Oxidative Stress Following Influenza Virus; An Overview
Negar Nayerain Jazi1, Omid Salahi Ardekani1 and Arash Letafati2*
1Department of Bacteriology & Virology, School of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran
2Department of Virology, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran
Submission:March 01, 2023; Published:March 20, 2023
*Corresponding author:Arash Letafati, Department of Virology, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran
How to cite this article: Negar Nayerain Jazi, Omid Salahi Ardekani and Arash Letafati. Oxidative Stress Following Influenza Virus; An Overview. Curr Trends Biomedical Eng & Biosci. 2023; 21(3): 556062. DOI:10.19080/CTBEB.2023.21.556062
Abstract
Oxidative stress causes from an imbalance between the cells’ capacity to remove them through detoxification processes and generation of reactive oxygen species (ROS). Apart from triggering the host’s immune response, the influenza virus has the ability to cause oxidative stress by directly targeting the cellular antioxidant systems. In this article, we will discuss the multifaceted role of oxidative stress in the pathogenesis of the influenza virus. On the one hand, oxidative stress can contribute to the clearance of the virus by stimulating the host’s immune response. On the other hand, excessive oxidative stress can also contribute to the severity of the disease by causing tissue damage and inflammation.
Introduction
The influenza virus belongs to the Orthomyxoviridae family, characterized by an enveloped structure and a negative-sense single-stranded RNA genome. In humans, Type A and Type B influenza viruses cause significant morbidity and mortality. Despite annual vaccination campaigns, the virus is capable of evading host immune responses by continually generating new strains via antigenic shift, drift, and mutation. These mechanisms enable the virus to establish infection and cause disease in susceptible populations [1].
Reactive oxygen species (ROS) are generated as a regular byproduct of cellular metabolism and participate in numerous biological pathways. However, an imbalance in the cellular redox state can result in oxidative stress, leading to deleterious effects such as DNA damage, inflammation, and dysregulation of cell proliferation. These consequences reflect the multifaceted nature of ROS and highlight the importance of maintaining redox homeostasis for proper cellular function [2].
Multiple studies have suggested that influenza viruses induce excessive ROS production, leading to oxidative stress and respiratory cell pathophysiology. Despite the virus’s ability to generate ROS, various cellular processes are activated to maintain a normal redox state during infection. Among these processes, the superoxide dismutase (SOD) family plays a crucial role as the most significant cellular antioxidant factor.
Notably, research indicates that influenza virus infection downregulates SOD1 levels, resulting in increased ROS production. Prolonged exposure to excessive ROS exacerbates the virus’s pathogenesis and contributes to tissue damage. These findings highlight the delicate balance between ROS and cellular redox state during influenza virus infection and underscore the significance of SOD as a key regulator of ROS production [3,4].
Findings
Several studies have shown that influenza viruses cause oxidative stress state within the cell [5]. In the following article, the possible mechanisms of the virus in inducing oxidative stress will be discussed. One mechanism for the Influenza A virus (IAV) to increase reactive oxygen species (ROS) is by downregulating superoxide anion dismutase 1 (SOD1). Influenza virus PB1-F2 induces cell death by inactivating matrix metalloproteinases and releasing proapoptotic proteins [6]. Through inhibiting SOD1, PB1-F2 contributes to alveolar epithelial cells production of mitochondrial ROS. Moreover, PB1-F2-induced ROS cause the inflammasome to activate and secrete IL-1β which is one of the inflammatory cytokines [3]. Furthermore, PB1-F2 represses the mitochondrial internal-membrane potential. PB1-F2 can trigger the permeabilization and instability of mitochondrial membranes by forming a variety of sized pores in planar lipid membranes [7].
The NOX family, which includes the members NOX1 to NOX5, DUOX1, and DUOX2, are present in cells and phagosome memAbstract branes [8]. It is seen that following A549 cell lines infection with H5N1 IV, NOX was upregulated whereas SOD1, SOD3, CAT, and Nrf2 were downregulated [9]. In IV infection, NOX2 and NOX4 appear to be implicated in the formation of ROS [10,11]. Activation of DUOX2-induced ROS production in airway cells was observed via IFN induction and stimulated the expression of the melanoma-associated differentiation gene 5 (MDA5) and the double-stranded RNA helicase enzyme RIG-1-like receptor (RIG- 1) [12].
Another mechanism involved in the OS establishment of the virus is through an surge in ROS and a decrease in intracellular glutathione (GSH). Glucose- 6-phosphate dehydrogenase (G6PD) regenerates the reduced form of GSH, which also maintains redox equilibrium. However, Infection with the influenza virus leads to a decrease in both the expression and activity of G6PD [13]. Moreover, sirtuin 2 (SIRT2) and nuclear factor erythroid 2-related factor 2 (NRF2) expression were both downregulated together with G6PD following infection [14].
