Green Synthesis of Iron Nanoparticles for Biomedical Applications
Mandeep Kaur and Dimple Sethi Chopra*
Department of Pharmaceutical Sciences and Drug Research, Punjabi University Patiala, India
Submission: August 15, 2018; Published: October 25, 2018
*Corresponding author: Dimple Sethi Chopra, Department of Pharmaceutical Sciences and Drug Research, Punjabi University Patiala, India.
How to cite this article: Mandeep K, Dimple S C. Green Synthesis of Iron Nanoparticles for Biomedical Applications. Glob J Nano. 2018; 4(4): 555643. DOI: 10.19080/GJN.2018.04.555643
Abstract
The use of plant extract is found to be a fascinating approach for non-toxic and efficient synthesis of iron nanoparticles. The components present in the extract replace the toxic reducing agents that are involved in the chemical synthesis of iron nanoparticles. This review article focuses on various plant extract used as reducing and capping agent for biosynthesis of iron nanoparticles. These iron nanoparticles were found to be hydrophilic and biocompatible. Iron nanoparticles prepared by green synthesis are thus highly suitable for biomedical applications. The plant extract used for preparation of iron nanoparticles were found to contain secondary metabolites like polyphenols, flavanoids. The polyphenols reduced Fe2+/Fe3+ to nanozerovalent iron or iron nanoparticles. Iron nanoparticles possess high surface to volume ratio along with significant magnetic properties in comparison to iron/iron nanoparticles. The nature and amount of second metabolites present in an extract varies from one plant to another. This in turn effects the composition of iron nanoparticles. Thus, iron nanoparticles can be designed by choosing appropriate plant extract (Figure 1).
Keywords: Iron nanoparticles; Green nanotechnology; Biocompatible; Drug delivery; Cell labeling
Abbrevations: XRD: X-Ray Diffraction; SEM: Scanning Electron Microscopy; TEM: Transmission Electron Microscopy; UV: Visible Spectroscopy; FTIR: Fourier Transform Infrared; VSM: Vibrating Sample Magnetometry; EDX: Energy Dispersive X-ray Spectroscopy; HNQ: Hydroxy Napthoquinone; TGA: Thermal Gravimetric Analysis; AFM: Atomic Force Microscopy; SPR: Surface Plasmon Resonance; FESEM: Field Emission Scanning Electron Microscopy, C: Catechin; GCg: Gallocatechin Gallate; EC: Epicatechin; EGCg- Epigallocatechin Gallate; EGC- Epigallocatechin; MG: Malachite Green; HR-TEM: High-Resolution Transmission Electron Microscopy; FE-SEM: Field Emission Scanning Electron Microscopy; EDX: Energy Dispersive X-Ray; NZVI: Nano Zero Valent Iron; VSM: Vibrating Sample Magnetometer; CGFE: Courpita Guianensis
Introduction
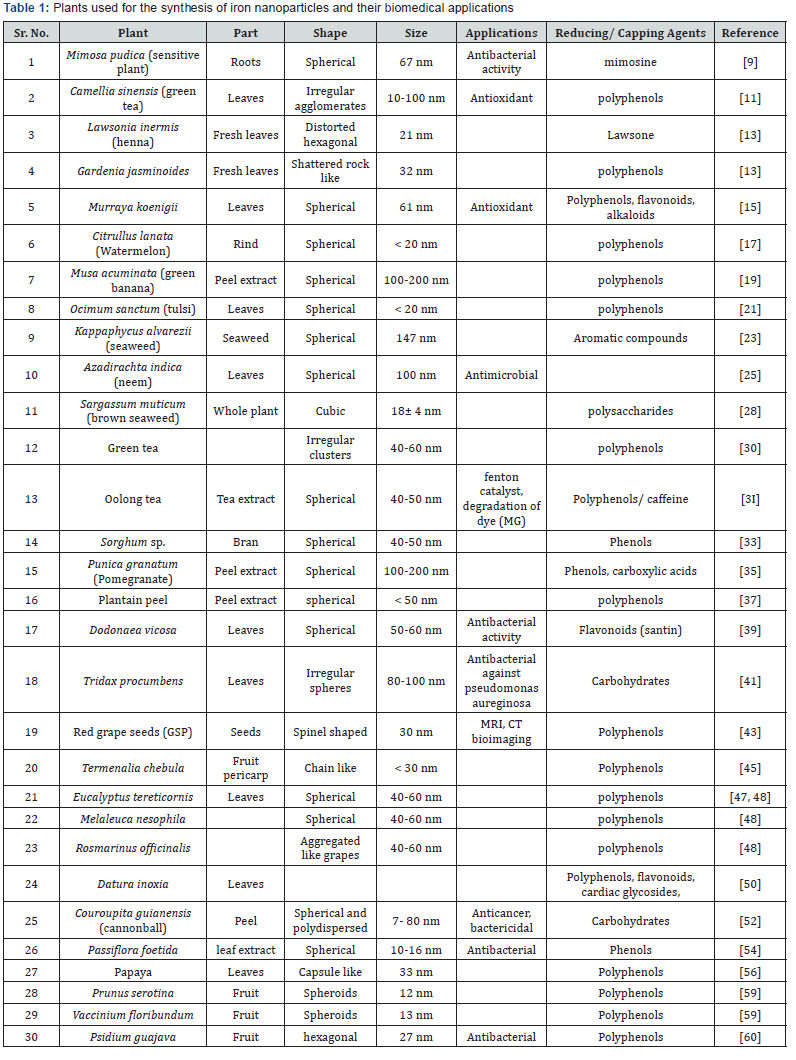
Nanoparticle are submicron moieties with diameters ranging from 1-100 nm made of inorganic or organic materials having novel properties as compared to the bulk materials [1]. Nanoparticles are used in biomedical applications as they offer many advantages to larger particles such as increased surface to volume ratio and increased magnetic properties such as cell labelling/cell separation, detoxification of body fluid, drug delivery, tissue repair, hyperthermia, magnetofection [2], blood clot lysis [3], blood purification [4] and leukemia [5], etc. Plant extract-mediated synthesis of nanomaterials is one of the more stable and suitable alternatives in comparison with those produced by physical, chemical and microbial methods. This method was adopted to develop a facile and rapid synthesis of iron oxide nanoparticles using a non-toxic, renewable plants [6,7] (Table 1).
