Dyeing of Cotton with Natural Colorants Extracted from Red Rose Flower
Mansoor Iqbal1, Saima Imad1, Razia Sultana1, Javaid Mughal M2, Aijaz Phanwar3 and Kamran Ahmed1
1Applied chemistry Research Centre, PCSIR Laboratories Complex, Off University Road, Pakistan
2Baddar Textile Processing Industries SITE, Pakistan
3Pakistan Standards & Quality Control Authority, Pakistan
Submission: June 13, 2023; Published: July 18, 2023
*Corresponding author: Kamran Ahmed, Applied chemistry Research Centre, PCSIR Laboratories Complex, Off University Road, Pakistan, Email: kamranfarooq20@hotmail.com
How to cite this article: Mansoor I, Saima I, Razia S, Javaid Mughal M, Aijaz P. Dyeing of Cotton with Natural Colorants Extracted from Red Rose Flower. Curr Trends Fashion Technol Textile Eng. 2023; 8(4): 555741. DOI: 10.19080/CTFTTE.2023.08.555741
Abstract
Cotton substrate was dyed with an extract of red rose petals. The dyeing was carried out with and without metallic mordants (Alum, Copper sulphate, Nickel sulphate, Ferric chloride, and Ferrous sulphate), using two different dyeing systems: pre-mordanting system, simultaneous mordanting system. The colour of the cotton substrate was measured by Data color in terms of K/S, and CIELAB values. In both mordanting system ferrous sulphate produced dark shade, the K/S value for simultaneous system is 16.76 and for pre-mordent K/S values is 12.05. It is also observed that the higher %fix is of simultaneous system for ferrous sulphate is 76.80% and for pre-moderating the % fix is 71.50%. But statistical analysis of both techniques tells us the reproducibility of copper sulphate is better to other metallic salts the S.D of K/S values for copper sulphate in simultaneous method is 0.2112 and for pre-mordent method the S.D is 0.1132.
Keywords: Natural colorants, Rose flower, Simultaneous mordanting system, Metallic salts
Introduction
Natural dyes, dyestuff and dyeing are as old as textile themselves. Man has always been interested in colours, the art of dyeing has a long past and many of the dyes go back into prehistory. It was practiced during the Bronze Age in Europe. The earliest written record of the use of natural dyes was found in china dated 2600 BC [1]. Upto the end of the 19th century natural dyes were the main colourants for textiles. The introduction of synthetic dyes led to an almost complete replacement of natural dyes, due to the favourable application properties of synthetic dyes. Besides a wide range of available colours, high reproducibility and improved quality of dyeing could be achieved at lower specific cost. The future demand for more sustainable processes can be seen as a driving force for new strategies, which could bring a revival of natural dyes in textile dyeing. However, the cost of such dyes has to be lowered considerably and the quality level of the dyeing needs substantial improvement [2,3].
Natural dyes contain natural coloring matter, which is neither carcinogenic nor hazardous to the environment, the colors are soothing to eyes, earthly, warm, and highly appealing. The dyes being natural are earth friendly and help maintain and ecological balance. They are non- allergic, non-toxic to the human body and perpetuate an ancient tradition [4].
Many natural dyestuff and stains were obtained mainly from plants and dominated as sources of natural dyes, producing different colours like red, yellow, blue, black, brown and a combination of these. Almost all parts of the plants like root, bark, leaf, fruit, wood, seed, flower, etc. produce dyes. It is interesting to note that over 2000 pigments are synthesized by various parts of plants, of which only about 150 have been commercially exploited [5,6]. Many plants used for dye extraction are classified as medicinal, and some of these have recently been shown to possess antimicrobial activity. There is clear evidence that some natural dyes by themselves have medicinal properties [7].
Despite several advantages of natural dyes over synthetics, the use of the former is still very limited due to the non-availability of standard shade cards and standard application procedures. Most of the natural dyes have no substantivity for the fibre and are required to used in conjunction with mordants. A mordant, usually a metallic salt, is regarded as a chemical, which is the dyestuff. A link is formed between the substrate and the dye, which allows certain dyes with no or little affinity for the substrate to be fixed [8]. This study aims to address the problems associated with natural dyes. Such as the nature of mordants, methods of moderating system and to investigate the CIE Lab values and K/S of the dyed samples and their fastness properties.
