A Cost Minimization Process of Heat and Energy Consumption for Direct Dyeing of Cotton Fabric Coloration with Triethanolamine
Anowar Hossain Md1* and Samanta AK2
1Department of Textile Engineering, City University, Bangladesh
2Department of Jute and Fibre Technology, University of Calcutta, India
Submission: August 09, 2019;Published: October 17, 2019
*Corresponding author: Anowar Hossain Md, Department of Textile Engineering, City University, Savar, Dhaka, Bangladesh
How to cite this article: Anowar Hossain Md ,Samanta AK. A Cost Minimization Process of Heat and Energy Consumption for Direct Dyeing of Cotton Fabric Coloration with Triethanolamine. Curr Trends Fashion Technol Textile Eng. 2019; 5(2): 555662. DOI: 10.19080/CTFTTE.2019.05.555662
Abstract
A cost minimization concept of heat and energy consumption for direct dyeing process of cotton fabric coloration was experimented with triethanolamine instead of sodium carbonate in the dyebath of direct dyeing with low temperature at 30℃, medium temperature at 60℃ and high temperature at 90℃ and compared with the conventional method of direct dyeing with high temperature at 90℃ where an acceptable range of increasing color strength (K/S) and color difference were observed for low temperature dyeing with triethanolamine comparing to the conventional dyeing process of high temperature with sodium carbonate. Considering heat and energy consumption in direct dyeing of cotton fabric coloration with triethanolamine method, the low temperature dyeing cost was minimized Rs. 2.24 comparing to high temperature sodium carbonate method dyeing cost.
Keywords:Direct dyeing; Cotton fabric; Sodium carbonate; Triethanolamine; Color strength; Cost minimization
Introduction
Dyeing and finishing of textiles requires large amount of heat and energy, which however cannot be eliminated but can be minimized with proper process selection, machine selection, dyes selection, auxiliaries selection as well as appropriate temperature selection in dyeing and finishing bath. In textile industry we often observe that huge amount of energy is consumed and wasted in the chemical processing of textiles. The question has been arised regarding the need to save energy and heat which can be done by adding some specific chemicals in the reaction, so that the processes may be carried out at relatively lower temperature. The purpose of the room/reduced temperature chemical processing is to carry out the feasibility of chemical processing and finishing at room temperature/reduced temperature to save energy and power which is an utmost importance to face the challenges of recent dyeing and finishing with competitive price. The fabric when exposed to lower temperature during chemical processing will definitely go through with less abrasion and breakages of fibres within it compared to those exposed at conventional higher temperature. The fabric chemically processed and finished at lower temperature might have the same colour, lustre and quality as compared to the one processed and finished at conventional higher temperatures. Both sodium carbonate and triethanolamine is toxic for the aquatic life. Sodium carbonate is normally applied for direct dyeing application on cotton fabric for fixation of dyes on cotton fabric and Triehanolamine is popularly used for cosmetic and medicine production for bonding of different chemical.
Malik S.K., Bhaumik S and Mukherjee R.N. studied that Low-temperature dyeing of cotton by direct dyes. The feasibility of dyeing of cotton using three different direct dyes at low temperature in the presence of organic base triethanolamine (TEA) has been studied. It is observed that the extent of dyeing depends upon the temperature of dyeing. TEA concentration and dye concentration. This method gives dyeing comparable to that of the conventional method at relatively very low temperature (50o). The fastness properties of the samples dyed using TEA are also comparable with those of the samples dyed in conventional manner and the dyeing cost is considerably less [1]. Triethanolamine-trisulphate (TES) was synthesized in good yield. Capillary electrophoresis (CE) was used to monitor TES synthesis, to follow TES hydrolysis and to analyse the residual products formed during the application of TES to cellulosic fabrics. TES was found to react covalently with the cotton fibre when applied in the presence of sodium hydroxide. Cotton fabrics treated with alkaline TES solutions by a pad-cure route at elevated temperatures, subsequently gave high wrinkle recovery angles, confirming that cross-linking of the cellulose molecular chains had been achieved. Significant yellowing was observed following these treatments, but fabric whiteness was improved by adding a reducing agent to the system; however, the latter addition gave a slight decrease in the wrinkle recovery values obtained from the treated cotton fabrics [2].
The synthesis of a new symmetric disazo direct dye containing the non {carcinogenic 4,40-diaminobenzanilide as the middle component and salicylic acid as the coupling component is presented. The synthesized dye is an analog of the benzidine based dye C. I. Direct Yellow 1 (C. I. 22,250)and was analyzed by thin layer chromatography (TLC), electronic spectra (VIS) and mass spectroscopy (FAB-MS). The coloristic and fastness properties of the synthesized dye were determined and compared with those of C. I. Direct Yellow 1. The toxicities of the dye and of its precursors were evaluated by biological tests, using the process of metamorphosis in the marine hydrozoon Hydractinia echinata. The concentrations (termed MRC50) at which the synthesized dye and its precursors antagonize metamorphosis induction were determined [3]. Direct dyes were applied on cellulosic material with triethanolamine [4]. Cotton fabric containing many neps was dyed with each of 17 azo direct dyes. Visible coverage of neps composed of immature fibres varied widely among the dyes. An evaluation of those structural characteristics of the dyes that might account for their differential ability to achieve coverage of neps was undertaken. Direct dyes containing more than one amino or amido group in their structure were found most likely to achieve relatively good nep coverage. Scanning electron micrographs revealed considerable variation in the types of fibres making up the individual neps in a single piece of cotton fabric. Differences in direct dye behaviour towards nep coverage were most evident with those neps composed of immature fibres with some secondary wall development [5].
