A Simple Method for Studying the Metallic Function Poisoning of Pt/Al2o3 Catalyst
Maria do Carmo Rangel1*, Francieli Martins Mayer1, Dino Sodré de Jesus1 and Genira Carneiro de Araújo1
1Instituto de Química, Universidade Federal do Rio Grande do Sul, Porto Alegre, RS, Brazil
2Instituto de Química, Universidade Federal da Bahia, Salvador, Bahia, Brazil.
Submission: September 01,2023; Published: September 25,2023
*Corresponding author: Maria do Carmo Rangel, Instituto de Química, Universidade Federal do Rio Grande do Sul, Porto Alegre, RS, Brazil, E-mail: maria.rangel@ufrgs.br
How to cite this article: Maria do Carmo R, Francieli Martins M, Dino Sodré de J, Genira Carneiro de A. A Simple Method for Studying the Metallic Function Poisoning of Pt/Al2o3 Catalyst. Eng Technol Open Acc. 2023; 5(4):555666. DOI: 10.19080/ETOAJ.2023.05.555666
Abstract
Alumina-supported platinum commercial catalysts usually deactivate with time due to poisoning and coke deposition. In naphtha reforming, for instance, the Pt-Re/Al2O3 catalyst seriously deactivates by coke and/or poisoning by sulfur. In addition, because of a new technology for oil drilling in deep water, silicon recently appeared as another poison to these catalysts. This is related to the injection of silicone in the wells, during oil drilling. Aiming to understand this poisoning, the influence of heating atmosphere on the poisoning of the metallic sites of alumina-supported platinum was studied in this work. This method may help the study of commercial catalyst poisoning in the laboratory. It was found that heating the contaminated catalysts (with methyl-substituted polyoxysilane, MSP) under both nitrogen and hydrogen flow , the metallic function deactivates, this effect being stronger in the case of nitrogen. This was assigned to the coverage of some platinum sites by silica under nitrogen flow. This methodology is more suitable to perform the silicon contamination in the laboratory to simulate the slow deactivation in industrial processes. Silicon was found to be a strong poison for Pt/2o3.
Keywords: Platinum Catalyst; Naphtha Reforming; Offshore Oil; Silicon; Poisoning
Introduction
Platinum catalysts with different supports (alumina, silica and others) and associated (or not) with other metals are widely used in chemical industries [1-3] and in the control of the emission of hydrocarbons and other pollutants [4]. The wide use of these catalysts is related to their high activity and selectivity in commercially important reactions such as hydrogenation, dehydrogenation, isomerization and oxidation, among others [1,5,6]. In all these applications, the catalysts are deactivated over the years, through different mechanisms, such as coke deposition, poisoning, incrustation and others, lead to a gradual and progressive loss of activity and/or selectivity [6-9].
One of the most severe deactivation problems has been noted over the commercial catalyst for the reforming of naphtha, an important fraction of petroleum, consisting mainly of C5-C12 paraffins, naphthenes, olefins, and aromatics. In this process, long-chain hydrocarbons are reconstructed through isomerization, hydrogenation, dehydrocyclization and dehydrogenation reactions, among others [6]. The reactions take place in acidic and/or metallic sites, requiring a bifunctional catalyst. The systems most used are Pt/2o3.
-Cl types, in which the metallic component has enough activity for hydrogenation and dehydrogenation reactions, while the support (chlorinated alumina) has sufficient acidity to promote isomerization reactions [6,7]. The cooperative action of the two sites promotes other reactions, such as dehydrocyclization, through a bifunctional mechanism [6-8]. Undesirable reactions also occur, which reduce the yield of the process, such as hydrocracking and hydrogenolysis, deactivating the catalyst by coke formation. This problem was significantly minimized by the introduction of a second and further a third metal, such as rhenium, iridium, germanium and tin [8,10,11] to the catalyst platinum monometallic. Bimetallic and trimetallic catalysts have shown greater resistance to coke deactivation and therefore are more stable under more severe operating conditions (higher temperatures and lower pressures).In industrial operations, a complex network of reaction occurs [12] during naphtha forming, favoring the catalyst deactivation by deposition of coke, sintering or poisoning by substances from the feed [6]. Coke production is by far the most important cause of the catalyst deactivation [6,13-17]. Both acid and metal sites catalyze coke formation, but the main amount is deposited over the acid sites [9,18]. The partial pressure of hydrogen and hydrocarbon, time on stream, gas–oil feed flow, and the reaction temperature are the main operation variables that affect the production of coke, as described by several authors [6,12-16].
