Monitoring of the Sound Velocity Variation In Rat Muscle and a Novel Approach to Quantify Muscle Injury
Zakir Hossain M1-3*
1Kinesiology, University of Calgary, Canada
2Health and Physical Education, Mount Royal University, Canada
3Zenith Consultancy & Technology Solution, Calgary, Canada
Submission:April 29, 2022;Published:May 30, 2022
*Corresponding author:Muhammad Zakir Hossain, Owner and CEO, Zenith Consultancy & Technology Solution, Calgary, AB, Canada
How to cite this article:RZakir Hossain M. Monitoring of the Sound Velocity Variation In Rat Muscle and a Novel Approach to Quantify Muscle Injury. Curr Trends Biomedical Eng & Biosci. 2022; 20(5): 556050. DOI:10.19080/CTBEB.2022.20.556050
Abstract
Detection of the degree of skeletal muscle injury is of utmost importance in medical treatment and rehabilitation. Ultrasound and magnetic resonance imaging are now been using to assess muscle injuries. Presented is the custom-built ultrasonic detection scheme that has been implemented to detect the degrees of muscle injuries. The changes in the velocity of ultrasound in muscle due to artificially administered injuries have been detected and compared. The purpose of this study is to present a novel high-resolution ultrasonic detection scheme in the field of skeletal muscle injury detection and research. To detect variations of the velocity of sound, the expansion of the muscle is suppressed by mechanical clamping. Under this condition, any variation in the time-of-flight of the ultrasonic signals can only be introduced by a variation of the velocity of sound along the path of the ultrasound transit signal. Opposite to the general behavior of healthy muscle, the injured muscle shows comparatively an increase in the time-of-flight, relating to a decrease in the sound velocity, with the levels of injuries. Since injured muscle increases the travel time of ultrasonic signal, the influence of artificial injury on the monitored muscle can be detected and quantified with the comparison measurements between healthy and injured muscle. The diminishing velocity value with the level of injuries is detected and quantified in this study. The observed rate of change in sound velocity and force for contraction and relaxation phases, for different activation frequencies, are found to be deteriorated with the degrees of injuries. The achieved resolution of the velocity of (longitudinally polarized) ultrasound in rat muscle is about 0.01% relating to the measurement on relaxed muscle by comparison to the well-known velocity of water. A resolution in time resolved measurements of about 0.01% is reached for each individual measurement, performed at a repetition rate of 50 M measurements per second, during monitoring of dynamic processes for the actual velocity of ultrasound traveling in the observed rat’s GM muscle.
Keywords: Variation of ultrasound velocity in rat muscle; Monitoring of the isometric muscle contraction; Quantification of rat skeletal muscle injury; Muscle injury dynamics; Real-time detection of the degree of muscle injury
Introduction
Muscle research has been around for over two thousand years, and has natural momentum due to its obvious necessity in life. Skeletal muscle is made up of thousands of cylindrical muscle fibers, often running all the way from origin to insertion. The fibers are bound together by connective tissue through which run blood vessels and nerves. Each muscle fiber contains an array of myofibrils that are stacked lengthwise and run the entire length of the fiber [1]. Rat medial Gastrocnemius (GM) muscle is a compartmentalized skeletal muscle. The functional properties and fiber type composition of the most proximal compartment contained predominantly fast twitch oxidative fibers and the distal compartment is composed of fast twitch glycolytic fibers [2]. The muscle–tendon unit is, in essence, the effort arm of the biomechanical system that enables locomotion. Locomotion is emerging through the interaction of complex neural, muscular, and skeletal systems. It arises from a complex orchestration of the neural, muscular, skeletal, and sensory systems. The nervous system controls muscle force generation in a quantal manner by successively recruiting motor units of increasing size. The muscle force is the sum of forces of multiple motor units (MUs), which have different contractile properties. During an isometric contraction, MUs develop infused tetani, which result from the summation of twitch-shape responses to individual stimuli [3-5].
Typically, skeletal muscle injury is defined with respect to morphological or physiological indices. Most frequently, quantitative decrements of muscle injury are determined on force production or disruptions in normal myofribilar structure. Allowing 4 minutes rest between each stretch, peak force was found to be the best predictor of muscle injury [6]. The most accepting assessments of injury rely on the quantization of the loss of muscle contractile function, such as reductions in force production relative to muscle cross section, increases in time-topeak force production, loss of peak tetanic force, or increases in fatigability. The decrements in muscle function by any physiological criterion or by disruptions of structure using morphological criteria is sufficient to show injury [7]. The ability to transmit maximum isometric force of injured muscle will be lesser than the tetanic force generated by healthy muscle [8,9]. Moreover, slowing down of relaxation speed is one of the anticipated changes in the fatigued skeletal muscle contraction [10].
The mean tetanic tension is greatest for type FF (Fast twitch, Fatigable) units, intermediate for type FR (Fast twitch, Fatigue resistant) units and smallest for type S (Slow twitch) units [11]. These differences may be attributed to differences in innervations ratio, i.e., the number of muscle fibers composing individual motor units, the mean cross-sectional area of the muscle fibers, and specific tension, i.e. the force output per unit cross-sectional area of muscle fibers. The difference among individual motor units was usually explained in terms of the first factor until the mid-1970s [12]. Burke and his co-workers estimated relative innervations ratio by an indirect approach and found that variation was very small among motor unit types [13,14]. They proposed that difference in specific tension of unit fibers (i.e. muscle fibers composing a motor unit) was an important factor determining the difference in tetanic tension between type FF (or FR) and S units in cat muscles. From the literature, it has been found that the healthy muscle’s contractile properties are affected by the injuries. The sarcolemmal integrity is also disturbed by the fiber damage [15]. Several mechanical properties of muscle are reported to be deteriorated due to the onset of fatigue [16,17]. There are many circumstances where it is desirable to obtain the contractile response of skeletal muscle under physiological circumstances: normal circulation, intact whole muscle, at body temperature. This includes the study of contractile responses like post-tetanic potentiation, muscle injury, and fatigue. Furthermore, the consequences of disease, disuse, injury, training and drug treatment can be of interest.
