Advanced Biomedical Devices Facilitated by Plasma Deposited Polyoxazoline Coatings
K Vasilev* and M Macgregor*
School of Engineering, Future Industries Institute, Australia
Submission: June 13, 2017; Published: July 24, 2017
*Corresponding author: K Vasilev and M Macgregor , School of Engineering, Future Industries Institute, University of South Australia, Mawson Lakes SA 5095 Australia, Email: Krasimir.vasilev@unisa.edu.au; Melanie.MacGregor@unisa.edu.au
How to cite this article: K Vasilev, M Macgregor. Advanced Biomedical Devices Facilitated by Plasma Deposited Polyoxazoline Coatings. Biostat Biometrics Open Acc J. 2017;2(3): 555586. DOI: 10.19080/BBOAJ.2017.02.555586.
Abstract
The development of suitable biomaterials is key to the progress of biomedical engineering. Plasma deposition is a versatile and waste free technique that can be used to produce surfaces with well denied chemical and physical properties. In this mini-review we show how well-char-acterised plasma deposited organic films have been used to screen and identify optimum substrate properties for several different biomedical applications. By providing as an example the plasma deposited poly oxazolines, we demonstrate the generation of advances biomaterial surfaces capable of limiting biofilm formation, guiding stem cell differentiation and even sensing cancer cells in urine.
Introduction
A large number of the recent improvements to healthcare services worldwide are relying on progress made in biomedical engineering. This growing field of research invaluably increase our quality of life by providing advanced products such as biocompatible prostheses, diagnostic devices, or targeted drug therapies. While generating such technologies is an inter disciplinary exercise in nature, the communality to all the example cited above is that biological entities, such as cells and proteins, do get in contact with a solid substrate and interact with it. This is why, in essence, the development of biotechnologies heavily relies on the properties of the biomaterial present at the interface with the biological system. In fact, it is well accepted that a surface chemical and physical properties play essential roles in dictating how cells and biomolecules interact with it. Though, the mechanisms by which surface cues govern the behaviour of cells, for instance, remain a fervent field of investigation. Advances in this field is somewhat hindered by the lack of model substrates capable to support large scale systematic studies in which single parameters are varied one at a time, to support robust statistical analysis of the biological behaviours. This is because, in most manufacturing processes, varying the topography of a biocompatible material; or its stiffness, porosity, or even transparency; is accompanied by an unavoidable and uncontrollable change in surface chemistry. A solution to these challenges can be the use of plasma deposition.
The deposition of nanothin organic coatings using plasma is a versatile and scalable approach to control the chemical composition and nano topography of the outermost layer of virtually any type of material [1-3]. Plasma polymers can be deposited from a variety of organic precursors and the deposition conditions can be varied in many ways so that chemistry and functionality of the resulting coatings can be finely tailored. Allylalcohol, allyl glycidyl ether, propanal, and allylamine are examples of commonly used organic precursor volatile enough to be introduced into a low vacuum chamber to produce thin polymeric films from their plasma phase. Plasma deposition also has the advantage to be a rapid “one-step” and waste free process. It is therefore particularly attractive for high throughput developmental studies involving cells, proteins, antibodies, bacteria, viruses, fungi and other bio-entities which typically require multiple biological and technological replicates as well as the screening of many (un)correlated parameters [4].
In the following, we review the particular case of organic films deposited from the plasma phase of alkyl-oxazolines. These new coatings have been used to speed up systematic fundamental research on
a. A selective cell capture platform.
b. Low fouling substrates and
c. Stem cell guidance surfaces.
The tangible outcome of these 3 research activities is the development of diagnostic devices, implant coating, and tissue engineering substrates, respectively.
Polyxazoline
Polyoxazolines (POx) are a class of biocompatible polymers under fervent investigation in the biomedical field as a possible alternative to polyethylene glycol [5] for drug delivery, tissue engineering and medical implant purposes [6,7]. Their conventional synthesis via ring opening polymerization results in a characteristic structure consisting of repetitive units of amide functions which presumably confer POx stealth properties [7]. However, generating POx thin films via wet organic chemistry is a delicate, mutli-step process which must be conducted under strict experimental conditions. These challenges have been circumvented by our group by discovering the polyoxazoline coating via plasma deposition (PPOx) [8]. As pioneers in the field we investigated the properties and functionality of the plasma PPOx film over a range of depositions conditions. The plasma ignition power, working pressure and deposition time were varied for methyl, ethyl and isopropyl oxazoline precurors [9,10]. The surface chemistry, wettability and stability of the PPOx films were systematically analysed by water contact angle measurement, FTIR, XPS, ToF SIMS, and ellipsometry. Under all deposition conditions, the chemical analysis demonstrated a rich film chemistry with signals undeniably corresponding to amine, amide, carbonyl, carboxyl and nitrile functions, as well as intact oxazoline rings. However, the relative concentration of the surface chemical groups, as well as the film wettability and stability vary with plasma power and precursor flow rate, as described in details previously [9,10]. For instance, the hydrophilicity of the films increases at lower plasma deposition powers, while surface functionality decreases with decreasing precursor flow rate (Figure 1).