The occurrence of sintering is mainly related to temperature, reaction atmosphere, kind and dispersion of the metal, promoters and impurities. The specific surface area, texture, and porosity of the support also affect the rates of sintering and redispersion [15,16]. For naphtha reforming catalysts, the most important problem of sintering is caused by the successive regeneration of the catalysts during coke burning, causing the slow sintering of the support, which can lead to catalyst deactivation over the years [6]. The other cause of the catalyst deactivation is the poisoning by impurities present in the feedstock or formed by secondary reactions during the process. It is known [8,10,12] that the poison is strongly chemisorbed on the active sites of the catalyst, causing a temporary or permanent decrease in the overall activity. Sulfur is probably the main poison for reforming catalysts, causing severe poisoning of the metal, but other impurities such as nitrogenous compounds, arsenic, sodium, copper, mercury and lead are also harmful. They can be irreversibly adsorbed and accumulated on the active sites of the catalyst [6,18]. Sulfur compounds are always present in the naphtha feedstock, requiring a hydrodesulfurization unit located upstream of the reforming reactor. The sulfur tolerance depends on the catalyst sensitivity, which is related with its composition (mono-, bi- and tri-metallic catalysts) and the presence of additives [6,8].
In recent times, new technologies have been proposed for oil drilling in deep waters, some of them injecting silicone into the wells. As a consequence, naphtha feedstock has been contaminated by small amounts of silicon (a few ppb). After some years, it was found that silicon also acts as a poison for Pt-Re/2o3 commercial catalysts in naphtha reforming. It was supposed that silicon compounds have been gradually and progressively deposited on the catalyst over the years, causing an irreversible deactivation. The poisoning finally led to the catalyst disposal, thus reducing its operating cycle in industrial plants. This drawback has aroused attention of the researchers [19] to understand the poisoning of reforming catalysts by silicon. An essential step for this study is the development of methodologies that can reproduce the poisoning of industrial catalysts, in the laboratory.
In a previous work [19], a new methodology for poisoning Pt/2o3 catalysts was developed under flow conditions. As far as we know, no other was carried out regarding silicon poisoning. In the present study, the poisoning was carried out under static conditions, aiming to find a faster and simpler method, which allows to simulate the industrial poisoning under conditions of less expenditure of time and reactants. With this goal in mind, the effect of heating atmosphere on the poisoning of the metallic function of industrial naphtha reforming was studied in this work. Pt/2o3 catalyst was used as a model system for naphtha reforming catalysts. As a poison, substituted methyl polyoxysiloxane (MPS) was used, which has a structure similar to the compounds used in oil drilling.
Experimental
The catalysts were prepared by impregnating a commercial support (γ -2o3, HIQ 20 Extruded TRILOB 1/16”, φ <100) with a platinum precursor. Alumina (10g) was calcined at 500°C for 2h, under air flow, and then dispersed in 21.84 mL of 0.4 mol. L-1 solution of chloroplatinic acid (H2PtCl6), 1 mL of hydrochloric acid and 5.2 ml of water. This suspension was kept in a rotary evaporator, for 6h, at room temperature. The system was then vacuum filtered and dried at 120°C, for 12h. The solid (PS sample) was then calcined under air flow (50 mL min-1) for 2h, to get platinum oxide supported on alumina.
The catalyst thus obtained was contaminated by silicon, using methyl polyoxysiloxane (MPS) as a model compound for the silicone injected in the wells. During contamination, 10g of the sample was dispersed in a solution of 3.4 g of substituted methyl polyoxysiloxane (MPS) in 34 mL of n-hexane, keeping the system under stirring, in a rotary evaporator, for 24h. The solid was dried in at 60°C, for 24h, producing the PC sample. A part of the material was heated at 500°C, for 2h under nitrogen flow (50 mL min-1), generating the PCN sample, and the other part was heated in the same conditions (500°C, 2h) but under hydrogen flow, producing the PCH sample.