The development of technologies suitable for monitoring of the muscle health is still progressing. We have lately contributed with a detection scheme suitable for detection of the muscle dynamics with high temporal resolution [18,19]. To observe variations of ultrasonic velocity in selected rat muscle, a detection method based on propagating longitudinal polarized ultrasonic bulk waves, suitable to determine the sound velocity across the selected Rat GM muscle, has been developed and applied. The artificial injuries we put to the rat GM muscle should potentially disrupt the contractile properties of that muscle. Such injuries would change in contractile properties of muscles that can alter or limit their functions those might manifest the alteration of propagation velocity of ultrasound in the muscle. With the miniaturized detection scheme presented here, the actual velocity of sound under activation of a muscle can be determined which on one hand can be used to justify the just mentioned simplifying assumption and on the other hand to gain additional information on the state of the muscle under observation. The rat medial GM muscle dynamics for an isometric contraction have been studied and presented in this study. Very important quantitative values of the injured muscle dynamics are revealed from this novel monitoring. The changes of contractile properties are observed depending on the level of imparted artificial injuries we administered.
Method and Protocol of Experiment
Protocol of experiment
This experiment is performed at the physiology laboratory of the kinesiology department of the University of Calgary, AB, Canada. The ethical procedure for animal experiment is approved by the University of Calgary research ethics board, animal care committee, and biosafety committee. To monitor the ultrasound velocity variation in muscle and to study the contractile responses of the rat medial GM muscle to artificial injuries, the animal is anesthetized, and the muscle is separated from surrounding tissue at its distal end. The Achilles tendon is attached to a force transducer, and the sonic sensors positioned opposite to the medial GM muscle, allowing measurement of the muscle’s contractile response and ultrasound velocity variation at 31 oC to 34 oC with an intact circulation. The medial GM muscle is surgically isolated, with the origin intact. Care has been taken to maintain the blood and nerve supplies. A long section of the sciatic nerve is cleared of connective tissue, and severed proximally. All branches of the distal stump that do not innervate the medial GM muscle are severed. The distal nerve stump is inserted into a cuff lined with stainless steel stimulating wires. The calcaneus is severed, leaving a small piece of bone still attached to the Achilles tendon. Immobilization by the metal probe in the femur and tibia prevents movement of the muscle origin. The Achilles tendon is attached to the force transducer and the loosened skin is pulled up at the sides to form a container that is filled with warmed paraffin oil. The oil distributes heat evenly and minimizes evaporative heat loss. A heat lamp is directed on the muscle, and the muscle and rat are allowed to warm up to 37oC. While it is warming, maximal voltage and optimal length can be determined. These are important initial conditions for any experiment on intact whole muscle. The experiment includes determination of standard contractile properties, like the force-frequency relationship, force-length relationship, and force - velocity (sound) relationship.
Monitoring the ultrasound velocity in muscle
The novel non-invasive high resolution sonic detection scheme based on chirped ultrasonic wave-trains passing the monitored muscle, earlier used for monitoring of the lateral muscle extension and sound velocity variation in contracted and relaxed human muscle [18,19], is employed here for monitoring of variations of the velocity of ultrasonic waves before, during and after activation of the observed animal muscle for healthy, mild (12 pokes of the needle to the muscle belly), moderate (22 pokes of the needle to the muscle belly), and severe (36 pokes of the needle to the muscle belly) injured states. The artificial injuries are administered with the sharp needle’s pokes. To keep a fixed distance between the transducers and to maintain the shape of the monitored muscle, a custom-designed structure, manufactured with the aid of stainless-steel outer support frame, was mounted on the monitored medial GM muscle of the Rat. The frame has an arrangement of controlling the distance between the ultrasonic transducers at any desired position. The emitting and receiving transducers, 1 mm sonomicrometry crystals (Sonometrics Corporation, London, ON, Canada) were mounted through bores (Figure 1 - right) of the custom-made frame to ensure direct contact to the belly of the monitored rat medial GM muscle. Experiments are performed on three Rats weight approximately (200±4) g each. With crystals across muscle fixed distance of 4.01 mm for Rat 1, and 6.73mm for Rat 2, and 6.65 mm for Rat 3. Tetanic contraction of 20, 40, 60, 100, 200 Hz for 300 ms were observed. Pause allowed between monitoring was 5 minutes. Low voltage (1.2 to 1.5) V with double-pulse, 5 ms delay, Temperature recorded for the medium 31°C, and for the Rat 34ºC. The primarily determined property was the variation of the TOF for an ultrasonic wave package traveling across the muscle.
