Abstract
As a result of increased consumer awareness of the organic nature of eco-friendly goods, renewed interest has been sparked in the use of textiles (preferably natural fiber products) dyed with eco-friendly natural dyes around the world. This research work was conducted in single stage/one bath dyeing which had been performed on 100% cotton knit fabric by applying Reactive and Mahogany (Swietenia mahagoni) leaf extracted dyes simultaneously. Because the mahogany plant leaf contains a triterpenoid group that consists of plenty of hydroxyl groups to be bonded with cotton, which has the same bonding nature of reactive dyeing on cotton, will create an opportunity to contract a revolutionary change in the dyeing sector as well as the textile industries. The dying procedure was carried out at 100oC temperature by the application of Reactive (red) dye and Mahogany leaf extract dye without using mordant. In the reactive-mahogany dyeing, reactive dye required conventional chemicals and auxiliaries like salt, soda etc, whereas mahogany dye did not require any chemicals and auxiliaries, which indicate eco-friendly and a scope to reduce the effluent load on account of the inclusion of natural dyestuff, energy saving dyeing process. Saving costs and dyeing costs can be more possible than double bath dyeing. Finally, an optimistic shade was observed after the research work and fastness properties, e.g. wash fastness rating 4 to 5, rubbing fastness was 4 and 5 (under wet and dry condition), perspiration fastness was showed 4. Evenness of shade along with the K/S values exhibited the as usual value like the conventional results. Because of the addition of mahogany dyes, the CIE L* a* b*detailed results kept proportionality with the rise of the depth of shades. From FTIR data, the characteristic peak for reactive-mahogany was approximately around 1190-1210cm-1, which represents confirming the bonding of dye with fiber. In addition, thermal degradation of dyed samples, the overall weight loss of the reactive-mahogany dyed samples was optimistic.
Keywords: Natural dye; Mahogany leaf; Single stage dyeing; Reactive dye; Eco-friendly
Abbreviations: TGA: Energetic Thermo Gravimetric Analysis; FTIR: Fourier Transform Infrared Spectroscopy
Introduction
Indisputably, wet processing is a fundamental part and parcel of textile industries and also can’t walk a single moment without synthetic dyes. However, synthetic dyes possess a number of demerits, like consuming a lavish amount of water and being not hygienic for the human body too, and are highly toxic as well, so they are not friendly to the environment. Moreover, synthetic dyes apply not only in single bath but also in double bath or two-bathdyeing process and this dyeing depends on what types of fabric are used, e.g. blend fabric, cotton, silk etc. and how many dyes are done. Besides, it can be said that there has been quite an abundant amount of research on the double bath or two-bath-dyeing process; so far, multiple colors and several other chemicals have been utilized step by step on textile materials in two-bath-dyeing. Following the conventional method, double bath dyeing was done in two stages on cotton fabric or blended fabric [1-11].
Consequently, energy losses, time-consumption, high costs along with other problems arise with double bath or two-bathdyeing [11]. For this, an alternative way is required to dispose of this problem, such as single bath or one-bath-dyeing can be considered. A lot of work had been done on single bath dyeing [12, 13]. Some researchers have shown that double bath dyeing is better for darker shade as well as single bath dyeing is better for lighter shade [11]. All the work was done with double bath dyeing by synthetic dyes only. But the inclusion of natural dyestuff with synthetic dyes is still a demand. Natural dyes have great potentiality over synthetic dyes. The importance of natural dyes is increasing day by day due to their eco-friendliness [1-10]. Natural dyes could be utilized as an alternative to synthetic dyes in fireproof garments, food garments, kids’ apparel, medical uniforms, sports costumes etc. for safety [14]. Natural dyes have also been applied both on synthetic (e.g. polyester etc.) and natural fiber like cotton; jute etc. [15-17].
As per our knowledge, no experimental work has been conducted on the application of synthetic and natural dyes simultaneously on the same fiber. In this current work, single bath or one-bath-dyeing has been done on cotton knit fabric by applying Reactive (red) and Mahogany leaf extract dye simultaneously. There are a tremendous number of mahogany plants in Bangladesh which develop a bounty of leaves each year. These leaves have no industrial uses in the growing zones and, subsequently, tons of tons of leaves are essentially wasted. On the other hand, the scientific use of these leaves for textile coloring purposes may be an efficient way to diminish the problem of such waste disposal and, at the same time, financial utilization in textile industries. The family of timber trees like mahogany (Meliaceae) leaf is the full source of triterpenoid, Scopoletin, melianone, cyclo mehogenol, swietenin, stigmasterol glucose, limonoids etc. [18- 20]. Triterpenoid is one of the major constitutional components of Mahogany plant leaf (Swietenia mahagoni) extract [21].
Most triterpenoids are alcohol and can combine with sugars to form glycoside [22]. Glycoside bonds are also [-O-] type, which resembles the bonding nature of reactive-cellulose. Due to the similarity of bonding nature, it seemed useful to have looked into the application outcomes of reactive and mahogany dyeing at the same time on cotton. That would be the scope to incorporate modification of color or a multicolor effect and also a scope to reduce the effluent load on account of the inclusion of natural dyestuff. In spite of the fact that a little research has been explored on the application of mahogany dye extraction from mahogany seed pods, rubiadin dye (from bark) both on synthetic (polyester, nylon) and natural fiber (silk) [23-25]; even mahogany leaf extract dye or other natural dyes have been applied on cotton, silk, wool or synthetic fabric by dint of mordant or without mordant [23-28].
