- Short stature,
- cognitive defects,
- Thick lips,
- alveolar ridge hypertrophy,
- thickening of palate,
- abnormal retinal pigmentation,
- retinal degenration,
- corneal opacities,
- heart, liver and spleen problems1
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Enlarged Vestibular Aqueduct
- An enlarged vestibular aqueduct apparently is the result of hydrodynamic changes and possibly labyrinthine membrane disruption.
- Early onset sensorineural hearing loss,
- Bilateral
- Often progressive
- May be accompanied by vertigo or incoordination.
- Autosomal dominant inheritance.
- Recessive inheritance is also possible
Semicircular Canal Malformations
Isolated lateral canal defects -most common inner ear malformations-identified on temporal bone imaging studies. Superior semicircular canal deformities are always accompanied by lateral semicircular canal deformities. Lateral canal deformities often occur in isolation.
a) Recently, the genes responsible for development of microstructures in the cochlea have been identified.
b) For example, connexins are the channels that connect neighboring cells and allow passive transfer of small molecules.
c) It can be inherited by Autosomal recessive as in most cases and Autosomal dominant as in few cases.
d) These gap junction are important for the electric and metabolic coupling of neighboring cells.
e) These connexins development are coded in genes like Connexin 26, 30, 31
f) When these genes get mutated there will be non syndromic hearing loss.
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Osteogenesis Imperfecta
Osteogenesis imperfecta is characterized by :-
- Bone Fragility
- Blue sclera
- Conductive, mixed, or sensorineural hearing loss
- Hyper-elasticity of joints and ligaments.
- Triangular face
- Short stature
- Hypermobile joints
- Cardiovascular abnormalities
- skin disorders.
Hearing loss is usually mixed and has a prevalence ranging from 26-78%. Autosomal dominant with variable expressivity and incomplete penetrance. Two genes for osteogenesis imperfecta have been identified,
1. COLIA1 on chromosome 17q
2. COLIA2 on chromosome 7q.
i. The hearing loss usually presents itself during the late 20s or early 30s.
ii. The conductive component of the hearing loss is attributed to the thickened and fixed stapes footplate, similar to what is seen in otosclerosis.
iii. The sensorineural component usually results from cochlear hair cell atrophy and atrophy of the stria vascularis.
iv. Also, anomalous bone formation in and around the cochlea may contribute to the sensorineural component of the hearing loss.
Ear anomalies
A. Michel Aplasia
a. Complete agenesis of the petrous portion of the temporal bone
b. External and middle ear may be unaffected.
c. Autosomal dominant inheritance has been observed, but recessive inheritance is also likely.
Treatment: Vibrotactile devices have been of some help
B. Mondini Aplasia
a) Mondini aplasia involves a developmentally deformed cochlea in which only the basal coil can be identified clearly.
b) Enlarged endolymphatic sac
c) Interscalar septum absent
d) Autosomal dominant
e) May not be bilateral.
f) Occurs with genetic syndromic disorders or can be with non genetic syndromic disorders.
Treatment
Early habilitative intervention and conventional amplification
C. Scheibe Aplasia
a) Cochlear-saccular dysplasia or pars inferior dysplasi
b) The bony labyrinth and the superior portion of the membranous labyrinth, including the utricle and semicircular canals are differentiated
c) Organ of Corti -poorly differentiated
d) Deformed tectorial membrane
e) Collapsed Reissner’s membrane, which compromises the scala media
f) Autosomal recessive nonsyndromic trait.
