Identifying Safe Operating Conditions for Chemical Reactors and Storage Facilities for Potentially Hazardous Chemical Products
Sharikov IV* and Sharikov FI
Saint Petersburg Mining University, Russia
Submission: August 31, 2019; Published: November 07, 2019
*Corresponding author:Sharikov IV, Saint Petersburg Mining University, Saint Petersburg, Russian Federation, Russia
How to cite this article: Sharikov IV, Sharikov FI. Identifying Safe Operating Conditions for Chemical Reactors and Storage Facilities for Potentially Hazardous Chemical Products. Eng Technol Open Acc. 2019; 3(2): 555609. DOI: 10.19080/ETOAJ.2019.03.555609
Abstract
In the Article the main sources of danger in the operation of technological equipment are considered. The main sources of hazards have been identified - this is the problem of the emergence and development of a thermal explosion during the operation of technological equipment and storage facilities. The basic conditions for the analysis of the occurrence and development of a thermal explosion are derived based on an analysis of the equations of thermal and material balance in reactors and other equipment, where exothermic reactions can occur.
Equations are obtained for a different mechanism of heat energy transfer, due to conduction, and convection, both forced and natural. The basics of numerical methods for solving the corresponding equations in partial derivatives are considered.
Introduction
During the operation of petrochemical complexes, various hazards arise associated with the possibility of the release of hazardous chemical products into the environment. To combat the harmful consequences of such situations, it is necessary to analyze the main situations that give rise to such situations. For solving this problem, we will consider the main situations capable of leading to the depressurization of chemical reactors and storage of fire and explosive substances and the formation of emissions of harmful substances into the environment. The starting materials, capable under certain conditions, lead to the destruction of reactors and storage facilities and the release of the contents into the environment, can be in a solid, liquid, or gaseous state. Sources of depressurization of chemically reactors and storage facilities may be external, as a result of the harmful effects of atmospheric conditions on storage material. They can also be internal, due to the processes of decomposition of stored substances with the release of a large amount of heat and gaseous decomposition products. The flow of exothermic processes in a closed volume leads to the emergence and development of a so-called thermal explosion. The rate of heat release during the process per unit volume of several exothermic reactions can be described by the following kinetic equation:
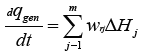
The heat dissipation rate will be equal to the value:
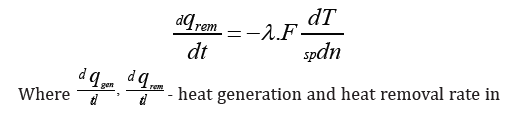
The heat dissipation rate will be equal to the value:
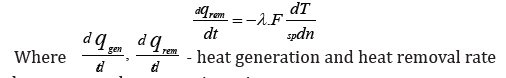
in element are volume reacting mixture
wjrate of j-й reaction kmol/(кмоль/m3·min)
ΔHjheat effect of j-th reaction /kJ/mol
dndT temperature gradient on normal to surface, covering the selected elementary volume, К/м
Fsp Specific surface, covering the allocated elementary volume, m2 / m3
Considering the elementary volume in the form of a sphere, a cylinder and a flat plate, we can obtain a generalized equation of heat and mass transfer in the following form:
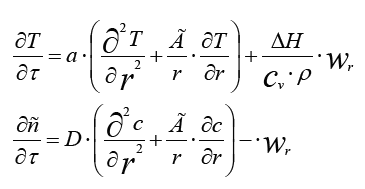
Here Г is a factor of a geometric form. Г = 0 for the plate, Г = 1 for the cylinder, and Г = 2 for the sphere. If the cylinder takes into account the distribution of heat along the axis of the cylinder, then this is equivalent to an additional plane flow along the axis, which must be added to the other two terms describing the change along the radius. As a result, for this case, we obtain the following system of equations models:
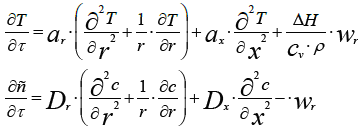
Where ar and ax are the coefficients of thermal conductivity along the radii r and along the longitudinal axis x;
Dr and Dx are the diffusion factors along the radii and along the longitudinal axis.
Dr and Dx are the diffusion coefficients along the radius r and along the longitudinal axis x.