Materials Used
Scoured and bleached cotton fabric was applied for this experiment and Atul direct orange 26, laboratory grade salt, sodium carbonate and triethanolamine(TEA) were used for dyeing.
Methodology
Scoured and bleached cotton fabric was dyed with 1% direct dye, orange-26 in twelve different formulation with 0%, 1%, 2% and 3% concentration of Triethanolamine for the newly proposed method of triethanolamine method as well as standard direct dyeing process of conventional method was trialed and compared with the process of direct dyeing with triethanolamine method where no sodium carbonate was applied. The same material and liquor ratio: 1:20 were maintained for all dye bath. Dyed fabric was tested after soaping. Measurement of color strength (K/S): K/S value was measured for analysis of surface colour strength of the dyed cotton fabric samples. K/S values of dyed cotton fabrics were determined by measuring surface reflectance of the dyed samples using computer aided Macbeth 2020 plus reflectance spectrophotometer followed by calculating the K/S using following Kulbeka-Munk equation with the help of relevant software.

Where, K is the co-efficient of absorption, S is the co-efficient of scattering and max Rλ is the reflectance value at maximum absorbance wavelength max Rλ The color fastness to washing was evaluated as per the ISO-2 and the color fastness to light was observed by sunlight and visual observation process.
Result and Discussion
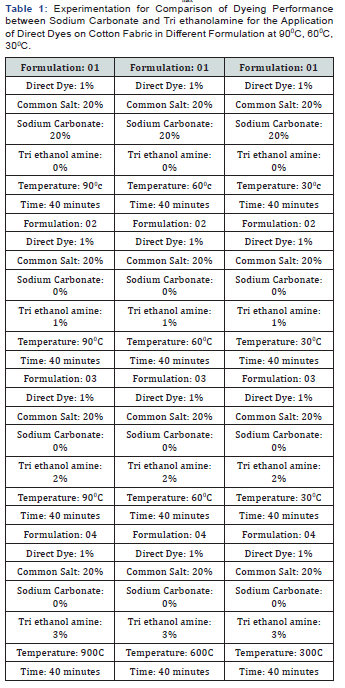
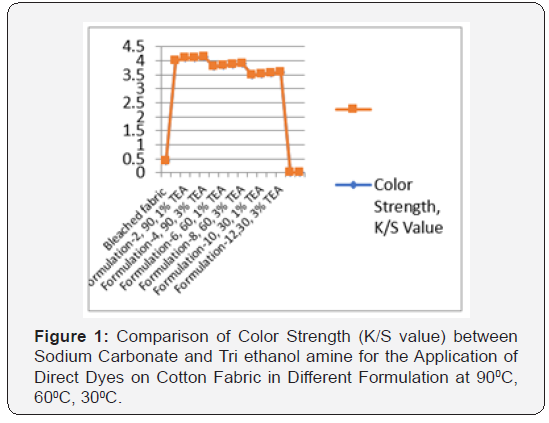
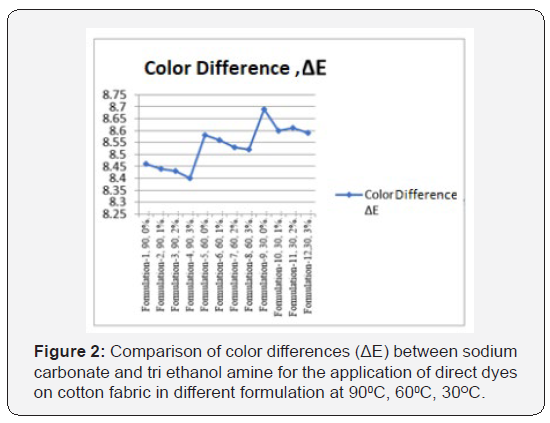
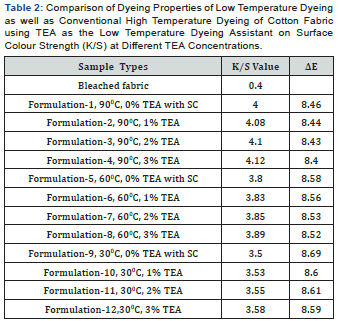
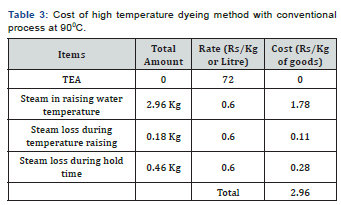
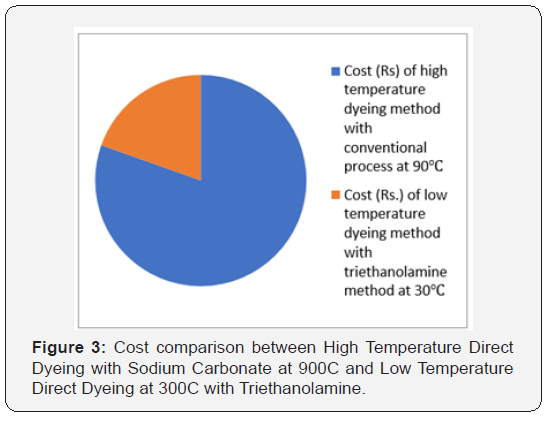
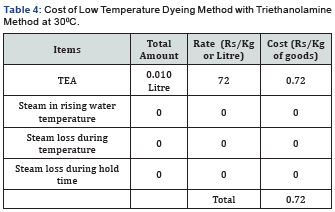
In [Table 2] and Figure [1,2] showed that Direct dyeing with low temperature at 30℃, medium temperature at 60℃ and high temperature at 90℃ and compared with the conventional method of direct dyeing with high temperature at 90℃ where an acceptable range of increasing color strength (K/S) and color difference were observed for low temperature dyeing with triethanolamine comparing to the conventional dyeing process for high temperature dyeing process with sodium carbonate. In [Table 3,4] and [Figure 3] showed that Considering heat and energy consumption in direct dyeing of cotton fabric coloration with triethanolamine method, the low temperature dyeing cost was minimized Rs. 2.24 comparing to high temperature sodium carbonate method dyeing cost.
Effect of Triethanolamine
Direct dyeing on cotton fabric observed that the dyeing with TEA gives better colour yield at low temperature comparing to conventional method. This enhancement in the colour strength could be associated with the favourable effect of TEA by enhancing the swellability and accessibility of the cellulose structure, modifying the state of the dye as well as dye structure, thereby enhancing its uptake and increasing the extent of dye fixation, thereby increasing the K/S values. [ΔE-Total colour difference, SC-Sodium Carbonate, TEA-Triethanol Amine, ΔETotal colour difference, K/S- Surface colour strength].