Treatment: Conventional amplification with habilitative intervention
D. Alexander Aplasia
a) Cochlear duct differentiation at the level of the basal coil is limited with resultant effects on the organ of corti and ganglion cells
b) Causes high frequency hearing loss with adequate residual hearing in the low frequencies
Treatment: use of amplification
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
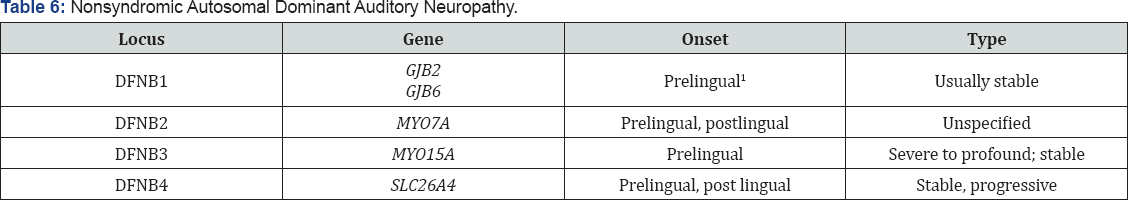
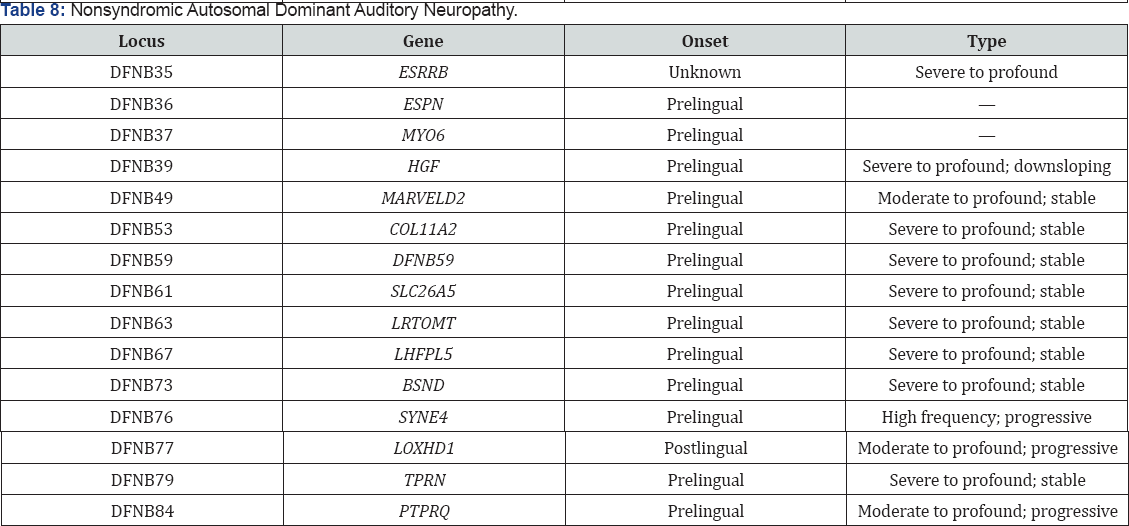
Autosomal Dominant Auditory Neuropathy
a) AN - clinically heterogeneous set of hearing disorders, neural functions are impaired but the OHCs of the cochlea appear to function normally.
b) Some families show autosomal dominant patterns of inheritance and affected members usually have peripheral neuropathy.
c) Kovach et al - mutation of PMP22 gene associated with Charcot- Marie - Tooth syndrome as a possible link to some of the characteristics of neuropathy, chromosome affected is 8q24.
I. PMP22 Gene
i. 'Peripheral Myeline Protein 22 gene' provides instructions for making protein called PMP22.
ii. This protein is major component of myelin. This is produced primarily by schwan cells.
iii. PMP22 may also play a role in regulating cell division and maturation, cell shape etc.
iv. Charcot-Marie-Tooth Disease and hereditary neuropathy are associated with PMP22 mutation
a) The early onset subtype usually progresses rapidly from onset at age 1 % years to profound loss by age 6.
b) Genetic linkage studies have identified at least 15 gene loci for recessive nonsyndromic hearing loss.
i. The gene DFNB2 on chromosome 13q may be the most common and has been identified as connexin 23.
ii. Another gene, DFNB1, also found on chromosome 13 codes for a connexin 26 gene gap junction protein.
II. Presbycusis
i. Another condition associated with acquired heteroplasmic mutations and hearing loss is presbycusis.
ii. Since mitochondrial DNA mutations and the resulting loss of oxidative phosphorylation activity seem to play an important role in the aging process, it is not unlikely that mitochondrial mutations in the auditory system can also lead to presbycusis).
iii. These mutations are thought to be associated with insidious decline in physiological and biochemical performance of an organ and to contribute significantly to the ageing process and ultimately death.
iv. Accordingly, suggests that presbyacusis is due to deletions and in particular a 4977 nucleotide deletion which is also called "common deletion".