Mimosa pudica (Sensitive plant)
The Mimosa pudica extract was used to prepare iron oxide nanoparticles through green route of synthesis. The plant extract contains mimosine, mimosinic acid, mimosinamine, tyrosine 3, 4- dihydroxypiridine [8]. The reaction was carried out by mixing iron precursor (aqueous ferrous sulphate solution and fresh root extract (reducing and stabilising agent) of M. pudica. This led to the formation of non-toxic and stable iron nanoconstructs. The resultant iron nanoparticles were characterised by XRD (X-Ray diffraction), SEM (Scanning electron microscopy), TEM (Transmission electron microscopy), UV-Visible spectroscopy, FTIR (Fourier transform infrared) analysis, VSM (vibrating sample magnetometry). The change in the colour of solution from yellow to brown that signifies the formation of iron nanoparticles was investigated through UV-visible spectra. Crystallinity of these particles was determined by XRD pattern. FTIR results revealed that mimosine was responsible for formation of the magnetic nanoparticles by reducing ferrous sulphate to nanosized iron. SEM and TEM images described the morphological properties and exact size of the iron nanoparticles respectively. VSM measured the magnetization value of the magnetic nanoparticles [9].
Camellia senesis (Green tea)
The major chemical constituents of plant are steroids, carotenoids, terpenoids, flavonoids, tannins, alkaloids and glycosides which act as important bioactive molecules [10]. The sustainable magnetic iron nanoparticles can be developed by the green tea leaf extract. The leaf extract was allowed to react with the aqueous ferric chloride solution which results in the formation of highly stable and cost- effective magnetic nanoparticles. It was the polyphenolic compounds present in the extract that cause the reduction or iron salts to iron nanoconstructs. EDX results exposed the elemental composition of the iron nanoparticle solution. SEM analysis was carried out to determine the structure and the appearance of the FeNPs and the nano size was analysed through TEM images [11].
Lawsonia inermis and Gardenia jasminoides
2-Hydroxy-1, 4-napthoquinone (HNQ; Lawsone) is the principle phytoconstituent present in the leaves of Henna. Other related compounds present in the leaves are: 1, 4dihydroxynaphthalene. 1, 4- naphthoquinone, 1,2-dihydroxy-glucoyloxynaphthalene and 2-hydroxy-1,4- diglucosyloxynaphthalene. Flavonoids (luteolins, apigenin, and their glycosides). Coumarins (esculetin, fraxetin, scopletin). Steroids (β- sitosterol). The leaves of Lawsonia inermis also reported to contain soluble matter tannin, gallic acid, glucose, mannitol, fat, resin and mucilage [12]. The ethanol extract of Gardenia jasminoides consists of number of phytoconstituents such as saponin, tannin, flavonoid, alkaloid, glycosides and phenols. The powder of the dried leaves of the Lawsonia inermis and Gardenia jasminodes used to prepare non-toxic, eco-friendly and cost- effective iron nanoparticles. These nanoparticles were characterised by thermal gravimetric analysis (TGA), transmission electron microscopy (TEM), scanning electron microscopy (SEM), Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), and atomic force microscopy (AFM).
The weight losses of iron nanoparticles synthesized from both plant extract was observed through TGA analysis. FT-IR was carried to determine the biomolecule that is responsible for the reduction reaction during the formation of iron nanoparticles. The Henna leaves extract consists of Lawson (2-hydroxy-1,4-naphthaquinone) as its main constituents that contain p-benzoquinone unit, benzene unit and phenolic group. The Feo Lawson complex results in the formation of the iron nanoparticles. The nature of the iron nanoparticles and their phase composition was identified by X-ray powder diffractometer. The exact size of the iron nanoparticles synthesized from both plants was determined by TEM analysis. SEM indicates the morphology or appearance of iron nanoparticles. The elemental composition was depicted through Energy dispersive X-ray Spectroscopy (EDX) analysis. These iron nanoparticles found to have antimicrobial activity. They are effective against all the bacterial strains [13].
Murraya koenigii
The most important bioactive constituents of present within leaf extract of Murraya koenigii are coumarins, carbazole alkaloids and flavonoids. It was reported that its hydro-methanolic extract consists of high percentage of phenolic content. The leaves of Murrya koenigii also contain proteins, fiber, carbohydrates, minerals, nicotinic acid, carotene, Vitamin C, Vitamin A, calcium and oxalic acid. Other related components are crystalline glycosides, carbazole alkaloids, koenigin, girinimbin, iso-mahanimbin, koenine, koenidine and koenimbine. Triterpenoid alkaloids cyclomahanimbine, tetrahydromahanimbine are also present in the leaves of M. koenigii. Murrayastine, murrayaline, pyrayafoline carbazole alkaloids and many other chemicals have been isolated from Murrya koenigii leaves [14]. The non-toxic, cost- effective iron nanoparticles were fabricated by adding ferrous sulphate solution to the Murraya koenigii broth. It was noticed that sudden change in colour from pale yellow to dark brown takes place indicating the formation of the FeNPs due to the bioreduction of ferrous ions by the polyphenols, flavonoids and alkaloids indicating the formation of iron nanoparticle and responsible of their stability which was monitored by UV-Visible spectroscopy and FTIR analysis. TEM study revealed the mean size and amorphous nature of the iron nanoparticles. Porous and agglomerated sponge like appearance was confirmed by the SEM images [15].
Citrullus lanatus (Watermelon)
The watermelon (Citrullus lanatus) rind extract have the reducing and stabilising potential to produce iron nanoconstruct with the aid of iron salts that act as precursor for the synthesis reaction. This addition changes the light brown colour solution into dark brown colour and hence colloids no iron oxide nanoparticles. The peel extract of watermelon rind considered as good sources of carotenoids (β-carotene), flavonoids, phytosterol (β-sitosterol), rutin and other bioactive compounds bearing functional groups like carboxylic and hydroxyl groups that are involved in the construction of zero valent iron nanoparticles and was verified by FTIR analysis [16]. Spherical shape, other morphological features and particle size distribution was carried out through SEM and TEM results. Face centered cubic structure was identified by XRD pattern. The resultant iron nanoparticles were found to have excellent catalytic activity in the synthesis of organic molecules [17].
