Application of Bauhinia Vahlii Bark Extract on Wool Fiber
Goutam Bar1* and Debojyoti Ganguly2
1Department of Textile Design, National Institute of Fashion Technology, Bhubaneswar, India
2Department of Textile Design, National Institute of Fashion Technology, Bhopal, India
Submission: November 09, 2022; Published: December 08, 2022
*Corresponding author: Goutam Bar, Department of Textile Design, National Institute of Fashion Technology, Bhubaneswar, India
How to cite this article: Goutam B, Debojyoti G. Application of Bauhinia Vahlii Bark Extract on Wool Fiber. Curr Trends Fashion Technol Textile Eng. 2022; 7(4): 555720. DOI: 10.19080/CTFTTE.2022.07.555720
Abstract
The increasing concern about synthetic dyes’ health hazards and environmental impact has sparked consumers’ interest in natural dyes. Natural dyes are sustainable, renewable, and biodegradable and do not contain toxic components commonly found in synthetic dyes. The extraction of colorants from natural substrates and their application on textiles are becoming popular and reinvestigated. Recently, several investigations have been carried out on the extraction of new natural dyes to apply to textile materials. In most cases, a metal salt-based mordant and synthetic axillaries are used to fix the dyes on textile materials. Applications of these metal salt base mordant and synthetic axillaries retards the sustainability of textile processing.
Considering the above, a bio-color was extracted from Bauhinia Vahlii (BV) bark and applied on wool fibers without using metal salt-based mordant and synthetic axillaries. The concentrated extract was applied on the superwash wool fibers with different extract concentrations (67% and 33% extract concentration) at varied pH [7, 6, 5.5, 5, 4.5, 4] at 90 °C temperature for 60 minutes with 1: 50 as material to liquor ratio. The pH of the dye bath was maintained using bio acids (citric acid) extracted from a lemon.
Introduction
The textile industry is the major consumer of synthetic dyes used for dyeing and printing textile materials [1]. Synthetic dyes are hazardous, carcinogenic to humans, destructive to ecosystems, and toxic to aquatic biodiversity [2]. Due to environmental awareness, the extraction of coloring matter from natural substrates and their application to textiles has recently received significant attention. Several studies have been conducted on the extraction of new kind of natural dyes and their application to textile materials. The demand for natural dyes is constantly increasing as the extraction of natural dyes and their use in textile materials are easy and do not require strong acids. In addition, natural dyes are biodegradable, non-toxic, non-carcinogenic, renewable, and eco-friendly and are readily available. They offer a wide range of shades with good color fastness properties [3]. Now, it is the need of the hour to use natural dyes instead of synthetic dyes for textile applications. Natural dyes can be extracted from various parts of plants, such as roots, bark, leaves, fruits, flowers, and other natural sources. Natural colorants are not only used for their medicinal and antioxidant properties but also to enhance the aesthetic value of textiles [4,5].
Wool and silk are natural protein fibers and have a general chemical structure of NH2-CHR-COOH. Amino (-NH2) and carboxyl (-COOH) groups present in the protein fiber take part in the dyeing process [6]. Acid and metal complex dyes have an affinity towards the protein fiber and amino group present in the protein fibers attached to the dye molecules by electrostatic forces and hydrogen bonds in acidic pH [7]. Acidic pH increases the amino group’s ionization, hence increasing the dye uptake. Natural dues also have a very good affinity towards wool and silk fibers and give a brighter range of shades [8]. Moreover, Uses of synthetic dyes for the dyeing of wool and silk materials retards the goal of sustainability.
To address some of the above issues, the current study aimed to dye wool fibers with eco-friendly natural dyes extracted from the bark of the Bauhinia Vahlii plant. Bauhinia Vahlii, as shown in Figure 1, is a giant climbing plant that can grow up to 10 to 30 m long and is one of the most abundant Indian Bauhinia species. This plant is distributed in the sub-Himalayan region, Assam, Central India, Bihar, Odisha, and the Eastern and Western Ghats of India. This plant is commonly known as c”, because of the characteristic shape of their leaves and is easily distinguished by the bifid (bilobed) leaves with showy flowers ranging from white, yellow to purple [3,9]. Bauhinia Vahlii is also known as Maloo Creeper or Kanchan Lata plant in India and is famous in Siali in Odisha. The woody stem can get as thick as 20 cm, and the plant bark is also rich source of fiber. This plant is a member of the Angiospermae class and the Leguminosaceae family [3,6,9]. Its leaves and stem bark have been used frequently in folk medicine as a therapeutic agent to treat diabetes, infections, pains, and inflammatory processes confirmed by numerous biological studies [6,7,9].