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Noise Induced Hearing Loss NIHL
Guy Van Camp
- KCNE1 show the version of the gene associated with increased risk to noise causes the encoded ion channel to open more rapidly than the normal version.»
- This defective gene could be responsible for making people sensitive to noise [30-35].
Otosclerosis
- Otosclerosis is caused by proliferation of spongy type tissue on the otic capsule eventually leading to fixation of the ossicles and producing conductive hearing loss.
- Hearing loss may begin in childhood but most often becomes evident in early adulthood and eventually may include a sensorineural component.
- Otosclerosis appears to be transmitted in an autosomal dominant pattern with decreased penetrance, so only 25% to 40% of gene carriers show the phenotype. The greater proportion of affected females points to a possible hormonal influence.
- Recent statistical studies suggest a role for the gene COLIA1 in otosclerosis, and measles viral particles have been identified within the bony overgrowth in otosclerotic foci, raising the possibility of an interaction with the viral genome.
Down's syndrome
- This is the most common of the chromosome abnormality syndromes typified by a wide range of abnormalities.
- Otolaryngologic findings are numerous in these patients and can affect every region of the head and neck.
- This includes small ears with overfolding of the superior helix, stenotic EAC and eustachian tube dysfunction [36,44].
- There is also an increased incidence of chronic ear disease in affected children due to increased incidence of upper respiratory infections, reduction of B and T cell function (immune system immaturity), and eustachian tube dysfunction.
- The hearing loss in DS is usually conductive secondary to the chronic middle ear disease but can also be due to ossicular chain abnormalities, especially the stapes.
- Upper airway obstruction are also problems encountered by children with DS due to the midface hypoplasia, and relative enlargement of the tongue, tonsils and adenoids in a constricted naso/oropharynx.
- In terms of speech and behavior, most Down's syndrome patients exhibit dysarthria and articulation deficits in conjunction with some degree of mental retardation (IQ 30-50).
- Trisomy of the 21st chromosome.
- Gene located on 21q22.3
- CHL, SNHL or mixed, chronic middle ear infection, serous otitis media and middle ear effusion, ET dysfunction, structural abnormalities of cochlea and decreased length
Symptoms
a) CNS impairment
b) Mental retardation
c) Alzheimer's disease
d) Hypotonia
e) Cerebral and cerebellar hypoplasia
f) Brachycephaly
g) Microcephaly
h) Macroglossia
i) Small ears, stature
j) Obesity
k) Small fingers
l) Branchydactyly
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Fetal Alcohol Syndrome (FAS)
a) Of children born to alcoholic mothers, 30-40% suffer this syndrome.
b) The amount of alcohol intake required to cause FAS has not been clearly established.
c) The alcohol induced developmental abnormalities can be the result of restriction of cell growth during critical periods.
d) Characteristics of the syndrome include prenatal and postnatal growth deficiency, microcephaly, and mental retardation (average IQ of 63).
e) Behavior is also affected as irritability and hyperactivity are common.
f) Neural tube defects and seizure disorder may also be present.
g) Almost no system is guaranteed to be spared as cardiac, renal and skeletal anomalies may manifest themselves as well as malignant neoplasms of embryonal origin.
h) Common facial dysmorphisms include narrow forehead, short palpebral fissures, ptosis of eyelids, midface hypoplasia, short nose, smooth philthrum, thin upper lip and hypoplastic mandible.
i) In addition, cleft palate or cleft lip may exist.
j) Ten percent of patients have hearing loss that may be either conductive or sensorineural.