Musa acuminate (Green banana)
Banana peel extract found to be rich source of phenolic compounds like naringenin, quercetin, chlorogenic acid. Other related compounds are minerals, vitamins, flavonoids, carbohydrates, etc. [18]. The filtrate of banana peel extract was employed to synthesize iron nano constructs that involve the use of no harmful or hazardous chemicals. Musa acuminate contains carotenoids bearing polyphenol moiety interacts with the iron precursor (ferrous sulphate) through hydroxyl groups when mixed with each other and reduces them to iron nanoparticles on steady and continuous stirring. This reduction results in the change in the brown colour solution to black, revealed from the intensity of the SPR (surface Plasmon resonance) peak through UV-spectroscopy indicating the formation of iron nanoparticles. Transmission electron microscopy (TEM) was done to investigate the size of formed iron nanoparticles. Crystallinity and Spherical shape were examined from XRD (X-Ray diffraction) pattern and SEM (Scanning electron microscopy) images respectively. Fourier transform infrared (FTIR) analysis proved the presence of the moiety that participates in the formation of iron nanoparticles [19].
Ocimum sanctum (Tulsi)
Fresh leaves and stem of Ocimum sanctum are great source of phenolic compounds such as circimaritin, cirsilineol rosameric acid, isothymusin and apigenin. It contains significant amount of eugenol [20]. The rapid biological synthesis of iron oxide nanoparticles using leaf extract of Ocimum sanctum provides an environment friendly, simple and efficient route. On the basis of the results of Fourier transform infrared spectroscopy, the presence of phenolic compounds and proteins were believed to be responsible for the formation and stabilization of synthesized iron oxide nanoparticles. Through XRD the average size was estimated to be 47 nm. SEM micrographs at different magnification levels showed that the synthesized iron oxide nanoparticles were in the form of irregular shapes and aggregated form with rough surfaces. The appearance of the nanoparticles found to be a porous and spongy. TEM analysis confirms that the size of the iron oxide nanoparticles was estimated to be less than 20 nm. Thus, the green synthesis using Ocimum sanctum leaf extracts can be cost-effective method for the synthesis of iron oxide nanoparticles [21].
Kappaphycus alvarezii (Seaweed)
Novel and bio-inspired magnetic nanoparticles were synthesized using Kappaphycus alvarezii seaweeds that are nontoxic, cheap and environment friendly. Number of chemical constituents such as alkaloids, flavonoids, steroids, glycosides, tannins, terpenes was found to present within the extract. It generally includes high concentration phenolic compounds (Chlorogenic acid, Sinapic acid, Gallic acid, Phloroglucinol, Vanillic acid, Cinnamic acid, Catechol, Ferulic acid) [22]. These seaweeds were used as stabilisers and reducing agent in the synthesis of iron nanoparticles. The Fe3o4 NPs were characterized by using transmission electron microscopy (TEM), X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR) and Ultraviolet- visible (UV-Visible) spectroscopy. On the addition of NaOH and with continuous stirring the solution, the change in the colour of the solution of ferric chloride and seaweed from light brown to black was observed that indicates that iron nanoparticles are formed due to the reduction cause by the biomolecules present within the extract. XRD studies revealed the high degree of crystalline and phase purity. FTIR analysis proved that particles are reduced and in solution by the capping agent that is likely to be the aromatic compounds. The synthesized nanoparticles are characterized by X-ray diffractometer to analyses the particle size of the synthesized iron nanoparticles. Morphological and size distribution was done with transmission electron microscope (TEM) images that found to be spherical and agglomerated [23].
Adazirachta indica (Neem)
The leaf extract of neem (Azadirachta indica) consists of various bioactive molecules such as glycosides, alkaloids, saponins, flavonoids, carbohydrates, tannins and phenolic compounds that play a major role in the development of iron nanoparticles [24]. On mixing the ferric chloride with plant extract in ratio 1:1, the reduction of Fe3+ to iron nanoparticles take place which results in change in colour (light green to black) and change in pH (high to low acidic conditions) immediately. These changes found to be the signal for the formation of the Nanoparticles present in zero valent oxidation state with no stability issue in the solution. The produced nanoparticles were characterized by various analysis techniques such as X-Ray Diffraction (XRD), UV-Visible Spectroscopy (UV-Visible) and TEM (Transmission Electron Microscopy). Morphology and appearance of the produced iron nanoparticles was determined by XRD analysis. The SEM images revealed the actual diameter of the nanoparticles. This synthesis proved to be eco-friendly, costeffective and have a significant role in biomedical applications [25].
Sargassum muticum (Brown seaweed)
Seaweed are a rich source of several phytochemicals including minerals, lipids, vitamins, and other bioactive constituents like polysaccharides, polyphenols and proteins [26]. Catechin, and quercetin, and phlorotannins, the phenolic compounds found to be present in brown seaweed [27]. The synthesis of iron nanoparticles from seaweeds (Sargassum muticum) is environment friendly, fast, single step, non- toxic, reliable method. The water extract of brown seaweed (BS, Sargassum muticum) contains sulphated polysaccharides which cause the reduction and act as stabilizer for iron nanoparticles and was confirmed by FTIR spectra. The reduction results in the colour change from yellow to brown that confirms the synthesis of iron nanoparticles. The phase analysis and the crystallinity of particles were identified by XRD (X-Ray diffraction). Morphology was determined by TEM analysis. It was clear from FESEM (field emission scanning electron microscopy) images that synthesized iron nanoparticles were cubic in shape. The magnetic behaviour of iron NPS were determined by VSM (vibrating sample magnetometry) [28].
Green tea
The contents of theanine, theobromine, caffeine, catechin (C), and gallocatechin gallate (GCg) were significantly decreased, whereas those of epicatechin (EC), epigallocatechin gallate (EGCg) and epigallocatechin (EGC) [29]. Green tea extract has been used as the reducing agent for the eco-friendly synthesis of iron nanoparticles or iron rods due to the presence of high amount of polyphenols and other organic groups in it. The synthesized NPs were then used as catalyst for the oxidation and reduction of organic solvents. When the green tea extract powder was mixed with ferric chloride (iron precursor), the colour of solution changes from yellow to dark greenish black colour indicating the formation of nanorods (at ambient temperature). The washing was done to remove excess precursors or polyphenols followed by freeze drying. The crystallinity of the iron nanoparticles was examined by XRD. FTIR analysis confirmed the biomolecules that were responsible for the reduction reaction. Morphology was determined through scanning electron microscopy (SEM) [30].