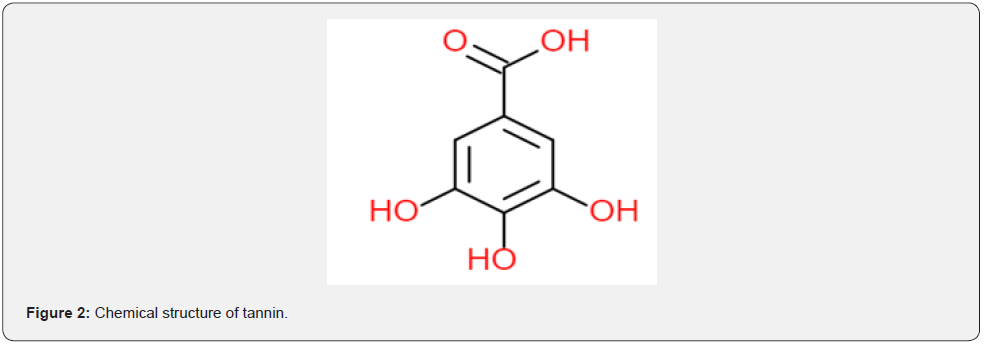
The bark of BV is composed of cellulose, hemicellulose, lignin, and pectin [10,11]. Bauhinia Vahlii bark extract contains various phytochemicals, such as alkaloids, carbohydrates, glycosides, saponins, flavonoids, triterpenes, anthraquinones, tannins, and phenolic compounds [3,11-13]. The presence of tannins and anthraquinones in BV bark extract indicates that it can be utilised as a coloring agent [3,4,13,14]. Tannin is the primary coloring matter found in aqueous BV bark extract, which contains a carboxylic group, as shown in Figure 2 (14,15). This coloring matter is a pHsensitive halocromic and polygenetic dyes. It changes its color with the change in pH. It has a high affinity towards silk and can dye silk fibers without a metal salt based mordanting agent, as tannin also acts as a self-mordanting agent. Dyeing silk fabric with mordants at different pH levels results in different color nuances [16,17]. In this current study, wool fibers were dyed with BV bark extract under different pH conditions, and colorimetric studies were performed.
Experimental
Materials
Superwash Wool fiber having 19.5 microns fineness is sourced from Jayashree Textile, India. Bauhinia Vahlii plant bark was supplied by District Rural Development Agency, Mayurbhanj, Odisha, India. The sodium hydroxide of Ramken was purchased from Bhubaneswar, India. Lemon juice was extracted from the fresh lemon collected from the local market of Bhubaneswar, Odisha, India.
Dye extraction from Bauhinia vahlii bark
The extraction process was performed under optimized [M:L ratio, temperature, time, and alkali concentration] conditions. Initially, BV bark is dried in the shed at ambient temperature and later finely chopped. The chopped fibrous material was soaked in distilled water at a material-to-liquor ratio of 1:40 for 30 minutes, followed by treatment with 20% sodium hydroxide solution for 2 hours at 95 °C in a Paramount water bath. The extract was collected by neutralization with citric acid (bio-acid).
Application of dyes
The superwash wool fiber samples were dyed with the BV plant bark extract by exhaust dyeing method. Extracted solutions were used directly, having a material-to-liquor ratio of 1:50 in two different concentrations with varied pH [7, 6, 5.5, 5, 4.5, 4]. In the method I, dye liquor was prepared by taking 67% aqueous BV bark extract and the rest water and lemon juice. In another way, 33% aqueous BV bark extract, the rest water, and lemon juice. The amount of lemon juice added is based on the acidic pH requirement. Dyeing was carried out in a Paramount water bath for 1 hour at 90 °C. Dyed samples were rinsed with cold and hot water to remove the superficial colorants.