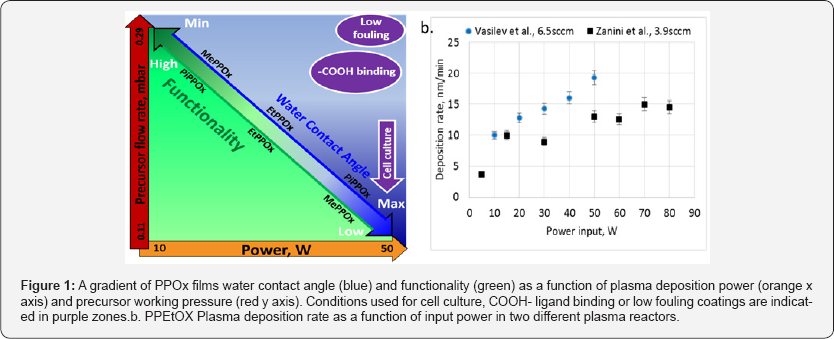
Around the same time, a study of thin films deposited from ethyl oxazoline in pulsed plasma mode by Bhatt et al reported comparable film chemistry [11]. However, the films reported by Bhatt et al. were deposited using lower plasma powers and pulsed cycles which led to non-negligible thickness loss after prolonged exposure to aqueous solvents. In contrast, the films we deposited in continuous wave mode at powers higher than 30W are stable in a range of solvent including acid, base, salt solution but also body fluids such as urine [10]. Later, Zanini et al. [12] also used low pressure plasma deposition to create polyethyloxazoline films using different powers and deposition time [12]. While the deposition conditions described in this work appear comparable to those used in our study, different plasma reactors were used, and so direct comparison between quantitative data may not be relevant.
For instance, in (Figure 1b) we plotted the deposition rates obtain by. Zanini et al. [12] for PEtOX deposited with increasing input powers together with those corresponding to our experimental conditions. The deposition rates are of the same order of magnitude despite differences in the precursor deposition flow rates. Most importantly, using a different set of analytical techniques to characterise the PPE tOX film chemistry and reactivity, namely NMR, profilometry, ATR/FTIR, and fluorescent confocal microscopy. Zanini et al. [12] confirmed our group's main original finding, specifically the partial retention of unopened oxazoline rings. Since conventional films are inherently created by ring opening polymerisation, retaining the oxazoline ring within the thin film is unique to plasma deposited PPOx and reproducible by different laboratories. This distinctive functionality is very valuable because the ring readily engage in covalent bonds with carboxylic acid groups which are present in biomolecules, and other ligands as demonstrated previously [10]. The reactivity of retained oxazoline ring with -COOH function was also confirmed in, Zanini et al. [12] study. Furthermore, they used NMR to estimate the typical amount of oxazoline ring retention and the degree of linear open ring structure in PPOx surfaces. For a PEtOX film deposited at 15 W they found up to 20% ring retention, while the remaining of the surface chemistry resulted from more complex plasma fragmentation and recombination processes. In good agreement with our initial analyses, these results are particularly interesting because they can be used to explain the unique properties of PPOx field for biomedical application. We were indeed able to create a matrix of the physico-chemical properties of PPOx as a function of the plasma deposition conditions that we used to design PPOx films tailored for cell guidance surfaces, biosensors and low fouling substrates as explained below.
