Recent Developments of Nanotechnology for Alzheimer’s Disease Diagnosis and Therapy
Xinyu Dai1, Yuntao Li1* and Yuejiao Zhong2
1 Department of Neurology, Second Affiliated Hospital of Nanjing Medical University, China
2Department of Oncology, The Affiliated Cancer Hospital of Nanjing Medical University, China
Submission: August 15, 2018; Published: October 25, 2018
*Corresponding author: Yuntao Li, Department of General Practice, Second Affiliated Hospital of Nanjing Medical University, No 121 Jiangjiayuan Road, Nanjing 210011, China.
How to cite this article: Xinyu D, Yuntao L, Yuejiao Z. Recent Developments of Nanotechnology for Alzheimer’s Disease Diagnosis and Therapy. Glob J Nano. 2018; 4(4): 555644. DOI: 10.19080/GJN.2018.04.555644
Abstract
Alzheimer’s disease (AD) is a common neurodegenerative disease-causing memory loss and deterioration of cognitive function. In addition, it displays other neurological symptoms such as delusions, deficiency in language, learning, and abstract thinking. The symptoms of the disease progress gradually, which can eventually lead to incapacitation of the individuals functioning. AD will have enormous social and economic impacts in the coming decades. Therefore, strategies for early detection and treatment of AD has become one of the most challenging in modern medicine. Drug and imaging agent’s delivery to the brain remains the main problems for the diagnosis and treatment of AD because of the protection of blood-brain barrier (BBB), however, nanoparticles can help overcome the limitations of BBB. In this article, we have explored latest developments in nanotechnology-based AD diagnosis and therapy
Keywords: Alzheimer’s disease; Nanoparticles; Nanotechnology; Targeting; Diagnosis; Therapy
Abbrevations: AD: Alzheimer’s Disease; BBB: Blood-Brain Barrier; NPs: Nanoparticles; APP: Amyloid Precursor Protein; PS1: Presenilin 1; PS2: Presenilin 2; Aβ: Amyloid-β Protein; CSF: Cerebral Spinal Fluid; MRI: Magnetic Resonance Imaging; SPIONs: Superparamagnetic Iron Oxide Nanoparticles; PEG: Polyethyleneglycol; PLA: Lactic Acid; AuNC: Gold Nanoclusters; AChE: Acetylcholinesterase
Introduction
Generally, nanotechnology refers to the measurement and modeling of substances in a nanoscale manner, which can be applied to engineering and technology by manipulating or modifying material materials to give them new properties. In current medical research, new diagnostic and therapeutic tools were developed combined with nanotechnology, which are beneficial to the specific transport and absorption of drugs and contrast agents to the brain and promote the regeneration of damaged neurons to limit or reverse neurological disorder. New Nano pharmaceutical technology that combines polymer nanoparticles, liposomes, micelles, dendrimers, and nanogels could enhance passage of drugs and small molecules a crossing the BBB.
Pathogenesis
AD as a common neurological disease there were 46.8 million people worldwide living with dementia in 2015 and this number will gradually increase to 131.5 million in 2050 [1]. There were three mutant genes have been reported to cause AD, including the gene encoding the amyloid precursor protein (APP) on chromosome 21, and the gene encoding presenilin 1 (PS1) on chromosome 14 and the gene encoding presenilin 2 (PS2) on the chromosome 1. These mutations can cause the amyloid-β protein (Aβ) to form senile plaques extracellularly, while the microtubule-associated protein tau (Tau) is hyperphosphorylated to form neurofibrillary tangles. These changes throughout the brain leads to extensive cortical damage and early loss of basal forebrain cholinergic neurons [2]. Among them, Aβ has become the main focus of neurodegenerative research and many current treatments are targeted to the production, fibrosis and clearance of Aβ. Although the treatment strategy is advanced, there is no obvious clinical benefit, and currently approved neurotransmitter-modulating drugs can only improve symptoms.