Twelve samples were developed, varying the pH and aqueous BV bark extract concentration. Six different pH levels were taken for the dyeing, and they are 7, 6, 5.5, 5, 4.5, and 4. Two different concentration levels of aqueous BV bark extracted were also taken. In the first option, dye liquor was prepared by mixing 67% aqueous BV bark extract and 33% mixture of water and lemon juice. The lemon juice quantity was varied to achieve the desired pH level, and six samples were developed, named A to F, with different pH levels, i.e., 7, 6, 5.5, 5, 4.5, and 4. mentioned earlier. Similarly, in the second option, 33% aqueous BV bark extract was mixed with a 67% mixture of water and lemon juice, and dye liquor was obtained. Sample G to L was developed using dye liquor prepared in 2nd option with varied pH, as mentioned earlier.
Measurement
Color strength
The conditioned wool fiber samples were scanned with a Color i7 spectrophotometer of Gretag Macbeth, USA. Scanning was performed using SAV aperture (6 mm) with specular reflectance (10° observer, D65 illuminant). These samples’ colorimetric values (L*, a*, b*, C, hº, and K/S) were measured as per the AATCC testing method 173-1998. The color strength of the dyed wool fiber samples was expressed as the K/S value. K/S value was calculated by the reflectance value of the samples using the Kubelka Munk Equation [18].
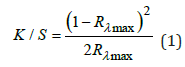
K, S, and Rλmax are the absorption, scattering coefficients, and reflectance value at the maximum absorbance wavelength (λmax).
Fastness properties
Wool fiber samples dyed with BV plant bark extracts were tested for their color fastness properties. The washing fastness of the samples were tested in a launder-o-meter following ISO 105 C06 test method at 50 °C for 45 min with 5 gram per liter nonionic detergent. Color change and the staining on the multifiber were accessed using AATCC grey scales. Colorfastness to light of the dyed samples was tested according to ISO 105 B02 test method, and the tested samples were assessed for change in color by AATCC blue wool scale.
Result and discussion
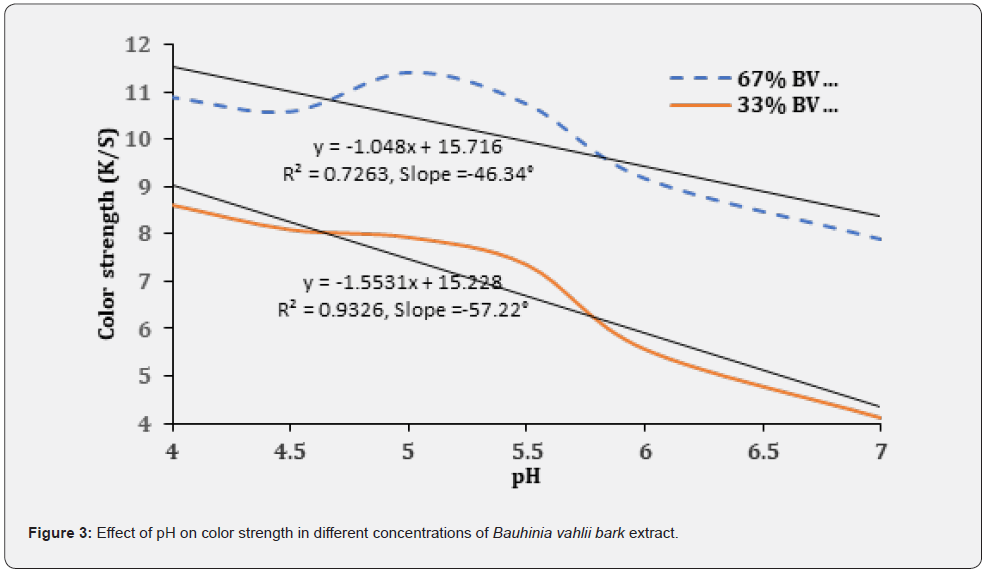
Colorimetric values [L*, a*, b*, c*, hº] and color strength (K/S) of wool fiber samples dyed with BV bark extract are tabulated in Table 1. The K/S value of all the dyed samples is significantly higher than that of raw wool fiber, which indicates wool fibers have absorbed a substantial amount of coloring matter. With the increase in shade depth, the reflectance percent value and the color strength (K/S) value increase. Single wavelength (SWL) color strength percentage value also depicts the same. K/S value and SWL color strength percentage confirm that sample