Exceptional Encouraged Results in the Characterisation and Applications of Semiconducting Ceo2 and Its Dopant Nanocomposites-A Review
Fowziya SA1,2, Uduman Mohideen AM1,2, Kavin Micheal M1,3, Ayeshamariam A1,3*, Mohamed Saleem A1,4, Seeni Mohamed Aliar Maraikkar SM5 and Jayachandran M6
1DepartmentResearch and Development Center, Bharathidasan University, India
2Departmnet of Chemistry, Khadir Mohideen College, India
3*Department of Physics, Khadir Mohideen College, India
4Department of Physics, Jamal Mohamed College, India
5Department of Computer Science, Sethu Institute of Technology, India
6Department of Physics, Sethu Institute of Technology, India
Submission: February 20, 2018; Published: April 30, 2018
*Corresponding author: A. Ayeshamariam, Department of Physics, Khadir Mohideen College, Adirampattinam 614 701, India,Tel: +919486738806; Email: aamariam786@gmail.com
How to cite this article: Fowziya SA, Uduman M AM, Kavin M M, Ayeshamariam A, Mohamed S A. et al. Exceptional Encouraged Results in the Characterisation and Applications of Semiconducting Ceo2 and Its Dopant Nanocomposites-A Review. Eng Technol Open Acc. 2018; 1(4): 555569. 10.19080/ETOAJ.2018.01.555569
Abstract
Cerium oxide has been widely investigated because of its multiple applications, such as a catalyst, an electrolyte material of solid oxide fuel cells, a material of high refractive index, Promising results have been obtained using cerium (Ce) oxide nanoparticles (CNPs) as antioxidants in biological systems. Oxide materials, its properties and applications were discussed. Most of the transparent conducting oxides have electrical and optical properties. Many applications were reported by these oxide semiconducting materials.
Keywords: Semiconducting oxides; Characterizations; Applications and devices
Abbreviations: NM: Nanomaterials; AgNW: Silver nano wire; UV: Ultraviolet; TiO2: Titanium dioxide; TiO2: titanium dioxide; ZnO : Zinc oxide; WO3: Tungsten oxide; SnO2: Tin oxide
Introduction
Nanomaterials (NM) exhibit novel physicochemical properties that determine their interaction with biological substrates and processes. Three metal oxides nano particles that are currently being produced in high tonnage, TiO2, ZnO and CeO2, were synthesized by flame spray pyrolysis process and compared in a mechanistic study to elucidate the physicochemical characteristics that determine cellular uptake, sub cellular localization, and toxic effects based on a test paradigm that was originally developed for oxidative stress and cytotoxicity in RAW 264.7 and BEAS-2B cell lines [1]. Silver can be used for the treatment of burns, wounds and several bacterial infections in the form of metallic silver, silver nitrate, silver sulfadiazine. In the anti micirobial activity, silver nano material and its nano composites plays vital role. Non-toxic silver nano materials was prepared in a simple and cost effective manner. E.coli bacteria was investigated in luria medium on solid agar plates and these nano particles were exposed to be effective bactericide [2].
Using silver nano particles, a pathogenic bacteria have developed against various antibiotics which have emerged up with assorted medical application such as silver based dressings, silver coated medicinal devices (nano gels, nano lotions etc)[3] . Silver nanowire (AgNW) networks are talented candidates to replace indium-tin-oxide (ITO) as transparent conductors. Highly conductive AgNW composite films with excellent optical transparency and mechanical properties were developed. It showed improved film conductivity and comparable performance to those based on commercial ITO substrates[4] . Titanium dioxide (TiO2) displays photocatalytic behavior under near-ultraviolet (UV) illumination and developed new phenomenon "plasmonic photocatalysis”. And enhances the degradation of methylene blue dye. The enhancement of the photocatalytic activity increases with a decreased thickness of the SiO2 shell. The plasmonic photocatalysis will be of use as a high performance photocatalyst in nearly all current applications but will be of particular importance for applications in locations of minimal light exposure [5]. Combining the magnetic iron oxide nanomaterials with semiconductor nanomaterials to form a magnetic iron oxide-semiconductor composite photocatalyst system becomes a simple and effective method. In an iron oxide- semiconductor system, iron oxide has many advantages, for example, low cost, high stability and compatibility, it not only plays the role of separating the photocatalyst from the solution, but also it can degrade organic pollutants. The common metal oxide semiconductors like titanium dioxide (TiO2), zinc oxide (ZnO), tungsten oxide (WO3), and tin oxide (SnO2) are proven to be dynamic photocatalysts for organic dyes and pollutants, these semiconductors not only destroy the conjugated chromophoric system, but also breakdown the molecular structure of the organic dyes and pollutants into harmless CO2 and H2O. As magnetic recovery photocatalytic materials, the iron oxide- semiconductor oxide photocatalyst system can effectively break through the bottleneck of low activity under visible light and demanding recycling processes from the products, and eventually become a potential visible light responsive MRPs in the future [6] (Figure 1).
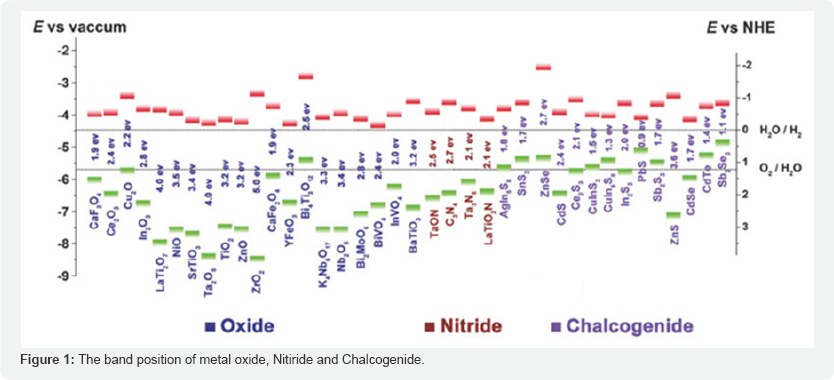
In the review of homogeneous and heterogeneous of CeO2 nanorods, nano flowers, nano cubes and its other different nano structure has attracted much attention due to improvement in its redox properties, transport properties, diffusion, absorption and surface to volume ratio with respect to bulk material. The CeO2 and doping nano composite may also be promising building blocks for nano scale devices. It is one of the most important rare earth oxide material and for its different applications as catalyst [7], oxygen sensor [8], electrolyte, material of solid fuel cell [9], and ultra violet light blocking material [10]. Many recent effort focused on an hierarchical assembly of one dimensional nano scale materials which are expected to have novel collective optical, mechanical, magnetic and electronic properties [11-13]. This significant properties meet out the challenges to purify waste water and degrading pollutants by its free methods of absorption mechanism.CeO2 hierarchical architecture and its composed dopants can be used to control by adjusting the reactant concentration to remove the pollutants from effluent water[0]. A complete understanding of CeO2 nano crystal and its synthesis method provides guidance for the removal mechanism of heavy metals from pollutant water. Because of lack of proper insitu-method to directly track