The catalysts were characterized by Fourier transform infrared spectroscopy (FTIR), chemical analysis, specific surface area measurements, and temperature programmed reduction (TPR). The activity of the metallic sites was evaluated in the cyclohexane dehydrogenation, a typical reaction of metal sites [14]. The effectiveness of the contamination was evaluated by FTIR. The spectra were obtained in the range of 4000 to 400 cm-1, using potassium bromide discs (0.05 g) and a model Valor-III Jasco equipment. The amount of silica and platinum in the catalysts was determined by inductively coupled plasma atomic emission spectroscopy (ICP-AES), using an ARL 3410 mini torch equipment. The solids were previously digested in a mixture of 1 mL of nitric acid, 3 mL of hydrochloric acid and 1 mL of sulfuric acid. The specific surface areas of the catalysts were measured using a TPD/ TPR 2900 Analyzer model Micromeritics equipment and the BET method. The sample (0.05 g) was heated under nitrogen flow up to 150°C and kept at this temperature for 1h. The measurements were carried out under a flow of a 30% N2/He mixture at liquid nitrogen temperature. The effect of the poisoning on the platinum reduction was studied by temperature programmed reduction using a TPD/TPR 2900 Analyser model Micromeritics equipment. In the experiments, the hydrogen consumption from a 5% H2/ N2 mixture was monitored, while the sample (0.15 g) was heated (10°C min-1) from 30 to 1000°C.
The catalysts were evaluated in cyclohexane dehydrogenation to benzene in a micro stainless steel fixed bed reactor operating at 315°C, 1 atm and WHSV=30 L-1. During the experiments, a stream of hydrogen (100 mL min-1) was flowed through a saturator, containing cyclohexane to obtain a ratio H2/cyclohexane=7 and then was fed to the reactor [20]. Prior the reaction, the sample (0.10g) was reduced under hydrogen flow, at 500°C, for 2h. The reactor effluent was analyzed by on line gas chromatography using a Varian 3400 CX chromatograph equipped with an FID and a packed column of FFAP on Chromosorb. The average error of the measurements was less than 3%.
Results and Discussion
(Figure 1) shows the FTIR spectra for the samples. For all cases, we can see a broad band at 3400 cm-1 and another one at 1600 cm-1, which are related to stretching and deformation vibrations, respectively, of O-H bonds in adsorbed water [21].
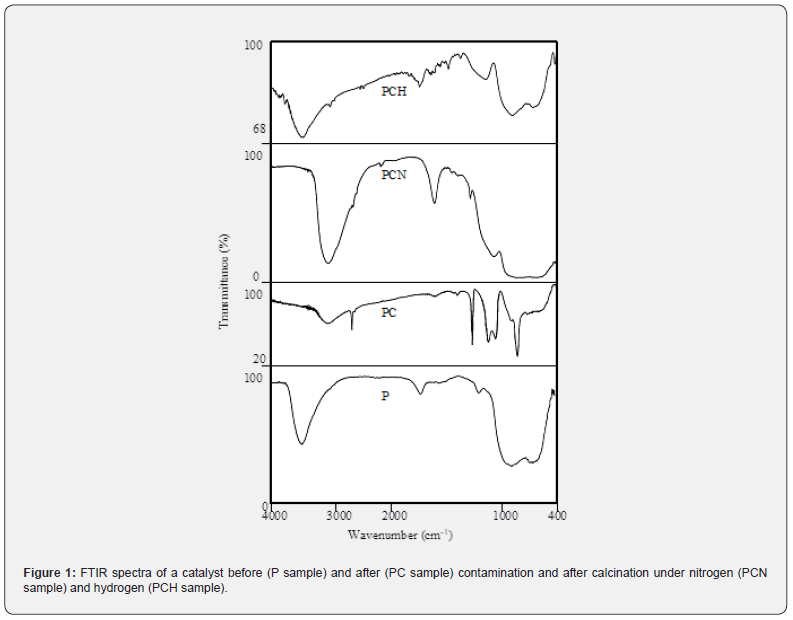
The sorption of silicone by the samples was confirmed from the characteristic bands of silicon-carbon and silicon-oxygen bonds. The species linked to silicon, such as the methyl group, Si(CH3)2, were identified by the band at 1262 cm-1, in addition to groups containing oxygen, such as Si-O-Si (1097 cm-1) [21]. These species are typical of the silicone used in the experiments, whose structure has methyl groups in addition to Si-O-Si bonds, which characterizes the compound. After heating, the silicon bands changed, indicating the transformation of organic silicon compounds. For the sample calcined under hydrogen flow, the band at 1262 cm-1 related to Si(CH3)2 