Materials
Flower Colour chosen
We have tried to take some advantage from the petals of red roses as you know in the wedding ceremonies there is a culture of using abundant number of roses in Pakistan. Approximately in a single wedding 5-10 kg of roses are usually used. But after the ceremony is over, these flowers are spread on the floor as garbage. So, to take advantage of that flower we did cotton dyeing by using extract of petals.
Studies on cotton substrate
Scoured and bleached, optical brightener free 100% woven cotton fabric (150g/m2, 1/1 plain weave) was used for natural dyeing purposes.
Chemicals and auxiliaries used
Metallic salts such as Alum, Copper sulphate, Ferric chloride, Nickel sulphate, Ferric sulphate were used analytical grade, purchased from Meark chemicals Pakistan, and soaping agent Sandopan DTC were used of commercial grades.
Equipment
Dyeing was carried out on IR dyeing machine of AHIBA. Colour matching system of Data colour SF 650X was used for the evaluation of colorimetric data. Light fastness was determined on ATLAS Ci 3000+ xenon weather-ometer. Samples were cured on curing machine of Rapid. Fastness rating was evaluated on colour matching cabinet by comparing with grey- scale. Rubbing fastness was determined on ATLAS crock meter.
Procedure
Extraction method: 200gm of dry red rose flowers were crushed and dispersed in 75% water and 25% ethanol in 1liter and heated to 100oC in a round bottom flask kept over water bath for quick extraction for 80mins. All the colours were extracted from flowers by the end of 1 hr. After extraction the extract was filtered through ordinary filter papers, the filtrate was collected, and the solvent was evaporated on rotatory evaporator and recovered to dryness (5.86gm). 100ml of distilled water was added to this extrated amount. The absorbance was recovered for determination of concentration of aqueous extract. The UVVisible spectrum of the extract showed higher color yield. This was further used for dyeing the fabric.
Preparation of cotton substrate: Cotton fabric was washed with a solution containing 0.5g/l of sodium carbonate and 2g/l of non-ionic detergent (Sandopan DTC) at 60oC for 30 min, keeping the material to ratio at 1:20. Then washed with distilled water twice then dry the bath substrate for dyeing purpose.
Dyeing without a mordant (control Sample)
The rose extract dyeing was carried out on an exhaust dyeing process. The cotton substrates were dyed at a liquor ratio of 1:20, with the dyebath containing 1g/l soda ash. The temperature was raised to 80oC over 30min and maintained at this level for 1 Hr. The dyed fabric was then rinsed with hot water temperature 60 oC, followed by soaping with non-ionic soaping agent 2g/l sandopan DTC. Finally, the fabric samples were washed thoroughly with cold water, squeezed and dried, and then finally cured at 155 oC for 60 sec on lab curing machine Rapid.
Dyeing with mordants
The two different methods of dyeing employed were premordanting and simultaneous mordanting system. Mordant concentration is 2 and 5g/l were used. In the pre-mordanting method, the fabric was first immersed in aqueous solution of alum, copper sulphate, Nickel sulphate, ferric chloride, and ferrous sulphate. The mordant was dissolved in distill water to make a liquor ratio is 40:1. The wetted sample was entered into the mordant solution and then brought to heating. The temperature of the dyebath was raised to 60 oC over 1h. The mordanted material was then rinsed with water thoroughly, sequeezed and dried. Mordanted cotton substrate needed to be used immediately for dyeing because some mordants are very sensitive to light [9]. All of the mordanted fabric was then dyed by the above method.
In the simultaneous mordanting system the five different mordants were also applied during the dyeing process outlined above.
Fastness testing
The washing fastness of the samples dyed under the optimized conditions was tested according to the ISO 105-CO3 method. The samples were washed in a standard soap solution at 60 oC for 30 min keeping the M:L ratio as 1:50. Dry and wet rubbing fastness of the dyeing was testing according to the ISO 105-X12 methods. Light fastness was tested according to the ISO 105-BO2 methods. The dyeing was exposed to xenon arc lamp for 24 hours at standard testing conditions [10].
Measurement of colour strength
The fixation of the dye in percentage was calculated first by determining the reflectance R of the dyed samples at the wavelength of minimum reflectance (maximum absorbance) on Data colour SF 650X spectrophotometer. The colour yield (K/S) value was calculated by using the Kubelka-Munk equation (eq-1) and the dye fixation % was evaluated using equation 2.
