Sample Size Estimation/Re-Estimation Under a Promising Zone Design in Clinical Trials
Tianxiao Zhang1,2*, Jingchuan Guo2 and Shein Chung Chow1
1Department of Biostatistics and Bioinformatics, Duke University School of Medicine, USA
2Department of Pharmaceutical Outcomes & Policy, University of Florida, USA
Submission: May 18, 2022; Published: May 30, 2022
*Corresponding author: Tianxiao Zhang, Department of Biostatistics and Bioinformatics, Duke University School of Medicine, 2424 Erwin Road, Durham, North Carolina, USA
How to cite this article: Tianxiao Z, Jingchuan G, Shein C C. Sample Size Estimation/Re-Estimation Under a Promising Zone Design in Clinical Trials. Biostat Biom Open Access J. 2022; 10(5): 555797. DOI: 10.19080/BBOAJ.2022.10.555797
Abstract
The promising zone design is a novel adaptive clinical trial design that focuses on the interim analysis test statistic and its conditional power. The term promising zone defined an interval of interim analysis test statistics and its corresponding conditional power. The use of promising zone design could reduce the cost and duration of the trials by adaptively reducing the sample size when the interim analysis result is satisfied. While it could also increase the probability of success of the trials by increasing the sample size when the interim analysis result is not sufficient to support the desired result [1]. Provided a general scheme to determine the promising zone by preserving the overall Type-I error rate [2] proposed more specified methodology and algorithm for clinical studies with time-to-event endpoint based on optimizing the conditional power while control the budget limit and power requirement. However, the validation of the concept of optimal promising zone is not fully discussed. This study mainly focuses on the validation of this method and formula derivation of conditional power and sample size.
Introduction
Pre-study power analysis for sample size calculation (power calculation) is an integral part of randomized clinical trial. In clinical trials, based on the primary study endpoint, power calculations are typically performed following the steps:
i. It is to set up appropriate hypotheses for clinical investigation
ii. It is to derive an appropriate statistic under a valid study design and the null hypothesis to be tested
iii. It is to evaluate power function under the alternative hypothesis.
iv. It is to select an appropriate sample size by fixing all the parameters in the power function for achieving a desired power (e.g., 80%) at a pre-specified level of significance (e.g., 5%).
Thus, power calculation depends upon the study design, the study objective (or hypotheses), and the primary study endpoint of the intended clinical trial. In practice, commonly used study designs for clinical investigation include, but are not limited to, a crossover design, a parallel design, or a complex innovative design such as an adaptive trial design. The study objectives could be the demonstration of non-inferiority, superiority, or equivalence (e.g., therapeutic equivalence or bioequivalence). The data types of the commonly considered study endpoints could be continuous variable, binary response, or time-to-event data. More details regarding power calculation for testing non-inferiority hypothesis, superiority hypothesis, and equivalence hypothesis with various data types of study endpoints can be found in [3].
In clinical trials, interim analysis is often performed regardless the study design used. The purpose of interim analysis is multi-fold. First it is to verify the assumptions made for power calculation at the planning stage of the intended study. Second, it is to make sure the intended trial will achieve the desired power if the observed clinically meaningful difference (or treatment effect) preserves until end of the study. Thus, a blinded sample size re-estimation is often performed at interim. Third, if an adaptive trial design is used, some adaptations (modifications or changes) may be recommended after the review of interim data. For sample size re-estimation [3] pointed out that one could consider alternative methods in addition to the method of conditional power. These alternative methods include, but are not limited to
i. Maintain treatment effect
ii. Controlling variability
iii. Reaching a desired probability of reproducibility.
Maintaining treatment effect is to make sure that the test treatment under investigation is efficacious while controlling variability is for post-approval quality control and assurance. Reaching a desired probability of reproducibility is to make sure the observed positive result is not by chance alone and hence is reproducible.
In practice, it is desirable to maintain treatment effect and control variability at the same time when performing sample size calculation at the planning stage and/or sample size re-estimation sample size for achieving the objectives of maintaining treatment effect and/or controlling variability is studied under a promising zone design. In the next section, the concept of promising zone is briefly outlined. Moreover, the construction of Brownian Motion of the promising zone will also be demonstrated in Section 2. Under a promising zone design, formulas of conditional power, adjusted critical value, adjusted sample size are derived in Section 3. Section 4 summarizes the results of simulated studies with parameters from two real protocol under a promising zone design. Some concluding remarks are given in Section 5.
Statistical Assumptions
Concept of Promising Zone Design