groups, became broader and was shifted to lower wavenumbers, overlapping with the band at 1097 cm-1 (Si-O-Si bonds). On the other hand, the band below 1000 cm-1, typical of metal-oxygen bonds i2n metal oxides [22], remained unaffected as compared to fresh catalyst. These findings suggest that no silicon oxide was formed in the catalyst. However, for the catalyst calcined under nitrogen flow, the band at 1262 cm-1 decreased while the other bands overlapped, producing the enlargement of the broad band below 1000 cm-1, which assumes the typical shape of the bands of Si-O bonds in silica [22] bonded to alumina (Figure 1) surface, through OH groups, according to the scheme shown in (Figure 2), in agreement with a previous work [23]. Under heating, some CH3 groups are eliminated, and the reminiscent silicon remains as organic compounds (PCH sample) or as silica (PCN). From these results, one can conclude that silicon is present mostly as organic compounds in PCH sample while for the PCN sample it is mostly as silica.

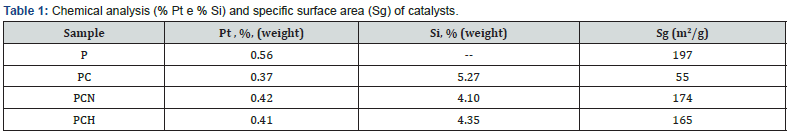
The chemical analysis results displayed in (Table 1) show that heating removes part of silicon in agreement with the FTIR spectra. Also, more silicon remained in the sample heated under hydrogen flow. The amount of platinum decreased indicating that some metal is loss during contamination. The specific surface area of the catalyst (P sample) largely decreased (PC) after contamination, as shown in (Table 1). As alumina is a non-porous solid, it is expected that its interparticle pores can accommodate the silicon compounds, which then are able to act as a ligand among the particles favoring their agglomeration and then decreasing the specific surface areas. However, during heating the specific surface areas are recovered, the differences being within the experimental error.
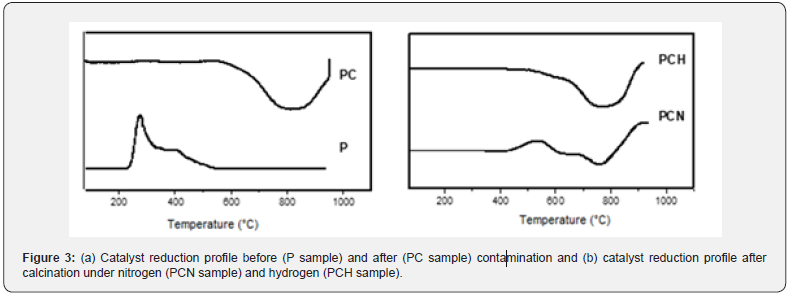
(Figure 3a) shows the TPR curves of the catalyst before and after contamination. Before contamination (P sample), the catalyst showed a typical reduction profile of platinum supported on alumina with a peak at 250 oC, assigned to platinum reduction [10]. The other peak at 360oC is related to platinum particles in strong interaction with the support [24]. After contamination, only one negative peak was noted at higher temperatures, related to hydrogen release from silicon compounds decomposition. It is probable that platinum could be reduced at lower temperatures, but no consumption was detected because of hydrogen generated by the decomposition of silicon compounds. After all platinum had been reduced a negative peak appeared related to the release of hydrogen from silicone. After calcination at 500oC, under nitrogen or hydrogen, the reduction patterns varied depending on the heating atmosphere, as shown in (Figure 3b). For the PCH sample, the profile was similar to PC sample. For the other catalyst, a peak at 540oC was noted, related to platinum in strong interaction with alumina and silica. At higher temperatures, the decomposition of silicon compounds occurs resulting in a negative peak.
