Abstract
A novel model of auxiliary blood circulation generated by a disc. pump was investigated using computational fluid dynamics, as well as in vitro and in vivo experiments. The structure of the developed model allowed to reproduce the physiology of natural blood flow, thereby minimizing blood cell trauma, ensuring adequate hemodynamics, and preserving homeostasis in the experimental animals.
According to the results of numerical modeling, in the selected model of auxiliary blood circulation the shear stress values did not exceed 70 Pa, which is significantly lower than the threshold level of 100 Pa and correlates with the maximum permissible hemolysis level of 0.2 g/L. The results of the physical stand experiments demonstrated complete agreement between the hydrodynamic parameters and those calculated using computational fluid dynamics methods. Acute experiments on mini-pigs confirmed the possibility of creating adequate hemodynamic conditions and maintaining hemolysis parameters in vivo, demonstrating the potential for clinical application of the new auxiliary blood circulation model.
Keywords:Computational fluid dynamics methods; disc. Pump; Blood circulation model; Hemolysis; Shear stress; Hemodynamic parameters; Experiments
Introduction
According to generally accepted approaches to modeling physiological systems, the choice of model architecture and the composition of its key parameters directly depend on the objectives established during its development [1–3]. The key requirements for such a model of the auxiliary blood circulation system are its safety for both the blood system and the organism as a whole. At the same time, the auxiliary circulation model should reproduce the natural physiology of blood flow. In this regard, numerical modeling of the physical processes of blood flow in vessels with complex vascular geometry is most effectively performed using computational fluid dynamics (CFD) methods [4], which make it possible to identify areas of turbulent flow and fluid stagnation, as well as vortical structures within the model. When modeling blood flow, the k–ε and k–ω turbulence models are primarily used [5]. The advantage of the k–ε model lies in its improved prediction of the propagation velocity of planar and round jets, as well as in its ability to evaluate flow characteristics in rotating flows with strong adverse pressure gradients, which is critically important for predicting hemolysis through the determination of shear stress levels. The k–ω model is applicable mainly for describing turbulence in the boundary layer. Therefore, the k–ε turbulence model was applied in our study.
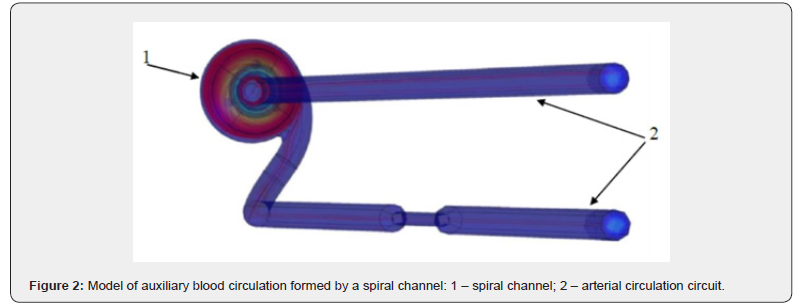
Hydrodynamic calculations form the basis of CFD and include solutions to the differential Navier–Stokes equations (equations describing the conservation of mass, momentum, and energy of the medium in a nonstationary formulation) with specified boundary and initial conditions. Verification of the parameters of the blood flow model is typically carried out by correlating the data obtained in modeling studies with the results of in vitro and in vivo experiments. If the disc..repancy between the modeling and experimental studies does not exceed 12%, the results are considered reliable [1]. A left ventricular bypass disc. pump [6– 7], which represents a spiral-shaped channel, was investigated using CFD and in vivo experiments (Figure 1). According to the hypothesis formulated at the initial stages of the study, the spiral channel would make it possible to reproduce the physiology of natural blood flow in the cardiovascular system, minimize blood cell injury, and improve hemodynamic parameters compared with existing systems. In the presented model, the spiral channel ceases to function merely as a mechanical component of the auxiliary blood circulation circuit and instead becomes an extension of the cardiovascular system (Figure 2). To assess the effectiveness and safety of the developed model, the following criteria were established: absence of stagnant zones and turbulent flows; shear stress should not exceed 100 Pa, since higher values lead to hemolysis in large vessels [8]; a cardiac output of 4.5–5.0 L/ min; and an outlet pressure within the range of 100–130 mmHg. Purpose of the study. To evaluate the degree of influence of the developed auxiliary blood circulation model, generated by a disc. pump, on blood system parameters and hemodynamics in in vitro and in vivo experiments.