Indeed, nowadays numerous parts of mahogany plants remain unexplored. Besides, there has been less work on hybrid dyeing (combination of two or more dyes) so far; like as- using two or more natural dyes extracted from Caesalpinia sappan and Leucaena leucocephale L. leaves or mango and turmeric leaf or other natural sources that were applied on natural fiber (cotton, silk) with help of mordant [29-31]. And there also had a small similarity between hybrid dyeing and reactive-mahogany simultaneous dyeing because of using two dyes or colors. In the actual study, reactive-mahogany dyeing was completed in single bath or one bath process without the assistance of any mordant and reactive dye required salt, soda, other auxiliaries, whereas mahogany dye did not require any auxiliaries. So, it was very important to see where the chemicals do any harm to the mahogany dyeing on cotton or not. Compatibility study of the dyes on cotton was crucial to reveal the dyeing properties. The improvement of new shades for dyeing materials incorporating modification of color was possible by reactive-mahogany dyeing simultaneously. Dyed fabric degradation [21] scopes will also be investigated.
Experimental
Materials and equipment (Table 1) Methods Preparation/extraction of Mahogany dye
Mahogany leaf/leaves were washed appropriately that no impurities could introduce on the leaves. After that clean leaves were blended by blender machine and turned into highly concentrated liquid color. To dry liquid color properly by using oven dryer machine so that no water could presence in the color and then it was turned into powder dye (Figure 1).
Dyeing procedure
At first sample cotton knit fabric and stock solutions were prepared as per recipe. Then stock solutions were taken into closed bath and required water was also added into the bath. It should be specified that the amount of glauber salt and soda ash would be changed according to the dye/shade percentage. Here, chemicals and other auxiliaries were used for reactive dye but no auxiliaries for mahogany dye. Then total dyeing was done at 100oC temperature for 1 hrs or 60 min. The material and liquor ratio was 1:15 (closed bath). After dyeing, hot wash was performed thrice. Then two times cold wash were done by normal water as if uniform shade performs. Finally, samples were dried by dryer machine to eliminate water.
Spectrophotometer operation
Instrumental color measurement/estimation of the conditioned colored samples were carried out using a Data color 650 spectrophotometer with 10oLAV (large area view) observer using different standard CIE illuminants: D65, TL83 and A. There were two softwares such as- data color tool (DCI- Tool) and data color match (DCI- Match) used to evaluate the measurement of different shades.
Wash fastness testing
The dyed samples were washed by washing & dry- cleaning color fastness tester; according to ISO 105 C10:2006 method. The sizes of samples were 10x4cm which were sandwiched between two adjacent fabrics and stitched. The material and liquor (M: L) ratio was 1:50 and the temperature of washing condition at 60oC and time of washing was 30 min. After washing, the samples were rinsed twice in cold water. Both washed and unwashed dyed samples were subjected to the wash fastness test utilizing a standard grey scale.


Rubbing fastness testing
The dyed samples were rubbed by Crock Master according to EN ISO 105X12:2001 standard under both condition (dry and wet). The sizes of the samples were 10x2cm, arm was weighted to provide a constant 9N load on the samples at all times and mechanical counter was kept track of completing 10 cycles. After completion of rubbing test, samples were compared AATCC grey scales for staining of color.
Perspiration fastness testing
Perspiration test Perspire Meter Phenolic Yellowing Tester & Incubator was performed according to ISO 105-E04 in alkali condition. The sizes of samples were 10x4cm which was attached the multi fiber adjacent fabric of the same size. Then switch on oven and pre heat perspire-meter and acrylic plates for 30 min. After that the samples were put in a disk with 100ml of sweat solutions and completely with the assistance of acrylic plate to wet the samples & multi fiber for 30 min pour off the excess solution from sample by utilizing glass or wringer. To keep each sample between two glass or acrylic plates and put them in the perspiremeter. Each perspire-meter could hold maximum 10 samples. In the event that all 11 plates were still need to be put within the perspire-meter. To put a pressure of 12.5kpa on the perspire-meter utilizing the load, lock it. 12.5kpa pressures must be equitably distributed on the sample surface. Plates must be parallel to each other; slant of plates due to thick & thin surface (uneven sample surface) must be avoided. To put the perspiration meter within the oven for 4 hours at (37±2) oC in upright position. After that, the samples were taken out from the oven and perspiration meter. Opened out each sample & dried those, with the sample as it were being in contact at the point of the stitching.
FTIR test
Parameters were utilized in estimations were: resolution 4cm-1, spectral range: 4000-600cm-1on FTIR spectrometer. Omic TM software computer program was performed together and processed the IR spectra. Samples were set on the FTIR spectrums and then were scanned. And the spectra were obtained in this work collection of FTIR spectra contained presently through and through many spectrums of several samples.
