Abstract
Sarcodes, potentized animal organs and tissues, are part of the clinical organo- therapy of homeopathic and anthroposophical medicine. The rationale for the indications of therapeutic applications differs between the various schools of thought on different continents. While anecdotal reports of clinical successes are frequent, only few clinical and preclinical studies have been published. In this in vitro experiment, we show for the first time that the sarcodes made from potentized liver, lung, heart, kidney, and spleen are able to inhibit to a statistically significant degree the proliferation of the LNCaP prostate, Hep3B liver, A-498 kidney and Y-1 adrenal cancer cell lines. We also find that potencies of the sarcodes can be both stimulating and inhibitory, which leads one to the necessity of determining experimentally the behavior of the individual potencies before they are clinically used.
Keywords:Sarcodes; Homeopathy; Anthroposophy; Y-1; Hep3B; LNCaP; A-498
Introduction
In general, sarcodes, potentized remedies made from healthy animal tissues, are seen as restorative remedies. They are most often not used as single treatments but help to augment sensitivity to the overall therapeutic plan [1]. The therapeutic rationale for using sarcodes has been approached in varying ways by homeopathic/anthroposophical practitioners [2,3]. The “French school” [4] uses only a few potencies of the various sarcodes (largely glandular) to accomplish its goals: 4CH and 5CH for stimulating organ function; 7CH, 9CH and 12CH for regulating organ function; and 15CH and 30 CH for inhibiting organ function. The assumption is being made that the potentized remedies contain corrective information for the respective organs.
Thus, for example, the above-mentioned potencies of the hypophysis are given to regulate abnormalities of the endocrine system. Potentized sarcodes are used in low potencies for local or limited symptoms; middle potencies for functional symptoms like spasms, headaches etc.; and higher dilutions are recommended for chronic diseases and behavior related symptoms. Potentized sarcodes are also used for “detoxifying the body physiology”, overcoming stress related physiological abnormalities, and more.
In homeopathic Materia Medica’s, the indication for the use of remedies is based on the so-called “provings” [5]. The remedies are sourced most frequently from the mineral and plant kingdom or from insect parts. In Phatak’s Materia Medica [6], only Thyroidinum, made from potentized dried thyroid gland of sheep or calf is listed as a sarcode coming from an animal organ. However, more recently, some Materia Medicas have been published specifically relating to sarcodes [7]. In India the followers of the Sankaran [8] school of thought apply sarcodes based on the so-called “sensations” that the patient presents with [9]. The choice of the remedy is made by understanding the overriding physiological perception that the patient has in his/her need to maintain a desired status quo.
Gaikwad [10] summarizes his impressions of the use of sarcodes by recommending that one could “Go for it when there is lack of reaction, and any particular organ, enzyme, hormone, or sphere of action needs to be stimulated; when mind and physical symptoms alternate; when there is lack of or exaggerated function of any particular organ/system or you are dealing with metabolic and hormonal disorders and need an intercurrent remedy. For each sarcode, you can read about what function it serves normally, what effect it has on the body, and clinical disease for which it is useful.” In all the schools mentioned above the use of sarcodes made from liver, lung, heart, spleen, or kidney is not reported.
This is different among the so-called anthroposophical doctors who follow the thinking of Rudolf Steiner [11]. Their widespread use of potentized sarcodes is intended to address body, mind, and spirit issues. Lower potencies (D7-D14) are understood to affect more metabolic processes; “midrange potencies” (D15-D21) rhythmical processes such as respiratory and circulatory; “high potencies” (potencies close to D30) the nervous -sense system. Sarcodes used in anthroposophical medicine are manufactured from nearly all organs and organ tissues such as nerves, endocrine glands, and from liver, lung, heart, spleen, or kidney. More than in other schools of thought anthroposophical doctors assign an important role to sarcodes for stimulating the immune system and supporting cancer care [12].