the growth of nano crystal in solution leads to removal mechanism after their reaction [14]. Ce can exist in two oxidation states; Ce3+ (electronic configuration:[Xe] 4f1 and Ce4+ (electronic con - figuration: [Xe]. Therefore, Ce oxide can have two different oxide forms, CeO2 (Ce4+) or Ce2O3 (Ce3+), in bulk material. At the nanoscale, the Ce oxide lattice has a cubic fluorite structure, and both Ce3+ and Ce4+ can coexist on the surface. Charge deficiency due to the presence of Ce3+ was compensated for by oxygen vacancy in the lattice; therefore, at nanoscale, Ce oxide contains intrinsic oxygen defects. These oxygen defects are actually 'hot spots' of catalytic reaction. The concentration of oxygen defects increases with a reduction in particle size [15]. Luminescent properties of CeO2 (Ce3+,Ce4+) reveals the unusual emission at low wavelength (320nm) coupled with a large stroke shift ( ~5000cm-1) and a high ion lattice coupling strength of S_30 which gives an insight into this unusual phenomena of removal mechanism and degrading properties showing some potential for lighting applications [16]. Cerium oxide is a semiconductor has a wide band gap of 3.19 eV [17], much effort has been made to develop potential uses by using this band gap and investigated many applications such as high storage capacitors, conductors, fuel cells, polishing materials and UV-blocks [18]. In oxidation states of Ce(lV) and Ce(lll) show strong absorption peak in ultra violet wavelength in range of 232-260nm and 300- 400nm. Materials containing Ce are also Having these properties including increase of optical band gap as a result of a quantum size effect at nano meter scale [19].The resemblance of energy levels of rare earth ion ( example Ce ) is an important different in emission properties. The difference between the adjacent state is large their energy corresponding to this transition within (4f shell) cannot be transferred to lattice and it's given out in the form of emission. Sometimes rare earth ions (example Ce) are usually trivalent, this element next to this three tends to exchange electron acquire this stable configuration which will be most suitable for luminescent properties of key energy levels. The energy level may be divided into three classes, those corresponding to 4fn configuration, 4fn-1configuration, and those corresponding to charge transfer involving the neighbouring ion [15]. Most of the elements exhibits energy transfer luminescence which are activated by sensitizers are co-activated. It is important to determine the optimum concentration of other elements with CeO2. Because of obtained efficient luminescence with a minimum energy loss.The purity of nano composites are the more important issues for Ce ions emissions can be from different two excited states. The way to keep this two emission from the upper states to purify the luminescence to quenches to emission via cross relaxation [16]. Figure 1 shows energy transfer of emission of lattice phonon relaxation intra system energy crossing of Ce3+and Ce4+ions. The efficiency of later process depends upon the magnitude of square overlap integrals between absorption and emission. Coordinate displacement between the equilibrium position of the ground and 5d excited state called the Franck-Condon shift can be adjusted in emission of Cerium by choosing suitable post ions to fix the emission frequency on to increase the emission efficiency. It is worth wide considering that the variation of the energy of f electron in the shell follows the variation of 3+ and4+ redox potential along the Ce series. It is related to the ability of the trivalent Ce3+ to lose (Figure 2).
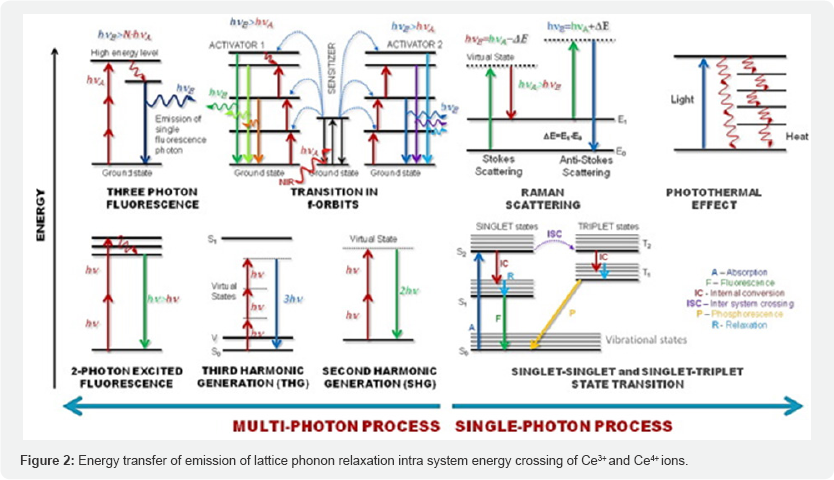
One electron and consequently to the stabilisation energy of the Ce4+ state which is clearly shown in Figure 2. Like Ce the large band gap material, the energy level 0f the impurity centre are distributed between valence and conductance band. This is suitable for Ce3+ earth ion with discrete atomic states displayed within the large forbidden band gap of materials but the propensity of their Ce ion to give up one electron should be regarded as its hole acceptor capability.
This is the case for Ce3+ andCe4+ with one more 4f electron than the empty half shell [17]. Careful analysis of the atomic lattices of Cerium oxide nano particles have clean surface and there is no passivation. But when they are doped with suitable elements the lattices from heterogeneous particles tend to align show they become to contact. Sometimes there is some point defect on the surfaces. Because the synthesis temperature suppose very low and there is not enough energy for the particles to achieve equilibrium shape and size .These type of particles try to attained higher energy than the spherical like particles [18].
Discussion
Among these NPs, titanium dioxide (TiO2), a noncombustible and odorless white powder, naturally exists in three modifications (anatase, rutile and brookite). Nowadays, nanosized TiO2 is produced abundantly and used widely because of its thermodynamic stability, anticorrosion and photocatalysis e.g.as pigments in paints, toothpastes, plastics, paper, ceramics, cosmetics and additives in food (E171) [19,20]. Its use as food additives was restricted by the US Food and Drug Administration e.g. the quantity of TiO2 does not exceed 1% by weight of the food. Studies on the potential toxicity of TiO2 NPs (focused on mammals such as mice and rats) showed that TiO2 NPs are able to accumulate in blood, liver and spleen [21-24]. Some TiO2 nanoparticles have been shown to have inflammogenic, oxidative, and genotoxic effects [25]. Johnston HJ et al. (2009) and the International Agency for Research on Cancer (lARC) classified TiO2 as possibly carcinogenic to humans (Group 2B) [26]. The element Ti consists of five naturally abundant isotopes (46Ti (8.0%), 47Ti (7.3%), 48Ti (73.8%), 49Ti (5.5%) and 50Ti (5.4%). Yet, 46Ti, 48Ti and 50Ti isotopes cannot be used for quantification due to strong isobaric and polyatomic interferences. A preliminary study conducted on the two other Ti isotopes on deionised water and biological samples using different spiked solutions with Ti (2, 5, 10, 20 and 50μg L"1) and different non target elements multi-elemental solution (10, 20, 40, 80 and 160μgL-1 of Li, Al, Cr, V, Ni, Zn, Co, Cu, Ge, Se, Sc, Mn, Fe, Sr, Mo, Y and 0.5,1 and 5mgL-1 of Ca, K, Mg, Na) showed that 47Ti and show the least interference in biological samples (see supplementary data). 71Ga and 115ln were tested as internal standards (lS) and both can be used for this application. So, for the remainder of this study, 47Ti, 49Ti and 71Ga as internal standard (4μgL-1) were used [27].
