Influence of Real CNG Engine Emissions On PdO/Al2O3 Methane Oxidation Catalysts
Sebastian Tomin1*, Uwe Wagner2 and Thomas Koch3
1Institute for Internal Combustion Engines (IFKM), Karlsruhe Institute of Technology (KIT), Germany
2Doctoral, Institute for Internal Combustion Engines (IFKM), Karlsruhe Institute of Technology (KIT), Germany
3Professor, Institute for Internal Combustion Engines (IFKM), Karlsruhe Institute of Technology (KIT), Germany
Submission: October 26,2023; Published: November 7, 2023
*Corresponding author: Sebastian Tomin, Institute for Internal Combustion Engines (IFKM), Karlsruhe Institute of Technology (KIT), Rintheimer Querallee 2, D-76131 Karlsruhe, Germany, Email Id: sebastian.tomin@kit.edu
How to cite this article: Sebastian T, Uwe W, Thomas K. Influence of Real CNG Engine Emissions On PdO/Al2O3 Methane Oxidation Catalysts. Eng Technol Open Acc. 2023; 5(4):555669. DOI: 10.19080/ETOAJ.2023.05.555669
Abstract
Natural gas engines play an important role in stationary energy generation, but also in mobile applications, especially in the maritime sector. However, this form of natural gas use produces methane emissions due to incomplete combustion, which must be removed because of the high greenhouse effect. This is done by means of oxidation catalysts. The PdO/Al2O3 investigated in this publication has emerged as an efficient catalyst material. Nonetheless, the water and sulphur present in real engine exhaust gas can lead to deactivation of the catalyst. Nitrogen oxides on the other hand can even be beneficial under certain circumstances. However, these findings are largely based on investigations with synthesis gas, which differs from real exhaust gas. In addition, various influencing variables such as temperature and space velocity are coupled with each other through engine control. For this reason, the investigation of the influences of real engine emissions and conditions on methane conversion discussed here is necessary as a supplement to laboratory investigations. Thus, a natural gas engine modified for lean-burn operation is connected to an innovative exhaust system in order to thereby measure catalyst samples on a laboratory scale under real conditions. The engine’s operating ranges analysed here extend from rich conditions required for the regeneration of deactivated catalytic converters to lean conditions such as are standard for combined heat and power plants. In addition, the influence of exhaust gas recirculation was considered. As a result, a noticeable minimum of methane conversion was observed in slightly lean conditions. Therefore, the influences of water, sulphur and nitrogen oxide emissions in particular are examined using exhaust gas measurement technology such as Fourier-Transform Infrared Spectroscopy.
Keywords: Methane Oxidation Catalyst (MOC); PdO/Al2O3; Water Inhibition; Sulfatisation; CNG Engine; Regeneration; Lean Burn Gas Engine
Abbreviations: AMA: Abgasmessanlage (Exhaust Gas Measuring System); bef. Cat.: Before Catalyst; CNG: Compressed Natural Gas; DFG: Deutsche Forschungsgemeinschaft (German Research Foundation); EGR: Exhaust Gas Recirculation; FTIR: Fourier-Transform Infrared Spectroscopy; GC: Gas Chromatograph; GHSV: Gas Hourly Space Velocity; MFB50: Mass Fraction Burned 50 %; T: Temperature; URAS: Ultrarot-Absorptions-Schreiber (Nondispersive Infrared Sensor)
Introduction
Despite the tight supply situation, natural gas is an important energy carrier that will remain relevant especially in the transition phase to climate neutrality. One form of using natural gas in this context is combustion in an engine. Natural gas engines play an important role above all in the area of stationary energy generation (combined heat and power plants) but also in mobile applications such as maritime transportation. Against the backdrop of supply difficulties and increasingly stringent exhaust gas standards, the further optimisation of engine processes and the associated exhaust gas aftertreatment is thus moving further into the focus of research activities. However, due to incomplete combustion, methane slip always occurs [1], which is very problematic due to the high influence of methane on the greenhouse effect [2] and thus requires the use of methane oxidation catalysts. Palladium-based catalysts with aluminium oxide as a carrier material have been shown to be effective, especially in lean-burn gas engines [3,4]. For