In our own work we have found indications for the use of sarcodes dictated by specific psychological moods [13]. The objective effectiveness of sarcodes has been considerably less researched than other potentized remedies derived, as mentioned above, from mineral, insect or plant sources. However, we have found several independent clinicians that have used sarcodes to improve the quality of life of patients. In a clinical study Ghare [14] concluded that Thyroidinum 3X has effect on anti TPO Ab titers and prevents progression to overt hypothyroidism. Khara and Whadhwani [15] reports outcome improvement in a patient where in home potentized mitral valve in ascending LM potencies was used.
Schmid et al. [16] showed that treatment of patients with ultrahigh dilutions of Thyrodinum 30CH contributed to weight reduction if the experimental parameters were well controlled. Lauppert [17] and Tadei [18] showed that potentized thyroxine had a curative effect in hyper stimulated frogs. Naidoo [19] demonstrated effectiveness of cat Saliva sarcode in Allergic adults. Finally, Verma [20] showed in a review article that sarcodes and nosodes represent valuable tools in the chronic management of renal stones within homeopathy.
A few animal studies have been published like the pre- clinical homeopathic study involving Insulinum 6CH, and Pancreatinum 6CH that exhibit antihyperglycemic effects in streptozotocin induced diabetic rats [21]. After an extensive literature search we have not been able to find any in vitro studies on sarcodes and especially not related to cancer applications. In the present study we are demonstrating a statistically significant effect of the potentized sarcodes of liver, lung, heart, kidney, and spleen on several cancer cell lines.
Materials and Methods
Sarcode Organ Potentization
The raw bovine organ material was obtained from New Zealand (There has never been a case of indigenous BSE in New Zealand). The organ material came with a certificate that the organs originated from cows that were 100% free range animals, 100% pasture fed, raised without genetically engineered feed (Non-GMO), raised without added hormones, and given no artificial ingredients. The organs were flaked, heated in water, then cooled, to remove as much fat as possible. These are then freeze dried, milled and sieved into a powder. Prior to use in the experiments the powder was then tested by an independent lab for heavy metals and microbiologic contamination.
The “potency”, a dilution prepared in “homeopathic” style, was designated as D1, and was made by adding 5 g of the bovine organ powder to 45 g lactose and mixed with a mortar and pestle for 1 hour. From D1, 5g was added to 45 g lactose and mixed as before; this was designated D2. From D2, 5g was added to 45 g lactose and mixed as before; this was designated D3. To make D4, 1 g of D3 was added to 9 ml sterile diH2O and mixed for 30 minutes and rested for 5 minutes then sterilized using a 0.22 μm filter (SimPure, B09B9QNXRL). Potencies D5 and D6 were made using 1 ml of the previous potency to 9 ml sterile diH2O, mixed for 30 minutes and rested for 5 minutes. D7-D30 were made using 1 ml of the previous potency to 9 ml sterile diH2O, mixed for 2 minutes and rested for 2 minutes.
Cell Culture
Y-1, a mouse adrenal normal cell line (ATCC, CCL-79), was typically seeded at 20% confluency in 75cm2 flasks, and grown to approximately 80% confluency at 37˚ C, 5% CO2, which typically took four days. The medium used was ATCC formulated F-12K (ATCC, 30-2004) supplemented with 2.5% Fetal Bovine Serum (FBS; ATCC, 30-2020), 15% horse serum (Gibco, 26050070) and 1% penicillin streptomycin (ATCC, 30-2300). Once confluent, the medium was removed, and the cells rinsed with 3 mL 0.25% Trypsin, 0.53 mM EDTA (TE; ATCC, 30-2101) and the cell monolayer was then harvested by adding 3 mL TE to the flask and incubated at room temperature for 5 minutes, followed by 6 mL fresh medium. The suspension was transferred to a 15-ml centrifuge tube, and sedimented at 130 x g for 7 minutes. The supernatant was removed, and the pellet resuspended in fresh media. The countess 3 (Invitrogen, AMQAX2000) was used to count cell inoculum and to check viability.
