A Review on Microwave Assisted Advanced Oxidation Process for Environmental Applications
Sridevi V*
Department of Chemical Engineering, Andhra University, India
Submission: February 11, 2020; Published: March 12, 2020
*Corresponding author:Sridevi V, Professor, Department of Chemical Engineering, AU College of Engineering, Andhra University, Visakhapatnam, 530003, Andhra Pradesh, India
How to cite this article:Sridevi V. A Review on Microwave Assisted Advanced Oxidation Process for Environmental Applications. Eng Technol Open Acc. 2020; 3(3): 555615. DOI: 10.19080/ETOAJ.2020.03.555615
Introduction
Presently, environmental deterioration, or the ecological crisis is the most serious problem faced by mankind. A major source of contamination is accidental spillage, or otherwise, on soils and waters, as exemplified by (sometimes) questionable manufacturing practices. Disposal of toxic substances, residues, solvents and the like are the major issues that industries and commercial establishments face continually. Patterson has noted that some 10.2 million metric tons of toxic substances were released into the environment in 1987 [1]; Unregulated and unsatisfactory disposal methods present long term unknown consequences of pollutants to health and to Earth’s ecology. Unfortunately, an ignorant chase for industrial and technological developments has created many environmental issues like global warming, loss of global diversity, forests and energy resources, depletion of the ozone layer, acid rain, etc. The more conventional methods of liquid/solid extraction, reverse osmosis, air stripping and bio-treatment or granular activated carbon can remove several toxic organic substances from polluted sites. However, despite claims to the contrary, these classical methods simply dispose the problem only to create a waste in a different ecosystem.
For example,
a) air stripping removes VOCs only to discharge them into the atmosphere;
b) reverse osmosis generates a reject stream of concentrated contaminants that require further treatment;
c) granular activated carbon requires either costly regeneration (through incineration) or burial in landfills;
d) liquid/solid separation methods generate sludge that also requires further disposal treatments [2].
Novel methods of water purification have moved from phase transfer processes to chemical and photochemical destruction of contaminants through ozonation for example. In this regard, utilization of engineered microbubble systems with ozone has proven to be a powerful method to decompose effectively chlorinated ethylenes (e.g. tetrachloroethylene, TCE) in situ to harmless HCl and CO2 [3].
Some of the common AOPs which have potential of application in environmental field include: ozonation, ultrasound, photocatalysis, Fenton, wet air oxidation, microwave radiation, ultraviolet radiation and hydrogen peroxide oxidation. AOPs are important tools for environmental technology, so they have to be placed on more sound scientific and engineering basis. However, the knowledge about the exact mechanisms of AOPs is still incomplete. The most difficult problem is how to choose or design the most efficient AOPs system for the given pollutant. So, the reaction mechanisms, efficiency improvement, and their mathematical modeling will be the key subjects of the future research. Some investigators called AOPs as ‘‘cold combustion’’. Just because, similar with combustion, AOPs is a kind of oxidation process which could oxidize and mineralize the organic matter under mild conditions. But we tried to discover more similarities between them and achieve some enlightenment which is helpful to analyze, predict, understand and improve the oxidation efficiency of AOPs. Here we report a similar phenomenon exist in the AOPs: induced effects, which is a general property of combustion reaction. In ancient times, human ancestors drilled wood to make fire. As a Chinese saying goes, ‘‘a single spark can start a prairie fire’’. Both of them mean that combustion can be induced by a small fire. Our experimental results indicated that AOPs may also have induced effects. The following experimental findings directly or indirectly reflect the induced effects of AOPs from different angles.