Functionalized TiO2 based nanostructured materials have positive effects in many biomedical applications such as bone scaffolds, vascular stents, drug delivery systems, and biosensors. For example, nano-TiO2 scaffolds accelerate the rate of apatite formation and enhance osteoblast adhesion, proliferation, and differentiation [28-31]. Possessing good blood compatibility and anti-coagulation characteristics, TiO2 nanotube arrays are promising for vascular implants, and nanostructured TiO2 has been widely reported as drug carriers as well [32,33]. ln particular, TiO2 nanotubes have been shown to be a superior platform for local drug delivery due to their excellent biocompatibility, controllable dimensions, surface chemistry, and large surface-to- volume ratio [34-36]. By changing the nanotube diameter, wall thickness, and length, the release kinetics of specific drugs can be tailored to achieve stable and sustained release [37]. Generally, the requirements for biosensors are good reproducibility and sensitivity to specific chemical and biochemical compounds, and owing to its high sensitivity to glucose, hydrogen peroxide, and cancer cells, nano-TiO2 has been extensively studied in bio sensing applications, for example, detection of blood glucose in diabetes mellitus patients and early monitoring of cancer [38-40].
The materials chosen as suitable dopants depend on the atomic properties of both the dopant and the material to be doped. ln general, dopants that produce the desired controlled changes are classified as either electron acceptors or donors. A donor atom that activates (that is, becomes incorporated into the crystal lattice) donates weakly-bound valence electrons to the material, creating excess negative charge carriers. These weakly-bound electrons can move about in the crystal lattice relatively freely and can facilitate conduction in the presence of an electric field. (The donor atoms introduce some states under, but very close to the conduction band edge. Electrons at these states can be easily excited to the conduction band, becoming free electrons, at room temperature.) Conversely, an activated acceptor produces a hole. Semiconductors doped with donor impurities are called n-type, while those doped with acceptor impurities are known as p-type. The n and p type designations indicate which charge carrier acts as the material's majority carrier. The opposite carrier is called the minority carrier, which exists due to thermal excitation at a much lower concentration compared to the majority carrier
Europium doped cerium oxide was synthesised by simple chemical precipitation method to enhance the emission properties and also concentration of oxygen ion vacancy increased with the increase in dopant concentration [41]. Nanocrystalline CeO2-x materials, pure or doped with 10%La or 15%Cu, were produced by magnetron sputtering from pure or mixed metal targets, followed by controlled oxidation. The effects of oxide nonstoichiometry and dopants on greater catalytic activity in oxidation reactions and the light-off temperatures for SO2 reduction by CO, CO oxidation, and methane oxidation and also the differences between the nanocrystalline and the precipitated materials are discussed in terms of the stoichiometry of these oxide catalysts were investigated [42]. The Fe3+, La3+ and Zr4+ incorporated in nano CeO2 lattice prepared by facile co precipitation method and nano-Au/CeO2 was synthesised by anion adsorption method and the catalytic performance of Au catalysts was investigated[43]. Nanocrystalline homogeneous Cu-doping of CeO2-x mixed-metal oxide was developed by magnetron sputtering from a mixed metal target and enhances the catalytic activity [44].
The synthesised nano-sized Ce0.8Sm0.2O1.9 particles form core like structures and their crystal size was investigated [45]. Titanium dioxide nanoparticles doped with Fe and Ce prepared by sonochemical method exhibit higher photocatalytic activity as compared to the catalysts prepared by the conventional methods. Also the Ce-doped TiO2 exhibits maximum photocatalytic activity followed by Fe-doped TiO2 and the least activity was observed for only TiO2. The presence of Fe and Ce in the TiO2 structure results in a significant absorption shift towards the visible region [46]. Ceria nano particles doped with Fe has been investigated for toluene total oxidation reaction [47]. The lattice constant of SnO2 increases and the grain size decreases with doping of Ni and Ce and gas sensing result revealed that the thick films deposited on alumina substrates using screen printing technique give selectively a high response with fast recovery towards acetone [48].
Thanks to the strength of its Research and lnnovation teams, and its expertise in Rare Earths, Solvay has developed separated rare earth polishing products with tightly-controlled composition, together with specifically designed morphology and characteristics.
ln recent years, Ce oxide nanoparticles (CNPs) have gained a lot of interest owing to their regenerative antioxidant property. Ce oxide is a very important material for industrial use, with applications in glass polishing, catalytic convertors for removing toxic gases, solid oxide fuel cells, electrochromic thin-film applications, sensors and catalysts [49].
The responses of cells exposed to nanoparticles have been studied with regard to toxicity, but very little attention has been paid to the possibility that some types of particles can protect cells from various forms of lethal stress. lt is shown here that nanoparticles composed of cerium oxide or yttrium oxide protect nerve cells from oxidative stress and that the neuro protection is independent of particle size. The ceria and yttria nanoparticles act as direct antioxidants to limit the amount of reactive oxygen species required to kill the cells. lt follows that this group of nanoparticles could be used to modulate oxidative stress in biological systems [50]. Non-stoichiometric oxides are widely used in high temperature energy applications, such as solid oxide fuel cells (SOFCs), oxygen permeation membranes, and gas conversion/reformation catalysis [51-53].
Biomedical applications
The focus of this review is to reveal a historic prospective of biomedical applications of nanoparticles (NPs) and an overview of the recent developments in this field, and then to discussion the commercialization of nanomaterials. Bones in our body is a nanocomposite material of hydroxyapatite crystallites in the organic matrix and composed of collagen [54].
Cancer therapy
Photodynamic cancer therapy is based on the destruction of the cancer cells by laser generated atomic oxygen, which is cytotoxic. A greater quantity of a special dye that is used to generate the atomic oxygen is taken in by the cancer cells when compared with a healthy tissue. Hence, only the cancer cells are destroyed then exposed to a laser radiation. Unfortunately, the remaining dye molecules migrate to the skin and the eyes and make the patient very sensitive to the daylight exposure. This effect can last for up to six weeks. To avoid this side effect, the hydrophobic version of the dye molecule was enclosed inside a porous nanoparticle [55]. The dye stayed trapped inside the ormosil nanoparticle and did not spread to the other parts of the body. At the same time, its oxygen generating ability has not been affected and the pore size of about 1 nm freely allowed for the oxygen to diffuse out.
Most major and established pharmaceutical companies have internal research programs on drug delivery that are on formulations or dispersions containing components down to nano sizes. Colloidal silver is widely used in antimicrobial formulations and dressings. The high reactivity of titania nanoparticles, either on their own or then illuminated with UV light, is also used for bactericidal purposes in filters. Enhanced catalytic properties of surfaces of nano-ceramics or those of noble metals like platinum are used to destruct dangerous toxins and other hazardous organic materials. Biomimetic nanostructures, which are synthetic products developed from an understanding of biological systems, biological nanostructures, the electronic-biological interface, devices for early detection of disease, lnstruments for studying individual molecules [56].
Potential risks
Nanotechnology applications have not been marketed long enough for claims to be corroborated about risks to human health or the environment. Still, small nanoparticles can enter the human body through pores and may accumulate in cells. The health effects of such nanoparticles are unknown. Historical experience with unintended consequences of technologies, such as drug resistance to antibiotics or the persistence of chemicals such as DDT in the environment, teaches us to take precautions.