Hep3B, a human liver cancer cell line (ATCC, HB-8064), was typically seeded at 20% confluency in 75cm2 flasks, and grown to approximately 80% confluency at 37˚ C, 5% CO2, which typically took four days. The medium used was Eagle’s Essential Medium (ATCC, 30-2003) supplemented with 10% FBS and 1% penicillin streptomycin. Once confluent, the medium was removed, and the cells rinsed with Dulbecco’s Phosphate Buffered Saline 1X (D-PBS) (ATCC, 30-2200). The cell monolayer was then harvested by adding 3 mL TE to the flask and incubated at room temperature for 5 minutes by 6 mL fresh medium. The suspension was transferred to a 15-ml centrifuge tube, and sedimented at 130 x g for 7 minutes. The supernatant was removed, and the pellet resuspended in fresh media. The Countess 3 was used to count cell inoculum and to check viability.
LNCaP clone FGC, a human prostate cancer cell line (ATCC, CRL- 1740), was typically seeded at 20% confluency in 75cm2 flasks, and grown to approximately 80% confluency at 37˚ C, 5% CO2, which typically took four days. The medium used was RPMI-1640 (ATCC, 30-2001) supplemented with 10% FBS and 1% penicillin streptomycin. Once confluent, the medium was removed, and the cells rinsed with D-PBS. The cell monolayer was then harvested by adding 3 mL TE to the flask and incubated at room temperature for 5 minutes, followed by 6 mL fresh medium. The suspension was transferred to a 15-ml centrifuge tube, and sedimented at 130 x g for 7 minutes. The supernatant was removed, and the pellet resuspended in fresh media. The Countess 3 was used to count cell inoculum and to check viability.
A-498, a human kidney cancer cell line (ATCC, HTB-44),), was typically seeded at 20% confluency in 75cm2 flasks, and grown to approximately 80% confluency at 37˚ C, 5% CO2, which typically took four days. The medium used was Eagle’s Essential Medium (ATCC, 30-2003) supplemented with 10% FBS and 1% penicillin streptomycin. Once confluent, the medium was removed, and the cells rinsed with 3 mL TE and the cell monolayer was then harvested by adding 3 mL TE to the flask and incubated at room temperature for 5 minutes, followed by 6 mL fresh medium. The suspension was transferred to a 15-ml centrifuge tube, and sedimented at 130 x g for 7 minutes. The supernatant was removed, and the pellet resuspended in fresh media. The countess 3 was used to count cell inoculum and to check viability.
Cell Viability Assay
Cells were plated in white, 96-well plates (Costar, CLS3917) at a density of 5 x 104 per well for Y-1 and Hep3B, 1 x 104 cells per well for LNCaP, and 8 x 104 cells per well for A-498 and a volume of 90 μl per well. The controls contained 10 μl ddH2O (0% and 100% controls) and the experimental wells contained 10 μl of the appropriate potency (D7-D30), with four replicates per treatment or control. Plates were left for 1 hour at room temperature, before subsequent incubation at 37˚ C and 5% CO2, to produce uniform cell distribution on the growth surface and reduce edge effect. Cell growth was assessed using the CellTiter-Glo luminescent cell growth assay (Promega, G9243), using 100 μl of reagent per well according to the manufacturer’s instructions. The luminescence was measured in a BioTek, Synergy LX plate reader. Cell growth was assessed for 0% control wells after an initial 18 hours of incubation, while sample and 100% control wells were assessed after an additional 48 hours of growth.
Statistical calculations
Cell count analysis was performed on four measurements, 16 wells in total (two sets of quadruplicates on two different plates). All samples were included in the analysis. Data are expressed as mean ± standard deviation. Significance (p < 0.05) and STDEV calculations were done with the Microsoft Excel software.