reasons of efficiency, natural gas engines are often operated lean. This also has positive effects on nitrogen oxide formation. Another measure to reduce nitrogen oxides is the use of Exhaust Gas Recirculation (EGR). Despite the abovementioned control measures, nitrogen oxide is still emitted. Although being harmful to the environment, they can have a positive influence on catalysis under certain circumstances [5-7]. An exhaust gas species that has a negative (inhibitory) effect on a methane oxidation catalyst is water. This results from competition between water and methane for the active sites on the catalyst surface [8] and hydroxyl adsorption on the support material and the active sites [9]. Besides inhibition effects, deactivation in the form of sulphur poisoning (sulphatisation) [8,9,11] occurs particularly in natural gas applications. Two sources of sulphur can be identified in the engine context. The first is sulphur-containing engine oil, which enters the cylinder during combustion, and sulphur-containing odorants, which are added to natural gas for safety reasons. Regeneration takes place via rich operation, as the resulting reducing conditions favour the removal of the sulphur on the catalytic converter surface [12]. Nevertheless, the findings of the literature cited so far were only obtained under laboratory conditions. In contrast to experiments with synthetic gases, real engine operation has a greater variety of exhaust gas species. In addition, important state variables that define the boundary conditions at the catalytic converter are coupled with each other. For example, lean-burn at constant engine load causes a change in exhaust gas temperature, exhaust gas composition and space velocity. Therefore, an investigation under real conditions is necessary as a supplement to laboratory tests. The aim of the tests discussed in this paper is thus to find out what influence real engine exhaust gas and conditions have on a palladium-based catalytic converter, especially with regard to NO, H2O and SO2 as well as the coupling of the state variables.
Materials and Methods
Engine Test Bench
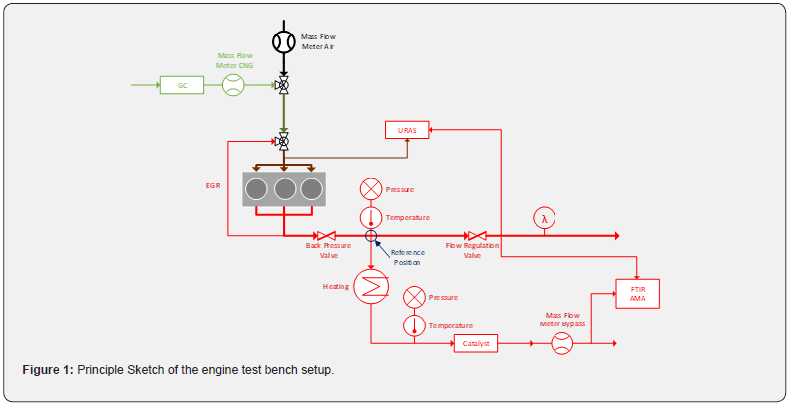
First, a stoichiometrically operated 2.2l-gas engine originally used in commercial and industrial applications was modified to enable lean-burn operation and exhaust gas recirculation (EGR). The modifications included a redesign of the intake manifold and the integration of an EGR section including cooler. In order to be able to handle higher engine loads, a connection to an external charge air supply is also necessary. The engine is coupled with an exhaust system whose integral component is a bypass line that enables catalytic converter samples to be measured. A backpressure flap simulates the flow resistance that a turbocharger would generate in reality. The mass flow through the bypass and thus the space velocity at the catalytic converter are controlled with the aid of a flow divider flap. Cooling of the exhaust gases up to the sample is prevented by using an adjustable heating band. The reference position, which is defined in such a way that it corresponds to the typical installation location (after the turbocharger) of a catalytic converter in series applications, serves as the basis for the setpoint values of the two aforementioned controls. The mass flow at the reference corresponds to the sum of the flows entering the engine (natural gas and air), both of which are measured using Coriolis scales. To determine the space velocities of the sample as well as the reference, the exhaust gas composition, pressures, temperatures as well as the catalyst volumes are missing. The exhaust gas composition is measured using Fourier transform infrared spectroscopes (FTIR) and an exhaust gas measurement system (AMA) and is the same at both locations. Pressure and temperature are recorded via thermocouples and pressure cells, respectively. The catalyst volume results from the geometry of the sample, which at the reference is defined as the volume of the series catalyst from the original engine (retrofit). The mass flow through the sample is recorded by a mass meter based on the differential pressure. In addition, the composition of the natural gas is measured using a gas chromatograph (GC). The EGR rate is determined by comparing the measured CO2 concentrations (URAS) in the exhaust gas and in the intake manifold (Figure 1).