A European Commission-National Science Foundation workshop held early this year discussed the societal aspects of nanotechnology. Participants concluded, "Nanobiotechnology could dramatically improve public health, but there is concern that technical developments could cause unforeseen adverse effects. Studies are needed to determine what environmental and health risks are associated with nano materials.” Medical nanotechnologies are entering industrial production, mainly for diagnostics, drugs, and therapies. ln the longer term, nanotechnology may help improve implants and even let blind people see. Nonetheless, governments must stimulate scientists to monitor possible health risks of nanoparticles that may accumulate in the body [56].
Fullerenes
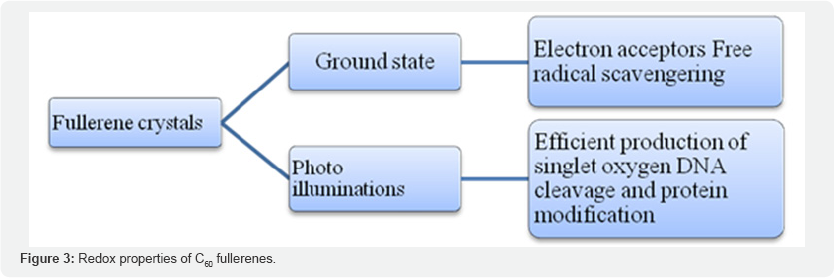
C60 fullerenes, popularly known as Bucky balls [57,58], are icosahedral (20 sided) structures made of 60 carbon atoms (Mr720), organized in a spherical arrangement of hexagonal and pentagonal groups. C60 molecules are 0.7 nm in size and can aggregate into larger crystalline structures, depending upon the medium in which they are suspended [59]. Some of the interesting properties of C60 include large electro negativity (high electron affinity). Second, crystals of fullerenes have strong photoluminescence (PL), which is sensitive to the exact "inter-ball” spacing, and this spacing is sensitive to the local pH and oxygen content. Third, fullerenes are amenable for chemical modifications which allow for the attachment of organic molecules of interest. Various types of C60 functionalization that enhance the hydrophilicity of fullerenes have been described (reviewed in) [57]. Hence, multiple reactive groups and functionalities, including antibodies, drugs, and metals may be attached to the core of C60 particles. Fourth, some studies indicate that fullerenes may selectively accumulate in tumors and are reported to be excreted in urine without accumulating in the tissues [60]. Some studies also have reported that fullerenes cross and protect the blood brain barrier [61,62], a property that can be exploited for certain applications. Among the various properties of fullerenes, redox properties (electron acceptor-donor) have been extensively studied and are particularly attractive for biomedical applications (Figure 3).
Carbon nanotubes
Nanotubes are another type of carbon nanoparticles that have been considered for a variety of biological applications, including as biosensors, drug delivery devices, and as therapeutic agents [63]. Carbon nanotubes are primarily composed of carbon atoms, arranged in benzene rings which form graphene sheets, and are rolled into tubes [64]. The single-walled carbon nanotubes (SWNT) consist of a single layer of graphene sheet, which are characterized by a high ratio of length over diameter (known as the aspect ratio). Thus, SWNTs can be several microns long with a diameter in the range of a few nanometers. Multiwalled nanotubes (MWNTs), on the other hand, consist of several concentric sheets, spaced at less than 1 nm. Nanotubes, because of their distinctive electrical, mechanical, and optical properties, are well suited for several biological applications. For example, nanotubes exhibit outstanding structural flexibility and fluidity and yet possess high mechanical strength. Nanotubes exhibit useful Raman scattering and fluorescence emission in the near infrared (nIR) spectrum between 900 and 1300nm.
Quantum dots
Quantum dots are fluorescent nanoparticles of about 2-10nm in size. The central core is composed of hundreds to thousands of atoms belonging to the groups of II-VI (e.g., cadmium, selenium) or III-V (e.g., indium) elements. The fluorescence emission spectrum of quantum dots is dependent on the size of the central nanocrystal core which can be adjusted by controlling the amount of precursors during the synthesis. The modest quantum yield of the core nanocrystal is substantially (up to 80%) enhanced by wrapping the core in a zinc sulfide shell. Since the coreshell of the nanocrystal is hydrophobic, it is rendered water soluble with bifunctional molecules (e.g., mercaptohydrocarbonic acid), surface silanization or coating the surface with amphiphilic polymers. The quantum dot crystals are functionalized by linking appropriate targeting molecules through either covalent or non- covalent methods [65].
Superparamagnetic nanoparticles
Super paramagnetic nanoparticles contain a magnetically active metal core and are widely used in MRI imaging for enhancing contrast. Iron oxide containing super paramagnetic nanoparticles has been commonly used as the core particle. The size and charge of iron oxide crystals and the nature of chemical coatings are some of the key determinants of the utility of these nanoparticles. Because ofthe excellent spatial resolution afforded by MRI, iron oxide nanoparticles have been used for diagnostic imaging of tumors, inflammatory and degenerative diseases. Iron oxide particles also have been conjugated to numerous antibodies for targeted molecular and cellular imaging [66].
Nanotoxicity
Nanomaterials may gain entry into human systems through inhalation, ingestion, or through dermal routes. Deposition and the physiological consequences elicited by inhaled nanoparticles are dependent on the size, shape (e.g., spherical, fibrous), surface charge, and the aggregation state of the nanoparticles. For example, smaller (<100 nm) particles are likely to travel deeper into the lung and deposit into the alveolar region.
Detection of Nanomaterials
It is important to correlate the biological/cytotoxic effects of nanoparticles with the actual intracellular and in vivo quantities that persist upon administration. Whereas a number of techniques, such as electron microscopy or atomic force microscopy, are used to detect nanoparticles in vitro, these techniques may not be suitable or convenient for the routine detection of nanoparticles and their load in tissues. While nanoparticles labeled with radioactivity, fluorescent probes, or chemicals (including drugs, enzymes, biotin, or other reagents) may be readily monitored, it is often difficult to detect low concentrations of unmodified nanoparticles or the metabolites of the conjugated particles. It is also important to consider whether in vivo interactions of nanoparticles (or their metabolites) with serum proteins or tissues would alter their physicochemical properties, thus, rendering their detection more difficult. Emerging research points to some success in this direction. For example, recent work demonstrates that the intrinsic IR fluorescence may be useful to detect and follow the pharmacokinetics of SWNTs [67]. Similarly HPLC detection of nanogram quantities of C60 fullerenes from protein containing samples has been reported [68]. ln addition, this method might be applicable for the detection of C60 in plasma and skin samples.