The Solution of the Obtained Equations of the Mathematical Model
The equations of the model obtained above are suitable for describing processes in solid materials or in highly viscous liquids, when the transfer of heat and mass by the movement of liquids can be neglected.
The equations should be supplemented by initial and boundary conditions, depending on the conditions of interaction of the apparatus with the environment.
To solve the obtained equations, the ThermEx software package can be used, which implements numerical grid methods for solving partial differential equations by the grid method with a variable grid step [1-3] This allows you to change the step near the borders to increase the accuracy of the solution. The following pictures show the dialog images when creating a calculation task and the results obtained using one of these (Figures 1-4).
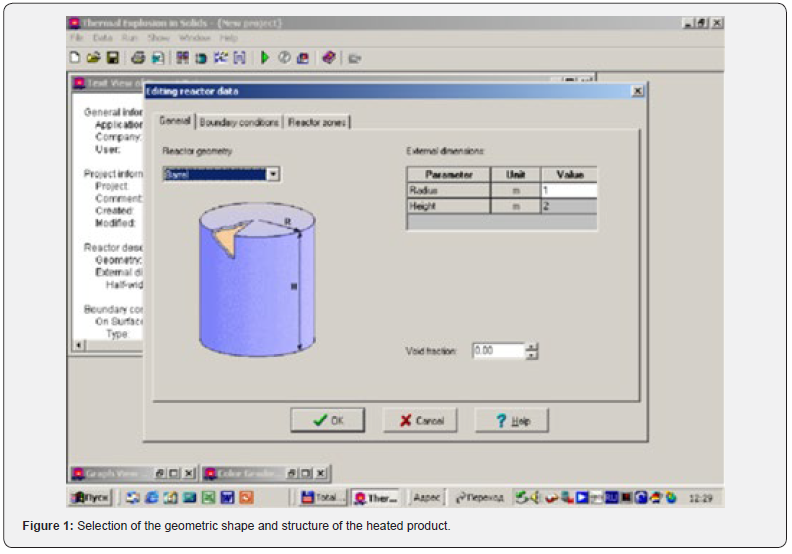
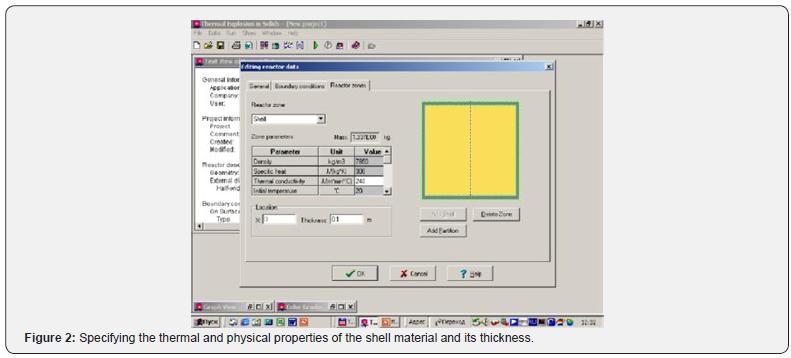
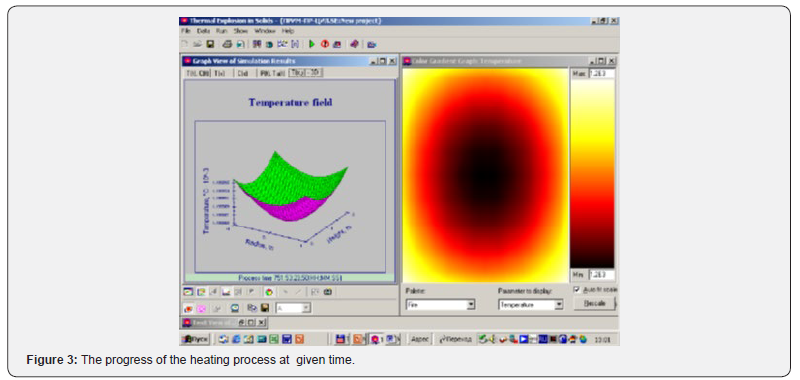
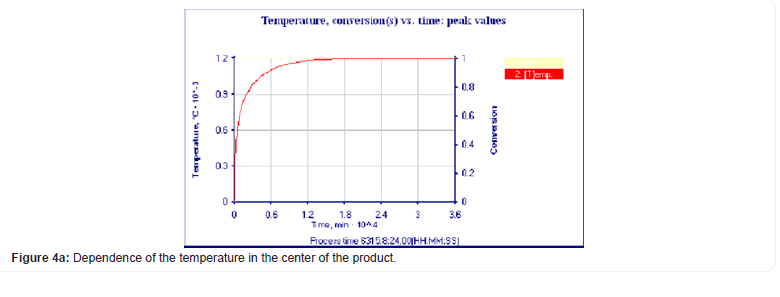
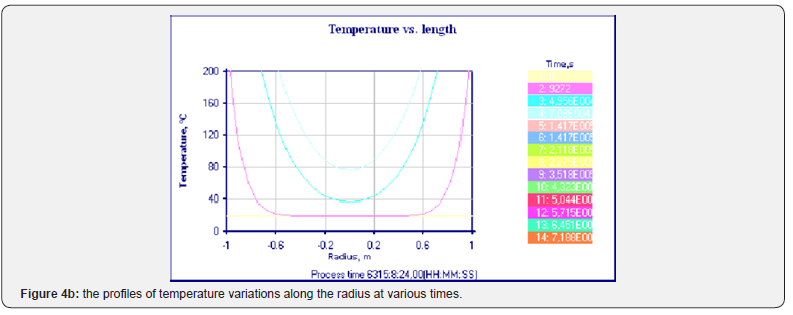
Using the Thermex software package, you can determine safe working conditions for solids and highly viscous liquids. When working with low-viscosity liquids, it is necessary to take into account convective transport, which leads to a decrease in temperature and concentration gradients, and therefore to a slowdown in the processes of thermal explosion [4]. Therefore, Thermex software package can be used to determine the boundaries of thermal