In this article, the promising zone design is based on a two-stage and two-arm parallel clinical trial design with continuous endpoints. However, this methodology is surely applicable for multi-stage trials and trials with binary and time-to-event endpoints. The statistical assumptions mainly follow the article of [1]. This two-arm, two-stage trial is assumed to study the responses of participants to a newly developed compound. Suppose that all the participants entered the study at the beginning of stages and no subjects were recruited in the middle of the study. After the recruitment, participants were randomly assigned to the control arm and experimental arm and there is no drop-out in this ideal trial. The responses of individuals are considered as independent variables that identically followed a normal distribution. To start with a simple case, the variables of responses are assumed to follow a normal distribution with equal variance. To be more specific, let  and
and  be the response of ith subject in two arms where i denote the ith subjects. Then, a treatment effect difference can be defined as
be the response of ith subject in two arms where i denote the ith subjects. Then, a treatment effect difference can be defined as  . To check whether this difference is statistical significance, hypothesis testing for difference is applied with a null hypothesis
. To check whether this difference is statistical significance, hypothesis testing for difference is applied with a null hypothesis  and a one-sided alternative hypothesis
and a one-sided alternative hypothesis  .The reason why the one-sided alternative hypothesis is considered is that non-at interim. However, applying multiple tests in one clinical trial might inflate the overall type-I error. Chen, Lan and DeMets have showed that the overall type-I error will not be inflated if the sample size is modified only when the interim analysis is promising using conventional Wald test. Rather than using weighted test which has been shown by [4], using adjusted critical value in conventional test is also applicable. To find the explicit form of adjusted critical value and other variables, the connection between variable of interest and normal distribution must be constructed. The most feasible way is to use the properties of stochastic process and Brownian motion which is also the validation of using the promising zone in clinical trial. In this article, the problem of selecting an appropriate inferiority is the desired target. Since this trial is a two-stage design, an interim analysis is performed after data was recorded with
.The reason why the one-sided alternative hypothesis is considered is that non-at interim. However, applying multiple tests in one clinical trial might inflate the overall type-I error. Chen, Lan and DeMets have showed that the overall type-I error will not be inflated if the sample size is modified only when the interim analysis is promising using conventional Wald test. Rather than using weighted test which has been shown by [4], using adjusted critical value in conventional test is also applicable. To find the explicit form of adjusted critical value and other variables, the connection between variable of interest and normal distribution must be constructed. The most feasible way is to use the properties of stochastic process and Brownian motion which is also the validation of using the promising zone in clinical trial. In this article, the problem of selecting an appropriate inferiority is the desired target. Since this trial is a two-stage design, an interim analysis is performed after data was recorded with  subjects and a final analysis is performed after
subjects and a final analysis is performed after  response data is accumulated. Considering the α-level and predefined power
response data is accumulated. Considering the α-level and predefined power  , the planned total sample size can be derived as
, the planned total sample size can be derived as  where the
where the  is the Z-score and
is the Z-score and  is a pre-specified, clinical-meaningful treatment effect. And define the maximal sample size
is a pre-specified, clinical-meaningful treatment effect. And define the maximal sample size  as the upper bound of sample size limited by the budget and regulation of sponsor.
as the upper bound of sample size limited by the budget and regulation of sponsor.
In terms of interim analysis and final analysis, let  where j denotes
where j denotes  stage,
stage, and
and Consequently, the mean of treatment difference between two groups
Consequently, the mean of treatment difference between two groups is also normally distributed and
is also normally distributed and where
where is the sample of data accumulated at Jthstage. Then the maximum likelihood estimates of treatment effect difference at two stages are
is the sample of data accumulated at Jthstage. Then the maximum likelihood estimates of treatment effect difference at two stages are and variance of
and variance of is var
is var  Considering a Wald statistic at jth stage denoted as
Considering a Wald statistic at jth stage denoted as .
.
The null hypothesis is rejected when  to assure the hypotheses testing reaches the α-level. And whether the interim analysis result is promising is defined by the power of hypotheses testing condition on the interim statistic W1 and the maximum loglikelihood estimator of treatment effect at stage one
to assure the hypotheses testing reaches the α-level. And whether the interim analysis result is promising is defined by the power of hypotheses testing condition on the interim statistic W1 and the maximum loglikelihood estimator of treatment effect at stage one 