Cancer therapy
ln most cases, cancer-related deaths occur due to the failure of chemotherapy and/or radiation therapy of the metastatic disease. Therefore, for a successful cancer treatment, it is critical to detect tumors early on during the disease progression, detect and ablate tumor metastasis. Nanotechnology has several applications in improving cancer therapy and some nanosized drugs are currently in clinical trials Brannon-Peppas L [69]. Prominent among those are liposomal doxorubicin and albumin conjugated Paclitaxel, which have been shown to reduce toxicity due to the adverse side effects of respective drugs. Liposomal doxorubicin (Doxil) has been shown to be an effective antineoplastic agent with improved biodistribution (longer plasma circulation times) which reduces severe dose-limiting cardiotoxicity associated with the drug treatment [70]. Like many drugs, Paclitaxel is poorly soluble in water and is administered as a formulation with Cremophor EL (polyethoxylated castor oil) and causes side effects such as hypersensitivity and nephrotoxicity and neurotoxicity. While several different formulations have been made to minimize the toxic effects of this drug, albumin- conjugated nano-sized paclitaxel (Abraxane), a Cremophor free- formulation, has been shown to be well-tolerated and yet more effective than the conventional drug [71]. Additionally, a number of nanosized formulations of other chemotherapeutic drugs such as 5-fluorouracil and camptothecin are being tested. Further, new formulations of aqueous compatible nanosized drugs (such as paclitaxel) are being developed through supercritical fluid technologies [72].
ln other applications, the unique properties of nanoparticles are being exploited for the treatment of cancer. ln this strategy, nanoparticles serve as multifunctional therapeutic agents, instead of being utilized as simple passive carriers of cargo. These multifunctional nanoparticles can potentially carry drugs to the target tissue, image the target tissues, and release the cargo in response to a signal or upon reaching appropriate cellular compartment. Further, the targeted nanoparticles may be used as therapeutic agents. Targeting the nanotherapeutic may be achieved by coupling a specific antibody or a small molecular weight ligand (e.g., folic acid) that recognizes a protein selectively expressed on tumor cells. Magnetic iron oxide particles [73]. Nanotubes [74] or other particles may be used as core nanoparticles, and the imaging can be accomplished through MRI (with iron oxide nanoparticles) or fluorescence methods if quantum dots [75] are incorporated into the multifunctional nanoparticle. For example, optical stimulation with an nlR laser of folic acid receptor targeted SWNTs has been shown to act as a trigger for the release of the internalized nanotube attached cargo (in this case oligodeoxy nucleotides) [76]. Further, nlR was also used to generate local heating and then cause photothermal ablation of the targeted cell populations. ln another example, tumor-targeting antibody, Herceptin, which is prepared against HER2 receptor (amplified in breast tumors), was coupled to gold nanoshells. This enabled the gold nanoshells to be targeted to HER2 over expressing tumor cells and when irradiated with lR, tumor cells were specifically killed [77]. Further, the ability of fullerenes to generate highly reactive singlet oxygen has also been tested as a potential photodynamic therapy of tumor cells. Thus, the concepts of multifunctional nanoparticles for cancer therapy have been validated in experimental systems.
Drug delivery systems
Medical devices such as orthopedic implants, dental implants, and vascular stents may require subsequent drug therapy to prevent infection or minimize inflammatio [78]. Drugs released directly from the implant surface rather than systemically can reduce unnecessary side effects. The aim of controlled drug delivery is to (i) administer the appropriate amount of drug to the relevant sites in the human body and (ii) regulate the drug delivery profile in order to optimize the therapeutic benefits [79]. TiO2 nanomaterials are suitable for drug-eluting implants and their drug delivery properties spur potential applications in the biomedical field [79]. Antibacterial Agent Delivery
Carrier of inorganic antimicrobial agents
The antibacterial properties of Zn-doped TiO2 nanomaterials have also been investigated in recent years [80]. Zn-doped Ti-based nanofibers promote antimicrobial effects against Staphylococcus aureus and Escherichia coli due to cell membrane disruption and cytoplasma leakage. The antibacterial activity of ZnCl2/TiO2, Zn(Ac)2/TiO2, Zn(NO3)2/TiO2, and ZnSO4/ TiO2, follows the subsequent order: ZnSO4 > ZnCl2 > Zn(NO3)2 > Zn(Ac)2, and the highest antibacterial activity observed for ZnSO4/TiO2 is possibly ascribed to the improved surface acidity [81]. lt has also been reported that zinc can be incorporated into TiO2 coatings to achieve a good bacterial inhibition ability, and the better antibacterial activity of Zn-incorporated TiO2 coatings may be attributed to the fact that Zn ions can be slowly and constantly released from the coatings [82,82].
Biosensors
A biosensor is a device incorporating a biological sensing element either closely connected to or integrated to a transducer Specific molecular recognition is a fundamental prerequisite on the basis of the affinity between complementary structures such as enzyme-substrate, antibody-antigen, and receptor-hormone, and this property is often used to generate the concentration dependent signals. The sensitivity and specificity depend on the biological recognition system [83,84]. Since the development of enzyme-based sensors for glucose by Clark and Lyons [85], improvements have been made in terms of the sensitivity, selectivity, and reproducibility as a result of rapid developments in nanotechnology and nanomaterials. Nanomaterial-based biosensors, which represent the integration of materials science, molecular engineering, chemistry, and biotechnology, improve the sensitivity and specificity of biomolecule detection and have great potential in biomolecule recognition and pathogenic diagnosis [86, 87].
In order to improve the performance of biosensors, TiO2 nanomaterials such as nanoparticles [88], nanotubes [89-90], nanofibers [91], HaoTang, gold nanoparticle encapsulated TiO2 nanoclusters [92], and TiO2/SiO2 nanocomposites [93,94], have been used in biosensing devices for enzymes, antibodies, microorganisms, and DNA. Nanostructured TiO2 based biosensors are sensitive, selective, fast, and reproducible for the detection of various chemical and biochemical compounds such as glucose, hydrogen peroxide, and cancer cells because of their superior properties including nontoxicity, large surface area, high adsorptivity, good uniformity, and excellent biocompatibility. In fact, TiO2 based biosensors have been proposed to be a prospective interface for the immobilization of biomolecules [95,96].
Enzymatic TiO2 based biosensors
Various types of enzymes such as glucose oxidase (GOD), horseradish peroxidase (HRP), urease, cytochrome C (cyt.c), and glutamate dehydrogenase can be immobilized on TiO2 nanomaterials [97]. When the two enzymes GOD and HRP are co-immobilized on the TiO2 based nanostructured surfaces, direct electron transfer between enzyme and electrodes is significantly enhanced due to the nanostructured environment of the TiO2 based layers [98-103]. These biosensors have a good sensitivity, low detection limit (≈10-6 M), and fast time response (few seconds) making them promising in low-cost, miniaturized multi-functional biosensors. In biosensing applications, novel nanostructures that can immobilize more enzymes on the surface of TiO2 and accelerate electron transfer between the biological components and electrode continue to be researched extensively.
Conclusion
CeO2 and its dopant semiconducting oxides were used for the various applications of nanocomposites. The properties of the materials were varied due to its doping ratio of the oxide materials. Some fundamental physical and chemical properties of ceria and its dopants were addressed by the many researchers and their recent progress in the size and shape-controlled synthesis and morphology-dependant performance of ceria and its doped oxide materials nano-microstructures was highlighted. Although, the nanostructures, morphologies, characterization approaches and many applications studies such as photocatlysts degradations processes of ceria-based nanostructured materials are extensively investigated in recent years so their properties and its applications with the achievement of some exceptional encouraged results were reported here.
References
- Xia T, Kovochich M, Liong M, Madler L, Gilbert B, et al. (2008) Comparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress properties. ACS nano 2(10): 2121-2134.
- Sondi I, Salopek-Sondi B (2004) Silver nanoparticles as antimicrobial agent: a case study on E. coli as a model for Gram-negative bacteria. Journal of colloid and interface science 275(1): 177-182.
- Rai M, Yadav A, Gade A (2009) Silver nanoparticles as a new generation of antimicrobials. Biotechnology advances 27(1): 76-83.
- Zhu R, Chung CH, Cha KC, Yang W, Zheng YB, et al. (2011) Fused silver nanowires with metal oxide nanoparticles and organic polymers for highly transparent conductors. ACS nano 5(12): 9877-9882.