Olong tea
The Fe3O4-NPs were prepared using ferric sulphate (FeSO4.7H2O) as iron precursor and olong tea extract as reducing agent and stabilizer. These synthesized FeNPs were characterised by X-Ray diffraction, SEM (scanning electron microscopy), UV-Visible spectroscopy, EDS, FTIR analysis. The structural and properties of the Fe3O4-NPs were investigated by X-ray diffraction. UV- V sible spectra confirmed the formation of iron nanoparticles. EDS was carried out to deter mine the elemental composition of the olong tea extract and iron nanoparticles developed from the extract. From the FTIR result, the biomolecules polyphenols/caffeine in oolong tea extract were found to play a relevant role in reduction of iron ions into iron nanoparticles. The size and final structure of FeNPs will depend upon the concentration of these biomolecules. The morphology and structure of the Fe3O4-NPs were further investigated by scanning electron microscopy (SEM). The iron NPs developed from olong tea extract found to act as fenton catalyst for oxidizing the dyes (cationic and anionic) and for reductive degradation of malachite green (MG) dye [31].
Sorghum bran
Sorghum known as functional food because of its richness tannins, phenolic acids, phytosterols, anthocyanins and policosanols [32]. Iron nanoparticles were synthesized from aqueous sorghum extract and ferric chloride. During the synthesis of nZVI both the precursor and the reducing agent were mixed in 1:1 proportion. The change in the colour from pale yellow to brown due to the reduction of ferric ions by the phenols indicating the formation of iron nanoparticles. UV– visible absorption is used to investigate SPR (Surface Plasmon Resonance). Characteristic surface plasmon absorption band was observed within the visible range. The characterization size and morphology of nZVI was performed using HR-TEM (high-resolution transmission electron Microscopy) and FE-SEM (field emission scanning electron microscopy) that determined the exact size and amorphous nature of the iron nanoparticles. EDX (Energy dispersive X-Ray) spectra of synthesized iron nanoparticles confirmed the presence of iron, phosphorous and oxygen [33].
Punica granatum (Pomegranate)
Ellagic tannins, ellagic acid and gallic acid are the dominant phenolic compounds found in peel extract of P. granatum. Flavonoids, tannins and alkaloids were also reported including gallocatechins, cyaniding, delphinidin, and sitosterol [34]. The mixture peel extract of pomegranate fruit was employed to develop eco-friendly, stable and cost-effective magnetic nanoparticle through green chemistry approach of chemistry. SEM, XRD, UV-Vis spectroscopy, FTIR analysis to determine the characteristics of synthesized iron nanoparticles. Morphology and the distribution of these FeNPs was characterized using SEM. It can be clearly seen that the FeNP synthesized using pomegranate peel extract are polydispersed and has a diameter of 15nm-34nm. The cystallinity and phase purity was determined by X-Ray diffraction study. UV-Visible spectroscopy results revealed the complete formation of iron nanoparticles. FTIR pattern proved that phenols and carboxylic acids are the key role for the reduction reaction for the synthesis of the iron nanoparticles [35].
Plantain
Plantain peel extract consists of substantial amounts of polyphenols such as quercetin, isoquercetin, catechin, astragalin, p-coumaric acid, rutin, carvacol, protocatechuic acid [36]. Magnetic nanoparticles can be synthesized by the use of plantain peel extract. FeCl3.6H2O and Sodium acetate was dissolved in the freshly prepared plantain peel extract solution. Black colour solution signifies the formation on magnetic nanoparticles and was demonstrated by UV-Visible spectroscopy. TEM, EDX, XRDdiffraction revealed the various characteristics of the resultant iron nanoparticles. Particles found to be spherical in shape, monodispersed and diameter less than 50nm. FTIR analysis demonstrated that carbohydrates, fibres, lipids, proteins, polyphenols contribute in the process of formation of MNPs. Polyphenols also act as reducing and capping agents. VSM (Vibrating Sample Magnetometer) used to study the Magnetic behavior of the resultant iron nanoparticles. They found to have excellent magnetic properties [37].
Dodonaea viscose
The leaf extract of D. viscose was known to contain alkaloids, tannins, carbohydrates, flavonoids, phenols, gums and mucilage, saponins, fixed oils and fats [38]. Fresh D. viscose leaves extract was used to develop biocompatible and non-toxic ZVI nanoparticles which have potential for antibacterial activity. The iron nanoparticles were synthesized by addition of leaf extract to FeCl3 solution. The mixture results in the sudden colour change (yellow to greenish black) at room temperature which gives the indication of formation of iron nanoparticles. The complete reduction of iron chloride into iron nanoparticles depends upon the quantity of the leaf extract of the plant. The Nano Zero Valent Iron (NZVI) were characterized by HRTEM, FTIR, AFM, XRD and UV- Visible spectroscopy. HRTEM (High Resolution Transmission Electron Microscopy) analysis was conducted that revealed the spherical nature and confirms that the particles lie within nanoscale range of the iron nanoparticles. FT-IR analysis was performed to establish the possible active biomolecules that are accountable for the capping and as stabilising agent by preventing further oxidation. The santin contains polyhydroxy groups that may as reduce agents for the development of the iron nanoparticles from the metal ions. FT-IR spectrum for the iron suspension was carried out to depict the functional groups involved in the reduction reaction [39].
Tridax procumbens
The phytoconstituents such as flavonoids (mainly epicatechin and kaemferol), alkaloids (mainly akuammidine), benzoic acid derivatives (mainly ferulic acid), carotenoids (mainly lutein), lignans (mainly galgravin), phytosterols (mainly stigmasterol) and hydroxycinnamates are present in the leaf extract of Tridax procumbens [40]. It also contains high content of carbohydrates that has potential to reduce iron salts to its nano forms. The resultant iron nanoparticles were examined by different techniques. Powder X-ray Diffraction (XRD) and Transmission Electron Microscope (TEM) analysis was done to check the crystallinity and size of the synthesized iron nanoparticles. Morphology was determined by SEM images. UVVisible spectra have shown the absorption lies within the visible range indicating the development of iron nanoparticles. On the basis of FTIR analysis it was obtained that aldehyde groups of carbohydrate are the candidates for the reduction and capping/ stabilising activity of iron nanoparticles. The iron nano forms synthesized from Tridax procumbens found to be effective against Pseudomonas aeruginosa [41].