One important requirement of clinical trials with multiple stages is that the overall type-I error should not be inflated. Small conditional power in the interim analysis will lead to an inflated overall type-I error since there are derived under the opposite hypothesis. On the contrary, too large conditional power, saying  will lead to the termination of the trials by efficacy. Thus,the sample size can only be increased when the conditional power is promising which means CP should fall in a certain interval. The detailed definition of promising zone design will be given next.
will lead to the termination of the trials by efficacy. Thus,the sample size can only be increased when the conditional power is promising which means CP should fall in a certain interval. The detailed definition of promising zone design will be given next.
Concept of Promising Zone Design
The collection of interim analysis test statistics  and corresponding conditional power
and corresponding conditional power  will be divided into three intervals. Within each interval, the way to modify sample size are different.
will be divided into three intervals. Within each interval, the way to modify sample size are different.
Unfavorable Zone
For the unfavorable zone which is defined by  where
where  should be specified before the beginning of the trial, conditional power in this interval is too low to meet the request of power. Continuing the trial might even lead to failure since the final power might be lower than the requirement by increasing the sample size from
should be specified before the beginning of the trial, conditional power in this interval is too low to meet the request of power. Continuing the trial might even lead to failure since the final power might be lower than the requirement by increasing the sample size from  to
to  . The trial could be terminated early for futility.
. The trial could be terminated early for futility.
Favorable Zone
For the favorable zone which is defined by  where
where  usually defined as 1-β, the conditional power in this interval indicates the contemporary trial result is so good that to meet the power request and there is no need to increase the sample size to the planned
usually defined as 1-β, the conditional power in this interval indicates the contemporary trial result is so good that to meet the power request and there is no need to increase the sample size to the planned  . The trial could proceed with the current sample size
. The trial could proceed with the current sample size  or be terminated for efficacy.
or be terminated for efficacy.
Promising zone
For the promising zone which is defined by  , the conditional power in this interval cannot lead to neither early futility termination nor efficacy termination of the trial. The trial needs to proceed to large sample size to detect sufficient evidence. However, with the conditional power and statistic derived in the interim analysis, the increased sample size could be adjusted to a new value
, the conditional power in this interval cannot lead to neither early futility termination nor efficacy termination of the trial. The trial needs to proceed to large sample size to detect sufficient evidence. However, with the conditional power and statistic derived in the interim analysis, the increased sample size could be adjusted to a new value  rather than n2 to avoid the futility or exceeding the budget. Since
rather than n2 to avoid the futility or exceeding the budget. Since  usually defined by the overall power requirement 1-β, it is more important to determine the lower bound of the interval. In this article, the works are mainly focus on determine the lower bound.
usually defined by the overall power requirement 1-β, it is more important to determine the lower bound of the interval. In this article, the works are mainly focus on determine the lower bound.
Construction of Brownian Motion
To get the explicit formula of conditional power, adjusted critical value and adjusted sample size, the hypothesis tests in promising zone design need to be justified to follow the properties of Brownian Motion. To construct the Brownian Motion, the concept of information will be introduced. This section is mainly developed from [5]. Statistically speaking, the progressing of clinical trial is measured better by the accumulated information rather than the participants enrolled. Considering the estimation of treatment effect in each stage , the information of
, the information of  define as
define as  . Next, the construction of Brownian Motion will be demonstrated. Considering in a clinical trial with k stages, then there will be k estimations of treatment effect
. Next, the construction of Brownian Motion will be demonstrated. Considering in a clinical trial with k stages, then there will be k estimations of treatment effect  . It can be shown that the sequence of
. It can be shown that the sequence of  satisfies three properties of stochastics process:
satisfies three properties of stochastics process:

 and the result of Central [5] Limit Theorem (when k is large enought), the sequence of estimations
and the result of Central [5] Limit Theorem (when k is large enought), the sequence of estimations can be approximate by Brownian Motion. Applying the general statement in the two-stage scenario, the mean treatment difference between two group,
can be approximate by Brownian Motion. Applying the general statement in the two-stage scenario, the mean treatment difference between two group, and
and are two components of a sequence of Brownian Motion, where
are two components of a sequence of Brownian Motion, where is the treatment difference detected with a modified sample size
is the treatment difference detected with a modified sample size [6] The variance here takes the fisher information that leads to
[6] The variance here takes the fisher information that leads to and
and can be easily justified by using the properties of Brownian Motion. On the contrary, under the alternative hypothesis,
can be easily justified by using the properties of Brownian Motion. On the contrary, under the alternative hypothesis, it gives
it gives .However, since
.However, since the difference of these two variables still has the Markov Properties,
the difference of these two variables still has the Markov Properties, and
and . The two different construction of variables following Brownian Properties will be used to separately derive the formula of type-I error and conditional power.
. The two different construction of variables following Brownian Properties will be used to separately derive the formula of type-I error and conditional power.
Proposed Method
Determine the Promising Zone by Controlling the Type-I Error Rate.
To preserve the overall type one error, the critical value of final analysis needs to be adjusted [6], the adjusted type-I error can be derived as follows,
Where  is the treatment effect under the null hypothesis,
is the treatment effect under the null hypothesis,  is the statistic in the final analysis with adjusted sample size
is the statistic in the final analysis with adjusted sample size  and the adjusted critical value
and the adjusted critical value  . More specifically, the left-hand side of the equation is
. More specifically, the left-hand side of the equation is