Hazardous organic wastes from industrial, military, and commercial operations represent one of the greatest challenges to environmental engineers. Advanced oxidation processes (AOPs) [4-7] are alternatives to the incineration of wastes, which has many disadvantages. Treatment options include biological treatment, mechanical treatment by ultrafiltration, treatment with active carbon filters, and reverse osmosis using disc tube module technology. It was observed that the majority of these approaches were only useful to transfer pollutants from aqueous to another phase; thus, generating pollution, which requires further processing of solid-wastes and regeneration of the adsorbent, which adds more cost to the process. Some other methods like, microbiological or enzymatic decomposition, biodegradation, ozonation, and advanced oxidation processes such as Fenton and photo- Fenton catalytic reactions, H2O2/UV processes, etc. have also been used for the removal of various contaminants from wastewaters [8,9]. In the last few decades, photocatalytic degradation processes have been widely employed for the destruction of organic pollutants in wastewater and effluents. Advanced oxidation processes (AOPs), have been developed as an emerging destruction technology resulting in the total mineralization of most of the organic contaminants. In the last few decades, photocatalytic degradation processes have been widely employed for the destruction of organic pollutants in wastewater and effluents. Advanced oxidation processes (AOPs), have been developed as an emerging destruction technology resulting in the total mineralization of most of the organic contaminants (Figure 1).
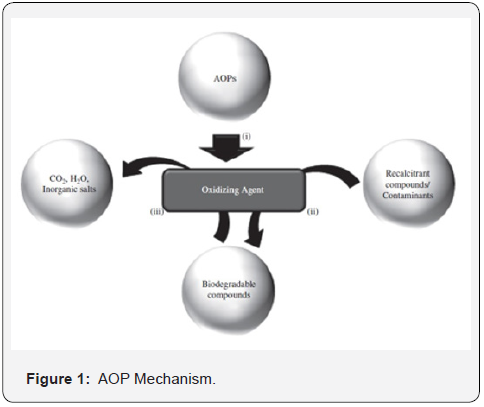
Advanced oxidation involves these basic steps:
i. The first step involves the formation of strong oxidants like •OH, HO2•, O2- etc.
ii. In the second step, these oxidants react with organic contaminants present in the Waste water converting them into biodegradable compounds.
iii. The last step is the oxidation of these biodegradable intermediates leading to complete
iv. Mineralization in water, carbon dioxide, and inorganic salts.
AOPs have many advantages over other traditional methods for the treatment of wastewater:
i. They have high reaction rates.
ii. They have a potential to reduce toxicity and completely mineralize organic contaminants
iii. They do not concentrate waste for further treatment like methods using membranes or activated carbon absorption.
iv. They do not create sludge as in the case of physical, chemical, or biological processes.
v. Their nonselective pathway allows for the treatment of different organics at a time.
vi. They are capital intensive installation; cost is relatively low as their chemistry can be tailored to have a specific application.
Microwave Radiation in AOP
Microwave radiation has found its niche in several domestic, industrial and medical applications. Interesting studies have explored applications of microwave radiation
a) in organic syntheses, as well as in polymerization and dehydration processes;
b) in inorganic syntheses by ceramic calcinations and solidification;
c) in environmental water treatments;
d) in safety and biological aspects;
e) in analyses and extraction processes; and
f) not least in food sterilization.
Microwaves can also decompose nondegradable materials from wastes. Microwave plasma has been used for etching materials, in chemical vapor deposition, and in surface processing of polymers and semiconductors, among others. It has even found a niche in the removal of NOx gases and in the degradation of chloro- and nitro-aromatics in an electrohydraulic discharge [10].
Microwave energy is being extensively used for the thermal processing of different kind of materials and products through heating, drying and pasteurizing of food and chemicals, sintering of ceramics, petroleum refining and many more things. Microwave is a non-ionizing radiation, and an integral segment of electromagnetic radiation lies in the frequency range of 300 MHz– 300 GHz. Microwave radiation has been developed and produced during World War-II and primarily used for the heating purposes only. However, with the time and research, it became famous for chemical transformations too. The extensive use of microwave ovens appeared after 1970s due to the emergence of improved and simplified magnetron that minimized its costs. Generally, industrial, commercial and domestic microwave ovens work at 2.45 GHz frequency as they are made to process food items having moisture/water and water absorbs a maximum of microwave irradiation at this frequency. The absorption of microwave radiation within the material increases the temperature of the substrate due to the conversion of absorbed microwave radiation into heat energy. A brief pictorial representation of the mechanism of microwave-induced heating of aqueous substrate is also illustrated with the essential steps involved in this process (Figure 2).