Red grape seeds
The seeds of red grape possess alkaloids, glycosides, phenols, saponins, flavonoids, tannins, steroids, monoterpenoids, lignins, and cardiac glycosides. They are rich source of phenolics and anthocyanins, the functional components [42]. Mixture of single iron precursor (ferric chloride) and grape seed proanthocyanidin (reducing agent and stabilizer) give rise to non-toxic, reliable and economical iron nanoparticles which was indicated by change in the yellow coloured solution into greenish-brown. Structural and physicochemical characterization proved to be crystalline, with inverse spinal morphology and particle size distribution through XRD, DLS and TEM analysis. FTIR analysis confirmed the involvement of polyphenol in the formation of iron nanoparticles. Magnetization power of GSP synthesized was measured by VSM (vibrating sample magnetometry). Grape seeds have got potent antioxidant property [43]
Terminelia chebula
Terminalia chebula extract consists of various constituents’ flavonoids, sterols, tannins, amino acids, resins, fructose, and fixed oils along with anthraquinones, 4,2,4 chebulyldglucopyranose, terpinenes and terpinenols. T. chebula extract contains tannin pyrogallol (hydrolysable) type, hydrolysable tannins (chebulic acid, gallic acid, punicalagin, chebulanin, neochebulinic, ellagic acid, corilagin, chebulegic acid, chebulinic acid, 1,2,3,4,6penta-Ogalloyl-ß-D-glucose, 1,6,-di-O-galloyl- D-glucose, casuarinin, 3,4,6-tri-O-galloyl-D-glucose and terchebulin) [44]. Terminelia chebula fruit extract, rich in polyphenols (in the form of hydrolysable tannins) was used as green reducing and stabilising/capping agent to form the iron nanoparticles. The immediate colour change from light yellow to dark brown due to the reduction of iron salts was noticed that signifies the formation of iron nanoparticles and was monitored by UV-Visible spectroscopy. Synthesized NPs were further confirmed with different characterization techniques such as XRD, SEM, TEM, EDS, FTIR analysis. X-ray Diffraction (XRD) analysis revealed that amorphous iron NPs were developed. SEM (Scanning Electron Microscopy) and Transmission Electron Microscopy (TEM) analysis helped to determine the size and morphology of the resultant iron nanoparticles. Presence of both Iron and oxygen found to be indicative of synthesis of iron nanoparticles through Elemental analysis of the extract [45].
Eucalyptus tereticornis, Melaleuca nesophila and Rosmarinus officinalis
The leaf extracts of these plants were used as reducing agent for the production of iron nanomaterials through greener route of synthesis. Polyphenol, the major constituent of leaves of these plants interact with iron ions to form black coloured complexes. UV-vis spectroscopy, XRD, SEM, TEM and FTIR analysis was carried for the characterization of the resultant iron nanoparticles. XRD patterns described their crystallinity and revealed ferric ion located in polyphenolic chelated globular nanoparticles. SEM and TEM analysis were performed to determine the diameter and structural properties of the green synthesized iron nanoparticles. The images confirmed the spherical shape of iron nanoparticles of eucalyptus and melaleuca and aggregated forms in case of rosamarinus. From the FTIR results it can be confirmed that the structure of polyphenols plays the key role in the formation of iron-polyphenol iron nanoparticles [46,47].
Datu Eucalyptus tereticornis, Melaleuca nesophila and Rosmarinus officinalis
The phytoconstituents present in leaf extract of D. inoxia are scopolamine, atropine, essential oils, flavonoids, saponins, phenols, and cardiacs glycosides [48]. The Ferric chloride tetra hydrate and ferrous chloride hexahydrate results in the generation of non-toxic, eco-friendly and stable Nano colloids when added to the aqueous leaf extract of DatuEucalyptus tereticornis, Melaleuca nesophila and Rosmarinus officinalis at room temperature. The presence of various bioactive molecules, cardiac glycosides, flavonoids and phenolic groups in the Datura inoxia leaf extracts that act as reductant and capping/stabilizing agents for the biosynthetic iron nanoparticles formation and this was investigated through FTIR. The change in color from yellow to greenish brown during the formation of iron nanoparticles was shown by UV-visible absorption spectra. HR-TEM image displayed the synthesized Fe3O4-NPs were mostly spherical in shape. Antimicrobial activity of these biosynthesized iron nanoparticles was comparably more than chemically synthesized iron nanoparticles [49].
Couropita guianensis (cannonball)
The extract composed of different active constituents such as steroids, glycosides, carbohydrates, couroupitone A, couroupitone B, isatin, triterpinoids, eugenol, linoleic acid, nerol, tryptanthrin linalool, phenolic resin substances. Rutin, Quercetin, Farmaricetin, Kaempherol, Ursolic acid, Lutolin, Saponines derivatives such as Hopanes [50]. Seaweed Courpita guianensis was used as green reducing and stabilishing agent to develop the iron nanoparticles through biosynthetic approach. The fruit extract of C. guianensis (CGFE) was added to iron solution. The resultant NPs were investigated with different characterization techniques such as XRD, DLS, FTIR, HRTEM, VSM, UV–visible spectroscopy, FT-IR, XPS, DLS and zeta potential analysis. Absorbance in visible region due to change in colour indicates the formation of iron nanoparticles and was monitored by UV-Visible spectra. Spherical and polydispersed nano size particles were investigated through TEM analysis. Crystallinity of magnetic NPs was depicted by X-Ray results. From FTIR results clearly proved the role of hydroxyl groups that intermingle with the iron precursor (ferric chloride) to produce iron Nano colloids. The super magnetic behavior of the resultant FeNPs was examined through VSM. They exhibit antibacterial activity. Cytotoxic effect was observed when human hepatocellular carcinoma cells (HepG2) were treated with resultant iron nanoparticles [51].
Passiflora foeitida
Passiflora foeitida leaf extract consists of alkaloids, flavonoids, quinines, resins, saponins, tannins, phenolic compounds, fixed oils, fats, proteins and carboxylic acids [52]. It is utilized for the fabrication of iron nanoparticles due to their reduction and capping/stabilising potential. The extract is composed of tannins and phenolic compounds (hydroxyl groups) and the role of these biomolecules on the formation and increased stability of nanoparticles was analyzed using the FTIR (fourier transform infrared) spectra. EDX (Energy dispersive x-ray) spectroscopy was performed to determine the elemental composition of resultant iron nanoparticles. TEM (Transmission electron microscopy) and SEM (Scanning electron microscopy) results revealed the exact size, spherical structure and crystallinity of the iron nanoparticles. These nanoparticles were observed for their antibacterial effect [53].