By the null hypothesis, 0δ=, by the assumption of Brownian Motion,W1

By the similar calculation,

By the identity of these two type-I error, the adjusted critical value can be derived as

By the properties of Brownian Motion under the alternative hypothesis, the conditional power can be presented as

By the similar calculation, the conditional power with adjusted sample size can be derived as follows:

Substituting the adjusted critical value in the formula of conditional power, the formula can be simplified as:

Set the conditional power with modified sample size  equals to 1-β, the modified sample size can be derived. It can be shown as
equals to 1-β, the modified sample size can be derived. It can be shown as

Moreover, the budget limit of the trial also needs to be considered in the design stage. Define the finally modified sample size with consideration of where
where
Determine the promising zone by constraints
Proposed one [2] new method to determine the promising zone of the conditional power. This method is like the method of controlling type-I error but easier to be conducted by investigators. The algorithm is given as following.

By maximizing the objective function, an interval of interim analysis can be derived denoted as  . Furthermore,
considering the scenario when
. Furthermore,
considering the scenario when  , sinceCPis determined by both
, sinceCPis determined by both  . In this case, there is need to increase the sample size anymore due to the consideration of trial budget limitation. With the increment of
. In this case, there is need to increase the sample size anymore due to the consideration of trial budget limitation. With the increment of  , to remain
, to remain  , the sample size should be reduced. Define the smallest interim analysis statistic
, the sample size should be reduced. Define the smallest interim analysis statistic  . And the algorithm for sample size is defined as following:
. And the algorithm for sample size is defined as following:
 By the formula of sample size and conditional power
By the formula of sample size and conditional power
Simulated Study
In this section, two simulated studies with parameters from real clinical trial protocol will be introduced. The first examples utilize parameters of a clinical trial of pancreatic cancer introduced by Hsiao [2]. The trial is a two-group placebo-control design. Participants were randomly assigned to treatment group using a recombinant human hyaluronidase and a concurrent placebo control group. The primary endpoint progression-free survival. Several literatures have shown that with the consideration of the uncertainty of parameters, the trial had to show desired power of hazard ratio in the range of [0,67,0.75]. Here, the simulated study will focus on the minimal treatment different  . By the requirement of sponsor, the
. By the requirement of sponsor, the  ,
, ,
,  ,
, ,
, ,
,  and
and  . In the simulated study, the relationship of modified sample size, modified power, original power, power increment, the probability of falling in the promising zone will be analyzed to evaluate the feasible and effect of promising zone design.
. In the simulated study, the relationship of modified sample size, modified power, original power, power increment, the probability of falling in the promising zone will be analyzed to evaluate the feasible and effect of promising zone design.
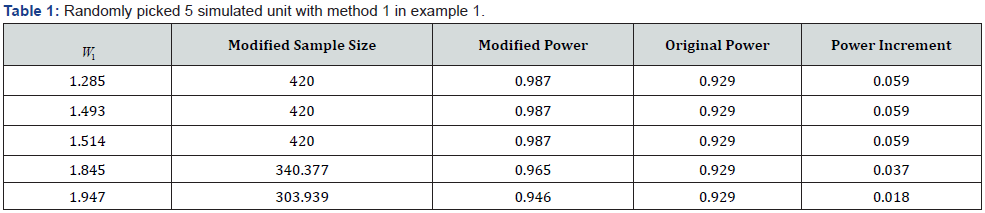
By method 1, determining the promising zone by preserving type-I error, the interim test statistic  and the promising zone is
and the promising zone is  . With such distribution and promising zone, the probability of falling in the promising zone is
. With such distribution and promising zone, the probability of falling in the promising zone is  . And the table with 5 randomly selected levels of the promising zone is shown in (Table 1) below. By the method 2, the promising zone in this example is
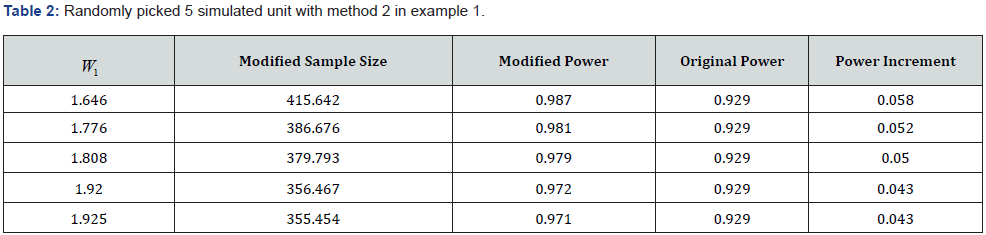
. And the table with 5 randomly selected levels of the promising zone is shown in (Table 1) below. By the method 2, the promising zone in this example is  . The table with 5 randomly picked W_1 presenting relationships between sample size,
. The table with 5 randomly picked W_1 presenting relationships between sample size, and CP is shown in (Table 2) below. The second example comes from a phase 3 Schizophrenia trial. By [7], participants with negative symptoms schizophrenia are randomized into two groups, one with the test drug and one with an active control. The primary endpoint is the increment of standardized score in the Negative Symptoms Assessment from the baseline to week 26. The planned trial size,
and CP is shown in (Table 2) below. The second example comes from a phase 3 Schizophrenia trial. By [7], participants with negative symptoms schizophrenia are randomized into two groups, one with the test drug and one with an active control. The primary endpoint is the increment of standardized score in the Negative Symptoms Assessment from the baseline to week 26. The planned trial size,  , was calculated based on 80% power to detect a mean score difference δ=2 with a one-sided level-0.025 test, assuming a score standard deviation σ = 7.5 in each treatment group.
, was calculated based on 80% power to detect a mean score difference δ=2 with a one-sided level-0.025 test, assuming a score standard deviation σ = 7.5 in each treatment group.  and the interim analysis will be conducted after 208 participants have been treated.
and the interim analysis will be conducted after 208 participants have been treated.
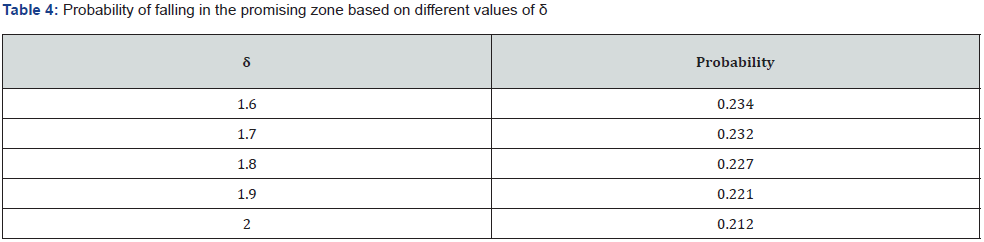
By the method 1, the promising zone is  . And a table with randomly picked 5 values are shown in (Table 3). In this example, the probability of falling in promising zone based on different values of δ are shown in the (Table 4). By the CPZ Design method, the promising zone is
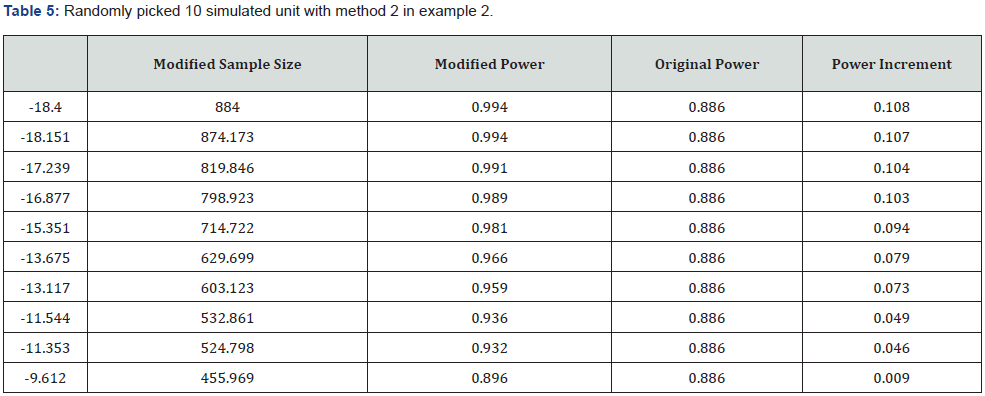
. And a table with randomly picked 5 values are shown in (Table 3). In this example, the probability of falling in promising zone based on different values of δ are shown in the (Table 4). By the CPZ Design method, the promising zone is  .And the table with 10 randomly selected
.And the table with 10 randomly selected  and corresponding sample size, power is shown in the (Table 5).
and corresponding sample size, power is shown in the (Table 5).