Materials and Methods
Computational simulations were performed using COMSOL Multiphysics software. The geometry of the spiral channel was imported into the COMSOL Multiphysics environment, input parameters were defined, and a computational mesh was generated.
The computational study included:
i. assessment of flow characteristics and flow distribution
within the auxiliary circulation model;
ii. identification of the safest auxiliary blood circulation
model;
iii. determination of shear stress levels.
Shear stresses were calculated using the Navier–Stokes system of equations, a well-known class of nonlinear partial differential equations describing the motion of liquid and gaseous media in the presence of viscosity [9].
The Navier–Stokes equations can be expressed as follows:
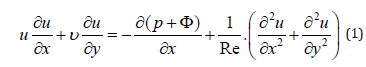
where:
u, v, p — basic unknowns, velocities and pressures;
F — potential of external forces, a given function;
Re — the Reynolds number, a positive parameter.
In a simplified version, the Navier-Stokes equations look like
this:
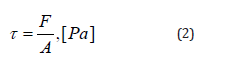
where:
F — the tangential force [N],
A — the area on which the force acts [m2].
Pa — shear stress level.
Resistance to blood flow through the vessels depends on the radius of the vessel , vessel lengths and blood viscosity η .
This dependence is described by the Poiseuille formula: R =
8lη/πr4,
where:
R — hydrodynamic resistance;
l — vessel length, m;
r — vessel radius, m;
η — blood viscosity.
Applied to the cardiovascular system, equation (2) can be expressed as:
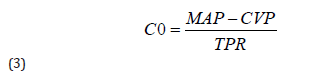
where:
CO – cardiac output;
MAP – mean arterial pressure;
CVP – central venous pressure;
TPR – total peripheral resistance.
The results of the numerical simulation were verified through physical experiments, the primary objective of which was to evaluate the hydrodynamic properties of the developed blood flow model (flow rate and pressure at a specified blood flow rate). For this purpose, a specialized experimental stand simulating systemic circulation was assembled. The schematic diagram of the experimental stand is shown in Figure 3.
The experimental stand is a closed hydraulic system that sequentially includes the following components: an expansion tank used to fill the system with fluid and remove air bubbles; a flexible sealed bag simulating the heart (2); elastic TYGON connecting tubing functioning as vessels (3); a Krohne VA-40 float rotameter for measuring fluid flow (4); a pressure sensor (5); a throttling device simulating the total peripheral resistance of the human cardiovascular system (6); a disc. pump connected at the bottom of the stand (10); a Biosoft-M Angioton pressure measuring device (7); and connecting tee connectors (9). A 40% aqueous glycerin solution was used as the working fluid, with viscosity closely approximating that of human blood.




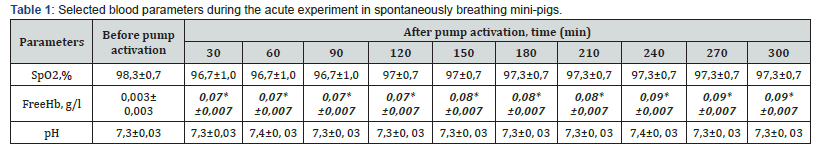
*Significant differences compared to before pump activation.

Acute in vivo experiments were conducted on mini-pigs with an average body weight of 68 ± 7 kg (n = 6). The duration of each acute experiment was 5 hours. The disc. pump was connected paracorporeally according to the “LV apex–descending thoracic artery” configuration. The functional vital parameters were recorded using an IntelliVue MP70 monitor (Philips). Blood analysis was performed using an HT-4000i hematology analyzer (Sysmex, Germany). Blood gas measurements were obtained using a RADIOMETER ABL800 FLEX system. The animals were maintained at all stages of the study (during preoperative preparation, surgery, and the postoperative period) in accordance with the requirements of the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (March 18, 1986).