Effect of Dyeing Temperature
The K/S values show that the colour yield at 60oC is highest with TEA, even almost nearest that in conventional method, but the increase in K/S with the increase in dyeing temperature from 30oC to 60oC was slightly increased in dye fixation for triethanolamine method. The increase in colour strength with the increase in temperature in case of dyeing with TEA appears to be due to the following reasons:
a. Opening up the cellulose structure
b. Increasing the accessibility of cellulose structure and
c. Overcoming the activation energy barrier of the dyeing process, thereby increasing the level of molecular activity of the dye-fibre system as well as dye-fibre interaction.
Fastness Properties
The color fastness to wash and color fastness to light (sunlight) was found negligible difference for low temperature dyeing comparing to high temperature dyeing in conventional method.
Cost Comparison of Low Temperature and High Temperature Method:
Cost of steam production for dyeing machine is Rs. 0.60/ Kg and cost of Triethanolamine is Rs. 72 Litre/Kg. [1] Proposed Reaction in Dyebath: The interaction between dye and fibre depends not only the chromophore group of dyes i.e. sulphonic acid group of dyes used in this experiment, but the structure of dye fixing agent is a very critical matter of color variation on the surface of fabric may be expected from this experiment of cotton fabric coloration which may be stated as Dye-Fibre- Triethanolamine.
Conclusion
Direct dyes at different concentration of TEA, it has been observed that the K/S values are almost same and does not show much difference when it is done in 30℃ and at 60℃ when compared to 90℃ .Thus, it has been proved that using TEA at room temperature shows similar results as 90℃ in the conventional method of direct dyeing. So, using TEA with Direct dyes at low temperature more energy and power can be saved, and thus huge amount of cost can be minimized.
References
- Malik SK, Bhaumik S, Mukherjee RN (2003) Low Temperature Dyeing of Cotton by Direct dyes. Indian Journal of Fibre and Textile Research28(4): 462-465.
- Lewis DM, Zhao XF (2004) Synthesis of Triethanolamine-Trisulphate and its Application to Cellulosic Fabrics. Coloration Technology 120(4): 172-179.
- Georgeta Maria Simu, Sergiu Adrian Chicu, Nicole Morin,Walter Schmidt, Eugen Sisu (2004) Direct Dyes Derived from 4,4’-Diaminobenzanilide Synthesis, Characterization and Toxicity Evaluation of a Disazo Symmetric Direct Dye. Turkish Journal of Chemistry 28: 579-585.
- Hans Rudolf Bolliger, Herbert AlexanderPotts (1951) Stable concentrated solutions of direct dyes and process for their manufacture. The chemical technology of dyeing and printing 2: 338.
- Lenore, Anne Wilcock (1988) Relationship between direct dye characteristics and coverage of immature fibre neps in dyed cotton fabric. Journal of the Society of Dyers and Colourists 104(12): 477-482.






