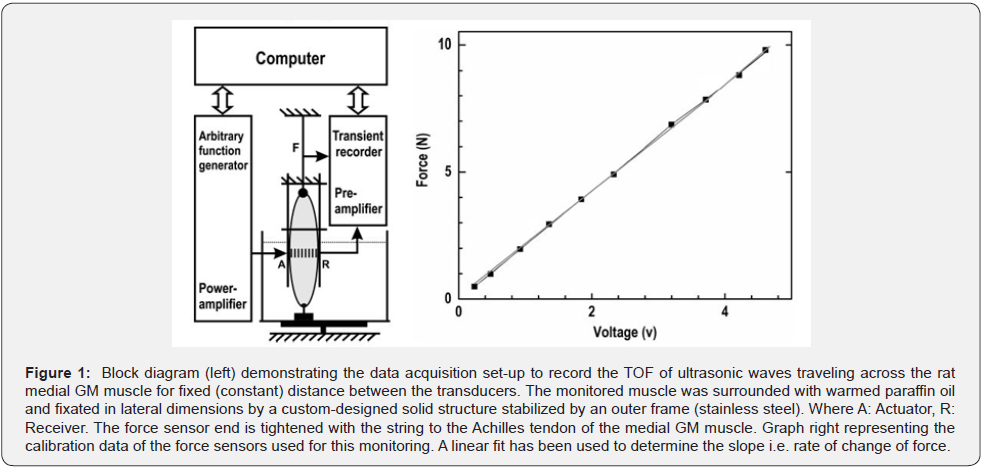
For an adjusted and fixed distance between the transducers the TOF observed for the passage in any object positioned in between the transducers including the instrumental delay can be determined directly from the time resolved recorded signals for excitation and detection of the acoustic waves [20]. The monitoring scheme is based on the detection of the transit time also addressed as Time-of-Flight (TOF) of ultrasound propagating laterally through the observed Rat’s GM muscle. The TOF from which all other data is derived is observed with the aid of a computer controlled arbitrary function generator and a synchronized transient recorder as illustrated in Figure 1- left. Detection of ultrasonic signal is carried out with a sample frequency of 50 MHz. The up chirped signals ranging from 1 to 3.7 MHz, and performing a cross correlation on the received transmitted signals, which leads to an improvement of the signal to noise ratio. Variations of the TOF of the ultrasound transmitted through the muscle can only originate from variations of the velocity since the muscle’s movement has been restricted with a suitable frame (Figure 1- left).
Prior to experiment, the force transducer was calibrated (Figure 1- right) to make sure that there is no difference in forces measured. The rate of change of force measured with the force transducer is determined to be 2.07±0.06 N/V through the followed calibration process. The calibrated force transducer was connected to the Achilles tendon of the monitored GM muscle with a string. The force data was recorded with the identical measurements system as used in sound velocity variation detection in muscle synchronously. The temporal variation of medial GM muscle force, sound velocity in muscle were determined at muscle-tendon complex lengths with tetani of 300 ms with stimulation frequencies of 40, 60, 100, 200·Hz at 1.2 to 1.5 V. Data were collected for healthy muscle and for 12/mild, 22/moderate, 36/severe artificial injuries. By subsequent data evaluation also based on data collected for the clamped muscle monitoring, not only the sarcomere dynamics of the activated muscle can be quantitatively monitored but also effects relating to variations of the velocity of sound can be evaluated on a quantitative basis.
Results
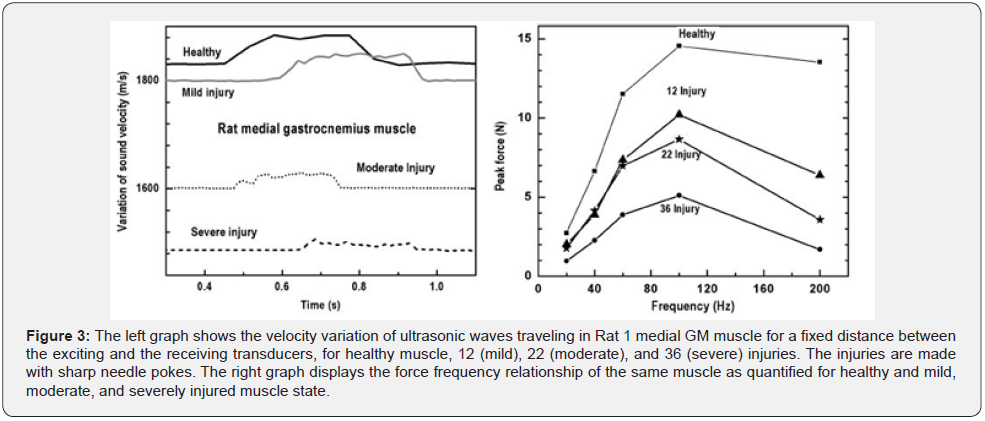
Figure 2 - left shows the results obtained for the Rat 1 muscle monitoring: Since the distance between transducers is fixed, the variations of the observed time-of-flight allow the determination of the variations of the sound velocity before, during, and after the contraction of the muscle. The temporal delay of the excitation signal is determined with a parabolic fit with the initial inflection points, the down headed arrow at the focal point of the fitted parabola as indicated 228.77 ns. The down- headed arrow at the point of intersection of the base line fit and the inflection data point line fit indicate the time of detection delay is 448 ns,as shown in Figure 2 - right. The difference between these delay times is the TOF value of the ultrasonic signal after passing through the healthy muscle: which is determined as 220.07 ns. The fixed distance between transducer is 4.02 mm, measured with Vernier Scale (Neoteck, Part nr. VTM-TL358). So, the velocity of the ultrasonic signal through the healthy rat GM muscle is quantified as 1826.69 m/s. Similarly, the results obtained for mild, moderate, and severely injured muscle are: 1816.75 m/s, 1612.38 m/s, and 1545.67 m/s respectively. These values are quantified when muscle was at inactive state that is at relaxed condition. The figure 3- left showing the values for sound velocity variation for relaxation phase-isometric tetani phase-tetani following relaxation phase, for healthy mild moderate and severely injured muscle. The isometric tetani phase is arbitrarily chosen for 300 ms.