Catalyst Probe
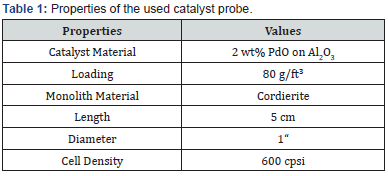
The catalyst material considered here is PdO on Al2O3 (Table 1), which, as explained at the beginning, is particularly well suited for methane oxidation. The catalyst diameter is 1”, the length 5 cm since a coating of near-series geometries is not feasible on a laboratory scale.
Measuring Program
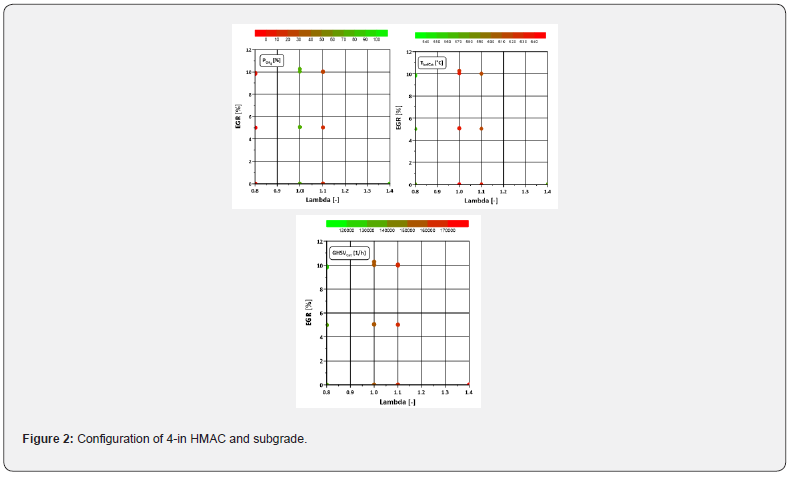
(Table 2) lists the operating points investigated. The tests were carried out with almost constant added fuel mass. A change in the operating parameters results in a change in efficiency, which causes the torque changes. The fuel-air-equivalence ratio λ was varied from rich (0.8) to lean (1.4). The rich operating points here represent regeneration, the remaining points normal operation in maritime or stationary applications. The oxygen content at stoichiometric conditions is slightly above zero due to incomplete combustion and the lambda control. The exhaust gas temperature is influenced by the position of the combustion phasing (MFB50). If it is shifted to later, the exhaust gas temperatures rise. Since, with an almost constant fuel mass, leaning can only occur through an increased air supply, therefore the exhaust gas mass flow also increases, which leads to an increase in the space velocity.

Results and Discussion
(Figure 2) shows the methane conversion as a function of lambda and EGR rate. Under lean conditions, the conversion rate is predictably poor due to the lack of oxygen for oxidation of methane. However, it is noticeable that a minimum seems to form in the lean range at λ = 1.1. This raises the question of where this can come from. Do the deactivation mechanisms mentioned in the introduction have an influence? What role does NO play? After all, it is known that raw nitrogen oxide emissions reach their maximum in slightly lean operation. These issues are discussed in more detail below.
Catalyst Deactivation
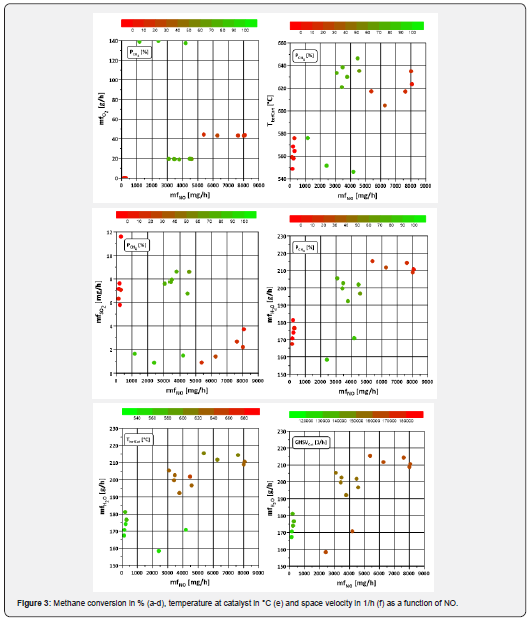
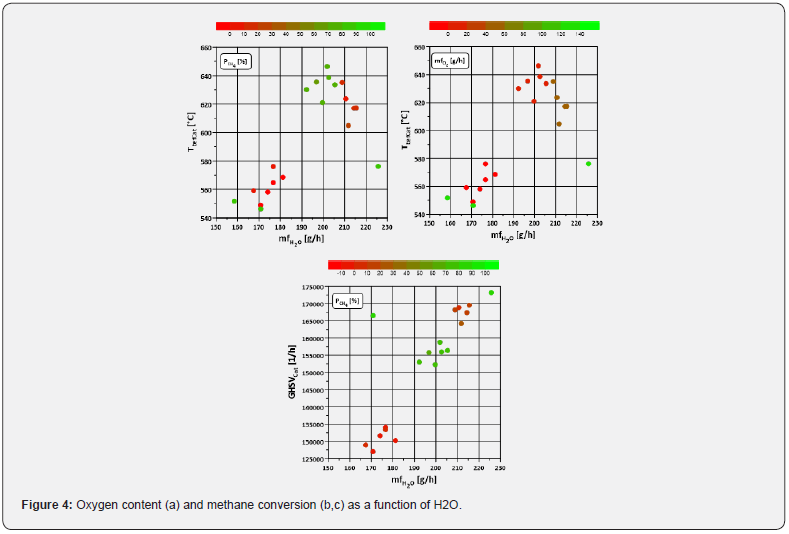
In order to shed more light on the issue of catalyst deactivation, it is necessary to take a look at the typical influencing variables known from the literature. As mentioned in the introduction, nitrogen monoxide influences catalysis. If the methane conversion is plotted against the NO mass flow, it can be seen that the decrease in CH4 conversion in the lean seems to be accompanied by an increased proportion of nitrogen monoxide (Figure 3 a-d). Another species known for its influencing (inhibitory) effect is water. A higher mass flow of both species is accompanied by a deterioration of the catalytic activity, which is clearly shown in (Figure 3d). In a comparison of H2O and NO (Figure 3e,3f), an influence of both temperature and space velocity is visible for water quantities above approx. 190 g/h and for O2 quantities between 0 and 50 g/h. The effect of water on the catalytic activity is also visible in the comparison of NO and H2O. A higher temperature in combination with a lower space velocity (and thus a higher residence time) has a positive effect on methane conversion (Figure 2b, c, Figure 3 d-f).