stability with some safety margin. To more accurately determine the conditions for the onset and development of a thermal explosion in well-flowing liquids, it is necessary to use the Convex software package, which uses a mathematical model that takes into account heat and mass transfer due to natural convection based on Boussinesq approximations [5,6]. Figures 5-8 show changes in the temperature’s fields taking into account natural convection [7].
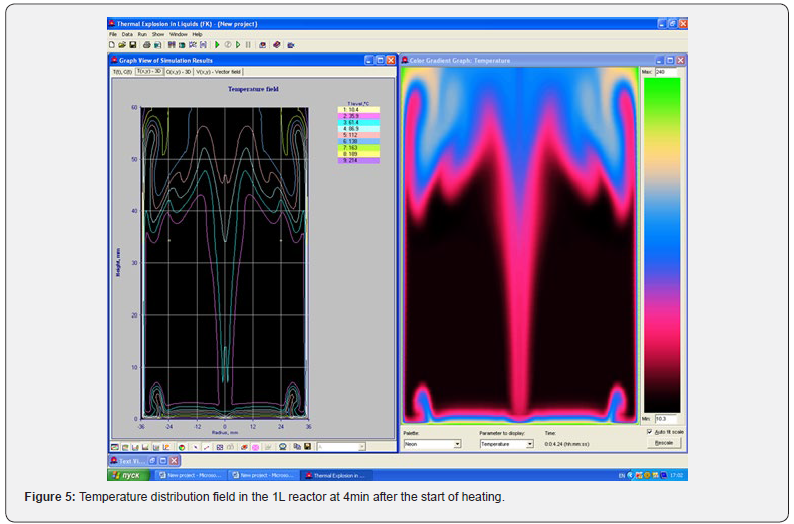
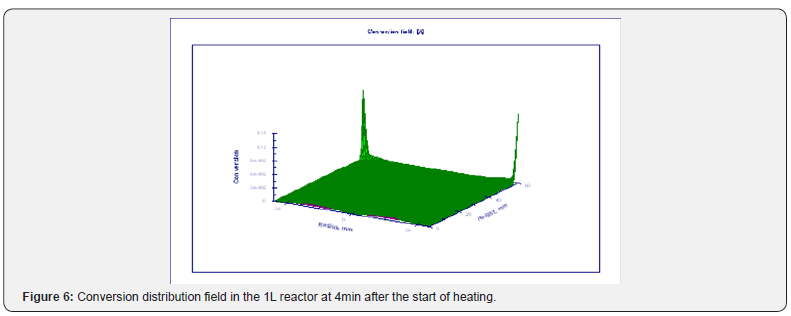
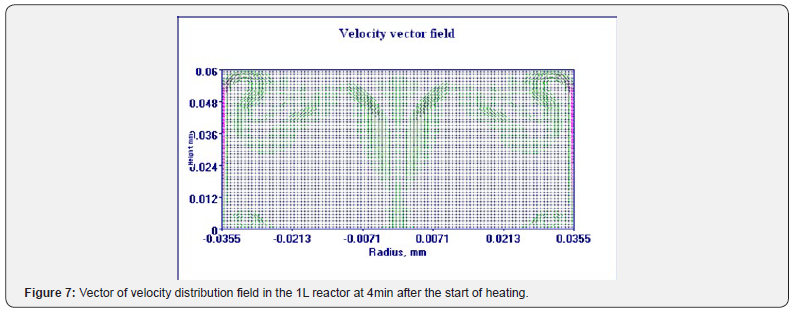
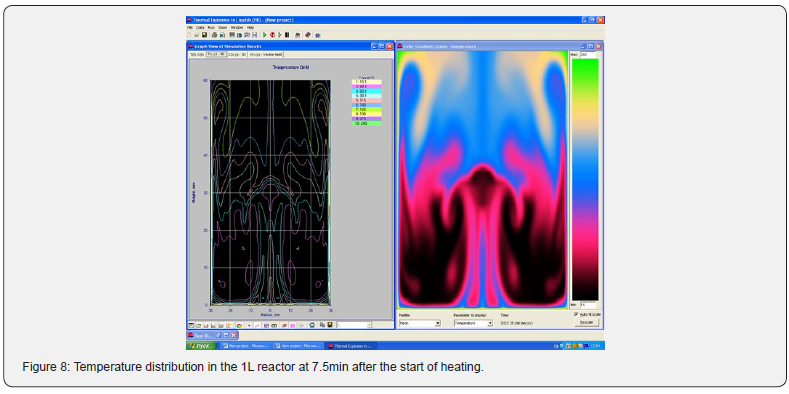
But in this way, it is possible to determine only the conditions for the onset of temperature rise, but not the development of the process. With the development of the process, evaporation of the liquid phase begins, a two-phase state arises, which is already described by other equations. However, to determine safe operating conditions, it is important to determine precisely the beginning of a thermal explosion and therefore the mathematical model, taking into account natural convection using the Boussinesq approximation, is quite suitable for these purposes.
Conclusion
1. Mathematical models that take into account the processes of heat and mass transfer taking into account internal sources of heat and mass due to chemical reactions allow us to determine the safe working conditions of chemical reactors and storages of potentially dangerous chemicals.