Results
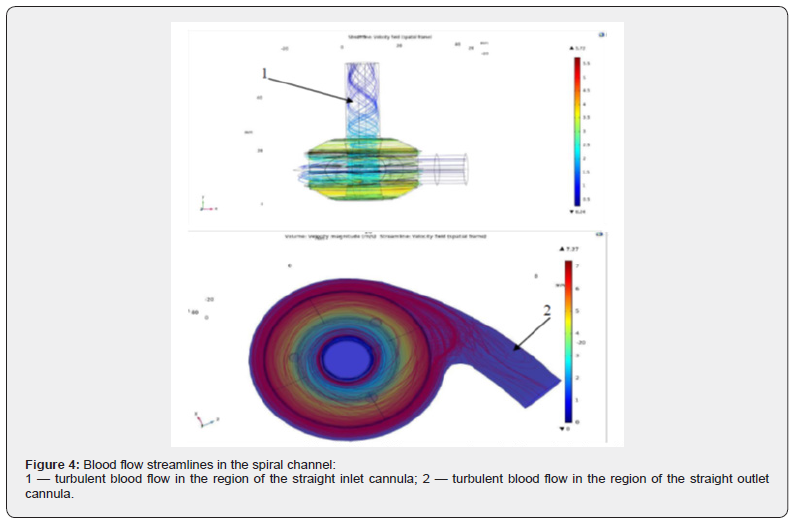
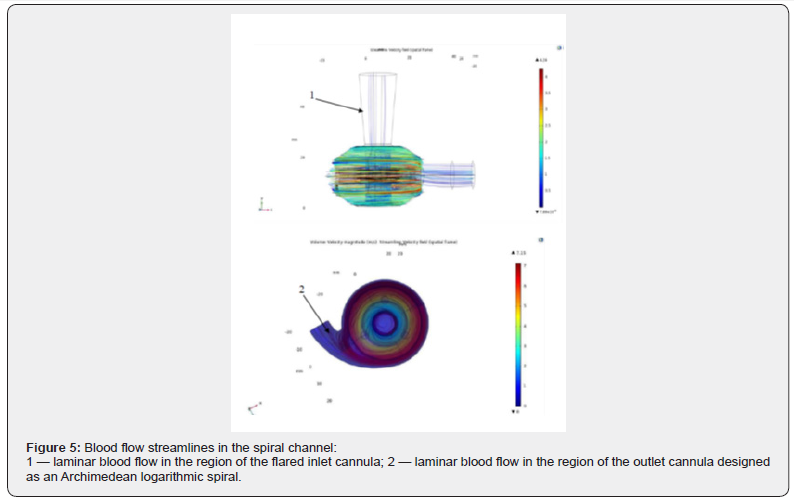
The first stage of the hydrodynamic study involved calculating the flow pattern in the developed auxiliary blood circulation model. It was found that turbulent flows were present in the regions of the inlet and outlet cannulas, associated with the straight geometry of the spiral channel cannulas (Figure 4). Subsequently, computational fluid dynamics analyses demonstrated that blood flow became more laminar and uniform when the inlet cannula was designed as an extended section (funnel-shaped inlet cannula) and the outlet cannula as an Archimedean logarithmic spiral outlet cannula (Figure 5) [6–7]. The hydrodynamic experiments showed a significant reduction in blood flow vortices, particularly in critical regions associated with the inlet and outlet channels. The use of an inlet channel with an increased diameter allowed to achieve more stable and uniform blood flow by eliminating abrupt velocity changes at the entrance to the spiral channel. In turn, the outlet cannula, designed according to the Archimedean logarithmic spiral principle, contributed to uniform blood flow as it exited the pump, thereby minimizing turbulence in the developed auxiliary blood circulation model. Within the second phase of the hydrodynamic analysis, primary attention was focused on the fluid flow rate and pressure characteristics in the developed auxiliary blood circulation model. Using COMSOL Multiphysics software, a simulation of the human systemic circulation was performed, into which the pump, designed in the form of a spiral channel, was integrated (Figure 6).