The pathogenesis of AD is multifactorial, and it is not easy to detect behavioral and memory changes caused by disease at an early stage. So, it is important to explore early markers to predict the onset of disease. As a biomarker, current research indicates that Aβ deposition is the first pathogenic event that occurs before clinical symptoms appear, and neurofibrillary tangles promote its development and exhibit detectable clinical symptoms [3,4]. Therefore, it can be speculated that Aβ and Tau proteins have been concentrated in cerebral spinal fluid (CSF) before clinical onset. In addition to exploring biomarkers, because the net charge of Aβ is negative, multivalent cations can bind Aβ and accelerate the process of aggregation and fibrillation, thus monitoring plasma multivalent cations (eg zinc, copper, iron, etc.) are expected in the early detection of AD [5].
Nanotechnology in the Diagnosis of AD
Since neuro invasiveness and degenerative changes have begun before the onset of AD symptoms, early diagnosis is the key to effective treatment of AD. Most studies focus on magnetic resonance imaging (MRI) using contrast-doped NPs or labeling NPs with fluorescent probes to detect and identify amyloid plaques.
Iron oxide NPs
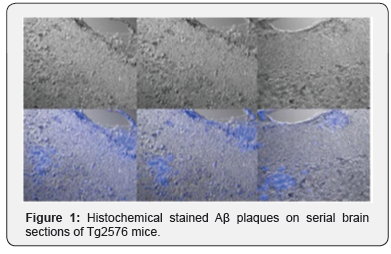
Magnetic iron oxide NPs have gained wide attention due to their large surface area, good magnetic properties, low toxicity, and good biocompatibility and degradability. For example, Cheng et al. [6] synthesized superparamagnetic iron oxide nanoparticles (SPIONs) connected with curcumin which coated with polyethyleneglycol (PEG)-lactic acid (PLA). The material is non-cytotoxic and has the ability to detect amyloid plaques in the brain of Tg2576 mice with AD [7] (Figure 1). Both top and bottom images were viewed by confocal microscopy. Top images were bright view and bottom images were fluorescence signal from stained chemical (from left to right stain by thioflavin T, curcumin or curcumin-MNPs) [7].
Gold NPs
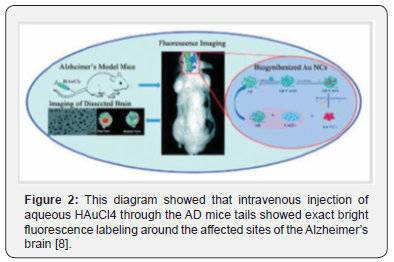
Lai et al exposed these sites to an aqueous solution of chloroauric acid to form a gold salt by targeting the site of infection [8]. These salts are assembled into gold nanoclusters (AuNC) for fluorescence imaging. Within a few hours after the injection of chloroauric acid by the tail vein of AD mice, there was a clear fluorescent label around the affected part of the brain, whereas the control mice did not show any fluorescent area after intravenous injection of chloroauric acid for 24 hr (Figure 2).
Nanotechnology in the Treatment of AD
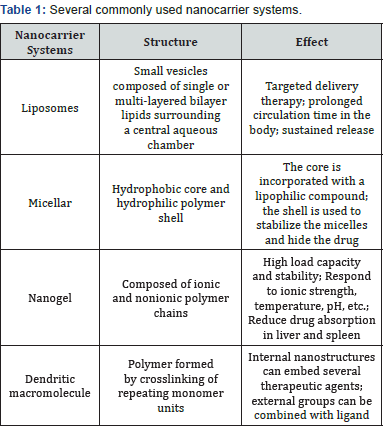
BBB is the primary barrier to the delivery of therapeutic drugs to the brain. Some treatments have forced them to open by causing structural damage to the BBB, at which point the BBB has lost its selectivity for drug passage. The carrier system combined with nanotechnology is the most promising treatment strategy for delivering drugs to the brain through the BBB. The current common delivery systems are shown in (Table 1) [9].