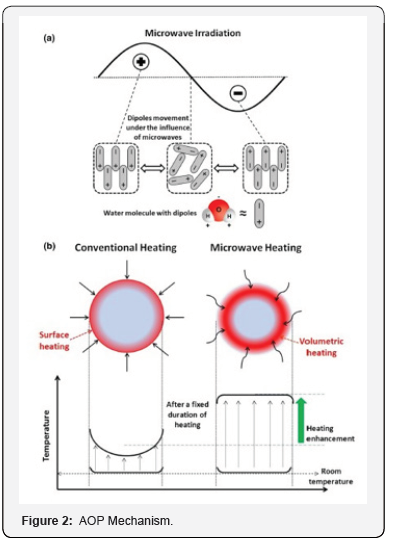
A. Mechanism of microwave-induced heating that involves several steps:
i. Microwaves penetrate through the substrate and create changing electric field.
ii. The polar molecules or dipoles such as water molecules repeatedly try to align with the external electric field of microwave.
iii. The resultant movement of dipoles causes some molecular frictions and time delay in dipoles that ultimately result in the heat generation.
iv. The generated heat used to be uniform depending on the uniformity of dipoles and microwave penetration depth of the medium.
B. Performance enhancement in microwave heating over conventional heating or surface heating methods. AOPs are to be important tools for environmental technology and they must be placed on more sound scientific and engineering basis.
Conclusion
This review acknowledged the applicability of microwave coupled advanced oxidation process-based techniques that offer a great edge over conventional methods used for the treatment of organic pollutants. The assistance of microwave to several advanced oxidation processes helps to speed up the process that improves the mineralization efficiency of the treatment method as well. Thus, microwave-enhanced processes coupled with advanced oxidation processes offer an environmentally benign and secured disposal of industrial dye waste. Moreover, the mechanism of action of various homogeneous and heterogeneous microwaveenhanced advanced oxidation processes with their advantages and limitations has also been elaborated with future directions of sustainable development in the microwave-enhanced advanced oxidation processes.
References
- Patterson JW (1989) Environ. Sci. Technol 23: 1032-1038.
- Zeff JD (1990) Ultrox operating experiences with UV/oxidation: Economics, Ultrox International, Santa Ana, CA, personal communication to N Serpone.
- Kerfoot WB (2003) ACS Symp. Ser 837: 86-108.
- Munter R (2001) Advanced oxidation processes: current status and prospects. Proc Estonian Acad Sci Chem 50(2): 59-80.
- Enoch SJ (2010) Chemical category formation and read across for the prediction of toxicity. In: Puzyn T, Leszczynski J & Cronin MTD (Eds.), Recent Advances in QSAR Studies: Methods and Applications. Springer, London.
- Oturan MA, Oturan N, Lahitte C, Trevin S (2001) Production of hydroxyl radicals by electrochemically assisted Fenton’s reagent: Application to the mineralizationof an organic micropollutant, pentachlorophenol. J Electroanal Chem 507: 96-102.
- Comninellis C, Pulgarin C (1991) Anodic oxidation of phenol for wastewater treatment. J Appl Electrochem 21: 703-708.
- Glaze WH, Kang JW, Chapin DH (1987) The chemistry of water treatment processes involving ozone, hydrogen peroxide and UV-radiation. Ozone Sci Eng 9(4): 335-352.
- Techcommentary (1996): Advanced Oxidation Processes for Treatment of Industrial Wastewater. An EPRI Community Environmental Center Publ.
- The UV/Oxidation Handbook (1994) Solarchem Environmental Systems, Markham, Ontario, Canada.






























