Nano-Carriers and Therapeutic Drug Delivery: Open Questions and Perspectives
Domenico Lombardo*
CNR-National Research Council, Institute for Chemical-Physical Processes, Italy
Submission: September 25, 2018; Published: October 23, 2018
*Corresponding author: Domenico Lombardo, CNR-National Research Council, Institute for Chemical-Physical Processes Chemical and Physical, Viale Ferdinando Stagno d’ Alcontres, 98158 Messina, Italy.
How to cite this article: Domenico L. Nano-Carriers and Therapeutic Drug Delivery: Open Questions and Perspectives. Glob J Nano. 2018; 4(3): 555640. DOI: 10.19080/GJN.2018.04.555640
Abstract
Recent achievements in nanotechnology has stimulated the development of a large variety of smart nano-platforms for the delivery of therapeutic drugs and smart applications in biotechnology and nano-medicine. A wide range of smart integrated Nano systems have proven their effectiveness for various types of biomedical tasks, thus paving the way for the modern approach of the personalized medicine. However, many of these nano-structured platforms present lack of experience between the pre-clinical and clinical studies, thus resulting in the huge difficulties to obtain regulatory and ethics approval. As a result, most of these smart nanostructured drug delivery systems are not currently approved for clinical use. In this article we highlight the critical issues and open questions which are still present when translating theragnostic approaches into the clinical practice.
Keywords: Nanomedicine; Nanocarriers; Drug delivery; Theragnostic approach; Personalized medicine
Introduction
Many conventional drug delivery systems present critical issues connected with side effects that limit their use in the clinical practice [1-3]. A large variety of nanoparticle-based platforms allow the design of the molecular architecture (size, shape, and surface functionalities) for the development of efficient therapeutic nanomaterials prototypes that include: organic (including polymer-based [4] such as block copolymers [5], dendrimers [6,7], hydrogels [8]), lipid nanocarriers [9-12]) and inorganic nanocarriers (such as metal [13] and silica [14,15] nanoparticles), as well as synthesized hybrid (organic/inorganic) nanostructure [16-18]. Moreover, the possibility to develop within the same nano-platform a diagnostic imaging system, allow the space-time evolution monitoring of the disease molecular response for each patient (theragnostic approach) (Figure 1) [1,19].
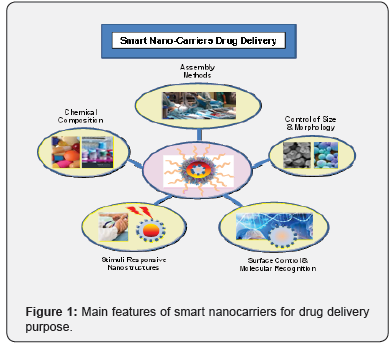
However, translating nanomedicine discoveries into the clinical practice request more innovative strategies and deeper investigation. In this article we highlight some critical issues and open questions that still limit the clinical applications of those novel technologies.
Active Targeting in Drug Delivery Nano-Platform: Critical Issues and Perspectives
The main challenge in drug delivery design is to selectively drive the drug molecules to the desired target (diseased) tissues, while avoiding the healthy ones. Locally activated drug release approaches offer the opportunity for the control of the active targeting of the diseased tissues, by employing specific ligand–receptor interactions. However, despite the significant progresses in the understanding the molecular basis of several pathology, molecular targeted therapy remains a promise of a future innovative approach [20]. Most of all drugs are associated, in fact, with undesired effects that create limitations to their clinical use. Moreover, the amount of drug delivered to tumor targets, in ligand–receptor mediated processes, is much less than 5% [21,22]. Thus, the mere presence of a ligand-receptor combination on a nanocarrier does not ensure a successful active targeting. This indicate that more efficient methods are required in order to improve the response rates to targeted drug delivery therapies.
A partial response to this limitation is provided by the employment of stimuli responsive nanocarriers [23]. Stimuli responsive nanocarriers aim to exploit the diseased tissues microenvironment (internal control factors such as the pH, enzyme and redox) to enhance the therapeutic effects [24]. However, optimizations and improvements experiments are needed before the translation of specific stimulus from preclinical models to the clinical practice. The main limitation of the “internal” stimuli responsive nano-systems is connected with the complexity of the biological micro-environment encountered by the nanocarriers within the (diseased tissues) biological microenvironment. Moreover, although the external stimuli responsive systems are much easier to be controlled, they present major problems related to normal tissue damage and tissue-penetration depth [25].
For this reason, a deeper (in vitro) investigation of the main effects in terms of structure-function relationship are of fundamental importance for the rational understanding of the main processes involved. Particularly interesting, in this respect, is the study of model bio-membranes and their interaction with nanoparticles, as they can be adopted as simplified models that mimic the relevant processes encountered in real cell membranes [26-39]. Those studies have given a strong input to the understanding of the complex processes driven by the interactions that a nanostructured material can develop toward biological systems [30-33].
Advanced Approaches: System Biology and Personalized Nanomedicine
Employment of highly sensitive nano-analytical and diagnostics techniques as well as new smart integrated medical nanosystems (such as biosensors) allow also to attain an early diagnostic and a global profiling of the (individual) patient disease, thus paving the way to the modern approach of personalised medicine [34,35].