The percent growth was measured according to the following
formula:
% Growth = 100 x (Sample – 0% Control)/(100% Control–
0% Control)
Results & Discussion
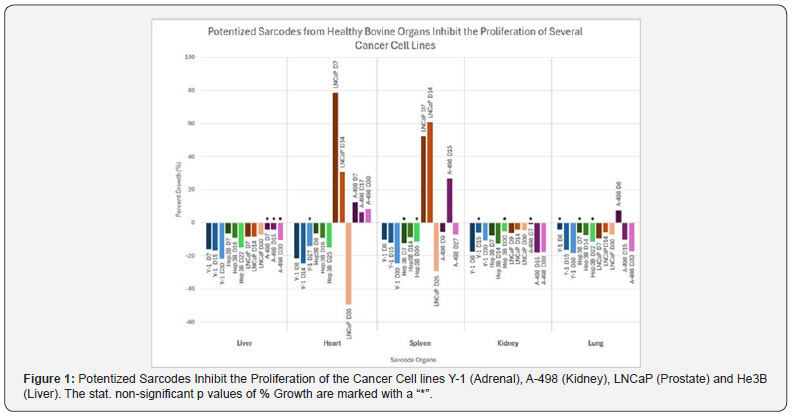
Here we are showing for the first time that the potentized sarcodes of liver, kidney, heart, lung, and spleen can inhibit the proliferation of multiple cancer cell lines in a statistically significant manner (Table 1 & Figure 1). Considering the worldwide search for additional cancer therapies that are better tolerated, this novel finding pointing to future possible therapeutic applications merits further study. However, it is important to note that in several of the experiments, potencies of the sarcodes could also stimulate the cancer cell line.
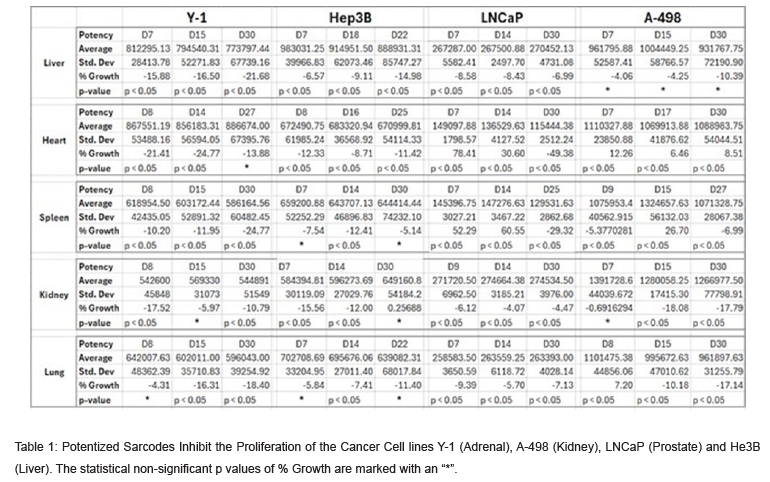
This confirms previous findings of various authors that each potency of a substance needs to be individually experimentally investigated to see in which direction – inhibiting or stimulating – it is active, or possibly not active at all [22-24]. We are the first to demonstrate in the study that sarcodes of potentized liver, kidney, lung, heart, and spleen, can inhibit the proliferation of cancer cells. From the point of view of conventional medicine sarcodes are seen as being of questionable value [25]. Only a more consistent effort in the direction of controlled studies, both basic science and clinical testing, will help to overcome this attitude. Consequently, studies such as the present one pointing to significant potential benefits for the use of sarcodes should continue in the future.
Authors Participation
i. R Rentea MD - designed the experimental project and wrote
the article.
ii. M Mueller M. Sc – executed, calculated the experimental
data, and wrote the Materials & Methods section.
iii. M Kamsler MD - participated in the potentization of the
sarcode material.
Conflict of Interest
The authors report no conflict of interest.
References
- Mashru R, Kajal Gandhi, Atodaria Ekta and Vedshree Raole (2017) Sarcode Therapy as a Restorative of Health. World Journal of Pharmacy and Pharmaceutical Sciences 6(8): 2579-2598.
- Sankar K, Jadhav AP (2017) Nosodes and sarcodes 16(1): 158-163.
- Tenka DrY (2021) Sarcodes and its homoeopathic application. International Journal of Homoeopathic Sciences 5(4): 51-54.
- Bosch J van den (2024) Sarcodes the Hidden Gems of Homeopathic Medicine. Emryss.