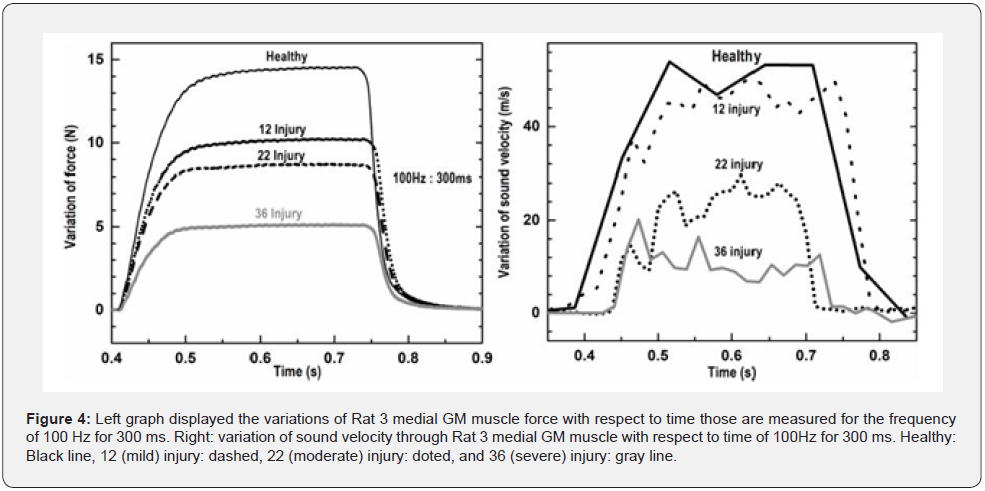
Similarly, calculated results obtained for the Rat 2 muscle monitoring: The velocity value through the severely injured muscle: The excitation signal delay: 228.77 ns, detection end delay: 676.77 ns, Distance: 6.87 mm, TOF: 448 ns, velocity: 1533.48 m/s. The velocity values for the mild, moderately injured, and healthy muscle are determined to be: 1604.34 m/s, 1805.33 m/s, and 1817.58 m/s respectively. The graphs in Figure 3 - right is representing the peak force value with respect to excitation frequency of 20, 40, 60, 100, and 200 Hz for healthy, 12, 22, and 36 injuries respectively, for Rat 2 medial GM muscle. Figure 4 - left displayed the variations of Rat 3 medial GM muscle force with respect to time those are monitored and quantified for the specific frequency of 100 Hz for 300 ms. Graph right representing the variation of sound velocity through rat medial GM muscle with respect to time for the same. Healthy: Black line, 12 injury: dashed, 22 injury: doted, and 36 injury: gray line. The quantitative values, the rate of change of Rat 3 GM muscle forces and the rate of change of sound velocities with the contraction and relaxation phases (for frequency 100 Hz and titanic duration of 300 ms) are presented in Table 1 and 2 respectively. These values are determined with slopes of the line fits, fitted with selective data points related to the rise and fall, of the respective curves of healthy, mild, moderate, and severe injuries.
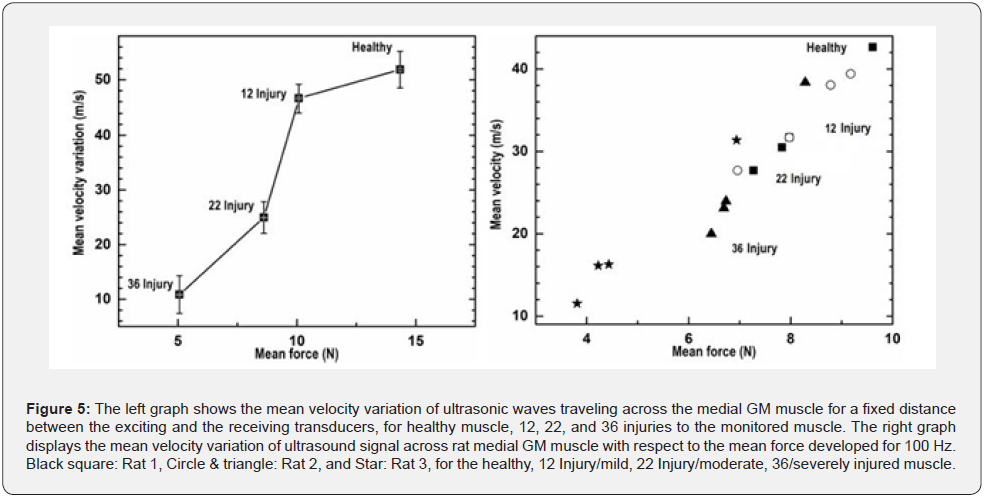
The left graph in Figure 5 shows the mean velocity variation of ultrasonic waves traveling across the medial GM muscle with respect to the mean muscle force developed for 100 Hz, for a fixed distance between the exciting and the receiving transducers, for healthy, mild, moderate, and severely injured muscle. The mean velocity and force variation quantified for healthy muscle: 51.84±3.35 m/s and 14.34±0.21 N; mildly injured muscle: 46.61±2.54 m/s and 10.08±0.15 N; Moderately injured muscles:24.96±2.91 m/s and 8.61±0.09 N; severely injured muscles: 10.85±3.45 m/s and 5.06±0.07 N respectively. The right graphs Figure 5 displays random the mean velocity variation of ultrasound signal in rat medial GM muscle with respect to the mean forces developed for 100 Hz and 300 ms excitation duration in rat medial GM muscle. Black squares are the represented quantitative values determined for the Rat 1, Circles & triangles are the represented quantitative values determined for the Rat 2, and Black Stars are the represented quantitative values determined for the Rat 3, for the healthy, 12 Injury/mild, 22 Injury/moderate, 36/severely injured muscle.
The left graphs in Figure 6 show the velocity variation recorded for the clamped muscle measurement on Rat 3 medial GM muscle. The vertical axis of the left graph is representing sound velocity variation for healthy, mild/12, moderate/22, and severely/36 injured muscle for three repetitions measured after 5 minutes of interval. The right graphs in figure 6 represent the mean velocity variations with respect to the mean forces for the same GM muscle monitoring for 100 Hz. The maximum sound velocity variation is determined with a line fit with the values observed for isometric tetani, excitation frequency of 100 Hz duration 300 ms. Quantitative values for 3 repetitive measurements on Rat 3 GM muscle at healthy condition, isometric contractile state, are observed: 1626.04±0.13 m/s, 1624.21±0.15 m/s, 1625.31±0.19 m/s respectively; for mildly injured state muscle are: 1618.03±0.14 m/s, 1619.17±0.12 m/s, 1620.04±0.23 m/s respectively; for moderately injured muscle are: 1613.02±0.15 m/s, 1615.11±0.18 m/s, 1617.04±0.21 m/s respectively; and for severely injured muscle are: 1604.11±0.13 m/s, 1606.25±0.12 m/s, 1608.21±0.16 m/s respectively.