TGA test
Energetic thermo gravimetric analysis (TGA) was performed by employing a TA Instrument SDT 650 Model that helped to change in transition of sample under conditions of controlled temperature, time and atmosphere. All samples analysis was conducted under a nitrogen atmosphere, and samples were filled with alumina crucibles. Temperature programs for dynamic tests were run from average 27oC to 500oC at temperature rate 5.00oC/ min. The samples weighting approximately 6mg to 20mg that arranged to prevent any thermoxidative degradation.
Results and Discussion
The color strength (K/S) value analysis
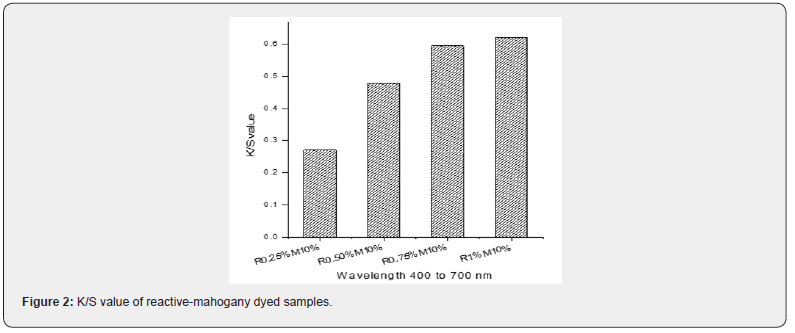
The co-efficient reflection of all dyed samples for the wavelength of 400 to 700nm was observed. K/S value represents the characteristics of dyed samples and it is a straightforward way to decide a color as a concentration. It is a single numerical value based on spectral data which represents the quantity of light absorbing substance (Figure 2). The K/S values of reactivemahogany dyed samples, R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10%, were presented at (Figure 3).
It is clear from (Figure 2) that, during the fixed amount application of mahogany leaf extract dye (10%), the k/s values of the samples are getting higher because of the gradual increment of individual reactive red color. When the samples were dyed with individual mahogany dye, the higher amount of dye applied ensured the higher k/s values. Thus, it can be stated that the mahogany and reactive dye application at a simultaneous method confirmed their coloration on the fabric proportionally on the basis of application concentration or depth of color.
Evaluation of CIE L* a* b* Value along with visual assessment
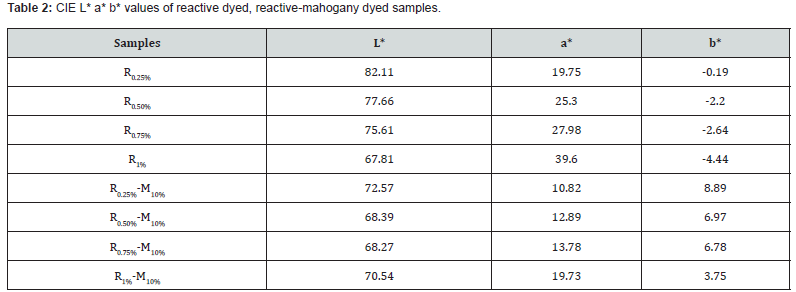
Data color 650 Spectrophotometer was used to estimate the CIE value of different samples which were dyed with reactive and mahogany leaf extract dye. Reactive samples were considered as standard, besides the CIE L* a* b* values of reactive-mahogany samples were also investigated. (Table 2) was displayed the entire color shade of L*, a*, b* values indicating that higher L* values indicated lighter shade and lower L* values indicated darker shades. Green and blue were represented by negative a*, b* values; whereas red and yellow were represented by positive a*, b* values. So, from this information it was clear that L*values of the resulted samples R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10% (reactive- mahogany dyeing) were decreasing than the L* values of standard samples R0.25%, R0.50%, R0.75% and R1% (reactive dyeing only); where increasing of reactive dye percentages were confirmed. At the same time a* values of resulted samples were also decreased proportionally from those of standard samples.
Therefore, a* values of resulted samples R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10% indicated inclination towards reddish color which was supposed to be happened. Whither, the difference of b* values was negligible as reactive red color was applied for reactive dyeing. Then b* values of resulted samples R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10%were increased gradually from those of standard samples R0.25%, R0.50%, R0.75% and R1%. So, b* values of all reactive-mahogany samples referred to yellowish color. Above the CIE value, an authentic color combination/improving new shade may be offered by reactive-mahogany dyeing. From the discussion it can be concluded that reactive dyeing and mahogany dyeing occurred at an independent mode on cotton, where the auxiliaries of reactive dye worked for the reactive dyeing and at the same time mahogany dye did not create any obstacle for reactive dyeing. Thus, the compatibility of the dyes is ensured. The visual evaluation is imperative to assess the dyed samples and enhancing properties from a common consumer’s point of view. Visual assessment of dyed samples especially reactive-mahogany and only mahogany samples was given below (Figure 3).
In R0.25%-M10% sample, mahogany dye may be relatively more prevalent than reactive dye. Then R0.50%-M10% sample may be equal mixing of reactive and mahogany dye properly. In R0.75%-M10% and R1%-M10% sample were being darker reddish color with regard to the reactive dye percentage was increased more than the previous sample.