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Goldenhar's Syndrome
a) Referred to as facioauriculovertebral dysplasia (FAVD) and hemifacial microsomia (HFM), this disorder results from aberrant development of the first and second branchial arches.
b) HFM is estimated to occur in 1 in 5600 live births, perhaps making it the most significant asymmetric craniofacial disorder.
c) Otologic manifestations include microtia/anotia, preauricular tags, ossicular abnormalities, abnormal facial nerve course, and hearing loss (conductive > sensorineural).
i. The hearing loss is predominantly conductive secondary to the abnormal development of the structures derived from the first and second branchial arches.
ii. Facial abnormalities include unilateral hypoplasia of the maxilla, malar and temporal bones in addition to mandibular ramus and condyle hypoplasia.
iii. Macrostomia or pseudomacrostomia (lateral cleft-like extension of the oral commisures), cleft lip or palate and delayed dental development.
iv. Lastly, the mastoid is poorly pneumatized and their may exist agenesis of the parotid gland or displacement of the gland.
v. In terms of non-head and neck features, affected individuals can also have cardiac abnormalities such as coarctation of the aorta, ventricular septal defect.
vi. Renal ectopia and hydronephrosis can encompass the renal abnormalities.
vii. Limb deformities can be present as well as cerebral malformation and mental retardation.
viii. Ocular abnormalities include blepharoptosis, microopthalmia, epibulbar tumors, and retinal abnormalities leading to reduced visual acuity.
20. Rubella
i. Consists of a triad characterized by deafness, congenital cataracts and heart defects.
ii. This disease is caused by an RNA togavirus and is transmitted postnatally via respiratory secretion, saliva, or direct contact.
iii. Transplacental transmission is the route responsible for congenital infection which can involve more sequelae if infection is present during the first trimester.
iv. In addition to the above listed triad, other abnormalities that may manifest are microcephaly, motor and neural retardation, hepatosplenomegaly, thrombocytopenia, encephalitis and interstitial pneumonitis.
v. The hearing loss in rubella is typically asymmetric and sensorineural with variable severity.
vi. The 500-2000 Hz frequencies are the most commonly affected.
vii. This hearing deficit usually manifests by 5 years of age and can be an isolated finding in 22%.
viii. Approximately 25% of patients will experience a progressive form of hearing loss.
- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
Cytomegalovirus (CMV)
- CMV has an incidence of 0.2%-2.3% of live births making it one of the most frequently occurring viruses worldwide and the leading cause of congenital malformations and mental retardation in developed countries.
- Of all the TORCH infections, CMV is the most common. Microcephaly, intrauterine growth restriction (IUGR), petechiae, encephalitis, hepatosplenomegaly, and deafness are some of the physical characteristics of a congenital CMV infection.
- CMV is estimated to account for 1/3 of sensorineural hearing loss in young children.
- Hearing impairment in CMV can be delayed (occurring months-years after birth), or fluctuating and progressive.
- Interesting to note, infants with petechiae and IUGR are 2- 3 times more likely to have sensorineural hearing loss.
- Post mortem temporal bone studies on infants who died from cytomegalic inclusion disease have revealed inclusion bodies in the stria vascularis, Reissner's membrane,
- Endolymphatic hydrops was noted in the cochlear ducts (Tables 3-10)

- Powerpoint presentation
- Introduction
- Need for knowing
- Genetic testing
- Types of Genetic Testing
- Procedure
- Results
- Mitochondrial Mutation and Ototoxicity
- Multifactorial Genetic Disorders
- Waardenburg Syndrome
- Stickler Syndrome
- Norrie Syndrome
- Charge
- Enlarged Vestibular Aqueduct
- Osteogenesis Imperfecta
- Autosomal Dominant Auditory Neuropathy
- Noise Induced Hearing Loss NIHL
- Fetal Alcohol Syndrome (FAS)
- Goldenhar's Syndrome
- Cytomegalovirus (CMV)
- References
References
- American College of Medical Genetics (2002) Genetics Evaluation Guidelines for the Etiologic Diagnosis of Congenital Hearing Loss. Genetic Evaluation of Congenital Hearing Loss Expert Panel. ACMG statement, Genet Med 4(3): 162-71.
- American College of Medical Genetics (2000) Statement on universal newborn hearing screening. Medical Genetics Translating Genes into Health, USA, p. 1-2.
- Journal of the American Medical Association (1979) Guide for the evaluation of hearing handicap. JAMA 241(19): 2055-2059.
- Arnos KS (2003) The implications of genetic testing for deafness. Ear Hear 24(4): 324-331.