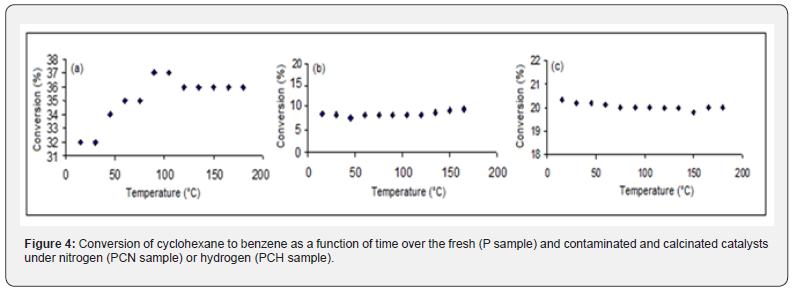
The conversion profiles, obtained in the dehydrogenation of cyclohexane, as a function of time over the fresh and contaminated catalysts are shown in (Figure 4). For the fresh catalyst, the values increase slowly at the beginning of reaction, reaching steady values after 100 min. On the other hand, contaminated catalysts lead to stable values since the beginning of reaction. Regardless the heating atmosphere, silicon decreased benzene conversion, indicating that silicon poisons the metallic sites. In addition, nitrogen atmosphere led to a stronger deactivation compared to hydrogen. In addition to the lower amount of platinum in the sample heated under nitrogen, this suggests that the release of organic silicon compounds at lower temperatures produces silica (hard to remove), which may cover some platinum atoms. In the case of catalysts heated under hydrogen, most of these compounds remain in organic form and are eliminated only in the reduction step that precedes the reaction. It is concluded, therefore, that silica promotes greater deactivation of metallic sites than organic silicon compounds.
Conclusion
In the development of contamination systems for modelling deactivation of naphtha catalysts, the use of hydrogen or nitrogen for heating the catalyst leads to different catalysts. Heating the catalysts under nitrogen flow is efficient for removing organic silicon compounds but silica is produced, and the activity of the catalyst is poor. Silica is difficult to remove and is supposed to cover some platinum sites. On the other hand, the catalyst heated under hydrogen showed no silica and then the loss of activity was slower. Because of this, the last methodology is more suitable to carry out the silicon contamination in the laboratory to simulate the slow catalyst deactivation in industrial processes. Silicon was found to be a strong poison for Pt/2o3 catalysts. In spite of the importance of silicon poisoning in industrial operations of naphtha reforming, very few studies were reported in the open literatures. Future research dealing with a real mixture used in the oil drilling mainly in deep waters is much needed to assure that this process will not damage the catalysts used downstream.
Acknowledgements
The authors are grateful for financial support from CNPq and FINEP. DSJ thanks CNPq for the scientific initiation scholarship. GCA thanks the RHAE/CNPq program for the grant.
Conflict of Interest
The authors declare no conflict of interest.
References
- Barnard AS and Chang LY (2011) Thermodynamic Cartography and Structure/Property Mapping of Commercial Platinum Catalysts. ACS Catal 1(2): 76-81.
- Li Z, Cui Y, Wu Z, Milligan C, Zhou L, et al. (2018) Reactive metal-support interactions at moderate temperature in two-dimensional niobium-carbide-supported platinum catalysts. Nature Catalysis 1: 349-355.
- Ballarini AD, Virgens CF, Rangel MC, De Miguel SR, Grau JM (2019) Characterization and behavior of Pt Catalysts supported on basic materials in dry reforming of methane. Brazilian Journal of Chemical Engineering 36(1): 275-284.
- Sun S, Jin C, He W, Li G, Zhu H, et al. (2022) A review on management of waste three-way catalysts and strategies for recovery of platinum group metals from them. Journal of Environmental Management 305: 14383.
- LS Carvalho, Conceição KCS, Mazzieri VA, Pieck CL, Rangel MC, et al. (2012) Pt-Re-Ge/Al2O3 catalysts for n-octane reforming: Influence of the order of addition of the metal precursors. Applied Catalysis A: General 419-420: 156-163.