D has a higher shade depth percentage than all dyed wool fiber samples. K/S value and SWL color strength percentage of sample D are 11.43 and 1574%, respectively, when dyed with 67% BV bark extract at pH 5. On the other hand, sample G has a lower shade depth percentage, having 4.12 and 567% K/S and SWL color strength percentage values, respectively. It is also clear from Table 1 and Figure 3 that the fiber absorbs less coloring matter at neutral pH (pH7) in both 67% and 33% BV extract concentration in dyeing and increases with a decrease in pH. The wool fiber absorbs tannin in BV bark extract due to the electrostatic interaction between the carboxylic group in the tannin and the amino group in the wool fiber. A low pH increases the degree of ionisation of the amino group in the wool fibers, resulting in improved dye uptake [8]. In the neutral pH zone, the ionic barrier retards the interaction between the carboxylic and amino groups, resulting in lower dye uptake. The slope of the color strength trend line when wool fibers dyed with 33% BV extract concentration is more than that of the 67% BV extract concentration, which indicates that the impact of pH on color strength is more in the former one. The higher color strength is obtained in a pH range of 4.5 to 5.5 in both options. Moreover, the R- squared value (R2) value of color strength graph when wool fiber dyed with 33% BV extract concentration is significantly higher than the other, which confirms that the color strength changes uniformly with the change in pH through pH range from 4 to 7. Images of all twelve dyed fiber samples are shown in Figure 4, and the color difference is also visible.
Colorfastness properties
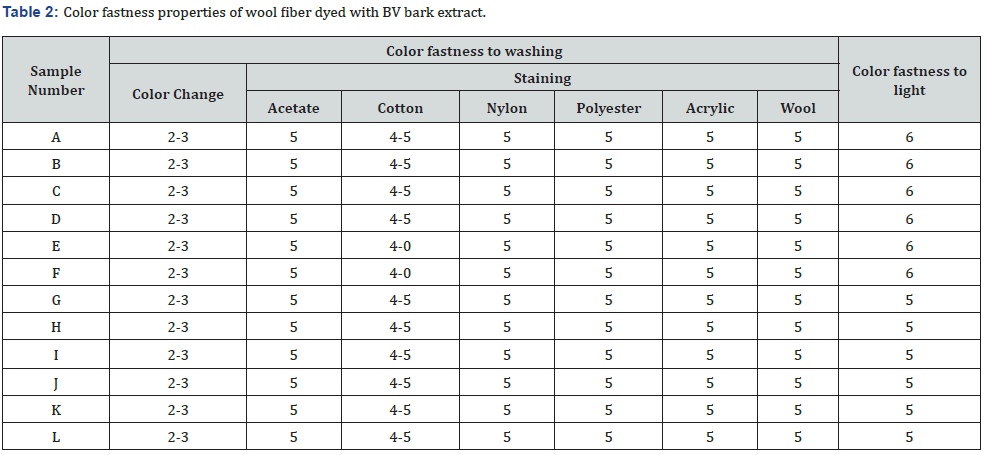
Table 2 illustrates the color fastness properties of wool fiber samples dyed with BV bark extracts. Irrespective of the shade depth and dyeing conditions, all the samples show similar test results. In colorfastness to washing, significant color change (rating 2-3) of the samples was observed; when washed with home laundry detergent. The color change happened due to the coloring matter’s halochromic effect, i.e., tannins. The halocromic effect occurred due to the deprotonation of the phenol oxygen present in the aromatic ring of the tannin by a detergent having an alkaline pH. Later, when samples are neutralized with acetic acid, they return to the original shade. In washing with detergent, neutral pH color change is minimal [rating 4]. All the samples exhibit very good to excellent results in staining to multifiber. Comparatively, darker samples show better colorfastness to light properties than lighter samples. Results are acceptable as per the standard norms.