- Squire B (2011) Repertory of Homoeopathic Nosodes and Sarcodes. B Jain Publishers Pvt Limited.
- Phatak SR (2013) Materia Medica of Homeopathic Medicine. B. Jain Publishers.
- Nayak C, Smita Brahmachari, Syed Afsar Ali (2016) Homoeopathic Materia Medica of Sarcodes. B. Jain Publishers.
- Rajan Sankaran (2005) The sensation in homoeopathy. Mumbai: Homoeopathic Medical Pub.
- Kalathia G (2011) The Potential of Sarcodes [Internet]. Homeopathic Links. Thieme.
- Gaurang Gaikwad (2022) Materia Medica of Nosodes and Sarcodes by Dr.– Reviewed by Vatsala Sperling. Hpathy.com.
- Steiner R (2010) Introducing Anthroposophical Medicine. Rudolf Steiner Press.
- Roemer F (2014)Therapiekonzepte der Anthroposophischen Medizin . Karl Haug Verlag.
- Rentea R (2025) Articles/Books - Lilly Kolisko Institute [Internet]. Lilly Kolisko Institute.
- Prajakta Ghare, Jadhav AB, Patil AV (2020) A Clinical Study to See the Effect of Thyroidinum, a Homoeopathic Preparation on Thyroid Peroxidase Antibody in Subclinical Hypothyroidism of Age Group between 18-70 Years. International Journal of Health Sciences and Research 10(2): 18-22.
- Kara S, Wadhwani G (2018) Two Clinical Experiences with a New Sarcode: Potentised Mitral Valve in LM Potencies. Homœopathic Links 31(04): 241-247.
- Schmidt J, Ostermayr B (2002) Does a homeopathic ultramolecular dilution of Thyroidinum 30cH affect the rate of body weight reduction in fasting patients? A randomised placebo-controlled double-blind clinical trial. Homeopathy 91(4): 197-206.
- Lauppert E (1998) Enhanced Inversion Effect of Thyroxine log10(-13) By Agitation Curative Effect Following Hyperstimulation in Frogs. In: High Dilution Effects on Cells and Integrated Systems. World Scientific Publishing.
- C Taddei-Ferretti, P Marotta, Istituto Italiano Per Gli Studi Filosofici (1998) High dilution effects on cells and integrated systems. Singapore; River Edge, Nj: World Scientific.
- Naidoo P, Pellow J (2013) A randomized placebo-controlled pilot study of Cat saliva 9cH and Histaminum 9cH in cat allergic adults. Homeopathy 102(2): 123–129.
- Verma A (2025) Role of Sarcodes and Nosodes in Chronic Renal Stone Management: A Review. World Journal of Pharmaceutical Research 14(9): 734-739.
- Aditya Dilipkumar Patil, Charusheela Dilipkumar Patil, Dilipkumar Kondiba Patil (2021) Evaluation of Antihyperglycemic potential of homeopathic medicines Insulinum, Pancreatinum and Uranium nitricum in Streptozotocin Induced diabetic Rats. International Journal of High Dilution Research 19(3): 02-17.
- Rentea R (2024) Anthromedics - Der Merkurstab: Rhythmical behavior of ultra-high potencies of substances in cell cultures: a pilot study. Anthromedics.org.
- Kolisko LN, Kolisko L (1960) Physiologischer und physikalischer Nachweis der Wirksamkeit kleinster Entitäten. Arbeitsgemeinschaft anthroposophischer Aerzte.
- Husemann F (2010) Rhythmusphanomene beim Wirksam - keitsnachweis potenzierter Heilmittel – nachgewiesen von Kolisko (1923) bis Cristea (1991). Der Merkurstab 25(45).
- Viktoria Vieracker (2015) Nosode and sarcode therapies and their history--a controversial inheritance. Medizin, Gesellschaft, und Geschichte : Jahrbuch des Instituts fur Geschichte der Medizin der Robert Bosch Stiftung 33: 155–177.






