Cell Guidance Surfaces
It is well accepted that cell adhesion to biomaterials rely on the initial formation of an adsorbed protein layer [13]. As they come in contact with the protein film, integrin receptors on the cells membrane recognise favourable binding sites which initiate the adhesion process. It is therefore essential for biomaterials to promote the adsorption of bioactive proteins. A surface capability to bind proteins is dictated by its wettability [14] charge [15,16] and polarity, all of which are inherent to the surface chemistry [17,18]. Plasma polymer deposition is a resourceful approach to modify a substrate chemical functionality due to the wide range of organic precursors that can be used to produce surfaces facilitating protein binding [19]. Oxygen-rich and nitrogen-rich plasma based surfaces have been particularly studied for their applications as biomaterials [20]. Oxygen rich chemistry are known to promote cellular attachment because they contain polar groups such as hydroxyls, [21] carboxyls [22,23] and carbonyls [24], which enter ionic interaction with cell adhesion mediating molecules. Amine rich substrates are also well amine monomers such as ethylendiamine [27], propylamine [28], butlyamine [29] and heptylamine [30] have also been used. The electrostatic adsorption of negatively charged proteins with amine known for enhancing cell growth and the most common plasma precursor used to confer amine functionalities to biomaterials is allylamine [25,26] Other volatile amine monomers such as ethylendiamine [27], propylamine [28], butlyamine [29] and heptylamine [30] have also been used. The electrostatic adsorption of negatively charged proteins with amine (NH2) groups which are positively charged in culture condition, is thought to confer the amine rich surfaces their biocompatiblity. Both oxygen and amine rich substrates can also form covalent bonds with carboxyl functions through the use of linking agents (e.g. carbodiimine, NHS, trifluoroacteic anhydride).
Plasma deposited polyoxazoline are both oxygen and nitrogen rich and we showed that PPOx deposited at high power (50W) with low precursor flow rate promoted the adhesion of human dermal fibroblast, [31] mesechemial stem cells and also kidney stem cells. Surface chemistry analysis indicated that oxygen and nitrogen are, in a PPOx film, engaged in many hydrophilic, H-donor groups which may explain their good ability to support mammalian cell growth.
The unique reactivity of PPOx films with -COOH chemical groups, enhanced when deposited at high power and moderate flow rate, has also been used to create surfaces with controlled nano topography by covalent binding of COOH- functionalised nano particles. In a recent study, we created in this way gradient of gold nano particles that were used to evaluate the influence of nano topography on the differentiation of kidney stem cells. We found that the stem cells preferentially differentiated into podocyte cell or the rougher part of the gradient while they were more inclined to differentiate into proximal tubule cells on smooth surfaces [32].
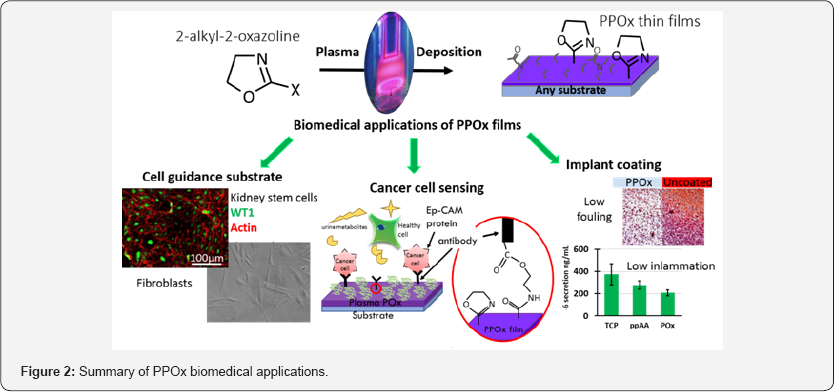
Diagnostic Devices
Additionally, PPOx ability to spontaneously form covalent bonds with the carboxylic acid groups present in biomolecules was used to generate immuno functionalised surface for the selective capture of cancer cells from urine [33]. Anti-epithelial cell adhesion molecules antibodies were covalently bound to PPOx substrate. Using PPOx to immobilise antibodies for diagnostic purposes is very useful because the strong covalent bond between the substrate and the sensing biomolecule is not disrupted by physiological variations in the composition, pH, and ionic strength of real body fluids such as urine [34].The biosensors developed in this way were able to detect cancer cells in model urine and also in real patient urine samples. The outcomes of this work are currently being commercialised (Figure 2).
Low Fouling Substrates
In another systematic study we investigated the fate of S. Epidermis on PPOx films across a range of deposition conditions [35]. The result of this work show that the most reactive PPOx films deposited at high power with high monomer flow rate were effectively hindering the formation of biofilms. This property of PPOx film is interesting for their use as low fouling substrates for implant coating. For this reason we also interrogated the response of immune cells to PPOx films. In this investigation, cytokine secretion from bone marrow-derived primary macrophages (BMDM) was measured in vitro. BMDM were selected as model immune cells because their function is to mediate early innate immune inflammatory responses. Compared to another nitrogen-rich plasma polymer and tissue culture plates, a marked reduction in the expression of TFN-a and IL-6 cytokine was observed on the PPOx substrates. Together, these results indicate that PPOx films could benefit several device types such as prosthesis, catheter, or even wound dressings.