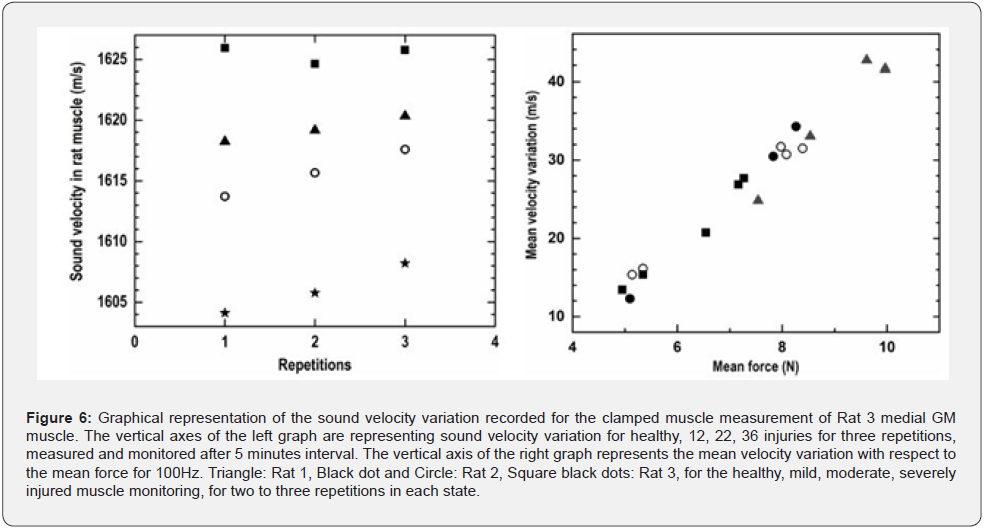
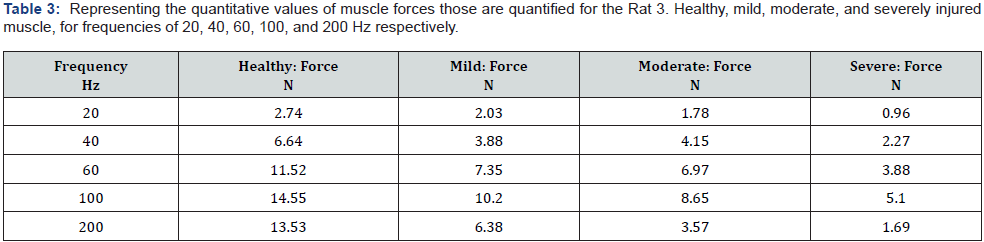
The mean force in Newton and the mean velocity variation in meter per second for healthy, mild, moderate, and severely injured muscle are shown in the left graphs of Figure 6. The quantitative results obtained for healthy: force 9.7 and 9.8 N, velocity 41and 43 m/s. Quantified sound velocity variations in mildly injured muscle are found between 31 to 37 m/s and force between 7.9 to 8.9 N;for moderately injured: velocity variations found between 21 to 29 m/s and force between 6.5 to 7.9 N; and for severely injured: velocity variations found between 10.5 to 17 m/s and force between 4.9 to 5.4 N. Values of muscle forces have been shown increments with the increased frequency steps for every state of status of the monitored muscle but the forces for severe stages for 200 Hz are recorded lesser that 100 Hz.
Evaluation
Effect of injuries on muscle’s contractile properties and sound velocity
To detect the velocity of ultrasonic waves traveling through muscle at the time of contraction and ‘before and after’ contraction, or ‘before and after’ controlled muscle activation the same settings and procedures concerning the employed excitation and correlation schemes were used. Achieved temporal resolution was up to 20ns. Spatial resolution of force and velocity are found to be up to 0.1 N and 0.01 m/s respectively. The value increment due to the temperature rise was considered negligible for little or no temperature raise or fall before, after, and during data collection. The ultrasound velocity values observed for Rat 1 and 2 GM muscle, healthy condition - relaxed state, were between 1828.69 m/s to 1817.58 m/s, lies between the velocity values in rubber to neoprene [21], which is close to the velocity value in human tendon [22]. Are these Rats GM muscle properties close to human tendon properties? The clarification of this finding requires further study. Because, the velocity value obtained for the Rat 3, healthy condition-isometric contraction state, were found similar to the velocity value observed for healthy human muscles those are between 1626.04±0.13 to 1625.31±0.19 m/s [19,23-24].
The velocities of ultrasound in severely injured muscle, relaxed state, were observed between 1533.48 m/s to 1545.67 m/s lies between the sound velocity values in water depth of 4.6 to 5.4 Km [25]. As we know, the acute injuries are largely attributable to damage to the muscle cell membrane, create unregulated influx of calcium through membrane lesions, including activation of proteases and hydrolases. The controlled injuries we administered to the Rat 3 GM muscle belly affects spindle’s hydrostatic pressure, create fiber discontinuity [26,27], the consequence is a hydrostatic pressure reduction so as of the sound velocity deterioration. The deteriorated sonic velocity values obtained for injured muscle in this study will help identifying and quantifying the level of muscle injury quantitatively. The quantitative values tabulated in Table 1 to 3 for healthy and injured muscle monitoring, Rat’s medial GM muscle are showing a clear deteriorations of muscle contraction and relaxation speed, sound velocities, and forces for different excitation frequencies with the level of injuries. The more the level of injury the lesser the muscle contraction and relaxation speed, sound velocities, and forces respectively are observed. These monitored and quantified deteriorated trends of muscle characteristic properties with respect to the level of injuries, we presented in this paper, opens the potentiality of using such variables in detecting the levels of muscle damages or injuries.