The third mechanism involved in OS induction is through NS1 protein. In chicken oviduct epithelial cells (COECs) treated with NS1 expression plasmids, the quantity of avian influenza virus NS1 protein was associated with elevated levels of ROS. It’s interesting to note that NS1-transfected COECs showed lower superoxide dismutase and catalase activity [2].
However, through the OS state within the cell, MicroRNAs (miRNAs) expression is changed. MiRs are one of the important noncoding RNAs that regulate a variety of cellular pathways by regulating gene expression. Interactions and regulation between Ros and miRNA can occur during influenza virus infection [4]. Different experimental models demonstrate differential expression of miRNAs under oxidative stress (mainly miR-9, -21, -220a, and -141) [15]. MiR-9 expression is stimulated by IAV through OS generation, which augments viral replication and mitochondrial dysfunction [4].
As a result of IAV induced ROS, miR-141 expression is enhanced to diminish cellular inflammation through the Nrf2 pathway [16]. In ROS conditions, miR-21 and miR-200a also seem to be upregulated which are involved in IAV infected cells-inhibited apoptosis [17].
IV infection can decrease downstream molecules of the Nrf2 pathway, such as SOD1, HO-1, NQO1, and CAT; thus, regulation of these molecules has a significant role in elevated ROS following IV infection [18,19].
IVs have the ability to control various oxidative-stress signaling pathways, such as PI3K/AKT, NF-κB, and MAPK, which ultimately enhance viral replication and contribute to the development of disease. Therefore, manipulating these signaling pathways may exacerbate the lung damage caused by IV infection [20] (Figure 1).
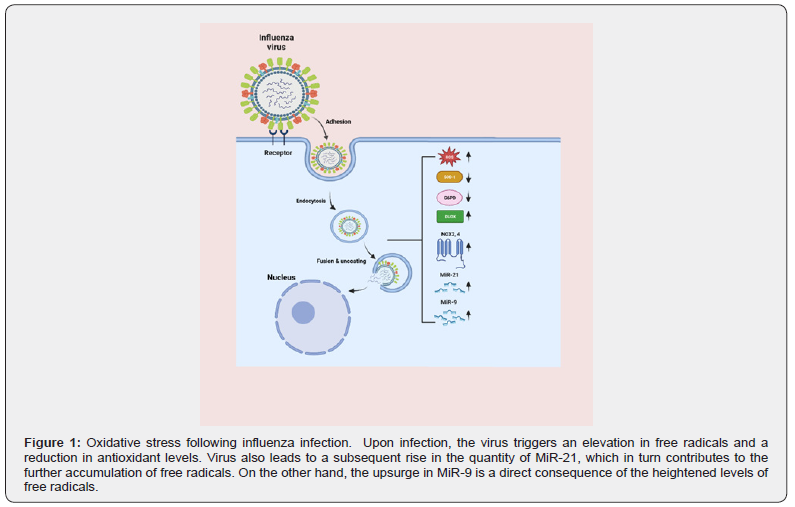
Discussion
Influenza virus infection is a major cause of respiratory illness worldwide. The virus infects the respiratory epithelial cells, leading to an inflammatory response and tissue damage. One of the mechanisms underlying this damage is oxidative stress induction, which occurs due to the excessive production of ROS by infected cells.
ROS and RNS are known to damage cellular macromolecules and disrupt cellular signaling pathways, leading to cellular dysfunction and death. Moreover, these species are also known to activate pro-inflammatory pathways, leading to an exacerbation of the inflammatory response. Several studies have demonstrated the induction of oxidative stress during influenza virus infection [11,21].
The induction of oxidative stress during influenza virus infection has important implications for disease pathogenesis and treatment. For example, antioxidants have been shown to reduce oxidative stress, influenza associated complications such as pneumonia and improve lung function [22]. Moreover, a recent study showed that treatment with the antioxidant N-acetylcysteine (NAC) reduced viral replication and inflammation in mice infected with the H5N1 highly pathogenic influenza virus [21,23]. Oxidative stress induction is a key mechanism underlying the pathogenesis of influenza virus infection. Targeting oxidative stress pathways may represent a promising approach for the treatment of this disease.
Conclusion
It is evident that augmenting comprehension of the oxidative stress mechanism of the influenza virus can yield novel insights into the pathogenesis of the virus. Additionally, it can offer a therapeutic avenue for curtailing and managing influenza virus infection, particularly in cases of highly virulent human avian influenza.
References
- McAuley JL, Gilbertson BP, Trifkovic S, Brown LE, McKimm-Breschkin JL (2019) Influenza virus neuraminidase structure and functions. Frontiers in microbiology 10: 39.
- Qi X, Zhang H, Wang Q, Wang J (2016) The NS1 protein of avian influenza virus H9N2 induces oxidative-stress-mediated chicken oviduct epithelial cells apoptosis. Journal of General Virology 97(12): 3183-3192.