The interaction of water with temperature and space velocity is shown separately in (Figure 4). The range of λ = 1.1 tends to show higher temperatures, whereby there is no clear limit as with the space velocity. This suggests that the influence of the GHSV weighs more heavily here. The findings from Sadokhina et al. [7] and Auvinen et al. [5] suggest that water inhibition is more powerful than the promoting effect of NO on methane conversion in the presence of H2O and SO2. So, following the findings from the literature, this deterioration means that the positive effects of NO (despite the high amounts) are overcompensated by water. The emissions occurring here in engine operation range from 500 to 4400ppm for λ ≥ 1 and thus significantly exceed the proportions (up to 500ppm) studied in Auvinen et al. [5]; Gremminger et al. [6]; Sadokhina et al. [7]. Since the deterioration of methane conversion is accompanied by a more significant change in nitrogen oxide amounts compared to the changes in water, temperature and space velocity, and the nitrogen oxide fractions differ significantly from those in the literature cited here, the exact influence of NO in real exhaust gas might need further investigation.
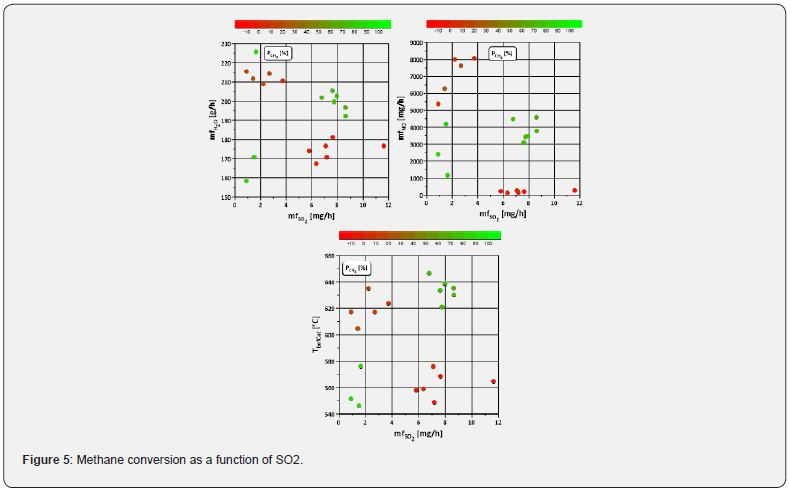
Another form of deactivation is sulphation, which can occur even with small amounts Lott et al. [11]. Since sulphur as well as nitrogen oxide and water raw emissions occur simultaneously, their effects on catalysis must also overlap. To assess which of the deactivation or promotional mechanisms dominate, the methane conversion is plotted against the SO2 mass flow (Figure 5). The first two plots in (Figure 5) indicate that sulphur poisoning has a lower effect on the methane conversion than water in the experimental period (10h in total), since the conversion rate in the range λ ≥ 1 shows high as well as low values at approximately equal amounts of SO2 (< 4mg/h). This is possibly due to the different nature of inhibition as opposed to poisoning. The latter needs more time to unfold its effect, whereas inhibition effects are characterised by a more abrupt occurrence. For example, in Lott et al. [11], a noticeable deterioration occurred at a temperature of 500°C from about 3h, whereas the test run time here per operating point is significantly lower at a higher temperature. Besides the temporal component, the amount of sulphur dioxide also plays a role. The SO2 concentrations measured in this publication are on average 2ppm and thus below the detection limit of the FTIR (4ppm) and below the concentration of 5ppm investigated by Lott et al. [11]. So, in order to investigate the influence of sulphur under real engine conditions more time is required.
