Nanomedicine – What’s in the Name?
Karel Petrak*
Chief Scientific Officer, USA
Submission: September 21, 2018; Published: October 10, 2018
*Corresponding author: Karel Petrak, Chief Scientific Officer, NangioTx Inc., Newark, New Jersey, USA.
How to cite this article: Karel Petrak . Nanomedicine – What’s in the Name?. Glob J Nano. 2018; 4(3): 555637. DOI: 10.19080/GJN.2018.04.555637
Keywords: Nanomedicine; Nanotechnology; Prevention; Treatment; Disease
Opinion
Nanomedicine is usually described as the application of nanotechnology, which in turn is a explained as “engineering of tiny machines” that could be used for the prevention and treatment of disease in the human body. The image has captured the imagination of science reporters. University of Mainz researchers have been reported [1] “using miniature drug-filled nanocarriers to target headaches and tumors” designed to “dock on to immune cells” and referred to as “miniature submarines”. Similarly, Science Magazine [2] reported that “nanomachines drill holes into cancer cells”. However, this perception of drug delivery is solidly based on science fiction [3].
The first nanodrug, Doxil (pegylated liposomal doxorubicin), was approved by FDA in 1995 to treat some types of cancers. As is always the case when drugs are developed and approved, this product was formulated to improve the balance between the efficacy and toxicity of doxorubicin therapy. Note that the drug is a liposome, a type of particle first described by British haematologist Alec D Bangham in 1961 [4] at the Babraham Institute, in Cambridge, some 30 years before the term “nanomedicine” was supposedly used for the first time [5].
Since that time, 50 nanodrugs have been approved by the FDA, with some 60 investigational nanodrugs being in the “pipe line [6]. It is interesting to note that the following “nanomachines” have been formulated as follows (Table 1).
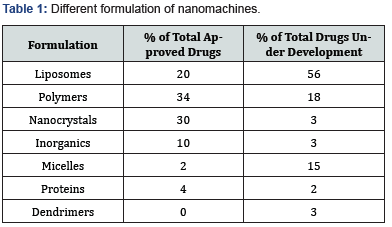
None of the approved products could be described as “nano machines”. The active components of these products, molecular drug entities, have always been at the nano scales. Similarly, all the particulate formulation aids used in the products approved to date were being used before the tag “nano” was added to their name.
Better, original rationales for converting newly acquired precision-medicine knowledge about biology and disease into effective human therapies are urgently needed. Giving old ideas new lease of life by changing their name does not in any way help our way forward.
References
- Using miniature drug-filled nanocarriers to target headaches and tumors.
- Meet the nanomachines that could drive a medical revolution.
- Petrak K (2012) Disease-target drug delivery – science or fiction?
- Bangham AD, Horne RW (1964) Negative Staining of Phospholipids and Their Structural Modification by Surface-Active Agents As Observed in the Electron Microscope. Journal of Molecular Biology 8(5): 660-668.
- Drexler KE, Peterson C, Pergamit G (1991) Unbounding the Future: The nanotechnology revolution.
- Ventola CL (2017) Progress in Nanomedicine: Approved and Investigational Nanodrugs. Pharmacy and Therapeutics 42(12): 742-755.






