Cholinergic anti-inflammatory effects play an important role in preventing the development of learning and memory disorders in AD patients, so acetylcholinesterase (AChE) inhibitors can be used for symptomatic treatment of AD. As an inhibitor of AchE and butyrylcholinesterase, rivastigmine was approved by the FDA in 2000 for the treatment of AD. Studies have shown that coating the drug in nanoparticles enhances the targeted delivery of rivastigmine and reduces the side effects of free drug administration [10-13].
Curcumin can enhance mitochondrial function and may be suitable for preventing AD. However, the bioavailability of curcumin is low. Some studies have suggested that the bioavailability of curcumin micelles is higher than that of natural curcumin, and the encapsulation of nanoparticles can increase the solubility of curcumin, prolong the circulation time in the body, and improve the Targeted release within brain [14-17]. There is also evidence that metals have the effect of damaging neurons. The level of metal ions can be reduced by the use of chelating agents that target the interaction between Aβ and metal ions in the brain of AD patients [18,19].
Limitations of Combining Nanotechnology to Treat AD
It is well known that NPs will be immediately covered by proteins to form protein crowns after coming into the biological environment [20], and the effect of protein crowns on Aβ fibrosis should be assessed. Different diseases may also alter the fate of NPs in the body, so NPs may also have different therapeutic effects or toxic effects after entering different patients. These studies suggest that in the future research of Nanotechnology, a variety of hidden factors in the nanobio interface should be considered. The potential toxicity of NPs is another important issue, and there are few reports around long-term toxicity after NPs use. In order to solve these problems, it has been expected to develop multifunctional NPs with multiple therapeutic capabilities, for example, comprehensive control of Tau protein phosphorylation, inflammatory response, redox reaction and improvement of mitochondrial.
Research undertaken so far to transport drugs via nanocarriers across the BBB, while considerable, has certainly not enough and has not met expectations [21], so there is an urgent need to re-examine current research ideas and methods to improve or find new ways.
Conclusions and Future Prospects
Since the discovery of central nervous system degenerative diseases, its diagnosis and treatment have become a huge medical challenge. In addition to research in the biological field, the combination with other fields, especially the integration of nanotechnology, has showed us a promising way. Current research indicates that the surface of NPs can be modified by covalent and non-covalent connection, giving it additional stability and drug affinity. In the future, we hope to see nanotechnology-based drug delivery systems can effectively treat more Invasive neurological disease or drug resistant disease. In addition, the functionalization of NPs and the combination of drugs indicate more possibilities, not only for drug system construction, but also for gene and protein delivery. Some nanoparticles also have special magnetic and optical properties, etc., giving them magnetic targeting and photothermal effects. With the advancement of nanotechnology, NPs will appear more in biomedical application.
Acknowledgement
This work was supported by the National Natural Science Foundation of China (grant number: 81301313; 81600159), the Natural Science Foundation of Jiangsu Province (grant number: BK20131015; BK20141015), Jiangsu Provincial College Students’ Practical Innovation Training Program (grant number: 201410312015Z), and Nanjing Developing Project of Medical Science (grant number: YKK13174).
References
- Martin P, Martin K, Maëlenn G, Adelina CH, Maria Karagiannidou (2016) World Alzheimer Report 2016: Improving healthcare for people living with dementia [OL]. Alzheimer’s Disease International (ADI).
- Sahni JK, Doggui S, Ali J, Baboota S, Dao L, et al. (2011) Neurotherapeutic applications of nanoparticles in Alzheimer’s disease. J Control Release 125(2): 208-231.
- Knopman DS (2016) Alzheimer disease: preclinical Alzheimer disease - the new frontier. Nat Rev Neurol 12(11): 620-621.
- Reiman EM (2016) Alzheimer’s disease: attack on amyloid-β protein. Nature 537(7618): 36-37.
- Azhdarzadeh M, Noroozian M, Aghaverdi H, Akbari SM, Baum L, et al. (2013) Serum multivalent cationic pattern: speculation on the efficient approach for detection of Alzheimer’s disease. Sci Rep 3: 2782.