- Bauer PW, Geers AE, Brenner C, Moog JS, Smith RJ (2003) The effect of GJB2 allele variants on performance after cochlear implantation. Laryngoscope 113(12): 2135-2140.
- Brookhouser PE, Smith SD (1998) Genetic hearing loss. In: Bailey BJ (eds). Head and Neck Surgery -Otolaryngology, (2nd edn), Lippincott - Raven, Philadelphia, USA.
- Brookhouser PE, Grundfast KM (1998) General sensorineural hearing loss. In: Cummings (eds). Pediatric Otolaryngology Head and Neck Surgery, (3ri edn), St Louis, Mosby, USA.
- Bussoli TJ, Steel KP (1998) The molecular genetics of inherited deafness-current and future applications. Journal of Laryngology and Otology 112(6): 523-530.
- Cryns K, Sivakumaran TA, Van den Ouewland JMW, Pennings RJE, Cremers CWRJ, et al. (2003) Mutational spectrum of the WFS1 gene in Wolfram syndrome, nonsyndromic hearing impairment, diabetes mellitus and psychiatric disease. Hum Mut 22(4): 275-287.
- Denoyelle F, Marlin S, Weil D, Moatti L, Chauvin P, et al. (1999) Clinical features of the prevalent form of childhood deafness, DFNB1, due to connexin-26 gene defect: implications for genetic counselling. The Lancet 353(9161): 1298-1303.
- Estivill X, Fortina P, Surrey S, Rabionet R, Melchionda S, et al. (1998). Connexin-26 mutations in sporadic and inherited sensorineural deafness. Lancet 351(9100): 394-398.
- Fischel Ghodsian N (1998) Mitochondrial mutations and hearing loss: paradigm for mitochondrial genetics. Am J Hum Genet 62(1): 15-19.
- Gantz BJ, Turner C, Gfeller KE, Lowder MW (2005) Preservation of hearing in cochlear implant surgery: advantages of combined electrical and acoustical speech processing. Laryngoscope 115(5): 796-802.
- Green GE, Scott DA, McDonald JM, Woodworth GG, Sheffield VC, et al. (1999) Carrier rates in the Midwestern United States for GJB2 mutations causing inherited deafness. JAMA 281(23): 2211-2216.
- Grundfast KM, Atwood JL, Chuong D (1999) Genetics and molecular biology of deafness. Otolaryngology Clinics of North America. 32(6): 1067-1088.
- Grundfast MD, Lalwani AK (1992) Practical approach to diagnosis and management of hereditary hearing impairment. Ear Nose and Throat Journal 71(10): 479-493.
- Heller AJ, Stanley C, Shaia WT, Sismanis A, Spencer RF, et al. (2002) Localization of biotinidase in the brain: implications for its role in hearing loss in biotinidase deficiency. Hear Res 173(1-2): 62-68.
- Hildebrand MS, Tack D, McMordie S, DeLuca A, Ae Hur I, et al. (2008) Audioprofile-directed screening identifies novel mutations in KCNQ4 causing hearing loss at the DFNA2 locus. Genet Med 10(11): 797-804.
- Hilgert N, Smith RJ, Van Camp G (2009) Forty-six genes causing nonsyndromic hearing impairment: Which ones should be analyzed in DNA diagnostics. Mutat Res 681(2-3): 189-196.
- Hoskins BE, Cramer CH, Silvius D, Zou D, Raymond RM, et al. (2007) Transcription factor SIX5 is mutated in patients with branchio-oto- renal syndrome. Am J Hum Genet 80(4): 800-804.
- Huyghe JR, Van Laer L, Hendrickx JJ, Fransen E, Demester K, et al. (2008) Genome-wide SNP-based linkage scan identifies a locus on 8q24 for an age-related hearing impairment trait. Am J Hum Genet 83(3): 401-407.
- Jackler RK, Luxford WM, House WF (1987) Congenital malformations of the inner ear: a classification based on embryogenesis. Laryngoscope 97(3): 2-14.
- American Academy of Otolaryngology and the American Council of Otolaryngology (1979) Guide for the evaluation of hearing handicap. JAMA 241(19): 2055-2059.