K/S = (1-R2)/ 2R ----------------(1)
% Dye fixation = K/S values of sample after soaping × 100 …………… (2)
K/S values of sample before soaping
Statistical Analysis
K/S, L, a, b, c, h, values of each dyeing condition were evaluated by analysis of variance and each treatment was replicated five times. The standard errors of the difference SED were calculated for each mordant and bath process.
Results and Discussions
Optimization of extraction conditions
The relative colour strength in terms of absorbance of the dye extracts obtained under different extraction conditions is given in Figure 1. As can be seen from figure 1, colour strength of dye extracts obtained at room temperature was minimum, slightly getting better at 60 oC and maximum when the extractions were carried out at boiling temperature.
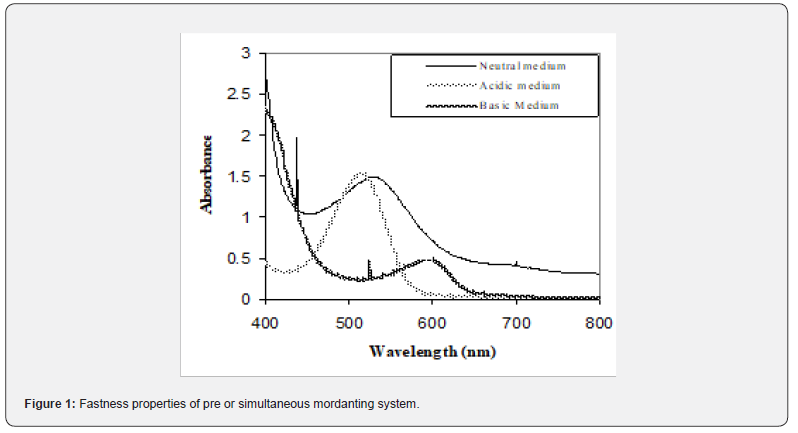
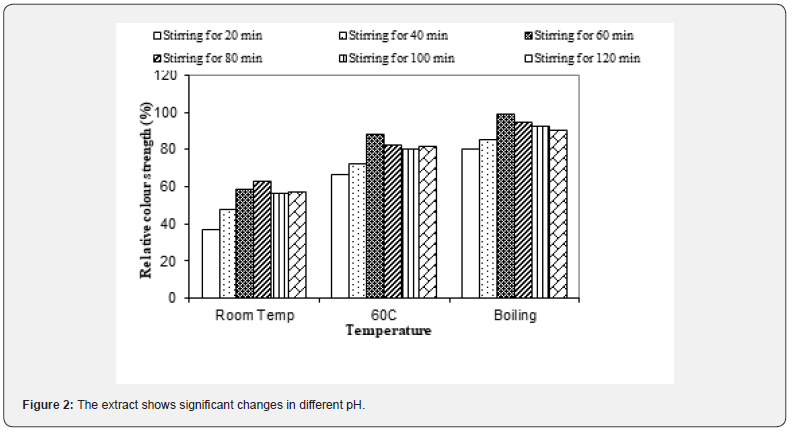
For extractions at room temperature, 60 oC and boiling temperature, the effect of stirring was sporadic. The maximum stirring time is 60min. After this time the colour strength was decreased, this is because after such a long stirring time, some impurities were also extracted along with the colouring components, thus decreasing the overall colour strength of the dye extract [11,12]. The aqueous extract showed one major Peak, λmax at nm in the visible region. The visible spectrum of extract of fresh flowers is shown in figure. The extract shows significant changes in different pH as shown in Figure 2. At pH 5, 7, 9, the λmax was also changed in case of at pH 9, it also indicates the colour of the extract is in greener region on the spectrum, and indicates the dyeing is favorable at alkaline condition and the resultant shade is green or like green.
Effect of Mordants on K/S values
Metallic mordants play an important role in the natural dyeing of textile substrate [13]. The colour intensity in terms of K/S values calculated from the colorimetric data for the dye applied on cotton substrate, the sample dyed without mordant i.e., controlled sample shows lighter shades as K/S value is only 2.41. The K/S values are increased while dyeing with different mordants as well as different method of mordent applying system. In fact, most of the natural dye extract have poor affinity for cotton fibers, their fastness is often enhanced by metal mordants, which form an insoluble complex with dye molecules [14].