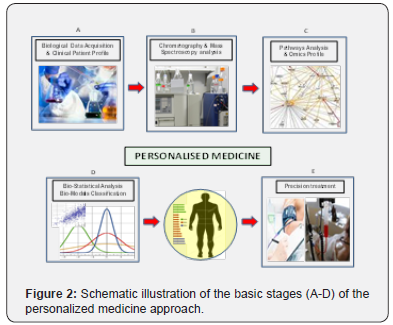
Modern approaches for personalized health monitoring and preventative medicine take advantage of the system biology approach, which is based on the (in parallel) collection of data from many components, using the so called “-omics” technologies, (such as proteomics, genomics, metabolomics). This modern approach aims to understand the complex functioning of the living systems, by inferring the complex pathways that regulates a specific physiological (or pathological) processes (Figure 2) [35]. However, the biocompatibility, toxicological aspects and ethical implications still represent critical issues to be resolved [36].
The complexity of the human genome due to the numerous genes involved, together with the large number of possible genetic variations, represent an important critical issue that impedes the routine application in the clinic of the personalised approach. Although these difficulties, we are confident that the personalized medicine approach could have a prominent role in the modern approaches and future practice of (nano)medicine [36]. In this respect the recognition of individual differences (of different patients) in drug response is an essential step toward the development of advanced medical approaches based of the optimized therapy. This challenge requires a synergistic effort between the different cross-disciplinary fields of nano-medicine, biochemistry and bioengineering.
Conclusions and Future Perspectives
The recent development of integrated drug delivery systems for biomedical applications has led to develop nano-platforms with improved therapeutic efficacy including target specificity, controlled drug release, lower therapeutic doses. In this respect, the modern approaches of supra-molecular self-assembly allow the construction of smart nanostructured materials with a large variety of chemical composition, architectures, and surface properties. The introduction of stimuli responsive (assembly/ disassembly) nanostructured systems in combination with the ligand–receptor recognition processes furnish a large variety of theragnostic solutions for the definition of specific bio-medical tasks such as the controlled release of drugs, imaging capabilities and multi-component (multi-functional) therapeutics.
Although modern approach to drug delivery evidence good performance against a large variety of specific diseases, a number of inherent drawbacks and critical issues are still present. Those drawbacks, that limit their translation in the clinic experience, are mainly connected with the low amount of drug delivered to diseased tissues (i.e. less than 5%) and the presence of possible severe side effects. Much efforts are then required to put these innovative nanocarriers into the clinical practice, by a deeper investigation of their physico-chemical behavior, therapeutic efficacy and safety.
Integrated nanocarrier systems will constitute an integral part of the future therapeutic intervention. New design and versatile control ability offered by smart nanostructures provides a wide range of solutions in placing the desired combination of functions into a single scaffold, thus paving the way for their translation into nanomedicine experience.
The next challenge consists, then, in the development of integrated nano-medical systems that allow to attain an early diagnostic and a global profiling of the health (and disease) of the individual patients, thus providing a novel approach for personalized health monitoring and preventive medicine.
References
- Liu D, Yang F, Xiong G, Gu N (2016) The Smart Drug Delivery System and Its Clinical Potential. Theranostics 6(9): 1306-1323.
- Allen TM, Cullis PR (2013) Liposomal drug delivery systems: from concept to clinical applications. Adv Drug Deliv Rev 65(1): 36-48.
- Joseph Francis PJ, Navas AA (2018) Versatility of Nanoparticles in the Modern Medicine. Glob J Nano 4(1).
- Hrubý M, Filippov SK, Štěpánek P (2005) Smart polymers in drug delivery systems on crossroads: Which way deserves following? Eur Polym J 65: 82-97.
- Mikhail AS, Allen C (2009) Block copolymer micelles for delivery of cancer therapy: transport at the whole body, tissue and cellular levels. J Control Release 138(3): 214-223.
- Jain K (2017) Dendrimers: Smart nanoengineered polymers for bioinspired applications in drug delivery. Biopolymer-Based Composites 169-220.
- Lombardo D (2014) Modeling dendrimers charge interaction in solution: relevance in biosystems. Biochem Res Int.
- Kopeček J, Yang J (2007) Hydrogels as smart biomaterials. Polym Int 56(9): 1078-1098.
- Xing H, Hwang K, Lu Y (2016) Recent Developments of Liposomes as Nanocarriers for Theranostic Applications. Theranostics 6(9): 1336- 1352.
- Lombardo D, Calandra P, Barreca D, Magazù S, Kiselev MA (2016) Soft interaction in liposome nanocarriers for therapeutic drug delivery. Nanomaterials 6(7): 125.
- Nirmal S, Dipti G, Avinashkumar S (2017) Nanostructured Lipid Carriers: As an Efficient Drug Delivery Carrier. Glob J Nano 3(3).
- Kiselev MA, Lombardo D, Lesieur P, Kisselev AM, Borbely S, et al. (2008) Membrane self-assembly in mixed DMPC/NaC systems by SANS. Chem Phys 345(2-3): 173-180.