FTIR analysis
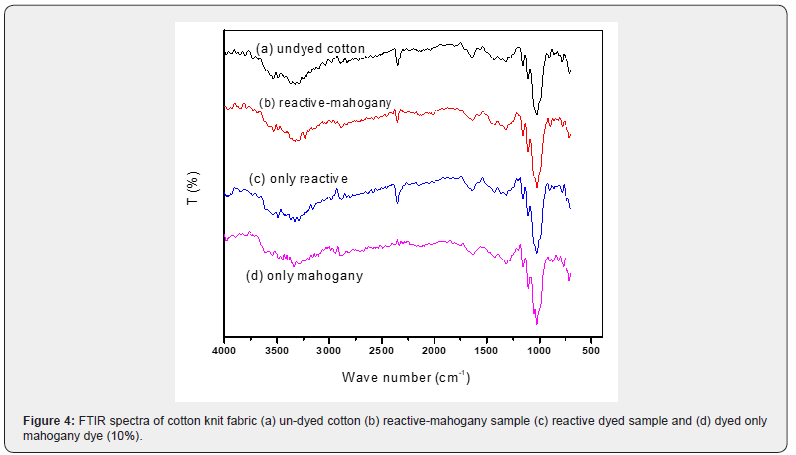
Fourier Transform Infrared Spectroscopy (FTIR) was used to evaluate the colorfastness and color intensity features of various samples. After coloring the samples by applying reactivemahogany leaf extract dye simultaneously, FTIR was performed. (Figure 4), FTIR spectral observations was used to easily measure the intensity of color utilizing an infrared spectroscopy with the facilities of FTIR equipment. Infrared rays could penetrate a few microns into the sample surface and generated spectrum wave number as results. A spectral scan was not possible since the dyed sample could not capture infrared light. The peak associated to the material from the spectrum scan was detected employing infrared light and the enduring spectral scan assessed. In the FTIR observation, the infrared spectrum range was from 4000-600cm-1 wave number.
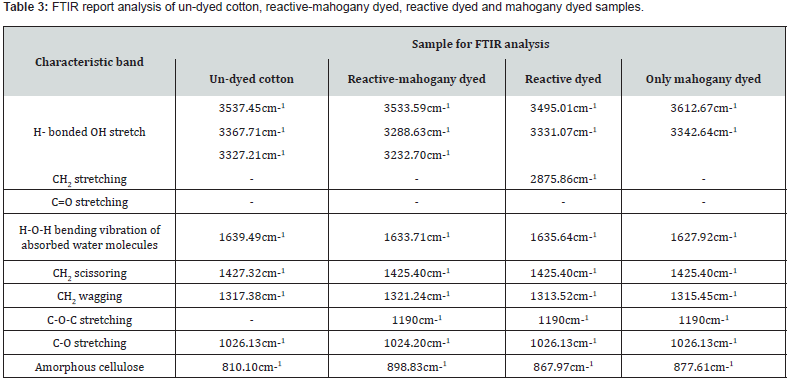
With regard to cotton fabric, the FTIR transmission spectra could say the distinction of 04 (four) samples such as- un-dyed cotton, reactive-mahogany shade sample, only reactive shade sample and only mahogany dyed sample due to estimate almost the chemical structure of fiber [32-35].
For un-dyed cotton, reactive-mahogany sample, only reactive dyed sample and only mahogany sample; the lowest and height peaks were 698cm-1 and 4000cm-1 respectively. These peaks were unique for identifying the chromophore present in the dyestuff that was responsible for certain colors shades on sample, and they clarified the existence of the color in the observable spectral range (Table 3). As per established theory of only reactive dyed sample the characteristic peak was at 1190-1210cm-1 represent the -C-O-C- stretching for the covalent bond formed with the cellulose ion of cotton and the cationic carbon of triazynyl reactive dye, or the terminal carbon of vinyl sulphone reactive dye [36].
Again, during dyeing with mahogany leaf extract dye, the sample exhibited the same characteristic peak at around 1200cm- 1, confirming the bonding of dye with fiber. It was known that the chemical component of mahogany leaf extract was mostly triterpenoids of limonoid class [35], where most triterpenoids were alcohol and could combine with sugars to form glycoside [22]. The repeating unit of cotton was cellubiose, where two β- glucose units were linked with 1-4 glucosidic linkage. At the region of 1200cm-1the characteristic band got stronger peak intensity than the peaks of the individually dyed (reactive-mahogany) samples. The peak was seemed to attain the collective shape because of the simultaneous dyeing with reactive and mahogany dye.
Color fastness to Wash
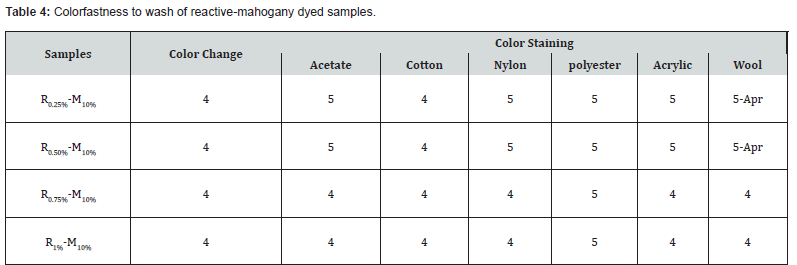
Color fastness to wash is the common quality parameter, which is considered expectation vital from the point of view of buyers. This test determines the loss and change of color within the washing process. For color staining and color change of samples, color fastness to wash was evaluated in two phases by using two separate grey scales. Test of color fastness to wash was carried out and the most excellent results were obtained (both in color change & color staining).