Caricaya papaya
Alkaloids, flavonoids, tannins, saponins, tannins, glycosides and steroids were found to present in the leaf extract of this plant [54]. Due to presence of these functional constituents the sustainable green chemistry approach was applied to generate magnetic nanoparticles using Caricaya papaya leaves (reducing and stabilising agent) and ferric chloride salts (precursor) was investigated by high throughput techniques such as XRD, SEM, TEM, UV-visible spectroscopy and FTIR. UV–visible spectrum of synthesized Fe3O4 NPs produces a strong absorbance in the visible region confirmed that the particles were stable and well dispersed in the solution. FTIR result also clearly depicts that carboxylate ions are in strong coordination with iron metals and reduces iron salts to iron nanoparticles. SEM and TEM images revealed the irregular shape and exact size of these nanoparticles. Crystalline nature was confirmed by XRD study. EDS was done to investigate the elemental composition of synthesized iron Nano constructs [55].
Prunus serotina and Vaccinium floribundum
The most abundant phenolic compounds present in Prunus serotina were cyanidin-3-O-rutinoside, chlorogenic acid, procyanidin B, hyperoside and quercetin, malonilglucoside. The presence of anthocyanins was also analysed [56]. Phenolic compounds that have been characterized in Vaccinium floribundum (berries) include gallic and ellagic acids, ellagitannins, flavan-3-ols, proanthocyanidins, flavonol glycosides, hydroxycinnamic acids and anthocyanins, and they are usually found in very high concentrations [57]. The fruit extract of these plants was utilized as reducing and stabilizing agent to prepare magnetic nanostructures from ferric salts. On mixing the fruit extract to the ferric chloride solution the black coloured precipitates were formed. The shift of the wavelength to the visible range signifies the formation of iron nanoparticles that was supported by UV-visible spectroscopy. Carboxylic and hydroxyl groups of polyphenols were confirmed to be the role model in reducing ferric ions and act as stabilizing agents for the synthesized iron nanoparticles through FTIR results. The number or the amount of iron nanoparticles formed were directly proportional to the concentration of the ferric chloride solution. The DLS analysis and TEM images describe the spherical shape and size distribution of the prepared iron nanomaterials. Morphological features were studied through SEM analysis. The synthesized iron nanoparticles from prunus serotina and vaccinium floribundum considered to be economical and non- toxic [58].
Psidium guajava (Guava)
Alkaloids, anthocyanins, carotenoids, fatty acids, lectins, essential oils, phenols, saponins, triterpenes, tannins, and vitamin C12 found to present in the leaf extract of this plant [59]. Iron nanoparticles can be fabricated by the treatment of aqueous extract of leaves of Psidium guajava with the aqueous FeCl3 solution. The yellow coloured mixture changes to black coloured indicating the generation of iron nanoparticles which was confirmed by UV-Visible spectroscopic study. Aqueous Fe ions were reduced by aqueous extract of the plant to develop stable iron nanoparticles in water. FTIR analysis revealed that the plant leaf extract consists of tannins, flavonoids, spanin, steroids, glycosides, polyphenols, carbohydrates that found to be responsible for the reduction of Fe3+. SEM-EDX was performed to determine the crystallinity and morphology. The images shown that resultant iron nanoparticles were agglomerated and irregular in shape. The particles found to be cubic through XRD analysis. The iron nanoparticles exhibit antibacterial activity against Bacillus cereus (gram positive), Escherichia coli (gram negative), Klebsiella pneumonia, and Staphylococcus aureus [60].
Conclusions and Future Prospect
Since FeNPs have been synthesized using chemical methods like Co-precipitation, Thermal decomposition these methods result in the significant oxidation of iron and special agents are required to improve shelf life of iron NPs. The chemical methods thus involve reagents and stabilizing agents which might be toxic when used for biomedical applications like magnetic resonance imaging (MRI), cell labelling. Therefore, this article was an effort to summarize data available in literature where plant extract have been used for synthesis of Fe NPs. The secondary metabolites like tannins, saponins, glycosides, flavonoids are principally responsible for reduction of Iron salts into Fe NPs. Moreover, they also act as stabilizing agent and prevent oxidation of Iron NPs. These iron NPs prepared using plant extract have better pharmacological activity and are safe to use. This novel, simple and economic preparation method will pave new way for the preparation of iron nanoparticles. This will boost applications of iron nanoparticles as biosensor, MRI contrast agent, theranostics.
References
- Wu W, He Q, Jiang C (2008) Magnetic Iron Oxide Nanoparticles: Synthesis and Surface Functionalization Strategies. Nanoscale Res Lett 3(11): 397-415.
- Gupta AK, Gupta M (2005) Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 26(18): 3395-4021.
- Voros E, Cho M, Ramirez M, Palange AL, Rosa ED, et al. (2015) TPA immobilization on iron oxide nanocubes and localized magnetic hyperthermia accelerate blood clot lysis. Advanced Functional Materials 1-10.
- Herrmann IK, Schlegel AA, Graf R, Stark WJ, Beck-Schimmer B (2015) Magnetic separation-based blood purification: a promising new approach for the removal of disease-causing compounds. J Nanobiotechnology 13: 49.
- Govind S, Yadav KS (2015) Applications of nanoparticles in treatment and diagnosis of leukemia. Materials Science and Engineering 47: 156- 164.
- Mohanraj S, Kodhaiyolii S, Rengasamy M, Pugalenthi V (2014) Green synthesized iron oxide nanoparticles effect on fermentative hydrogen production by clostridium acetobutylicum. Appl Biochem Biotechnol 173 (1): 318-331.
- Huang L, Weng X, Chen Z, Megharaj M, Naidu R (2014) Green synthesis of iron nanoparticles by various tea extracts: Comparative study of the reactivity. Spectrochim Acta A Mol Biomol Spectrosc 130: 295-301.
- Azmi L, Singh MK, Akhtar AK (2011) Pharmacological and biological overview on Mimosa pudica Linn. International Journal of Pharmacy & Life Sciences 2(11): 1226-1234.
- VA Niraimathee, Subha V, Ernest Ravindran RS, S Renganathan (2016) Green synthesis of iron oxide nanoparticles from Mimosa pudica root extract. International Journal of Environment and Sustainable Development 15(3): 227-240.
