A Preliminary Investigation of CO2 Adsorption on Palladium Catalysts in Mesoporous Silica Support
Naif Alarifi*
*Institute of Refining and Petrochemicals Technologies, King Abdulaziz City for Science and Technology (KACST), Riyadh, Saudi Arabia
Submission: August 18, 2024;Published:March 19, 2025
*Corresponding author:Naif Alarifi, Institute of Refining and Petrochemicals Technologies, King Abdulaziz City for Science and Technology (KACST), Riyadh, Saudi Arabia
How to cite this article:Naif A. A Preliminary Investigation of CO2 Adsorption on Palladium Catalysts in Mesoporous Silica Support. Eng Technol Open Acc 2025; 6(3): 555687.DOI: 10.19080/ETOAJ.2025.06.555687
Introduction
Increasing interest in carbon capture, sequestration, and CO2 capture are instrumental processes in the reduction of greenhouse gas emissions. Palladium-supported silica with 0.5 and 1% Pd/SiO2 were examined for CO2 sorption at various temperatures and pressures.
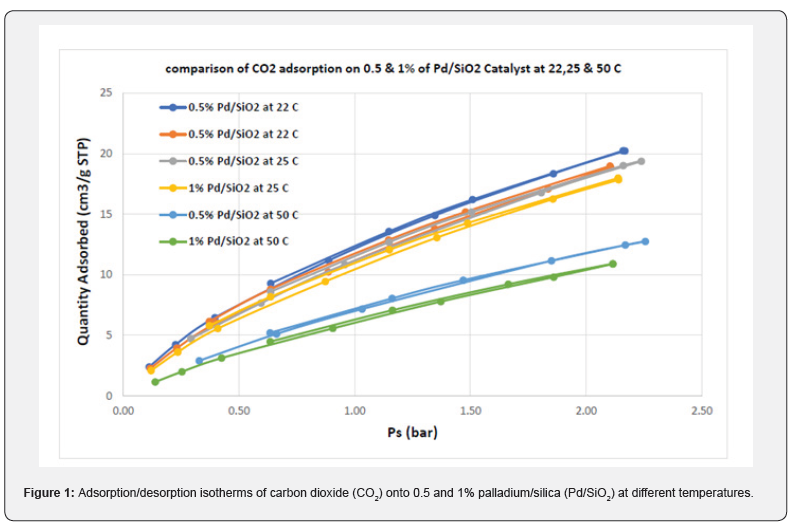
CO2 gas was studied for sorption onto Pd/SiO2 with temperatures between 22, 25, and 50°C and pressures between 0.3 and 2.25 bar, where the equilibrium adsorption capacity in CO2 reached its highest value at 0.5 Pd/SiO2 and 22°C.
On the flip side, a significant amount of CO2 is a greenhouse gas and a significant factor in global warming and climate change. In recent years, there has been a rise in interest in the development of a material that can act as a sorbent for CO2 storage at lower temperatures due to the possibility of mitigating CO2 emissions in order to protect the environment.
The aim of this work is to investigate how to improve the pathways used to capture CO2 and remove toxic gases from the environment. For this purpose, the adsorption-desorption experiments were conducted at different temperatures and pressures. This primary study presents a behavior of the used material for studying their CO2 adsorption capabilities
Adsorption Study
The CO2 storage studies were performed using a High Pressure Volumetric Analyzer (HPVA), that measured the adsorption/desorption isotherms of CO2 gas at different temperatures and pressures. Before the adsorption measurements, the samples were degassed at 350°C for 5 hours and exposed to flotation in a helium at atmosphere to inspect the mass and skeleton volume of the loaded sample.
(Figure 1) shows the measured adsorption/desorption isotherms of CO2 at different temperatures; 22, 25, and 50°C. Overall, it revealed that the adsorption capacity of CO2 decreases by increasing the temperature and loaded percentage of Palladium. Furthermore, the adsorption/desorption capacity of CO2 gas onto Pd/SiO2 increases by increasing the pressure at all temperatures. The highest and lowest adsorption capacity for CO2 on Pd/SiO2 at a pressure of ~2.15 bar were 20 cm3/g and 12 cm3/g at 22 and 50°C, respectively. It is to be noted that the adsorption and desorption data were almost identical, without any noticeable lag. According to the data, Pd/SiO2 performs well in the adsorption of CO2 and has a type-I adsorption isotherm, which allows for its regeneration by decreasing pressure or increasing temperature. At high temperatures in a small pressure range, there are small deviations from experimental data observed, The Langmuir model fitting was generally in good agreement with experimental data at all temperatures and throughout the entire pressure range, as observed in (Figure 1).
Conclusion
Stable palladium nanoparticles supported on mesoporous silica (Pd/SiO2) were assessed for their ability to capture CO2 at different temperatures and pressures. The absorption of CO2 gas by Pd/SiO2 was effective, particularly at low temperatures (such as 22°C). It would be feasible to suggest using the Pd/SiO2 material for CO2 adsorption. The absolute experimental CO2 concentrations were accurately correlated using the Langmuir adsorption isotherm model with temperature and pressure. The findings indicate that Pd/SiO2 catalysts are capable of providing low-cost and environmentally friendly methods for capturing CO2 at low temperatures.