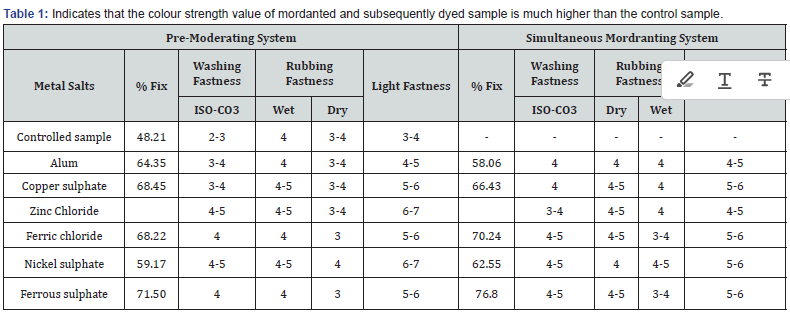
Table 1 indicates that the colour strength value of mordanted and subsequently dyed sample is much higher than the control sample. On applying mordant, the increasing value of K/S shows the increasing dye affinity towards cotton fibre. The mean values of K/S are higher as sequence in simultaneous mordant is Ferrous sulphate > Ferric chloride > Copper sulphate > Nickel sulphate > Alum which are 16.76,14.388, 6.746, 5.018 and 3.154 respectively. While in case of pre-mordent system the sequence is Ferrous sulphate > Ferric chloride > Copper sulphate > Alum > Nickel sulphate which are 12.058, 8.704, 7.822, 5.448 and 3.484 respectively. The increasing of K/S value from 2.7 of controlled sample to maximum K/S 16.76 of Ferrous sulphate, can be attributed that metal mordants, as a polyvalent metal ion forms coordination complexes with certain dyes. During dyeing two types of bonds are involved in the fundamental reaction between a mordants dye and a mordent. One is a covalent bond with usually hydroxyl oxygen and the metal atom, while the other is a coordinate bond with the metal with the double bonded other oxygen also referred as chelation. By comparing the two mordanting techniques, the simultaneous mordanting system performs better visualized in terms of higher color strength values. It may be explained that in simultaneous mordanting system the metal ion has equal opportunity to make a chelate compound between dye and cotton fiber, while in pre-mordant system the mordant first makes the chelate compound with the dye molecule and may have limited option to fix on the cotton substrate.
Fastness properties of dyed substrate
The washing and light fastness of the dyed sample are shown in (Table 1). The dyed samples without mordanting i.e., controlled sample showed poor fastness properties as compared to mordanting system. The washing fastness rating of the controlled sample is 2-3. The addition of mordant, the washing fastness improves good with a rating of 4-5, 4 for simultaneous mordanting system and pre-mordants respectively. In case of light fastness, the controlled sample has a 3-4 rating. When pre-mordanting system applied the light fastness is significantly increased, the rating is 6-7, but in simultaneous mordant, the rating is 4-5. In natural dyeing the moderating agent is directly influenced by their dyeing properties as well as fastness properties. The higher light fastness properties of reactive dyes can be attributed to the strong intermolecular H-bonding which exists in the form of six membered rings. This enhances the stability of the compound by a reduction in electron density at the chromophore. As a result, sensitivity of dye towards photochemical oxidation becomes reduced. But such strong interaction is not present in the rose extract. In fact, fastness properties can also be evaluated in terms of fixation. The controlled sample has very poor fastness as it has very minimum fixation i.e., 48.21% only. While on applying mordant, the fixation increases which also increases the fastness properties. A slight difference in fixation can be observed in (Table 1) between simultaneous mordanting system and premordent system. Iron metal has maximum fixation in both mordant systems. The order of fixation in pre-mordanting system is Ferrous sulphate > Copper sulphate > Ferric chloride > Alum > Nickle sulphate which are 71.50, 68.45, 68.22, 64.35 and 59.17 respectively. In case of simultaneous mordant system, the order of fixation is Ferrous sulphate > Ferric chloride > Copper sulphate > Nickle sulphate > Alum which are 76.80, 70.24, 66.43, 62.55 and 58.06 respectively.