- JM Parera, NS Fígoli (1995) Catalytic Naphtha Reforming. In: Antos GJ, Aitani AM, Parera JM (Eds.), Marcel Dekker, New York, 45-78.
- Barbier J (1986) Deactivation of reforming catalysts by coking-a review. Applied Catalysis 23(2): 225-243.
- LS Carvalho, LC Pieck, MC Rangel, NS Fígoli, JM Parera (2004) Sulfur Poisoning of Bi- and Trimetallic Gamma-2o3 Supported Pt, Re and Sn Catalysts. Industrial & Engineering Chemistry Research 43(5): 1222-1226.
- SM Domingues, JM Brito, De Oliveira AS, Valentini A, Reyes P, et al. (2001) Coke Formation on H-Mordenite Catalyst during the Benzene Transalkylation with C9+ Aromatics. Studies in Surface Science and Catalysis139: 45-52.
- Carvalho LS, Pieck CL, Rangel MC, Fı́goli NS, Grau JM, et al. (2004) Trimetallic naphtha reforming catalysts. I. Properties of the metal function and influence of the order of addition of the metal precursors on Pt-Re-Sn/γ-Al2O3-Cl. Applied Catalysis A: General 269(1-2): 105-116.
- LS Carvalho, CL Pieck, MC Rangel, NS Fı́goli, CR Vera, et al. (2004) Trimetallic naphtha reforming catalysts: II. Properties of the acid function and influence of the order of addition of the metallic precursors on Pt-Re-Sn/γ-Al2O3-Cl. Applied Catalysis A: General 269(1-2): 105-116.
- Chen Z, Wang Y, Li Y, Wu J, Zhang L (2023) Explicit molecule-based reaction network simplification: Theory and application on catalytic reforming. Chemical Engineering Science 277: 118833.
- Rahimpour MR, Jafari M, Iranshahi D (2013) Progress in catalytic naphtha reforming process: A review. Applied Energy 109: 79-93.
- Richardson JT (1992) Principles of Catalyst Development. Springer Science+Business Media, New York, pp.185-224.
- Farauto RJ, Bartolomew CW (1997) Fundamentals of Industrial Catalytic Processes. Champamn & Hall, London, pp. 265-334.
- MD Argyle, CH Bartholomew (2016) Heterogeneous Catalyst Deactivation and Regeneration: A Review. In: Bartholomew H, Argyle MD (Eds.), MDPI, Basel, 1-128.
- Meng S, Li W, Xu H, Li Z, Li Y, et al. (2021) Non-thermal plasma assisted catalytic reforming of naphtha and its model compounds with methane at near ambient conditions. Applied Catalysis B: Environmental 297: 120459.
- J Barbier (1985) Deactivation and Poisoning of Catalyst. In: Oudar J, Wise H (Eds.), Marcel Dekker, New York, 109-150.
- Souza MOG, Reyes P, Rangel MC (1999) Silicon poisoning of Pt/Al2O3 catalysts in naphtha reforming. Studies in Surface Science and Catalysis 126: 469-472.
- B Ayo, BA Susu, (1988) Platinum-rhenium/alumina catalyst III. Mechanism of C6-ring hydrogenation/dehydrogenation reactions. Applied Catalysis 40: 1-16.
- Vander Maas JH (1969) Basic Infrared Spectroscopy. Heyden & Son, Great Britain, UK.
- Niquist RA, Kagel RO (1971) Infrared Spectra of Inorganic Compounds. Academic Press, Orlando, USA, PP. 1-500.
- Beguin B, Garbowiski E, Primet M (1991) Stabilization of alumina toward thermal sintering by silicon addition. Journal of Catalysis 127(2): 595-604.
- Lietz G, Lieske H, Spindler H, Hanke W, Völter J (1983) Reactions of platinum in oxygen- and hydrogen-treated Ptγ -Al2O3 catalysts: II. Ultraviolet-visible studies, sintering of platinum, and soluble platinum. Journal of Catalysis 81(1): 17-25.






