To investigate the auxiliary blood circulation model formed by a spiral channel, including its hemodynamic characteristics, an approach was applied that involved varying the level of hydraulic pressure within a specific segment, indicated in Figure 6 as section 3. This enabled to regulate and adjust the pressure at the terminal part of the system, varying it from 40 to 200 mmHg, which directly influenced the hydrodynamic characteristics of the auxiliary blood circulation model. The third stage of the hydrodynamic study focused on determining shear stress levels. The equations specified in the Methods section were used for the numerical solution of problems related to this spiral channel model, followed by the implementation of these solutions in COMSOL Multiphysics software. As a result of the calculations, a detailed analysis of the shear stress distribution was conducted in three key regions of the disc. pump (Figure 7): at the base of the inlet cannula (1), in the lower part of the pump—specifically in the flow divider region (2), and in the flow distribution region at the outlet, upstream of the outlet cannula (3). The maximum shear stress level was recorded in the inlet cannula region (70 Pa) and in the flow divider region (70 Pa), while in the region upstream of the outlet cannula the shear stress was even lower (50 Pa). In all regions, the measured shear stress levels did not exceed the permissible threshold of 100 Pa [8].
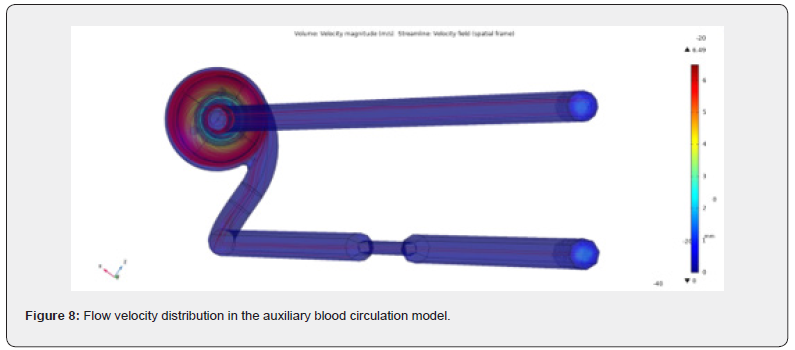
The fourth stage of the hydrodynamic study focused on analyzing the flow velocity distribution in the developed auxiliary blood circulation model. The maximum recorded flow velocity was 6 m/s. The presence of inlet and outlet lines did not alter the velocity within the model, indicating uniform flow inside the lines. The velocity in the spiral channel was found to be 2.8 m/s, suggesting the absence of stagnant zones. As shown in Figure 8, where the flow streamlines are presented in red, no flow vorticity was detected. The computational studies enabled to determine the flow and pressure characteristics of the spiral channel (Figure 9). It is well known that the pressure gradient in the system varies depending on the rotor speed. A rotor speed in the range of 2000– 2500 rpm was found to be the most physiologically favorable, generating a pressure of 100–135 mmHg at the inlet and 73–113 mmHg at the outlet.
When the disc pump operated within the experimental setup at a speed of 2000–2500 rpm, the actual fluid flow rate was 5.7– 6.7 L/min with a pressure drop of 91–123 mmHg. The physical experiments demonstrated a high degree of agreement with the calculated values: at a speed of 2000–2500 rpm and a flow rate of 6–7 L/min, the pressure drop ranged from 84 to 119 mmHg. The discrepancy between the modeling and experimental studies did not exceed 8%, indicating a high level of reliability of the obtained results. The discrepancy below 10% confirms the adequacy of the developed model [1]. Further, the developed auxiliary blood circulation model was investigated in a vivo experiment on minipigs with an average body weight of 68 ± 7 kg (n = 6), breathing spontaneously. As shown in Table 1, a slight tendency toward decreased blood oxygen saturation was observed alongside an increase in the concentration of free hemoglobin (FreeHb) in the animals’ blood. At the same time, the pH remained stable and did not differ from baseline values in any of the experiments. Based on the presented data, although intravascular hemolysis occurred during the in vivo experimental procedures, its level was relatively low and did not exceed the threshold value of 0.2 g/L [10]. The low level of hemolysis indicates good hemocompatibility of the developed pump. The studies also revealed a slight decrease in hemodynamic parameters (Table 2), particularly invasive arterial pressure. This tendency was likely associated with incomplete hemodynamic recovery following surgical intervention within the observed time period.