Colour Values
Effect of pre-mordanting and simultaneous mordanting system on L*, a*,b*, c* and h with different metallic mordants at optimized conditions is given in (Table 1) a ,b, c. L* represents lightness values, the higher the lightness values represents lower the colour yield, a* and b* represent the tone of the colour, positive values of a* and b* represents redder and yellower tones while negative values shows greener and bluer tones. C* represents chroma or purity of colour, h represents hue, shade of colour [15]. Thus tones, hue and chroma of different experiment interpreted from (Table-1). The L values of dyed sample indicated that the extracted dyes were producing only light shades on cotton substrate. The addition of mordants increased the darkness of the colour by changing the nature of mordants. The L values of pre-modern and simultaneous mordanting system, both methods show the ferrous sulphate is produced darker shade the L values of simultaneous system for ferrous sulphate is 33.726, but in premordent system the copper sulphate produced darker shade the L value is 49.936.
The a* and b* values indicate the tone of the shade. Alum and nickel sulphate produce lemon yellow shade in both dyeing systems but in case of copper sulphate they produced greener tone. In the case of ferrous sulphate in simultaneous mordanting system the b* value is -14.4, they show bluer tone the overall shade is grayish violet, in pre-mordent system they produced less grayish violet having b* value is -6.68. The values of all dyeing experiments show redder tone.
Conclusion
Extraction and dyeing conditions of a natural dye from red rose petal were optimized. The dye obtained displays good saturation on cotton substrate. They also have the potential to replace some of the toxic, sensitizing, and carcinogenic dye and intermediates. Rose leaf as a natural dye has greater potential because it is grown widely for its commercial and medicinal importance. These do not cause damage to the environment during their extraction and may be used satisfactorily with mordants. These dyes also improve the depth of shade. The light, washing and rubbing fastness properties of the fabric dyed with rose petal can be improved by different mordants systems. The fastness results indicated that the rose petal dye on cotton substrate is sufficiently good. The above results have strongly indicated that the fabric dyed with rose petal dye can find a great value in textile coloration and export market.
References
- Moiz A, Aleem Ahmed M, Kausar N, Ahmed K, sohail M (2010) study the effect of metal ion on wool fabric dyeing with tea as natural dye. Journal of Saudi chemical society 14(1): 69-76.
- Bechtold T, Mussak R, Mahmud-Ali A, Ganglberger E, Geissler S (2006) Extraction of natural dyes for textile dyeing from coloured plants wastes releases from the food and beverages industry. Journal of the science of food and Agriculture 86(2): 233-242.
- Mahatma D, Tamaria SC (2005) Natural dye yielding plants and indigenous knowledge on dye preparation in acronyehal Pradesh. northeast India 88(9): 1474-1480.
- Chandramouli KV (1995) Sources of natural dyes in India-A compendium with regional names. PPST foundation, Chennai pp. 116.
- The wealth of India-A dictionary of Indian raw materials, and industrial products, Council of Scientific research and industrial research, national institute of science communication and information resources, New Delhi, 2003.
- Singh R, Jain A, Panwar S, Gupta D, Khare Sk (2005) Antimicrobial activity of natural dyes. Dyes and Pigment 66(2): 99-102.
- Gulrajani ML, Gupta D (1992) Natural dyes and their application to textiles. Indian institute of technology, India.
- Shanker R, Vanker PS (2007) Dyeing cotton, wool, and silk with Hibiscus mutabilis (Gulzuba). Dyes & Pigment 74(2): 464-469.
- Anonymous, Methods of test for colour fastness of textiles and leathers, the society of Dyers and Colourists. UK, 1990.
- Nagia FA, El-Mohamedy RSR (2007) Dyeing of wool with natural anthraquinon dyes from fusarium oxysporum. Dyes & Pigments 75(3): 550-555.
- Boling Y, Qunling W, Leilan Y (2005) Mordant dyeing with rutin and troxerution. International dyer 190(6): 23-27.
- Deo HT, Desai BK (1999) Dyeing of cotton and jute with tea as a natural dye. JSDC 115(7-8): 224-227.
- Jothi D (2008) Extraction of natural dyes from African marigold flower (Tagetes exected) for textile coloration. AUTEX Research Journal 8(2): 49-53.
- Iqbal M, Aleem A, Mughal J, Siddiqui MQ (2006) Light fastness of Bi-functional reactive dyes with pad-Batch and pad-Dry Cure methods on cellulosic substrate. Jour Chem Soc Pak 29(3): 195-197.






