- Awazu K, Fujimaki M, Rockstuhl C, Tominaga J, Murakami H, et al. (2008) A plasmonic photocatalyst consisting of silver nanoparticles embedded in titanium dioxide. Journal of the American Chemical Society 130(5): 1676-1680.
- Laurent S, Forge D, Port M, Roch A, Robic C, et al. ( 2008) Magnetic iron oxide nanoparticles: synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chemical reviews 108(6): 2064-2110.
- Zhang F, Jin Q, Chan SW (2004) Ceria nanoparticles: size, size distribution, and shape. Journal of applied physics 95(8): 4319-4326.
- Wang ZL, Feng X (2003) Polyhedral shapes of CeO2 nanoparticles. The Journal of Physical Chemistry B 107(49): 13563-13566.
- Zhou K, Wang X, Sun X, Peng Q, Li Y (2005) Enhanced catalytic activity of ceria nanorods from well-defined reactive crystal planes. Journal of Catalysis 229(1): 206-212.
- Mai HX, Sun LD, Zhang YW, Si R, Feng W, et al. (2005) Shape-selective synthesis and oxygen storage behavior of ceria nanopolyhedra, nanorods, and nanocubes. The Journal of Physical Chemistry B 109(51): 24380-24385.
- Kehoe AB, Scanlon DO, Watson GW (2011) Role of lattice distortions in the oxygen storage capacity of divalently doped CeO2. Chemistry of Materials 23(20): 4464-4468.
- Rodriguez JA, Liu P, Hrbek J, Evans J, Perez M (2007) Water Gas Shift Reaction on Cu and Au Nanoparticles Supported on CeO2(111) and ZnO(000$\bar1$): Intrinsic Activity and Importance of Support Interactions. Angewandte Chemie International Edition 46(8): 13291332.
- Yang Z, Woo TK, Hermansson K (2006) Adsorption of NO on unreduced and reduced CeO2 surfaces: A plane-wave DFT study. Surface science 600(22): 4953-4960.
- Da Silva RO, Gonsalves RH, Stroppa DG, Ramirez AJ, Leite ER (2011) Synthesis of recrystallized anatase TiO2 mesocrystals with Wulff shape assisted by oriented attachment. Nanoscale 3(4): 1910-1916.
- Deshpande S, Patil S, Kuchibhatla SV, Seal S (2005) Size dependency variation in lattice parameter and valency states in nanocrystalline cerium oxide. Appl. Phys. Lett 87(13): 133113.
- Ono Y, Takayama K, Kajitani T (1996) X-Ray Diffraction Study of LaBSiO5. Journal of the Physical Society of Japan 65(10): 3224-3228.
- Lin HC (2005) Production, application and market of cerium oxide. Hydrometallurgy of China 24(1): 10.
- Fu Q, Saltsburg H, Flytzani-Stephanopoulos M (2003) Active nonmetallic Au and Pt species on ceria-based water-gas shift catalysts. Science 301(5635): 935-938.
- Abecassis Wolfovich M, Jothiramalingam R, Landau MV, Herskowitz M, Viswanathan B, et al. (2005) Cerium incorporated ordered manganese oxide OMS-2 materials: lmproved catalysts for wet oxidation of phenol compounds. Applied Catalysis B: Environmental 59(1-2): 91-98.
- Cao, Shixiu, Ravikumar B, Shahid Hussain, A. Ayeshamariam, Nimra Aslam, and Khalid Naseer (2016) Synthesis and characterization of CeO2 and ZnCeO2 nanomaterials and exposure to photocatalytic activity. Journal of Materials Science: Materials in Electronics 27(2): 1873-1880.
- Nakazawa E, Shionoya S (1970) Relaxation between excited levels of Tb3+ ion due to resonance energy transfer. Journal of the Physical Society of Japan 28(5): 1260-1265.
- Jia D, Jia W, Evans DR, Dennis WM, Liu H, Zhu J, Yen WM (2000) Trapping processes in CaS: Eu2+, Tm3+. Journal of Applied Physics 88(6): 3402-3407.
- Wang ZL, Kang ZC (1998) Structure Analysis of Functional Materials. In Functional and Smart Materials Springer, Boston, MA.
- Wang J, Zhou G, Chen C, Yu H, Wang T, Ma Y, Jia G, Gao Y, Li B, Sun J, Li Y. (2007) Acute toxicity and biodistribution of different sized titanium dioxide particles in mice after oral administration. Toxicology letters 168(2): 176-185.
- Ju-Nam Y, Lead JR (2008) Manufactured nanoparticles: an overview of their chemistry, interactions and potential environmental implications. Science of the total environment 400(1-3): 396-414.
- Barlow S, Chesson A, Collins JD, Flynn A, Hardy A, Jany KD, Knaap A, Kuiper H, Larsen JC, Le Neindre P, Schans J. (2009) The potential risks arising from nanoscience and nanotechnologies on food and feed safety. The EFSA Journal 7(3): 958.
- Sugibayashi K, Todo H, Kimura E (2008) Safety evaluation of titanium dioxide nanoparticles by their absorption and elimination profiles. The Journal of toxicological sciences 33(3): 293-298.
- Van Ravenzwaay B, Landsiedel R, Fabian E, Burkhardt S, Strauss V, Ma- Hock L (2009) Comparing fate and effects of three particles of different surface properties: nano-TiO2, pigmentary TiO2 and quartz. Toxicology letters 186(3): 152-159.
- Peralta-Videa JR, Zhao L, Lopez-Moreno ML, de la Rosa G, Hong J, Gardea-Torresdey JL (2011) Nanomaterials and the environment: a review for the biennium 2008-2010. Journal of hazardous materials 186(1): 1-5.
- Johnston HJ, Hutchison GR, Christensen FM, Peters S, Hankin S, et al.(2009) Identification of the mechanisms that drive the toxicity of TiO2 particulates: the contribution of physicochemical characteristics. Part Fibre Toxicol 6: 33.
- lARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Titanium Dioxide (2010) lARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Carbon Black, Titanium Dioxide and Talc. lARC Monographs 93: 193-214.
- Nia Y, Millour S, Noel L, Krystek P, de Jong W, Guerin T (2015) Determination of Ti from TiO2 nanoparticles in biological materials by different lCP-MS instruments: method validation and applications. J Nanomed Nanotechnol 6(2): 1-8.
- Gongadze E, Kabaso D, Bauer S, Park J, Schmuki P, lglic A. (2013) Adhesion of osteoblasts to a vertically aligned TiO2 nanotube surface, Mini Rev Med Chem 13(2): 194-200.
- Tan AW, Pingguan-Murphy B, Ahmad R, Akbar SA. (2013) Advances in fabrication of TiO2 nanofiber/nanowire arrays toward the cellular response in biomedical implantations: a review. Journal of materials science 48(24): 8337-8353.
- Brammer KS, Choi C, Frandsen CJ, Oh S, Johnston G, Jin S. (2011) Comparative cell behavior on carbon-coated TiO2 nanotube surfaces for osteoblasts vs. osteo-progenitor cells. Acta biomaterialia 7(6): 2697-703.
- Dong W, Zhang T, Epstein J, Cooney L, Wang H, et al. (2007) Multifunctional nanowire bioscaffolds on titanium. Chemistry of Materials 19(18): 4454-4459.