- Tariq AL, Reyaz AL (2012) Phytochemical analysis of camellia sinensis leaves. International Journal of Drug Development & Research 4(4): 311-316.
- Ulker O, Fadime K, Esra K, Savaskoparal A (2016) Green synthesis of iron nanoparticles using Camellia sinesis extract. International Journal of Green and Herbal Chemistry 5: 29-37.
- Borade AS, Babasaheb NK, Rajkumar VS (2011) A phytopharmacological review on Lawsonia inermis (Linn.). International Journal of Pharmacy & Life Sciences 2(1): 536-541.
- Keerthana K, Jothi G, Sridharan G, Agnel AJN (2018) Microscopic, Physicochemical and Phytochemical Analysis of Gardenia jasminoides (Ellis). International Journal of Pharmacy and Pharmaceutical Sciences 10(1): 97-102.
- Farrukh TN, Farrukh MA (2015) Antibacterial activity of green synthesis of iron nanoparticles using Lawsonia inermis and Gardenia jasminoides leaves extract. Journal of Chemistry: 1-7.
- Noolu B, Ajumeera R, Chauhan A, Nagalla B, Manchala R, et al. (2013) Murraya koenigii leaf extract inhibits proteasome activity and induces cell death in breast cancer cells. BMC Complementary and Alternative Medicine 13(7): 1-17.
- Abu-Hiamed HA (2017) Chemical Composition, Flavonoids and β-sitosterol Contents of Pulp and Rind of Watermelon (Citrullus lanatus) Fruit. Pakistan Journal of Nutrition 16(7): 502-507.
- Prasad C, Gangadharal S, Venkateswarlu P (2016) Bio-inspired green synthesis of Fe3O4 magnetic nanoparticles using watermelon rinds and their catalytic activity. Applied Nanoscience 6(6): 797-802.
- Merlene AB, Suriyakala MA, Gothandam KM (2012) Varietal Impact on Phytochemical Contents and Antioxidant Properties of Musa acuminata (Banana). Journal of Pharmaceutical Sciences & Research 4(10): 1950-1955.
- Sudha K, Anitta S, Mythilidevi P, Thejomayah G (2015) Biosynthesis of iron oxide nanoparticles from green banana peel extract. International Journal of Social Science & Interdisciplinary Research 4(6): 165-176.
- Garg P, Sardana S (2016) Pharmacological and therapeutic effects of Ocimum sanctum. European Journal of Pharmaceutical and Medical Research 3(8): 637-640.
- Balamurughan MG, Mohanraj S, Kodhaiyolii S, Pugalenthi V (2014) Ocimum sanctum leaf extract mediated green synthesis of iron oxide nanoparticles: spectroscopic and microscopic studies. Journal of Chemical and Pharmaceutical Sciences 4: 201-204.
- Sumayya SS, K Murugan (2017) Phytochemical screening, RP-HPLC and FTIR Analysis of Kappaphycus alvarezii (Doty) Doty EX P.C Silva: Macro red algae. Journal of Pharmacognosy and Phytochemistry 6(1): 325-330.
- Yew YP, Shameli K, Miyake M, Kuwano N, Bt Ahmad, et al. (2016) Green Synthesis of Magnetite (Fe3O4) Nanoparticles Using Seaweed (Kappaphycus alvarezii) Extract. Nanoscale Research Letters 11(1): 276.
- Prashanth GK, Krishnaiah GM (2014) Chemical composition of the leaves of Azadirachta Indica Linn (Neem). International journal of advancement in engineering technology, management and applied sciences 1(5): 21-31.
- Pattanayak M, Nayak PL (2013) Green Synthesis and Characterization of Zero Valent Iron Nanoparticles from the Leaf Extract of Azadirachta indica (neem). World Journal of Nano Science & Technology 2(1): 6-9.
- Puthamohan Vinayaga Moorthi, Chelliah Balasubramanian (2015) Antimicrobial properties of marine seaweed, Sargassum muticum against human pathogens. Journal of Coastal Life Medicine 3(2): 122- 125.
- Namvar F, Mohamad R, Baharara J, Zafar-Balanejad S, Fargahi F, et al. (2013) Antioxidant, antiproliferative, and antiangiogenesis effects of polyphenol-rich seaweed (Sargassum muticum). BioMed Research International 1-9.
- Mahdavi M, Namvar F, Ahmad MB, Mohamad R (2013) Green biosynthesis and characterization of magnetic iron Oxide (Fe3O4) nanoparticles using Seaweed (Sargassum muticum) aqueous extract. Molecules 18(5): 5954-5964.
- Lee LS, Kim SH, Kim YB, Kim YC (2014) Quantitative analysis of major constituents in green tea with different plucking periods and their antioxidant activity. Molecules 19(7): 9173-9186.
- Herlekar M, Barve S, Kumar K (2014) Plant-mediated green synthesis of iron nanoparticles. Journal of Nanoparticles 1-9
- Huang L, Weng X, Chen Z, Megharaj M, Naidu R (2013) Synthesis of iron-based nanoparticles using oolong tea extract for the degradation of malachite green. Spectrochim Acta A Mol Biomol Spectrosc 117: 801-804.
- Awika JM, Rooney LW (2004) Sorghum phytochemicals and their potential impact on human health. Phytochemistry 65(9): 1199-1221.
- Njagi EC, Huang H, Stafford L, Genuino H, Galindo HM, et al. (2011) Biosynthesis of iron and silver nanoparticles at room temperature using aqueous Sorghum bran extracts. Langmuir 27(1): 264-271.
- Jayaprakash A, Sangeetha R (2015) Phytochemical screening of Punica granatum Linn. Peel Extracts. Journal of Academia and Industrial Research 4(5): 160-162.
- Salim, Saleh, Al-Timmimi (2017) Characterization of iron nanoparticle preparation from Punica granatum peel. International Journal of Science and Nature 8(2): 213-216.
- Ganiyu O, Oduje AA, Stephen AA, Ayodele JA (2015) Interaction of plantain (musa paradisiaca) peel extracts (unripe, ripe and over ripe) with key enzymes linked to hypertension (angiotensin-I converting enzyme) and their antioxidant activities (in vitro): a nutraceutical approach. Advances in Food Sciences 37(2): 1-9.