Conclusion
Bauhinia Vahlii bark extract is an excellent source of natural dye. Wool fibers can easily be dyed with BV bark extract without metal salt-based mordants. The shade percentage is enhanced when dyed in acidic pH. Colorfastness to washing of the dyed samples shows a satisfactory result and is within the acceptable limit when washed with neutral pH detergent. Colorfastness to light of the dyed fiber samples is found satisfactory.
References
- Sudha M, Saranya A, Selvakumar G, Sivakumar N (2014) Microbial degradation of azo dyes: a review. International Journal of Current Microbiology and Applied Sciences 3(2): 670-690.
- Dan'Azumi S, Bichi M (2010) Industrial Pollution and Heavy Metals Profile of Challawa River in Kano, Nigeria. Journal of Applied Sciences in Environmental Sanitation 5: 56-62.
- Bar G, Bar M (2020) Dyeing and flame-retardant finishing of silk fabric: an eco-friendly approach. SN Applied Sciences 2(3): 1-9.
- Teklemedhin TB, Gopalakrishnan LH (2018) Environmental Friendly Dyeing of Silk Fabric with Natural Dye Extracted from Cassia singueana Journal of Textile Science & Engineering S3: 001.
- Das D, Datta DB, Bhattacharya P (2014) Simultaneous Dyeing and Finishing of Silk Fabric with Natural Color and Itaconic Acid. Clothing and Textile Research Journal 32(2): 93-106.
- Silva AB, Silva MG, Arroyo PA, and Barros MASD (2013) Dyeing Mechanism of Wool and Silk with Extract of Allium Cepa. Chemical Engineering Transactions 32: 715-720.
- Shenai VA (2006) Chemistry of Dyes and Principles of Dyeing, (5th edn), Sevak Publications, Mumbai, India.
- Zhou Y et al., (2014) Simultaneous dyeing and functionalization of silk with three natural yellow dyes. Industrial Crops and Products 64: 224-232.
- Shukla D and Gahlot K (2018). Studies on Antimicrobial Potential of Bauhinia Vahlii Stem Extracts. Journal of Drug Delivery & Therapeutics 8(3).
- Ray R, Das SN, Mohapatra A, Das HC (2020) Comprehensive characterization of a novel natural Bauhinia Vahlii stem fiber. Polymer Composites 41(9): 3807–3816.
- Bar G and Chaudhary K (2022) Characterization of Textile Grade Novel Bauhinia Vahlii Journal of Natural Fibers.
- Shukla D and Shukla K (2017) Pharmacognostical Standardization and Phytochemical Screening of Bauhinia Vahlii Leaves. World Journal of Pharmacy and Pharmaceutical Sciences 6(8): 2635-2643.
- Chauhan R, Saklani S (2013) Bauhinia vahlii; Plant to be explored. International Research Journal of Pharmacy 4(8): 5-9.
- Chavan RT, Kadam AS (2012) Preliminary phytochemistry and antimicrobial activity of bark of Bauhinia racemosa Current Botany 3(4): 31-33.
- Elbanna AH, Mahroua EA, Khaleel AE, El-alfy TS (2016) Morphological and anatomical features of Bauhinia vahlii Wright & Arnoot Grown in Egypt. Journal of Applied pharmaceutical Science 6(12): 084-093.
- Samanta AK, and Konar A (2011) Dyeing of Textiles with Natural Dyes. In Natural Dye, edited by Emriye Akcakoca Kumbasar 1: 29–56.
- Meyer TD, Iline Steyaert, Karen Hemel soet, Richard Hoogenboom, Veronique Van Speybroeck, et al. (2016) Halochromic properties of sulfonphthaleine dyes in a textile environment: The influence of substituents. Dyes and Pigments 124: 249-57.
- Ganguly D, Mondal C, Choudhury AKR (2017) Application of syntan and sodium sulphate to improve the solid dyeing effect of wool-silk blends dyed with milling type acid dyes. Research Journal of Textile and Apparel 21(3): 219-238.






