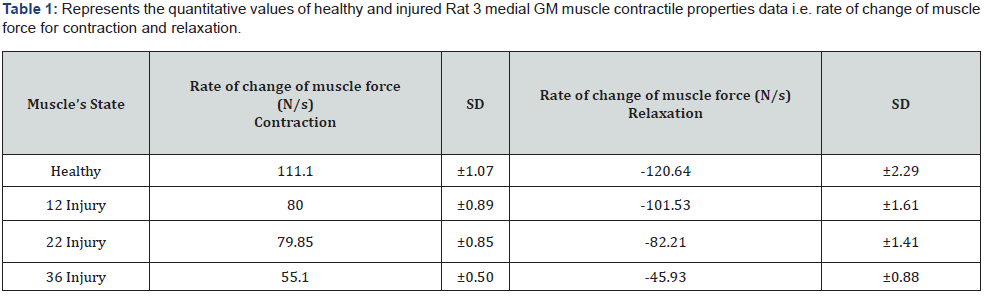
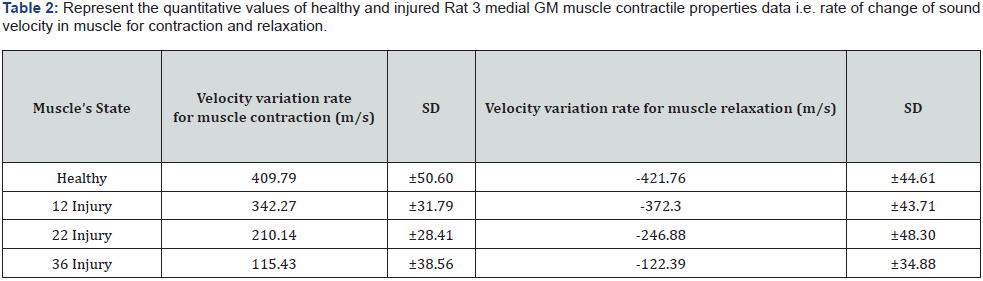
It has been observed that at the isometric contraction phase, initiated with 100 Hz for 300 ms, the sonic velocity and force are increased for every stage of muscle health tested. It is also observed that the degree of the increment of sound velocity and force reductions are inversely related to the severity of injuries administered. The graphical representation of the monitored rat medial muscle dynamics data presented in Figure 3 left nicely support above statements. As we know muscle stiffness and force increased with contraction and decreased with relaxation. The degree of increase and decrease of the stiffness and force are depending on the number of muscle fiber recruitment during contraction phase and de-recruitment during relaxation phase. The force and stiffness development relates to the hydrostatic pressure in muscle spindle as well. It is believed that the muscle force and stiffness both are inversely related with the level of injuries as injury hinders the recruitment rate of actomyocin complex. Therefore, the quantitative values obtained from the slopes of increment and decrement of muscle forces and sound velocities and their relationship with the level of injuries could be of very potential quantitative indicator for the detection of muscle injury and for the rate of the recruitment and de-recruitment of muscle fibers as well.
The force frequency relationship of healthy, mild, moderate, severely injured muscles, those are shown in Figure 3 right, indicate that the healthy muscle developed comparatively higher force values for each level of activations than the severely injured muscle. The severely injured muscle showed the lowest force development in every activation levels. It is observed that mild and moderately injured muscle forces often coincide with the forces those developed for the frequency of 20, 40, and 60 HZ except 100 Hz. At 100 Hz, all levels of forces are clearly distinguishable. Therefore, it could be suggested that the force frequency relationship observed near the vicinity of 70 to 100 Hz are expected to detect the levels muscle injuries. The force values monitored for 200 Hz are found comparatively lower than the 100 HZ. This was expected to be opposite. This could result due to the tears of muscle fibers that might occur due to the excessive tensile force developed at this range. With the degrees of tears, that is with the amounts of the discontinuation of myofribilar junctions, the recruitment rate of actomyocin complex is suffered. Results observed for 200 Hz showed lesser and lesser force development with the levels of muscle injuries. The concept of tearing muscle fibers might support this progressive reduction of the forces with respect to the level of muscle injuries.
In Figure 4 - right it is shown that the rapid increment in sound velocity lies between the onsets of contraction to the maximum isometric contraction. In Figure 4 - left it is shown that the rapid increment of muscle force also lies between the onsets of contraction to maximal isometric contraction. Almost similar patterns of curve are observed in both states. Values observed here for muscle forces and sonic velocity variations in time could be of great interests in the approach of the quantification of the levels of muscle injuries. The quantitative analyses with respect to different levels of injuries and on the basis of the functional characteristics of the focused muscle are depicted in the graphs of Figure 5. The left graph represents the mean velocity variation of ultrasonic waves traveling across the medial GM muscle, for healthy, mild, moderate, and severely injured muscle. The right graph in Figure 5 displays the mean velocity variation of ultrasound signal across rat medial GM muscle with respect to the mean force developed for 100 Hz. These quantitative results of sound velocity and muscle forces are also showing very linear relationship with the levels of injuries; hence support the idea of the quantification muscle injuries with the presented highresolution ultrasonic detection scheme.