SO2 Desorption or Post-Oxidation
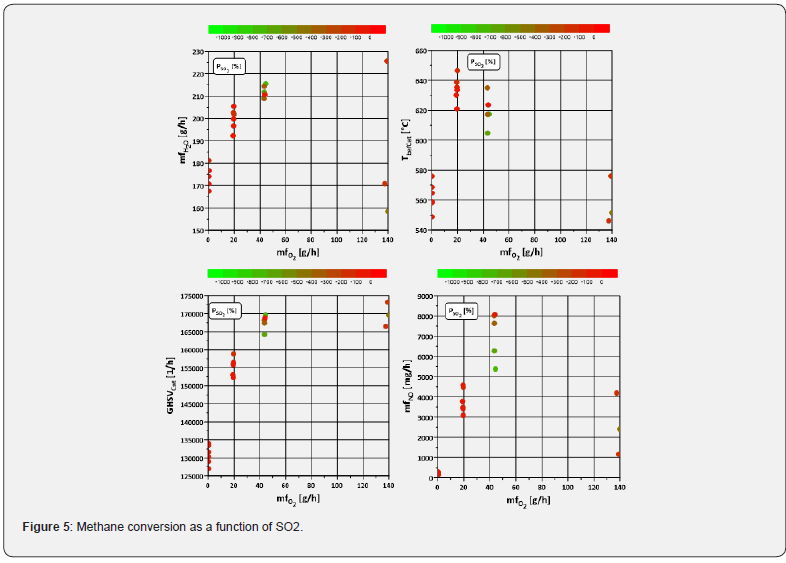
If deactivation phenomena occur, the question also arises as to which measures can be used to at least partially reverse them. One way of assessing the effectiveness of regeneration measures is the desorption of sulphur-containing species from the catalytic surface Lott et al. [11], which results in an increase in SO2 emissions downstream of the catalyst. Lott et al. [11] found that such desorption is enhanced in the presence of water and under rich conditions. (Figure 6) a seems to partially confirm this. In this diagram, the SO2 formation rate is shown as a function of the amount of O2 and H2O. The highest increase rates occur - except for one outlier - at higher water quantities, but under lean conditions, which is why it is still unclear whether and to what extent the SO2 formation in the catalyst is not due to post-oxidation of other sulphur species not detected here. There does not seem to be a clear influence of both temperature and space velocity, since in both cases both high and low formation rates occur in a similar range of values (Figure 6b,6c). Interesting could be a possible influence of NO, which is shown in (Figure 6d). It can be seen that higher NO emissions lead to higher rates of increase, whereby a maximum seems to be formed in the range between 5000 and 7000 mg/h. However, since increased water and nitrogen monoxide emissions occur simultaneously, as described above, it is also not entirely certain here whether the desorption-promoting effect can be attributed to only one of the two species.