- Adeyemi OS, Sulaiman FA (2015) Evaluation of metal nanoparticles for drug delivery systems. J Biomed Res 29(2): 145-149.
- Li Z, Barnes JC, Bosoy A, Stoddart JF, Zink JI (2012) Mesoporous silica nanoparticles in biomedical applications. Chem Soc Rev 41(7): 2590- 2605.
- Pasqua L, Leggio A, Sisci D, Andò S, Morelli C (2016) Mesoporous silica nanoparticles in cancer therapy: Relevance of the targeting function. Mini-Rev Med Chem 16(9): 743-753.
- Li F, Liang Z, Ling D (2017) Smart Organic-Inorganic Nanogels for Activatable Theranostics. Curr Med Chem.
- Bonaccorsi L, Lombardo D, Longo A, Proverbio E, Triolo A (2009) Dendrimer Template Directed Self-Assembly during Zeolite Formation. Macromolecules 42(4): 1239-1243.
- Morelli C, Maris P, Sisci D, Perrotta E, Brunelli E, et al. (2011) PEGtemplated mesoporous silica nanoparticles exclusively target cancer cells. Nanoscale 3(8): 3198-3207.
- Sathishkumar G, Sivaramakrishnan S (2018) Theranostic Nanomedicine: State of the Art Technology. Glob J Nano 3(5): 555621.
- Bae YH, Park K (2011) Targeted drug delivery to tumours: Myths, reality and possibility. J Control Release 153(3): 198-205.
- Pirollo KF, Chang EH (2008) Does a targeting ligand influence nanoparticle tumor localization or uptake? Trends Biotechnol 26(10): 552-558.
- Kirpotin DB, Drummond DC, Shao Y, Shalaby MR, Hong K, et al. (2006) Antibody targeting of long-circulating lipidic nanoparticles does not increase tumor localization but does increase internalization in animal models. Cancer Res 66(13): 6732-6740.
- Mura S, Nicolas J, Couvreur P (2013) Stimuli-responsive nanocarriers for drug delivery. Nature Mater 12: 991-1003.
- Dai Y, Xu C, Sun X, Chen X (2017) Nanoparticle design strategies for enhanced anticancer therapy by exploiting the tumour microenvironment. Chem Soc Rev 46(12): 3830-3852.
- H Thakur, D Sahu (2016) Biological Effects of Electromagnetic Waves: Case Studies and Safety Standards. Indian Journal of Science and Technology 9(47): 1-7.
- Sackmann E (1995) Physical basis of self-organization and function of membranes: Physics of vesicles. In: Lipowsky R, Sackmann E, (Eds,) Handbook of Biological Physics, Elsevier: Amsterdam, 1: 213-303.
- Lombardo D, Calandra P, Bellocco E, Laganà G, Barreca D, et al. (2016) Effect of anionic and cationic polyamidoamine (PAMAM) dendrimers on a model lipid membrane. Biochim Biophys Acta 1858(11): 2769- 2777.
- Katsaras J, Gutberlet T (2000) Lipid bilayers. Structure and Interactions. Springer-Verlag: Berlin Heidelberg.
- Kiselev MA, Janich M, Hildebrand A, Strunz P, Neubert RHH, Lombardo D (2013) Structural transition in aqueous lipid/bile salt [DPPC/NaDC] supramolecular aggregates: SANS and DLS study. Chem Phys 424(6): 93-99.
- Bourgaux C, Couvreur P (2014) Interactions of anticancer drugs with biomembranes: What can we learn from model membranes? J Control Release 190(28): 127-138.
- Kiselev MA, Lombardo D (2017) Structural characterization in mixed lipid membrane systems by neutron and X-ray scattering. Biochim Biophys Acta Gen Subj 1861(1): 3700-3717.
- Santos S, Medronho B, Santos T, Antunes FE (2013) Amphiphilic Molecules in Drug Delivery Systems. In: Coelho J, (Eds,) Drug Delivery Systems: Advanced Technologies Potentially Applicable in Personalised Treatment. Advances in Predictive, Preventive and Personalised Medicine, Springer, Dordrecht, 4: 35-95.
- Lombardo D, Calandra P, Magazù S, Wanderlingh U, Barreca D, et al. (2018) Soft nanoparticles charge expression within lipid membranes: The case of amino terminated dendrimers in bilayers vesicles. Colloids Surf B Biointerfaces 170: 609-616.
- Vinita V (2017) Potential Devices of Nanostructured Nanomaterials in Drug Delivery Systems. Glob J Nano 3(1)
- Zhang XQ, Xu X, Bertrand N, Pridgen E, Swami A, et al. (2012) Interactions of nanomaterials and biological systems: Implications to personalized nanomedicine. Adv Drug Deliv Rev 64(13):1363-1384.
- Soares S, Sousa J, Pais A, Vitorino C (2018) Nanomedicine: Principles, Properties, and Regulatory Issues. Front Chem 6: 360.






