The quantitative analyses with respect to different levels of injuries and on the basis of the functional characteristics of the focused muscle are depicted in the graphs of figure 6. The left graph shows the quantitative values of the sound velocity variation recorded for the clamped muscle measurement on Rat 3 medial GM muscle for healthy, mild, moderate, and severely injured muscle, recorded for three repetitions. The right graph represents the mean velocity variation with respect to the mean force developed for 100 Hz activation frequency. These quantitative results presented here are also showing very clear influences of the level of muscle injuries on sound velocity and muscle force. Thereby shows that the measurement and monitoring of sound velocity and muscle force in muscle could be of great interest in muscle injury detection.
Comparative characterization of sound velocity and muscle force
The left graphs in Figure 3 showed the ultrasound velocity variation through rat GM muscle in time. The observation includes and inactivated phase, relaxed to the isometric contraction phase, the isometric contraction phase, and then the isometric contraction following relaxation phase. It has been observed that at the inactive phase the sound velocity values are deteriorated with the level of injuries. The more the numbers of pokes the less the velocity values are observed. Again, with the muscle activation, that is when muscle reached from relaxed to the isometric contraction phase, the sound velocity increased further. This phenomenon is observed for healthy, mild, moderate, severely injured muscle. But the amplitude of sound velocity in the isometric contraction phase showed a clear decrease with the level of injures as well. The right graph in Figure 3 displayed the variation of peak muscle force with respect to the level of activation frequencies. Peak force value showed an increase with the increased frequencies up to 100 Hz, for every monitored level. The highest values of net forces with frequencies are observed for healthy muscle. Lowest are observed for the severely injured state. Mild and moderate injury state’s net force values coincide for 20, 40, and 60 HZ but showed clear differences for 80 Hz and above. An exception is observed for 200 Hz. At 200 Hz, net force values were found lower than the values observed for 100Hz activation frequency. The amount of net force deterioration found dependent with the levels of injuries.
A clear GM muscle’s force and ultrasound velocity reduction with the level of injuries are observed, as shown in Figure 4 healthy muscle force of contraction as well as the sonic velocity are observed maximum, the lowest contractile force and ultrasonic velocity variation are observed for the most severely injured muscle. The comparative pictures of the mean sound velocity variation with respect to the mean muscle force are presented in Figure 5. The sound velocity variation and muscle force both are found deteriorated with the degrees of injuries administered. The graphs in Figure 6 showed the comparative characterization of sound velocity variations those measured and monitored for three repetitions on healthy, mild, moderate, and severely injured muscle. It has been observed that sound velocity in monitored GM muscle has been deteriorated with the level of artificial injuries we administered, the more the number of pokes the less the sound velocity in muscle. The other comparative quantitative values of healthy and injured medial GM muscle contractile properties data i.e. rate of change of muscle contraction and relaxation speed are presented in Table 1 and 2, shows deterioration with the degree of injuries. The quantitative values of muscle forces those quantified for the Healthy, Mild, Moderate, and severe injuries for the frequencies of 20, 40, 60, 100, and 200 Hz respectively presented in Table 3, also showed the similar relationship with respect to the level of injuries.
Conclusion
The developed monitoring scheme is capable of determining the velocity of (longitudinally polarized) ultrasound in rat muscle with an accuracy of about 0.01% relating to the measurement on relaxed muscle by comparison to the well-known velocity of water. A resolution in time resolved measurements of about 0.01% is reached for each individual measurement performed at a repetition rate of 50 M measurements per second during monitoring of dynamic processes for the actual velocity of ultrasound traveling in the observed GM muscle. The variation of ultrasound velocity in time, variation of muscle force variations in time, the mean velocity variation with the mean muscle force are quantified with a very high resolution. To our knowledge the variation of the velocity of sound has not yet been monitored in life rat muscle under contraction and subsequent relaxation as involved in predefined and controlled condition. The results obtained here for healthy muscle give a range of variation under contraction for the rat GM muscle of only 2.7% and for severe injured muscle 0.8%. Identification of all the influential factors responsible for above demonstrated variations in sonic velocity for a contracted and a relaxed muscle could be the basis for a reliable quantitative indicator for muscle injury levels. Furthermore, as compared to its initial values, the velocity of sound was found to decrease with the severity of imparted injuries. The monitored muscle force and sound velocity in each frequency state showed inverse relationship. These factors might turn out to be the reliable quantitative indicators for the levels of muscle injuries detections. The slope of contraction could possibly be used to quantify the velocity of the recruitment of contractile element and the relaxation slope for the measurement of the counter effect of such isometric contraction, which similarly relates to contraction and relaxation speed of muscle.
The quantitative values of healthy and injured rat medial GM muscle’s contractile properties data i.e., rate of change of muscle force for contraction and relaxation. The velocity of sound changes with injuries at relaxed state as well as in the isometric contractile state and shown deterioration with the degrees of injuries. Rate of change of muscle force, contraction and relaxation slope could be considered as a quantitative indicator of the level of muscle injury, since the sound velocity increases with its increased stiffness and stiffness depends on the recruitment of muscle fibers, since the functions and the recruitment of muscle fiber are affected by the levels of muscle injury. Presently practiced palpation method in muscle injuries detection could be very subjective. Therefore, we have presented this systematic quantitative detection approach to quantify the types and level of muscle injury. The quantitative values of ultrasound velocity, and its inverse relationship with muscle force dynamics with respect to the level of muscle injury presented in this paper identifies the potential utility of the ultrasound signals in types and degrees of muscle injury detection.
The developed detection scheme is already portable, could be controlled remotely, and very accurate in levels of skeletal muscle injuries.
According to the literature, MRI and Ultrasound Imaging are the two commonly recommended noninvasive technologies are advised in diagnosing the skeletal muscle injury [28]. The presented results showed that the novel detection scheme implemented in this study could be of additional potential and comparatively economic technology addition in this intriguing muscle injury diagnostic field. Further studies on rat and human muscles are planned to be establishing the utility of this presented novel high-resolution ultrasonic injury detection technology. In this age of quantum technology, the rapid development of the algorithm related to big data analysis, and artificial intelligence, our future detection scheme with multiple sensors array will add up a wireless, noninvasive, and quantitative detection scheme in muscle injury assessment.