Discussion
Feasibility of promising zone design
From the two examples of [8] simulated studies, we can see that the power increment in the first example is relatively small. And with the increase of interim analysis, the power increment drops. Furthermore, the probability of falling into the promising zone remains lower than 30%. Whether it is necessary to conduct an extra interim analysis remains to be discussed. And it needs to be treated separately by different trials and different parameters.
Drawbacks of Promising Zone Design
As with all designs with early determination, promising zone lost chances to detect further information. The most one among all information is the potential adverse events. Since promising zone design is aiming to increase the probability of success of the trial at phase III and toxicity [9] analysis is usually applied at phase I, it is not an obstacle that hardly stops investigators from using promising zone design.
Particularly for the method of determining the promising zone by preserving the type-I error, the formula gives identity conditional power regardless of the treatment effect. This identity is caused by the construction of the Brownian Motion. However, whether it will still show this identity when using different tests and constructing the Brownian Motion by other methods remains discussion. In terms of the constraint optimization method, there is no modification in critical values which means there is no constraint for type-I error. The type-I error might not be preserved under some circumstances. Further research could focus on the combination of the two methods above.
References
- Mehta Cyrus, Pocock Stuart (2011) Adaptive Increase in Sample Size When Interim Results are Promising: A Practical Guide with Examples. Statistics in med 30(28): 3267-3284.
- Hsiao S T, Liu L, Mehta C R (2019) Optimal promising zone designs. Biometrical journal. Bio Zeitschrift 61(5): 1175-1186.
- Chow S C, Shao J, Wang H, Lokhnygina Y (2017) Sample Size Calculation in Clinical Research. Third Edition, Taylor & Francis, New York, USA.
- Cui L, Hung H J, Wang S J (1999) Modification of sample size in group sequential clinical trials. Biometrics 55(3): 853-857.
- Lan K G, Zucker D M (1993) Sequential monitoring of clinical trials: the role of information and Brownian motion. Stat in Med 12(8): 753-765.
- Mörters P, Peres Y (2010) Brownian motion (Vol 30) Cambridge University Press UK.
- Gao P, Ware J H, Mehta C (2008) Sample size re-estimation for adaptive sequential design in clinical trials. J Bio Stat 18(6): 1184-1196.
- Jennison C, Turnbull B W (1999) Group sequential methods with applications to clinical trials CRC Press USA.
- Whitehead J (1997) The design and analysis of sequential clinical trials. John Wiley & Sons, USA.






