Conclusion
Overall, plasma deposited thin films, and more specifically PPOx films present many advantages for the rapid development of biomedical products. While a thorough understanding of the surface physical and chemical properties as a function of the deposition conditions is a pre-requisite, it is possible to rapidly produce tailored substrates for high throughput bioanalysis and coatings for a wide range of medical devices covering areas from implants and tissue engineering constructs to diagnostics.
References
- Vasilev K, Michelmore A, Martinek P, Chan J, Sah V, et al. (2010) Early stages of growth of plasma polymer coatings deposited from nitrogen- and oxygen-containing monomers. Plasma Processes and Polymers 7(9-10): 824-835.
- Michelmore A, Martinek P, Sah V, Short RD, Vasilev K (2011) Surface morphology in the early stages of plasma polymer film growth from amine-containing monomers. Plasma Processes and Polymers 8(5): 367-372.
- Vasilev K, Michelmore A, Griesser HJ, Short RD (2009) Substrate influence on the initial growth phase of plasma-deposited polymer films. Chem Commun (Camb) 24: 3600-3602.
- Delalat B, Rojas-Canales DM, Rasi Ghaemi S, Waibel M, Harding FJ, et al. (2016) A combinatorial protein microarray for probing materials interaction with pancreatic islet cell populations. Microarrays (Basel) 5(3): E21.
- Hoogenboom R (2009) Poly(2-oxazoline)s: a polymer class with numerous potential applications. Angew Chem Int Ed Engl 48(43): 7978-7994.
- Knop K, Hoogenboom R, Fischer D, Schubert US (2010) Poly(ethylene glycol) in Drug Delivery: Pros and Cons as Well as Potential Alternatives. Angew Chem Int Ed Engl 49(36): 6288-6308.
- Chapman RG, Emanuele Ostuni, Takayama S, Holmlin RE, Yan L, et al. (2000)_ Surveying for Surfaces that Resist the Adsorption of Proteins. Journal of the American Chemical Society 122(34): 8303-8304.
- Ramiasa MN, Cavallaro AA, Mierczynska A, Christo SN, Gleadle CJ, et al. (2015) Plasma polymerised polyoxazoline thin films for biomedical applications. Chemical Communications 51: 4279-4282.
- MacGregor Ramiasa MN, Cavallaro A, Visalakshan RM, Gonzalez L, Vasilev K (2016) Plasma deposited Polyoxazoline coatings, a versatile new class of biomaterials, in CHEMECA 2016: Chemical Engineering - Regeneration, Recovery and Reinvention,Engineers Australia: Adelaide 302-312.
- Macgregor-Ramiasa MN, Cavallaro AA, Vasilev K (2015) Properties and reactivity of polyoxazoline plasma polymer films. Journal of Materials Chemistry B 3(30): 6327-6337.
- Bhatt S, Pulpytel J, Mirshahi M, Khonsari FA (2015) Cell Resistant Peptidomimetic Poly (2-ethyl-2-oxazoline) Coatings Developed by Low Pressure Inductively Excited Pulsed Plasma Polymerization for Biomedical Purpose. Plasma Processes and Polymers 12(6): 519-532.
- Zanini S, LucaZoia, Dell'Orto EC, Natalello A, Villa AM (2016) Plasma polymerized 2-ethyl-2-oxazoline: Chemical characterization and study of the reactivity towards different chemical groups. Materials & Design 108: 791-800.
- Ruoslahti E, Pierschbacher MD (1987) New perspectives in cell adhesion: RGD and integrins. Science 238(4826): 491-497.
- van Wachem PB, Beugeling T, Feijen J, Bantjes A, Detmers JP, et al. (1985) Interaction of cultured human endothelial cells with polymeric surfaces of different wettabilities. Biomaterials 6(6): 403-408.
- Pasche S, Voros J, Griesser HJ, Spencer ND, Textor M (2005) Effects of Ionic Strength and Surface Charge on Protein Adsorption at PEGylated Surfaces. J Phys Chem B 109(37): 17545-17552.
- Hallab NJ, Bundy KJ, O'Connor K, Clark R, Moses RL (1994) Cell adhesion to biomaterials: correlations between surface charge, surface roughness, adsorbed protein, and cell morphology. J Long Term Eff Med Implants 5(3): 209-231.