Discussion
In this study, a new auxiliary blood circulation model generated by a disc. pump was investigated. The model is distinguished by the presence of a flow divider, a specific rotor design, an inlet cannula configured as a flared section (“funnel”), and an outlet cannula designed as an Archimedean logarithmic spiral (“snail-shaped”) [6–7]. It has previously been noted that the threshold shear stress level for an auxiliary circulation model should be considered 100 Pa, and exceeding this value leads to membrane damage. Importantly, not only the threshold level but also the duration of exposure to shear stress plays a critical role in its effect on red blood cells [6]. In our study, the maximum shear stress level was recorded in the regions of the inlet cannula and the flow divider, reaching 70 Pa and therefore remaining below the threshold value of 100 Pa.
Physical experiments confirmed a high degree of agreement with the calculated values, making it possible to proceed to in vivo studies following the wet-lab phase. The results of six in vitro tests showed that at a pump rotational speed of 2500 rpm, a gradual increase in the level of free hemoglobin (FreeHb, g/L) in donor blood plasma began after 2 hours of pump operation. By the end of the experiment, it reached 0.04 ± 0.01 g/L compared to a baseline value of 0.03 ± 0.01 g/L. In acute experiments with spontaneously breathing animals, the observed intravascular hemolysis did not exceed the threshold value of 0.2 g/L [10], indicating a high level of safety of the developed auxiliary blood circulation model. In chronic animal studies, an increase in FreeHb was observed on days 1–2 of the experiment, ranging from 0.01 ± 0 g/L to 0.05 ± 0.1 g/L. From day 3 until the end of the observation period, fluctuations in free hemoglobin levels remained within 0.03–0.04 g/L, with a baseline value of 0.01 ± 0 g/L [11]. Thus, based on the results of computational analysis, physical experiments, and in vivo studies, the newly developed auxiliary blood circulation model driven by a disc. pump demonstrated the ability to achieve hemodynamic parameters consistent with physiological conditions while maintaining acceptable hemocompatibility.
References
- Defares JG, Osborn JJ, Hara UU (1963) Theoretical synthesis of the cardiovasc. system. Acta Physiol. Pharmacol 12(3): 189-265.
- Feller ED, Sorensen EN, Haddad M, Pierson RN, Johnson FL, et al. (2007) Clinical outcomes are similar in pulsatile and nonpulsatile left ventricular assist de-vice recipients. Ann Thorac Surg 83(3): 1082-1088.
- Gohean JR, George MJ, Pate TD (2013) Verification of a Computational Cardiovas-cular System Model Comparing the Hemodynamics of a Continuous Flow to a Synchronous Valveless Pulsatile Flow Left Ventricular Assist Device. ASAIO J 59(2): 107-116.
- Gauthier SV (2017) Innovations in transplantology: the development of the problem of heart transplantation in the Russian Federation. Pathology of blood circulation and cardiac surgery 3S (21): 61-68.
- Fraser KH, Zhang T, Taskin ME, Griffith BP, WU JZ (2012) A quantitative comparison of mechanical blood damage parameters in rotary ventricular assist devices: shear stress, exposure time and hemolysis index. J Biomech Eng 134(8): 081002.
- Golovin AM, Golovina KO (2021) Patent of the Russian Federation No. 205275. The pump of internal auxiliary blood circulation. Byul p. 19.
- Golovin AM, Golovina KO RF (2022) Patent No. 210144 Left ventricular bypass disc. pump for maintaining mechanical heart function. Byul p. 10.
- Pushina DM, Salikhova T, Yu, Biryukova LS, Gurii GT (2020) Loss of stability of the liquid state of the blood and assessment of the risk of hydrodynamic activation of throm-bosis. Izvestiya vuzov. Radiophysics 9(63): 894-918.
- Koptev AV (2013) Method of constructing solutions of Navier-Stokes equations. Pro-ceedings of the Russian State Pedagogical University named after A.I. Herzen 154: 16-23.
- Malinauskas A (2017) FDA benchmark medical device flow models for CFD val-idation. ASAIO Journal 63(2): 150-160.
- Golovina KO, Golovin AM, Aizman RI (2023) The effect of the left ventricular by-pass mechanical circulatory support device on some blood parameters. Clinical physiology of blood circulation 1(20): 78-88.






