- Shin N, Pyo CW, Jung KI, Choi SY (2015) Influenza A virus PB1-F2 is involved in regulation of cellular redox state in alveolar epithelial cells. Biochemical and biophysical research communications 459(4): 699-705.
- Haque MM, Murale DP, Lee JS (2020) Role of microRNA and oxidative stress in influenza A virus pathogenesis. International journal of molecular sciences 21(23): 8962.
- Lee C (2018) Therapeutic modulation of virus-induced oxidative stress via the Nrf2-dependent antioxidative pathway. Oxidative Medicine and Cellular Longevity 2018: 6208067
- Chen KK, Minakuchi M, Wuputra K, Ku CC, Pan JB, et al. (2020) Redox control in the pathophysiology of influenza virus infection. BMC microbiology 20(1): 1-22.
- Yamada H, Chounan R, Higashi Y, Kurihara N, Kido H (2004) Mitochondrial targeting sequence of the influenza A virus PB1-F2 protein and its function in mitochondria. FEBS letters 578(3): 331-336.
- Panday A, Sahoo MK, Osorio D, Batra S (2015) NADPH oxidases: an overview from structure to innate immunity-associated pathologies. Cellular & molecular immunology 12(1): 5-23.
- Lin X, Wang R, Zou W, Sun X, Liu X, et al. (2016) The influenza virus H5N1 infection can induce ROS production for viral replication and host cell death in A549 cells modulated by human Cu/Zn superoxide dismutase (SOD1) overexpression. Viruses. 8(1): 13.
- Bedard K, Krause KH (2007) The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiological reviews 87(1): 245-313.
- Amatore D, Sgarbanti R, Aquilano K, Baldelli S, Limongi D, et al. (2015) Influenza virus replication in lung epithelial cells depends on redox‐sensitive pathways activated by NOX4‐derived ROS. Cellular microbiology 17(1): 131-45.
- Kim HJ, Kim CH, Kim MJ, Ryu JH, Seong SY, et al. (2015) The induction of pattern-recognition receptor expression against influenza a virus through Duox2-derived reactive oxygen species in nasal mucosa. American Journal of Respiratory Cell and Molecular Biology 53(4): 525-535.
- De Angelis M, Amatore D, Checconi P, Zevini A, Fraternale A, et al. (2022) Influenza virus down-modulates G6PD expression and activity to induce oxidative stress and promote its replication. Frontiers in cellular and infection microbiology 11: 804976.
- Baird L, Yamamoto M (2020) The molecular mechanisms regulating the KEAP1-NRF2 pathway. Molecular and cellular biology 40(13): e00099-00020.
- Dong C, Sun X, Guan Z, Zhang M, Duan M (2017) Modulation of influenza A virus replication by microRNA‐9 through targeting MCPIP1. Journal of medical virology 89(1): 41-8.
- Li Y, Li J, Belisle S, Baskin CR, Tumpey TM, et al. (2011) Differential microRNA expression and virulence of avian, 1918 reassortant, and reconstructed 1918 influenza A viruses. Virology 421(2): 105-113.
- Li Y, Chan EY, Li J, Ni C, Peng X, et al. (2010) MicroRNA expression and virulence in pandemic influenza virus-infected mice. Journal of virology 84(6): 3023-3032.
- Pyo CW, Shin N, Jung KI, Choi JH, Choi SY (2014) Alteration of copper–zinc superoxide dismutase 1 expression by influenza A virus is correlated with virus replication. Biochemical and biophysical research communications 450(1): 711-716.
- Shoji M, Arakaki Y, Esumi T, Kohnomi S, Yamamoto C, et al. (2015) Bakuchiol is a phenolic isoprenoid with novel enantiomer-selective anti-influenza A virus activity involving Nrf2 activation. Journal of Biological Chemistry 290(46): 28001-28017.
- Nencioni L, De Chiara G, Sgarbanti R, Amatore D, Aquilano K, et al. (2009) Bcl-2 expression and p38MAPK activity in cells infected with influenza A virus. Journal of Biological Chemistry 284(23): 16004-16015.
- Geiler J, Michaelis M, Naczk P, Leutz A, Langer K, et al. (2010) N-acetyl-L-cysteine (NAC) inhibits virus replication and expression of pro-inflammatory molecules in A549 cells infected with highly pathogenic H5N1 influenza A virus. Biochemical pharmacology 79(3): 413-420.
- Uchide N, Toyoda H (2011) Antioxidant therapy as a potential approach to severe influenza-associated complications. Molecules 16(3): 2032-2052.
- Beck MA (2001) Antioxidants and viral infections: host immune response and viral pathogenicity. Journal of the American College of Nutrition 20(sup5): 384S-388S.






