The observation an effect on the colorfastness of reactivemahogany dyed samples were R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10% when washed, based on the above (Table 4) demonstrated that colorfastness to wash had an excellent grading for color staining with a rating of almost 4 to 5. Where, the color staining, acetate, nylon, polyester, acrylic was nearly same. As reactive dye identically created covalent bond with cellulose fiber, thus the fastness properties would be better in all the cases. Similarly, mahogany leaf extract dyes also created bond with cellulose [basically triterpenoid reacts with glucose, which had mentioned earlier] around the zone about 1200cm-1. FTIR data ensured that the reactive-mahogany dyed sample was exhibited extended peak at the targeted zone, and thus the color fastness to wash was reasonably provide better values.
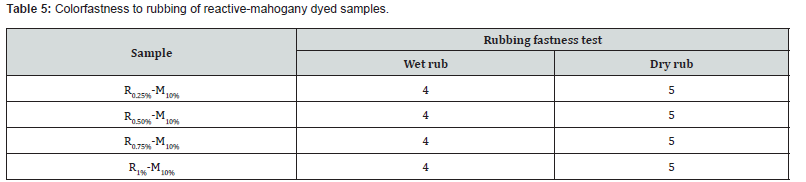
Color fastness to rubbing
Fastness is the elemental necessity that dyed samples ought to display, to the conditions in which they will undergo during the processes that follow coloring and even during their subsequent utilization. This test was outlined to determine the amount of color transferred from the surface of dyed cotton knit fabric to other surface by rubbing. Best results of color fastness to rub (under wet and dry condition) were obtained (Table 5). The grey scale rating of rubbing fastness was showed 4 & 5 (under wet and dry condition) for both shades such as sample R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10%. Here, 4 and 5 were indicated absolutely good-excellent level too. Samples fastness properties could vary depending both on color strength (K/S) value and fixation percentage. In addition to fastness of dyed samples were also connected to FTIR data because of the peak for the most intense hue reactive- mahogany dye was almost around 1200cm-1 that confirming the bonding of dye with fiber. Basically, triterpenoid reacts with glucose, which had mentioned earlier that improved dye fastness. Due to this region the rubbing fastness of reactivemahogany samples were provided outstanding performance.
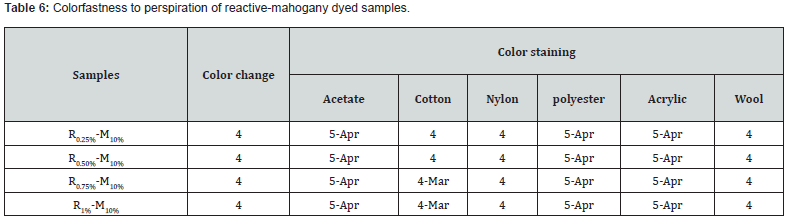
Color fastness to perspiration
Apparels that come into contact with the body and are heavily perspired may suffer from extensive local discoloration. The purpose of this test was measured the color resilience of the dyed samples to effect of alkali perspiration. Color stain and color change were done employing individual grey scales (Table 6).
The grey scale rating of perspiration fastness was showed 4 for sample R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10%. Hither, 4 were indicated good as opposed to 4/5 was indicated absolutely good to excellent level also. From FTIR data, the wave number for the most intense hue reactive-mahogany was approximately around 1200cm-1 which confirming the bonding of dye with fiber [Basically triterpenoid reacts with glucose that had mentioned earlier] was the main factor to improve the dye fastness. That’s why colorfastness to perspiration properties was good to excellent according to the (Table 6).
Uniformity Assessment
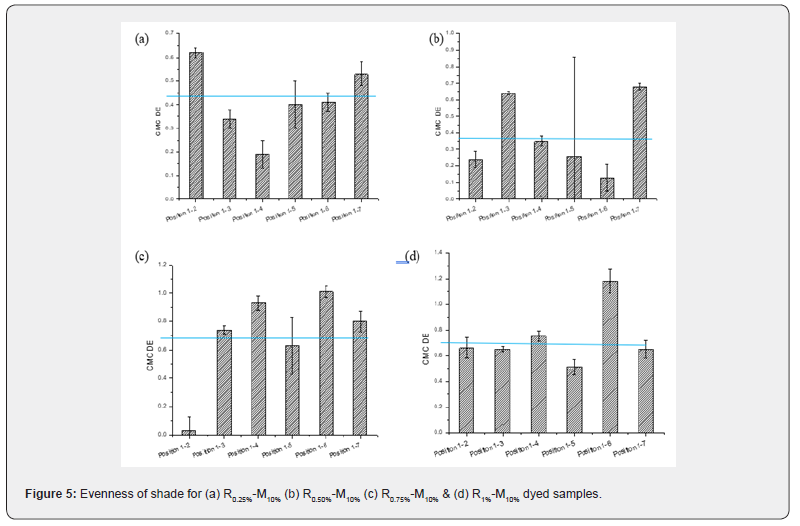
Comparison from visual assessment is not sufficient to observe accurate color difference in dyed samples. Also, evenness or uniformity of shade is a crucial criterion of natural dyed material for their performance. The dye sources are natural and the content of dye molecule per unit amount of the plant part is not confirmed to be evenly proportioned. Hence, forth CMC DE values were calculated to determine the color difference between each dyed samples at different positions to justify their evenness. Commercially, acceptable CMC DE values range up to 1. The color difference (CMC DE values) of reactive-mahogany dyed samples [R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10% respectively] have been investigated within the samples at random check basis for the observation of color evenness or uniformity of the dyed samples.