- Yang Y, Lai Y, Zhang Q, Wu K, Zhang L, et al. (2010) A novel electrochemical strategy for improving blood compatibility of titanium-based biomaterials. Colloids and Surfaces B: Bio interfaces 79(1): 309-313.
- Weng Y, Song Q, Zhou Y, Zhang L, Wang J, et al. (2011) lmmobilization of selenocystamine on TiO2 surfaces for in situ catalytic generation of nitric oxide and potential application in intravascular stents. Biomaterials 32(5): 1253-1263.
- Vasilev K, Poh Z, Kant K, Chan J, Michelmore A, et al. (2010) Tailoring the surface functionalities of titania nanotube arrays. Biomaterials 31(3): 532-540.
- Shokuhfar T, Sinha-Ray S, Sukotjo C, Yarin A L (2013) lntercalation of anti-inflammatory drug molecules within TiO2 nanotubes. Rsc Advances 3(38): 17380-17386.
- Peng L, Mendelsohn AD, La Tempa TJ, Yoriya S, Grimes CA, et al. (2009) Long-term small molecule and protein elution from TiO2 nanotubes. Nano letters 9(5): 1932-1936.
- Popat KC, Eltgroth M, LaTempa TJ, Grimes CA, Desai TA (2007) Titania nanotubes: a novel platform for drug-eluting coatings for medical implants? 3(11): 1878-1881.
- Srivastava S, Ali MA, Solanki PR, Chavhan PM, Pandey MK, et al. (2013) Mediator-free micro fluidics biosensor based on titania-zirconia nanocomposite for urea detection. RSC Advances 3(1): 228-235.
- Doong RA, Shih HM (2010) Array-based titanium dioxide biosensors for ratio metric determination of glucose, glutamate and urea. Biosensors and Bioelectronics 25(6):1439-1446.
- Qiu J, Zhang S, Zhao H (2011) Recent applications of TiO2 nanomaterials in chemical sensing in aqueous media. Sensors and actuators B: Chemical 160(1): 875-890.
- Kumar A, Babu S, Karakoti AS, Schulte A, Seal S (2009) Luminescence properties of europium-doped cerium oxide nanoparticles: role of vacancy and oxidation states. Langmuir 25(18): 10998-11007.
- Tschope A, Liu W, Flytzanistephanopoulos M, Ying JY (1995) Redox activity of non stoichiometric cerium oxide-based nano crystalline catalysts. Journal of catalysis 157(1): 42-50.
- Sudarsanam P, Mallesham B, Reddy P S, GroRmann D, Grunert W, et al. (2014) Nano-Au/CeO2 catalysts for CO oxidation: Influence of dopants (Fe, La and Zr) on the physicochemical properties and catalytic activity. Applied Catalysis B: Environmental 144: 900-908.
- Tschope A, Ying JY (1995) The effects of non-stoichiometry and dopants in nanocrystalline cerium oxide-based catalysts on redox reactions. Nanostructured Materials 6(5-8): 1005-1008.
- Cheng MY, Hwang DH, Sheu HS, Hwang BJ (2008) Formation of Ce0.8Sm0.2-O1.9 nanoparticles by urea-based low-temperature hydrothermal process. Journal of Power Sources 175(1): 137-144.
- Shirsath SR, Pinjari DV, Gogate PR, Sonawane SH, Pandit AB (2013) Ultrasound assisted synthesis of doped TiO2 nano-particles: characterization and comparison of effectiveness for photocatalytic oxidation of dyestuff effluent. Ultrasonics sonochemistry 20(1): 277286.
- Meledina M, Turner S, Galvita VV, Poelman H, Marin GB, et al. (2015) Local environment of Fe dopants in nanoscale Fe: CeO2- x oxygen storage material. Nanoscale 7(7): 3196-3204.
- Bagal LK, Patil JY, Mulla IS, Suryavanshi SS (2012) Studies on the resistive response of nickel and cerium doped SnO2 thick films to acetone vapor. Ceramics International 38(8): 6171-6179.
- Sun C, Li H, Chen L (2012) Nanostructured ceria-based materials: synthesis, properties, and applications. Energy & Environmental Science 5(9): 8475-8505.
- Schubert D, Dargusch R, Raitano J, Chan SW (2006) Cerium and yttrium oxide nanoparticles are neuroprotective. Biochemical and biophysical research communications 342(1): 86-91.
- Adler SB, Lane JA, Steele BC (1996) Electrode kinetics of porous mixed- conducting oxygen electrodes. Journal of the Electrochemical Society 143(11): 3554-3564.
- Sugiura M (2003) Oxygen storage materials for automotive catalysts: ceria-zirconia solid solutions. Catalysis Surveys from Asia 7(1): 77-87.
- Dyer PN, Richards RE, Russek SL, Taylor DM (2000) Ion transport membrane technology for oxygen separation and syngas production. Solid State Ionics 134(1-2): 21-33.
- Salata OV (2004) Applications of nanoparticles in biology and medicine. J Nanobiotechnology 2(1): 3.
- Roy I, Ohulchanskyy TY, Pudavar HE, Bergey EJ, Oseroff AR, et al. (2003) Ceramic-based nanoparticles entrapping water-insoluble photosensitizing anticancer drugs: a novel drug-carrier system for photodynamic therapy. J Am Chem Soc 125(26): 7860-7865.
- Schultz J, Mrksich M, Bhatia SN, Brady DJ, Ricco AJ, et al. (2006) Biosensing: international research and development. Springer Science & Business Media, Germany
- Bosi S, Da Ros T, Spalluto G, Prato M (2003) Fullerene derivatives: an attractive tool for biological applications. Eur J Med Chem 38(11-12): 913-923.
- Jensen AW, Maru BS, Zhang X, Mohanty DK, Fahlman BD, et al. (2005) Preparation of fullerene-shell dendrimer-core nanoconjugates. Nano Lett 5(6): 1171-1173.
- Dresselhaus MS, Dresselhaus G, Eklund PC (1996) Science of fullerenes and carbon nanotubes. Academic Press, San Diego, CA, USA.
- Tabata Y, Murakami Y, Ikada Y (1997) Antitumor effect of poly(ethylene glycol)modified fullerene. Fullerene Sci Technol 5(5): 989-1007
- Tsao N, Kanakamma PP, Luh TY, Chou CK, Lei HY (1999) Inhibition of escherichia coliinduced meningitis by carboxyfullerence. Antimicrob Agents Chemother 43(9): 2273-2277.
- Yamago S, Tokuyama H, Nakamura E, Kikuchi K, Kananishi S, et al. (1995) In vivo biological behavior of a water-miscible fullerene: 14C labeling, absorption, distribution, excretion and acute toxicity. Chem Biol 2(6): 385-389.
- Katz E, Willner I (2004) Biomolecule-functionalized carbon nanotubes: applications in nanobioelectronics. Chem phys chem 5(8): 1084-1104.
- Bianco A, Hoebeke J, Kostarelos K, Prato M, Partidos CD (2005) Carbon nanotubes: on the road to deliver. Curr Drug Deliv 2(3): 253-259.
- Alivisatos AP, Gu W, Larabell C (2005) Quantum dots as cellular probes. Annu Rev Biomed Eng 7: 55-76.