2. Thermex and ConvEx software packages created on the basis of grid calculation methods allow us to determine the safe working conditions for reactors and storages of potentially hazardous chemicals in solid and liquid states.
References
- Yu V Sharikov, Beloglazov IN (2011) Modeling of systems. Part 1. Synthesis of models of technological objects on the basis of equations of hydrodynamics and chemical kinetics. Petersburg Mining University, Russia.
- Yu V Sharikov, Beloglazov IN (2012) Modeling of systems. Part 2 Methods of numerical implementation of mathematical models. Petersburg Mining University, Russia.
- Benin AI, Kosoy AA, Sharikov YuV (1990) Automated system for studying the thermal explosion of chemical-technological processes. Journal of the All-Union, Сhemical Society N4: 428-433.
- Benin AI, Kossoy AA, Smikalov Pyu (1992) Automated system of kinetic research in thermal analysis. I. General description of automated system. Journal of Thermal Analysis 38(5): 1151-1165.
- Benin AI, Kossoy AA, Sharikov FYu (1992) Automated system of kinetic research in thermal analysis. II. Organization of kinetic experiment in ASKR. Journal of Thermal Analysis 38: 1167-1180.
- Kossoy AA, Sheinman I (2007) Comparative Analysis of the Methods for SADT Determination. Journal of Hazardous Materials 142(3): 626-638.
- Sharikov IV (2018) The scailing problem of oxide materials hydrothermal synthesis for various autoclave reactors with considering natural convection. Journal of Applied Mechanical Engineering 7: 39-40.






