(Figure 5) demonstrates the comparative CMC DE values where some random check points were selected for investigating the color differences of reactive-mahogany dyed samples [R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10% respectively]. All the dyed samples exhibited CMC DE values within compatible ranges that affirm the evenness of shade. It was previously mentioned that there are huge triterpenoids of luminoid class in mahogany leaf extract that is able to form glycoside linkage with glucose of cotton cellulose. For even shade it is important that sufficient functional groups should be there also in cotton fiber. But conventionally, basically for reactive dye, alkali medium is formed by the active hydroxyl ion provided by sodium carbonate. Most probably within the huge alcoholic content, there might be some active hydroxyl group that could initiate the formation of cellulosate ion, and then uniform distribution of fiber-dye bonding is thought to be happened.
TGA analysis
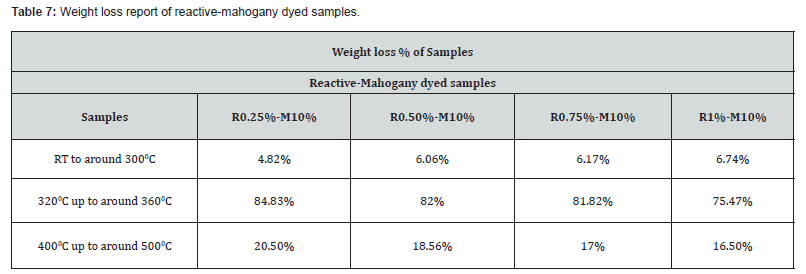
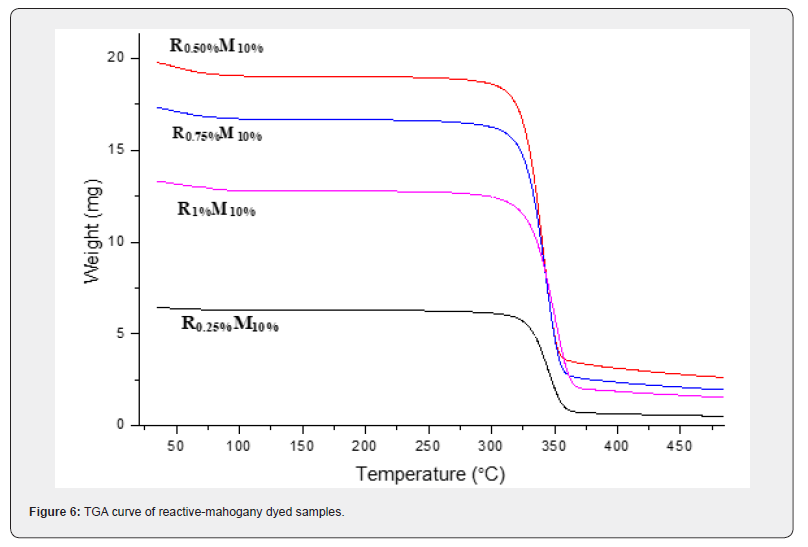
TGA is broadly required to explore the thermal decomposition of polymer or materials to decide the thermal decomposition kinetic parameters. These parameters can be needed to get a better understanding of thermal stability of polymer or materials too. TGA enables to that obtained by ordinary process. In order to consider the evaluation of thermal analysis data were permitted to record the diminishment in weight percentage versus increment in temperature plot for reactive-mahogany dyed samples were respectively R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10%.
As evident from (Figure 6), the onset temperature (of degradation) was found to improve within the case of sample R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10% of reactivemahogany dye. This can be due to the protective effect of different shade percentage of reactive dyes with mahogany 10% shade. (Table 7) The weight loss within the to begin with step compares to the evaporation of bound water. This procedure was made a wide endothermic peak and ends at around 300oC. Therefore, a preliminary weight coming about from moisture departing was showed up beginning from room temperature up to around 300oC. The weight loss% of all dyed samples were mostly similar up to around 300oC, in any case the weight loss% at 350oC were exceptionally diverse. The weight loss of sample R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10% were respectively 4.82%, 6.06%, 6.17% and 6.74%. Reactive-mahogany samples (from room temperature to around 300oC) were loss their weight gradually by the rising of temperature and reactive dye% was also increased where mahogany dye was stationary.