- Arima Y, Iwata H (2007) Effect of wettability and surface functional groups on protein adsorption and cell adhesion using well-defined mixed self-assembled monolayers Biomaterials 28(20): 3074-3082.
- Gray J J (2004) The interaction of proteins with solid surfaces. Curr Opin Struct Biol 14(1): 110-115.
- Coad BR (2013)Controlled covalent surface immobilisation of proteins and peptides using plasma methods. Surface and Coatings Technology 233(0): 169-177.
- Ratner BD (1993) Plasma deposition for biomedical applications: J Biomater Sci Polym Ed 4(1): 3-11.
- Lee J H, Park JW, Lee HB (1991) Cell adhesion and growth on polymer surfaces with hydroxyl groups prepared by water vapour plasma treatment. Biomaterials 12(5): 443-448.?
- Francesch L, Garreta E, Balcells M, Edelman ER, Borros S (2005) Fabrication of Bioactive Surfaces by Plasma Polymerization Techniques Using a Novel Acrylate-Derived Monomer. Plasma Processes and Polymers 2(8): 605-611.
- Detomaso L (2005) Plasma deposited acrylic acid coatings: Surface characterization and attachment of 3T3 murine fibroblast cell lines. Surface and Coatings Technology 200(1-4): 1022-1025.
- Ertel SI, Chilkoti A, Horbett TA, Ratner BD (1992) Endothelial cell growth on oxygen-containing films deposited by radio-frequency plasmas: the role of surface carbonyl groups. J Biomater Sci Polym Ed 3(2): 163-183.
- Shard AG, Whittle JD , Beck AJ , Brookes PN, Bullett NA (2004) A NEXAFS examination of unsaturation in plasma polymers of allylamine and propylamine . The Journal of Physical Chemistry B 108(33): 12472-12480.
- Andrew HL, Helmut T, Jamie Q, Nicolas VH (2008) Comparison of the binding mode of plasmid DNA to allylamine plasma polymer and poly (ethylene glycol) surfaces. Surface Science 602(10): 1883-1891.
- Choukourov A, Birderman H, Kholodkov I, Slavinska D, Trchova M (2004) et al. Properties of amine-containing coatings prepared by plasma polymerization. Journal of applied polymer science 92(2): 979-990.
- Fally F, Doneux C, Riga J, Verbist JJ (1995) Quantification of the functional groups present at the surface of plasma polymers deposited from propylamine, allylamine, and propargylamine. Journal of applied polymer science 56(5): 597-614.
- Gancarz I, Bryjak J, Pozniak G, Tylus W (2003) Plasma modified polymers as a support for enzyme immobilization II Amines plasma. European polymer journal 39(11): 2217-2224.
- Gengenbach TR, Chatelier RC, Griesser HJ (1996) Characterization of the Ageing of Plasma-deposited Polymer Films: Global Analysis of X-ray Photoelectron Spectroscopy Data. Surface and Interface Analysis 24(4): 271-281.
- Visalakshan RM, MacGregor-Ramiasa ACM , Hayball J, Vasilev K (2016) Downstream influences of oxazoline plasma polymerisation conditions on chemical and biological interactions, in Proceeding of CHEMECA 2016:Chemical Engineering - Regeneration, Recovery and Reinvention Engineers Australia pp: 840-848.
- MacGregor-Ramiasa, Hopp I, Bachhuk A, Murray, Vasilev K (2017) Surface nanotopography guides kidney-derived stem cell differentiation into podocytes. Acta Biomaterialia 56: 171-180.
- Macgregor-Ramiasa M, McNicholas K, Ostrikov K, Li J, Michael M, et al. (2017) A platform for selective immuno-capture of cancer cells from urine. Biosens Bioelectron 96: 373-380.
- Ostrikov K, MacGregor-Ramiasa MN, Vasilev K (2016) Biomaterial Platform for Cancer Diagnostic from Patient Urine in Proceeding of CHEMECA 2016:Chemical Engineering - Regeneration, Recovery and Reinvention. Engineers Australia: Adelaide.
- Cavallaro A A, Macgregor-Ramiasa MN, Vasilev K (2016) Antibiofouling Properties of Plasma-Deposited Oxazoline-Based Thin Films. ACS Applied Materials & Interfaces 8(10): 6354-6362.






