- Sada Venkateswarlu, Rao Y Subba, Balaji T, Prathima B, Jyothi NVV (2013) Biogenic synthesis of Fe3O4 magnetic nanoparticles using plantain peel extract. Materials Letters 100: 241-244.
- Ramamurthy V, Rajeswari DM, Gowri R, Vadivazhagi MK, Jayanthi G, et al. (2013) Study of the phytochemical analysis and antimicrobial activity of Dodonaea viscosa. International journal of pure and applied zoology 1(2): 178-184.
- Kiruba Daniel SCG, Vinothini G, Subramanian N, Nehru K, Sivakumar M (2013) Biosynthesis of Cu, ZVI, and Ag nanoparticles using Dodonaea viscosa extract for antibacterial activity against human pathogens. Journal of Nanoparticle Research 15: 1319-1328
- Catherine CI, Jude CI, Mercy OI (2015) Phytochemical Composition of Tridax procumbens Linn Leaves: Potential as a Functional Food. Food and Nutrition Sciences 6: 992-1004.
- Senthil M, Ramesh C (2012) Biogenic synthesis of Fe3O4 nanoparticles using Tridax procumbens leaf extract and its antibacterial activity on Pseudomonas aeruginosa. Digest Journal of Nanomaterials and Biostructures 7(3): 1655-1660.
- Rekha SS, Bhaskar M (2013) Screening and identification in vitro antioxidant activities of phytochemical compounds in ethanolic grape (vitis vinifera) seed extract. International Journal of Pharma and Bio Sciences 4(3): 609-617.
- Narayanan S, Sathy BN, Mony U, Koyakutty M, Nair SV, et al. (2011) Biocompatible magnetite/gold nanohybrid contrast agents via green chemistry for MRI and CT bioimaging. ACS Applied Material and Interfaces 4(1): 251-260.
- Riaz M, Khan O, Sherkheli MA, Khan MQ, Rahid R (2017) Chemical constituents of Termenalia chebula. Natural Products: An Indian Journal 13(2): 112.
- Mohan Kumar K, Mandal BK, Siva Kumar K, Sreedhara Reddy P, Sreedhar B (2013) Biobased green method to synthesise palladium and iron nanoparticles using Terminalia chebula aqueous extract. Spectrochim Acta A Mol Biomol Spectrosc 102: 128-133.
- Wang T, Jin X, Chen Z, Megharaj M, Naidu R (2014) Green synthesis of Fe nanoparticles using eucalyptus leaf extracts for treatment of eutrophic wastewater. Science of the Total Environment 466-467: 210-213.
- Wang Z, Fang C, Megharaj M (2014) Characterization of Iron- Polyphenol Nanoparticles Synthesized by three Plant Extracts and Their Fenton Oxidation of Azo Dye. ACS Sustainable Chemistry & Engineering 2 (4):1022-1025.
- Ayuba VO, Ojobe TO, Ayuba SA (2011) Phytochemical and proximate composition of Datura innoxia leaf, seed, stem, pod and root. Journal of Medicinal Plants Research 5(14): 2952-2955.
- Amlan KD, Marwal A, and Verma R (2014) DatuEucalyptus tereticornis, Melaleuca nesophila and Rosmarinus officinalis Leaf Extract Mediated One Step Green Synthesis and Characterization of Magnetite (Fe3O4) Nanoparticles. Research and Reviews: Journal of Pharmaceutics and Nanotechnology 2: 21-24.
- Sundararajan R, Koduru R (2014) A complete profile on couroupita guianensis- traditional uses, pharmacological activities and phytoconstituents. Pharmaphore 5(1): 147-159.
- Sathishkumar G, Logeshwaran V, Sarathbabu S, Jha PK, Jeyaraj M (2017) Green synthesis of magnetic Fe3O4 nanoparticles using Couroupita guianensis Aubl. fruit extract for their antibacterial and cytotoxicity activities. Artif Cells Nanomed Biotechnol 46(3): 589-598
- Birudu RB, Naik JM, Siddappa RD, Jilani SK, Janardhan M (2015) Phytochemical screening of ethanolic extract of Passiflora foetida (Linn) and medicinal importance. Indian Journal of Research in Pharmacy and Biotechnology 3(4): 324-327.
- Suganya D, Rajan MR, Ramesh R (2016) Green synthesis of iron oxide nanoparticles from leaf extract of Passiflora foetida and its antibacterial activity. International Journal of Current Research 8(11): 42081-42085
- Alorkpa EJ, Boadi NO, Badu M Saah, SA (2016) Phytochemical screening, antimicrobial and antioxidant properties of assorted carica papaya leaves in Ghana. Journal of Medicinal Plants Studies 4(6): 193- 198.
- Latha N, Gowri M (2014) Biosynthesis and Characterization of Fe3O4 Nanoparticles Using Caricaya Papaya Leaves Extract. International Journal of Science and Research 3(11): 1551-1556.
- Luna-Vazquez FJ, Ibarra-Alvarado C, Rojas-Molina A, Rojas-Molina JI, Yahia EM, et al. (2013) Nutraceutical Value of Black Cherry Prunus serotina Ehrh. Fruits: Antioxidant and Antihypertensive Properties. Molecules 18(12): 14597-14612
- Vasco C, Riihinen K, Ruales J, Kamal-Eldin A (2009) Chemical Composition and Phenolic Compound Profile of Mortino (Vaccinium Floribundum Kunth). J Agric Food Chem 57(18): 8274-8281.
- Murgueitio E, Debut A, Landivar J, Cumbal L (2016) Synthesis of Iron Nanoparticles through Extracts of Native Fruits of Ecuador, as Capuli (Prunus serotina) and Mortino (Vaccinium floribundum). Biology and Medicine 8(3): 1-3.
- Gayathri V, Kiruba D (2014) Preliminary Phytochemical Analysis of Leaf Powder Extracts of Psidium guajava L. International Journal of Pharmacognosy and Phytochemical Research 6(2): 332-334.
- Jeyasundari J, Praba PS, Jacob YBA, Vasantha VS, Shanmugaiah V (2017) Green Synthesis and Characterization of Zero Valent Iron Nanoparticles from the Leaf Extract of Psidium guajava Plant and their Antibacterial Activity. Chemical Science Review and Letters 6(22): 1244-1252.






