- Cheng KK, Chan PS, Fan S, Kwan SM, Yeung KL, et al. (2015) Curcuminconjugated magnetic nanoparticles for detecting amyloid plaques in Alzheimer’s disease mice using magnetic resonance imaging (MRI). Biomaterials 44(44): 155-172.
- Panahi Y, Mohammadhosseini M, Abadi AJ, Akbarzadeh A, Mellatyar H (2016) An Update on Biomedical Application of Nanotechnology for Alzheimer’s Disease Diagnosis and Therapy. Drug Research 66(11): 580-586.
- Lai L, Zhao C, Li X, Xiaoli Liu, Hui Jiang, et al. (2016) Fluorescent gold nanoclusters for in vivo target imaging of Alzheimer’s disease. Rsc Advances 6(36): 30081-30088.
- Panahi Y, Mohammadhosseini M, Abadi AJ, Akbarzadeh A, Mellatyar H (2016) An Update on Biomedical Application of Nanotechnology for Alzheimer’s Disease Diagnosis and Therapy. Drug Res 66(11): 580- 586.
- Yang ZZ, Zhang YQ, Wang ZZ, Wu K, Lou JN, et al. (2013) Enhanced brain distribution and pharmacodynamics of rivastigmine by liposomes following intranasal administration. Int J Pharm 452(1-2): 344-354.
- Ismail MF, Elmeshad AN, Salem N (2013) Potential therapeutic effect of nanobased formulation of rivastigmine on rat model of Alzheimer’s disease. Int J Nanomed 8(1): 393-406.
- Mutlu NB, Değim Z, Yilmaz Ş, Eşsiz D, Nacar A (2011) New perspective for the treatment of Alzheimer diseases: liposomal rivastigmine formulations. Drug Dev Ind Pharm 37(7): 775-789.
- Fazil M, Md S, Haque S, Kumar M, Baboota S, et al. (2012) Development and evaluation of rivastigmine loaded chitosan nanoparticles for brain targeting. Eur J Pharm Sci 47(1): 6-15.
- Sancini G, Gregori M, Salvati E, Ilaria Cambianica, Francesca Re, et al. (2013) Functionalization with TAT-peptide enhances blood-brain barrier crossing in vitro of nanoliposomes carrying a curcuminderivative to bind amyloid-peptide. J Nanomed Nanotechnol 4: 1-8.
- Mourtas S, Lazar AN, Markoutsa E, Duyckaerts C, Antimisiaris SG, et al. (2014) Multifunctional nanoliposomes with curcumin-lipid derivative and brain targeting functionality with potential applications for Alzheimer disease. Eur J Med Chem 80: 175-183.
- Lazar AN, Mourtas S, Youssef I, Parizot C, Dauphin A, et al. (2013) Curcumin-conjugated nanoliposomes with high affinity for Ab deposits: possible applications to Alzheimer disease. Nanomedicine 9(5): 712-721.
- Kakkar V, Muppu SK, Chopra K, Kaur IP (2013) Curcumin loaded solid lipid nanoparticles: an efficient formulation approach for cerebral ischemic reperfusion injury in rats. Eur J Pharm Biopharm 85(3 Pt A): 339-345.
- Roberts BR, Ryan TM, Bush AI, Masters CL, Duce JA (2012) The role of metallobiology and amyloid-β peptides in Alzheimer’s disease. Journal of Neurochemistry 120(Suppl1): 149-166.
- Bush AI (2013) The metal theory of Alzheimer’s disease. J Alzheimers Dis 33(Suppl 1): S277-S281.
- Anna Salvati, Andrzej S Pitek, Marco P Monopoli, Kanlaya Prapainop, Francesca Baldelli Bombelli, et al. (2013) Transferrin-functionalized nanoparticles lose their targeting capabilities when a biomolecule corona adsorbs on the surface. Nat Nanotechnol 8(2): 137-143.
- Ansari SA, Satar R, Perveen A, Ashraf GM (2016) Current opinion in Alzheimer’s disease therapy by nanotechnology-based approaches. Curr Opin Psychiatry 30(2): 128-135.






