- Kelley PM, Harris DJ, Comer BC, Askew JW, Fowler T, et al. (1998) Novel mutations in the connexin 26 gene (GJB2) that cause autosomal recessive (DFNB1) hearing loss. Am J Hum Genet 62(4): 792-799.
- Kenneson A, Cannon MJ (2007) Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev Med Virol. 17(4): 253-276.
- Kokotas H, Petersen MB, Willems PJ (2007) Mitochondrial deafness. Clin Genet 71(5): 379-391.
- Konings A, Van Laer L, Michel S, Pawelczyk M, Carlesson PI, et al.(2009) Variations in HSP70 genes associated with noise-induced hearing loss in two independent populations. Eur J Hum Genet 17(3): 329-335.
- Konigsmark BW, Gorlin RJ (1979) Genetic and metabolic deafness, Philadelphia, 1976, WB Saunders. 4(3): 1-310.
- Middleton A, Hewison J, Mueller RF (1998) Attitudes of deaf adults toward genetic testing for hereditary deafness. Am J Hum Genet. 63(4): 1175-1180.
- Morton CC, Nance WE (2006) Newborn hearing screening-a silent revolution. N Engl J Med 354(20): 2151-2164.
- Northern JL, Downs M (2002) Hearing in Children. Baltimore, MD: Lippincott, Williams, and Wilkins, Philadelphia, Pennsylvania, USA, pp. 720.
- Pandya A, Xia XJ, Erdenetungalag R, Amendola M, Landa B, et al. (1999) Heterozygous point mutations in the mitochondrial tRNA Ser(UCN) precursor coexisting with the A1555G mutation in deaf students from Mongolia. Am J Hum Genet. 65(6): 1803-1806.
- Ruf RG, Xu PX, Silvius D, Otto EA, Beekmann F, et al. (2004) SIX1 mutations cause branchio-oto-renal syndrome by disruption of EYA1- SIX1-DNA complexes. Proc Natl Acad Sci USA. 101(21): 8090-8095.
- Smith RJH, Shearer AE, Hildebrand M, Van Camp G (1999) Deafness and Hereditary Hearing Loss Overview Gene Reviews, University of Washington, USA.
- Shearer AE, DeLuca AP, Hildebrand MS, Taylor KR, Gurrola JG II, et al. (2010) Comprehensive genetic testing for hereditary hearing loss using massively parallel sequencing. Proc Natl Acad Sci USA 107(49): 21104-21109.
- Smith RJH, Bale JF, White KR (2005) Sensorineural hearing loss in children. Lancet. 365(9462): 879-890.
- Steel KP (1998) A new era in the genetics of deafness. New England Journal of Medicine 339(21): 1545-1547.
- Toriello HV, Reardon W, Gorlin RJ, (eds) (2004) Hereditary Hearing Loss and Its Syndromes. Oxford University Press, New York, USA.
- Van Camp G, Smith RJH (2010) The Hereditary Hearing Loss Homepage. Science open, Burlington, USA.
- Van Camp G, Willems PJ, Smith RJ (1997) Nonsyndromic hearing impairment: unparalleled heterogeneity. Am J Hum Genet. 60(4): 758764.
- Vore AP, Chang EH, Hoppe JE, Butler MG, Forrester S, et al. (2005) Deletion of and novel missense mutation in POU3F4 in 2 families segregating X-linked nonsyndromic deafness. Arch Otolaryngol Head Neck Surg 131(12): 1057-1063.
- Weegerink NJ, Huygen PL, Schraders M, Kremer H, Pennings RJ, et al. (2011) Variable degrees of hearing impairment in a Dutch DFNX4 (dFN6) family. Hear Res 282(1-2): 167-1677.
- Wolf B, Spencer R, Gleason T (2002) Hearing loss is a common feature of symptomatic children with profound biotinidase deficiency. J Pediatr 140(2): 242-246.
- Zelante L, Gasparini P, Estivill X, Melchionda S, D Agruma L, et al. (1997) Connexin 26 mutations associated with the most common form of non- syndromic neurosensory autosomal recessive deafness (DFNB1) in Mediterraneans. Hum Mol Genet 6(9): 1605-1609.





