Acknowledgement
The development of the hard and software employed here has been supported by the ASI, Germany, and Zenith Consultancy & Technology Solution, Calgary, Canada. Helpful laboratory support, cooperation and discussion with Brian MacIntosh, HPL, Kinesiology, University of Calgary is gratefully acknowledged.
Funding
The author does not have any financial and personal relationships with other people or organizations that could inappropriately influence or bias of this work.
References
- Bodine SC, Roy RR, Meadows DA, Zernicke RF, Sacks RD, et al. (1982) Architectural, histochemical, and contractile characteristics of a unique biarticular muscle: the cat semitendinosus. J Neurophysiol 48(1): 192-201.
- De Ruiter CJ, Habets PE, de Haan A, Sargeant AJ (1906) In vivo IIX and IIB fiber recruitment in gastrocnemius muscle of the rat is compartment related. J Appl Physiol 81(2): 933-942.
- Adrian ED, Bronk DW (1929) The discharge of impulses in motor nerve fibers: Part II. The frequency of discharge in reflex and voluntary contractions. J Physiol 67(2): i3-i151.
- Henneman E (1957) Relation between size of neurons and their susceptibility to discharge. Science 126(3287): 1345-1347.
- Liddell EG, Sherrington CS (1925) Recruitment and some other factors of reflex inhibition. Proc R Soc Lond B Biol Science 97(686): 488-518.
- Warren GL, Hayes DA, Lowe DA, Armstrong RB (1993) Mechanical factors in the initiation of eccentric contraction-induced injury in rat soleus muscle. J Physiol 464: 457 - 475.
- Street SF, Ramsey RW (1965) Sarcolemma: Transmitter of active tension in frog skeletal muscle. Science 149(3690): 1379-1380.
- Dellorusso C, Crawford RW, Chamberlain JS, Brooks SV (2001) Tibialis anterior muscles in mdx mice are highly susceptible to contraction induced injury. J Muscle Res Cell Motil 22(5): 467-475.
- Harris JB (2003) Myotoxic phospholipases A2 and the regeneration of skeletal muscles. Toxicon 42(8): 933-945.
- Westerblad H, Lannergren J, Allen DG (1997) Slowed Relaxation in Fatigued Skeletal Muscle Fibers of Xenopus and Mouse. Contribution of [Ca2+]i and cross-bridges. J Gen Physiol 109(3): 385-399.
- Burke RE, Levine DN, Salcman M, Tsairis P (1974) Motor units in cat soleus muscle: physiological, histochemical and morphological characteristics. J Physiol 238(3): 503-514.
- Henneman E, Olson CB (1965) Relations between structure and function in the design of skeletal muscles. J Neurophysiol 28: 581-598.
- Burke RE, Tsairis P (1973) Anatomy and innervation ratios in motor units of cat GM. J Physiol 234(3): 749-765.
- Steegmaier M, Blanks JE, Borges E, Vestweber D (1997) P-selectin glycoprotein ligand-1 mediates rolling of mouse bone marrow-derived mast cells on P-selectin but not efficiently on E-selectin. Eur J Immunol 27(6): 1339-1345.
- Matsuda R, Nishikawa A, Tanaka H (1995) Visualization of dystrophic muscle fibers in mdx mouse by vital staining with Evans blue: Evidence of apoptosis in dystrophin-deficient muscle. J Biochem 118(5): 959-964.
- Merletti R, Lo Conte LR, Orizio C (1991) Indices of muscle fatigue. J Electromyogr Kinsiol 1(1): 20-30.
- Brenner B (1990) Molecular mechanisms in muscular contraction. In: Squire JM (Ed.), Muscle mechanics and biological kinetics. Mac Millan Press, London, UK, pp. 77-149.
- Zakir Hossain M, Grill J, Grill W (2019) Monitoring skeletal muscle dynamics and modelling the non- linear response. SN Appl Sci 1: 1538.
- Zakir Hossain M, Grill W (2019) High-resolution monitoring of the velocity of ultrasound in contracted and relaxed human muscle. Comp Methods Biomech Biomed Engg: Imaging & Visualization 7(4): 414-419.
- Zakir Hossain M, Voigt H, Grill W (2009) Monitoring of variations in the velocity of sound in contracting and relaxing muscle. Health monitoring of structural and biological systems. Proc SPIE 7295: 72951Q.
- Class Instrumentation Ltd Ultrasonic Sound Velocity Table.
- James S, Scott W, Locke S, Sue H (2012) Measuring mechanical properties of tendon in vivo. J Sci Med Sport 15 (1): S62-S63.
- Topchyan A, Tatarinov A, Sarvazyan N, Sarvazyan A (2006) Ultrasound velocity in human muscle in vivo: Perspective for edema studies. Ultrasonics 44(3): 259-264.
- Mol CR, Breddels PA (1982) Ultrasound velocity in muscle. J Acoust Soc Am 71(2): 455-461.
- Velocity of sound as a function of depth at a position north of Hawaii in the Pacific Ocean derived from the 2005 World Ocean Atlas the SOFAR channel spans the minimum in the velocity of sound at about 750-m depth.
- James GT (2011) Mechanisms of muscle injury, repair, and regeneration. Compr Physiol 1(4): 2029-2062.
- Tamura Y, Hatta I, Matsuda T, Segi H, Tsuchiya, T (1982) Changes in muscle stiffness during contraction recorded using ultrasonic waves. Nature 299: 631 - 633.
- Rubin SJ, Feldman F, Staron RB (1995) Magnetic resonance of muscle injury. Clinical Imaging 19(4): 263-269.






