Conclusion
The minimum in methane conversion in the range of λ = 1.1 could be due to several effects:
i. The results seen in this paper together with the findings from Sadokhina et al. [7] and Auvinen et al. [5] suggest that water inhibition is more powerful than the promoting effect of NO on methane conversion in the presence of H2O and SO2. So, following the discoveries from the literature, this deterioration means that the positive effects of NO (despite the high amounts) are overcompensated by water. The emissions occurring here in engine operation range from 500 to 4400 ppm for λ ≥ 1 and thus significantly exceed the proportions (up to 500 ppm) studied in Auvinen et al. [5], Gremminger et al. [6]; Sadokhina et al. [7].
ii. The influence of SO2 on methane conversion is minimal, as the measurement period may be too short to achieve a strong effect.
iii. One way of assessing the effectiveness of regeneration measures is the desorption of sulphur-containing species from the catalytic surface Lott et al. [11], which results in an increase in SO2 emissions downstream of the catalyst. The highest rates of increase occur - apart from one outlier - at higher water amount, but under lean conditions, which is why it is still unclear whether and to what extent the SO2 formation in the catalyst is not due to post-oxidation of other sulphur species not detected here.
The results show a strong influence of the operating strategy on PdO/Al2O3 catalysts. In order to avoid too high methane emissions, the slightly lean range would thus have to be left out and the engine would have to be operated outside of it.
Acknowledgements
Funding by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) Project ID 426888090-CRC 1441 (“TrackAct”) Project C5 is gratefully acknowledged.
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Ushakov Sergey, Stenersen Dag, Einang Per Magne (2019) Methane slip from gas fuelled ships: a comprehensive summary based on measurement data. Journal of Marine Science and Technology 24(4): 1308-1325.
- Howarth Robert W (2014) A bridge to nowhere: methane emissions and the greenhouse gas footprint of natural gas. Energy Science & Engineering 2(2): 47-60.
- Ciuparu Dragos, Lyubovsky Maxim R, Altman Eric, Pfefferle Lisa D, Datye Abhaya (2002) Catalytic combustion of methane over palladium-based catalysts. Catalysis Reviews 44(4): 593-649.
- Gélin Patrick, Primet Michel (2002) Complete oxidation of methane at low temperature over noble metal-based catalysts: a review. Applied Catalysis B: Environmental 39(1): 1-37.
- Auvinen Paavo, Kinnunen Niko M, Hirvi Janne T, Maunula Teuvo, Kallinen Kauko, et al. (2021) Effects of NO and NO2 on fresh and SO2 poisoned methane oxidation catalyst-Harmful or beneficial? Chemical Engineering Journal 417: 1385-8947.
- Gremminger Andreas Thomas, Pereira de Carvalho Hudson Wallace, Popescu Radian, Grunwaldt Jan-Dierk, Deutschmann Olaf (2015) Influence of gas composition on activity and durability of bimetallic Pd-Pt/Al2O3 catalysts for total oxidation of methane. Catalysis Today 258: 470-480.
- Sadokhina Nadezda, Smedler Gudmund, Nylén Ulf, Olofsson Marcus, Olsson Louise (2017) The influence of gas composition on Pd-based catalyst activity in methane oxidation- inhibition and promotion by NO. Applied Catalysis B: Environmental 200: 351-360.
- Gélin Patrick, Urfels Laetitia, Primet Michel, Tena Emmanuel (2003) Complete oxidation of methane at low temperature over Pt and Pd catalysts for the abatement of lean-burn natural gas fuelled vehicles emissions: influence of water and sulphur containing compounds. Catalysis Today 83(1-4): 45–57.
- Gholami Rahman, Alyani Mina, Smith Kevin (2015) Deactivation of Pd Catalysts by Water during Low Temperature Methane Oxidation Relevant to Natural Gas Vehicle Converters. Catalysts 5(2): 561-594.
- Escandón Lara S, Ordóñez Salvador, Vega Aurelio, Díez Fernando V (2008) Sulphur poisoning of palladium catalysts used for methane combustion: effect of the support. Journal of Hazardous Materials 153(1-2): 742-750.
- Lott Patrick, Eck Mario; Doronkin Dmitry E, Zimina Anna, Tischer Steffen, et al. (2020) Understanding sulfur poisoning of bimetallic Pd-Pt methane oxidation catalysts and their regeneration. Applied Catalysis B: Environmental 278: pp. 119244.
- Gremminger Andreas, Lott Patrick, Merts Menno, Casapu Maria, Grunwaldt Jan-Dierk, et al. (2017) Sulfur poisoning and regeneration of bimetallic Pd-Pt methane oxidation catalysts. Applied Catalysis B: Environmental 218: 833-843.






