The main cracking stage for all dyed samples were approximately at temperature 320oC up to around 360oC with a weight loss rate were rapidly increased by increasing temperature. For reactive-mahogany dyed samples, weight loss% were comparatively 84.83%>82%>81.82%>75.47% which were decreased by the rising of reactive dye% where mahogany dye was constant. It was evident that the difference between the weight losses% at this organized compared to reactive-mahogany shades. The rest cracking stage for the samples (reactivemahogany) R0.25%-M10%, R0.50%-M10%, R0.75%-M10% and R1%-M10% were approximately at temperature 400oC up to around 500oC with the nearly same weight loss%. The value of the overall weight loss at the last of the thermal degradation of the dyed samples were under evaluated, appeared that the overall weight loss of the reactive-mahogany dyed samples was optimistic.
Conclusion
A Nobel approach in textile coloration was dyed cotton knit fabric with reactive and natural dyes extract from mahogany leaf/ leaves simultaneously by single bath process. Mahogany dye had shown similar bonding nature as like as reactive-cellulose bonding and to improve modification color or incorporate multicolor effect. During the fixed amount application of mahogany leaf extract dye, the k/s values of the samples are getting higher because of the gradual increment of individual reactive red color. So, the mahogany and reactive dye application at a simultaneous method confirmed their coloration on the fabric proportionally on the basis of application concentration or depth of color. The CIE L* a* b* detailed, reactive dyeing and mahogany dyeing occurred at an independent mode on cotton, where the auxiliaries of reactive dye worked for the reactive dyeing and at the same time mahogany dye did not create any obstacle for reactive dyeing. So, mahogany dyeing samples were represented darker reddish yellow with enhancing of mahogany dye percentage individually.
Thus, the compatibility of the dyes is ensured. From FTIR data, the characteristic peak for reactive-mahogany was approximately around 1190-1210cm-1, which represents confirming the bonding of dye with fiber. The peak seemed to attain a collective shape because of the simultaneous dyeing with reactive and mahogany dye. Finally, an optimistic shade was observed after the research work and fastness properties e.g. wash fastness rating 4 to 5, rubbing fastness was 4 and 5 (under wet and dry condition), perspiration fastness was shown as 4. Evenness of shade along with the K/S values exhibited the as usual value like the conventional results. In addition, thermal degradation of dyed samples, the overall weight loss of the reactive-mahogany dyed samples was optimistic. So, extraction and refinement of natural dyes and their application could be of incredible centrality in the future of the commercial and domestic textile industries as well.
Acknowledgements
This research and work have been carried out the lab of Dhaka University of Engineering & Technology. It is a pleasant aspect that now the scope has come to offer gratitude for all of them. Special credit goes to the person, Md. Abdul Hannan, Associate Professor of Textile Engineering Department; Dhaka University of Engineering & Technology (DUET), Gazipur, Bangladesh and thanks to total textile department also.
<-- dropdown Start here
References
- El-Nagar, SH Sanad, AS Mohamed, A Ramadan (2005) Mechanical properties & stability to light exposure for dyed Egyptian cotton fabrics with Natural & Synthetic dyes. Polymer Plastics Technology &Engineering 44(7): 1269-1271.
- Tutak, NE Korkmaz (2012) Environmentally Friendly Natural Dyeing of Organic Cotton. Journal of Ntural Fibers 9(1): 51-59.
- M Kumaresan, PN Palanisamy, PE Kumar (2011) Application of ecofriendly natural dye on silk using combination of mordants. International Journal of Chemistry Research 2(1): 11-14.
- N Punrattanasin, M Nakpathom, B Somboon, N Narumol, NR Kitkrai et al. (2013) Silk fabric dyeing with natural dye from mangrove bark (Rhizophoraapiculata Blume) extract. Industrial Crops and Products 49: 122-129.
- PS Vankar, R Shanker, D Mahanta, SC Tiwari (2008) Ecofriendly sonicator dyeing of cotton with Rubiacordifolia Linn. using biomordant. Dyes and Pigments 76(1): 207-212.
- B Sanda, L Indrie (2021) Natural Dye Extraction and Dyeing of Different Fibers: A Review. Innovative and Emerging Technologies for Textile Dyeing and Finishing Pp: 113-135.
- SS Affat (2021) Classifications, Advantages, Disadvantages, Toxicity Effects of Natural and Synthetic Dyes: A review. University of Thi-QarJournal of Science 8(1): 130-135.
- MAS Barrozo, KG Santos, FG Cunha (2013) Mechanical extraction of natural dye extract from Bixaorellana seeds in spouted bed. Industrial Crops and Products 45: 279-282.
- DA Peggie, AN Hulme, H McNab, A Quya (2008) Towards the identification of characteristic minor components from textiles dyed with weld (Reseda luteola L.) and those dyed with Mexican cochineal (Dactylopius coccus Costa). MicrochimicaActa 162(3): 371-380.
- E Mikropoulou, E Tsatsaroni, EA Varella (2009) Revival of traditional European dyeing techniques yellow and red colorants. Journal of cultural heritage 10(4): 447-457.
- HI Patil, VA Dorugade, VS Shivankar (2016) Single & double bath dyeing of Polyester/Cotton blended fabric using disperse & reactive dye. International Journal on Textile Engineering& Process 2(3): 29-33.