- Corot C, Robert P, Idee J-M, Port M (2006) Recent advances in iron oxide nanocrystal echnology for medical imaging. Adv Drug Deliv Rev 58(14): 1471-1504.
- Cherukuri P, Gannon CJ, Leeuw TK, Schmidt HK, Smalley RE, et al. (2006) Mammalian pharmacokinetics of carbon nanotubes using intrinsic near-infrared fluorescence. Proc Natl Acad Sci USA 103(50): 18882-18886.
- Xia XR, Monteiro-Riviere NA, Riviere JE (2006) Trace analysis of fullerenes in biological samples by simplified liquid-liquid extraction and high-performance liquid chromatography. J Chromatogr A 1129(2): 216-222.
- Brannon-Peppas L, Blanchette JO (2004) Nanoparticle and targeted systems for cancer therapy. Adv Drug Deliv Rev 56(11): 1649-1659.
- O'Brien MER, Wigler N, Inbar M, Rosso R, Grischke E, et al. (2004) Reduced cardiotoxicity and comparable efficacy in a phase III trial of pegylated liposomal doxorubicin HCl (CAELYXTM/Doxil(R)) versus conventional doxorubicin for first-line treatment of metastatic breast cancer. Ann Oncol 15(3): 440-449.
- Moreno-Aspitia A, Perez EA (2005) Nanoparticle albumin-bound paclitaxel (ABI-007): a newer taxane alternative in breast cancer. Future Oncol 1(6): 755-762.
- Pathak P, Prasad GL, Meziani MJ, Joudeh AA, Sun YP (2007) Nanosized paclitaxel particles from supercritical carbon dioxide processing and their biological evaluation. Langmuir 23(5): 2674-2679.
- Gupta AK, Gupta M (2005) Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 26(18): 3995-4021.
- Klumpp C, Kostarelos K, Prato M, Bianco A (2006) Functionalized carbon nanotubes as emerging nanovectors for the delivery of therapeutics. Biochim Biophys Acta 1758(3): 404-412.
- Yezhelyev MV, Gao X, Xing Y, Al-Hajj A, Nie S, et al. (2006) Emerging use of nanoparticles in diagnosis and treatment of breast cancer. The Lancet Oncol 7(8): 657-667.
- Shi Kam NW, O'Connell M, Wisdom JA, Dai H (2005) Carbon nanotubes as multifunctional biological transporters and near-infrared agents for selective cancer cell destruction. PNAS 102(33): 11600-11605.
- Loo C, Lin A, Hirsch L, Lee MH, Barton J, et al. (2004) Nano shell enabled photonics-based imaging and therapy of cancer. Technol Cancer Res Treat 3(1): 33-40.
- Wu S, Weng Z, Liu X, Yeung KW, Chu P (2014) Functionalized TiO2 based nanomaterials for biomedical applications. Advanced functional materials 24(35): 5464-5481.
- Signoretto M, Ghedini E, Nichele V, Pinna F, Crocella V, et al. (2011) Effect of textural properties on the drug delivery behaviour of nanoporous TiO2 matrices. Microporous and mesoporous Materials 139(1-3): 189-9(2.
- Amna T, Hassan MS, Barakat NA, Pandeya DR, Hong ST, et al. (2012) Antibacterial activity and interaction mechanism of electrospun zinc- doped titania nanofibers, Appl Microbiol Biotechnol 93(2): 743-751.
- Wang Y, Xue X, Yang H (2014) Modification of the antibacterial activity of Zn/TiO2 nano-materials through different anions doped. Vacuum 101: 193-199.
- Hu H, Zhang W, Qiao Y, Jiang X, Liu X, et al. (2012) Antibacterial activity and increased bone marrow stem cell functions of Zn-incorporated TiO2 coatings on titanium. Acta biomaterialia 8(2): 904-915.
- Thevenot DR, Toth K, Durst RA, Wilson GS (1999) Electrochemical biosensors: recommended definitions and classification. Pure and applied chemistry 71(12): 2333-2348.
- Buch RM, Rechnitz GA (1989) Intact chemoreceptor-based biosensors: responses and analytical limits. Biosensors 4(4): 215-30.
- Clark LC Jr, Lyons C (1962) Electrode systems for continuous monitoring in cardiovascular surgery, Ann N Y Acad Sci 102(1): 29-45.
- Zhang Y, Yang M, Portney NG, Cui D, Budak G, et al. (2008) Zeta potential: a surface electrical characteristic to probe the interaction of nanoparticles with normal and cancer human breast epithelial cells. Biomed Micro devices 10(2): 321-328.
- Cui D (2007) Advances and prospects on biomolecules functionalized carbon nanotubes. Journal of Nanoscience and Nano technology 7(4- 5): 1298-1314.
- Jiang LC, Zhang WD (2009) Electro deposition of TiO2 nano particles on multi walled carbon nano tube arrays for hydrogen peroxide sensing. Electro analysis 21(8): 988-993.
- Pang X, He D, Luo S, Cai Q (2009) An amperometric glucose biosensor fabricated with Pt nanoparticle-decorated carbon nanotubes/TiO2 nanotube arrays composite. Sensors and Actuators B: chemical 137(1):134-138.
- Kang Q, Yang L, Cai Q (2008) An electro-catalytic biosensor fabricated with Pt-Au nanoparticle-decorated titania nanotube array, Bio electrochemistry 74(1): 62-65.
- Liu X, Zhang J, Liu S, Zhang Q, Liu X, et al. (2013) Gold nano particle encapsulated-tubular TiO2 nano cluster as a scaffold for development of thiolated enzyme biosensors. Analytical chemistry 85(9): 43504356.
- Tang H, Yan F, Tai Q, Chan HL (2010) The improvement of glucose bio electro catalytic properties of platinum electrodes modified with electrospun TiO2 nanofibers. Biosensors and Bioelectronics 25(7): 1646-1651. 2
- Shu X, Chen Y, Yuan H, Gao S, Xiao D (2007) H2O2 sensor based on the room-temperature phosphorescence of nano TiO2/SiO2 composite. Analytical chemistry 79(10): 3695-3702.
- Li Y, Liu X, Yuan H, Xiao D (2009) Glucose biosensor based on the room-temperature phosphorescence of TiO2/SiO2 nano composite. Biosensors and bioelectronics 24(12): 3706-3710.
- Mathur S, Erdem A, Cavelius C, Barth S, Altmayer J (2009) Amplified electrochemical DNA-sensing of nano structured metal oxide films deposited on disposable graphite electrodes functionalized by chemical vapor deposition. Sensors and Actuators B: Chemical 136(2): 432-437.
- Topoglidis E, Cass AE, Gilardi G, Sadeghi S, Beaumont N, et al. (1998) Protein adsorption on nano crystalline TiO2 films: an immobilization strategy for bioanalytical devices. Analytical chemistry 70(23): 51115113.
- Li Q, Cheng K, Weng W, Du P, Han G (2013) Titanium dioxide nano rod- based ampere metric sensor for highly sensitive enzymatic detection of hydrogen peroxide. Microchimica Acta 180(15-16): 1487-1493.
- Viticoli M, Curulli A, Cusma A, Kaciulis S, Nunziante S, et al. (2006) Third-generation biosensors based on TiO2 nanostructured films. Materials Science and Engineering 26(5-7): 947-951.






