- CR Meena, A Nathany, RV Adivarekar, N Sekar (2013) One bath dyeing process for Polyester/Cotton blend using physical mixtures of Disperse/Reactive dyes. European International Journal of Science &Technology. 2(2): 6-16.
- F Sarkar, RK Prasad, MR Howlader, N Abir, Nurnnabi, R Aktar (2015) Scope of dyeing Polyester Cotton (PC) blended fabric in single bath process for water, energy & time saving. ISOR Journal of Polymer &Textile Engineering 2(3): 12-16.
- AK Samanta, A Konar (2011) Dyeing of textiles with natural dyes. Institute of Jute Technology 3(1): 29-30.
- MAR Bhuiyan, A Ali, A Islam, MA Hannan, SMF Kabir, et al. (2018) Coloration of polyester fiber with natural dye henna (LawsoniainermisL.) without using mordant: a new approach towards a cleaner production. Fashion and Textiles 5(2).
- MF Shahin, RM Ahmed (2014) Optimizing the dyeing process of alkali treated polyester fabric with dolu natural dye. International journal of engineering research & application 4(6): 35-40.
- A Davulcu, H Benli, Y Sen, MI Bahtiyari (2014) Dyeing of cotton with thyme & pomegranate peel. Cellulose 21 4671-4680.
- RP Rastogi, BN Mehrotra, S Sinha, P Pant, R Seth (1993) Compendium of Indian medicinal plants. Central drug research institute Vol 2.
- SAM Abdelgaleil, M Doe, M Nakatani (2013) Rings B, D-secolimonoidantifeedants from Swieteniamahogani. Phytochemistry 96: 312-317.
- SAM Abdelgaleil, M Doe, Y Morimoto, M Nakatani (2006) Rings B, D- secolimonoids from the leaves of Swieteniamahogani. Phytochemistry 67(5): 452-458.
- SZ Moghadamtousi, BH Goh, CK Chan, T Shabab, HA Kadir (2013) Biological activities and Phytochemicals of Swieteniamacrophylla King. Molecules 18(9): 10465-10483.
- PK Mukherjee (2019) Quality Control and Evaluation of Herbal Drugs. Evaluating Natural Products and Traditional Medicine 237-328.
- MAA Mamun, MM Hossain, MA Khan (2020) Dyeing of polyester fabric with natural colorants extracted from mahogany (Swieteniamahagoni) seed pods. Journal of Engineering Science 11(1): 37-42.
- AA Mamun, MM Bashar, S Khan, MN Roy, MM Hossain, et al. (2021) Mordant free dyeing of nylon fabric with Mahogany (Swieteniamahagoni) seed pods: A cleaner approach of synthetic fabric coloration. Textile Research Journal 92: 17-18.
- MA Haque, GMA Khans, SAA Razzque, K Khatun, AK Chakraborty et al. (2013) Extraction of rubiadin dye from Swieteniamahagoni and its dyeing characteristics onto silk fabric using metallic mordants. Indian Journal of Fibre and Textile Research 38: 280-284.
- A Hossain, AKMS Islam, AK Samanta (2018) Pollution Free Dyeing on cotton fabric Extracted from Swieteniamacrophylla and Musa Acuminate as Unpolluted Dyes and Citrus. Limon (L) as Unpolluted Mordanting Agent. Trendsin Textile Engineering & Fashion Technology 3(2): 286-293.
- S Adeel, S Ali, I A Bhatti, F Zsila (2009) Dyeing of Cotton Fabric using Pomegranate (Punicagranatum) Aqueous Extract. Asian Journal of Chemistry 21(5): 3493-3499.
- X Xu, J Gong, Z Li, Q Li, J Zhang, et al. (2020) Mordant free Dyeing and Functionalization of Wool fabrics with Bio Colorants Derived from Apocynumvenetum L.Bast. ACS Sustainable Chemical Engineering 8(33): 12686-12695.
- N Kusumawati, A Kistyanto, Samik (2019) Exploration of natural dyes by using a combination of Caesalpiniasappan and Leucaenaleucocephala L. leaves. Atlantis highlights in chemistry and pharmaceutical sciences 1: 33-37.
- N Kusumawati, AB Santosa, A Wijiastuti (2018) Development of textile natural dyeing using hybrid dyes from mango leaves turmeric. Advances in engineering research 171: 50-55.
- Y Zhou, J Zhang, R Tang, J Zhang (2015) Simultaneous dyeing and functionalization of silk with three natural yellow dyes. Industrial crops and products 64: 224-232.
- MI Kiron (2013) Physical, Chemical and Morphological Structure of Cotton Fiber. Textile learner.
- MI Kiron (2014) Chemical Composition of Cotton Fiber. Textile learner.
- A Roy, S Saraf (2006) Limonoids: Overview of significant Bioactive Triterpenes distributed in plants kingdom. Biological & Pharmaceutical Bulletin 29(2): 191-201.
- SZ Moghadamtousi, BH Goh, CK Chan, T Shabab, HA Kadir (2013) Biological activities and Phytochemicals of Swieteniamacrophylla King. Molecules 18(9): 10465-10483.
- J Shore (1995) Cellulosics Dyeing. Society of Dryers and Colourists Pp: 180.






























