Predictive Potential of Interleukin-6, Pentraxin-3, and Procalcitonin in Identifying Sepsis and Septic Shock Among High-Risk Fever and Neutropenia Pediatric Cancer Patients Running Head: Biomarkers Predictive Value for Septic Shock
Mahmoud Kamal1, Samah Elkhaiat2, Noha Radwan3 and Ahmed EL-hemaly1,2*
1Department of Pediatric Oncology, National Cancer Institute (NCI)-Cairo University, Cairo, Egypt
2Department of Pediatric Oncology, Children's Cancer Hospital of Egypt (CCHE), Cairo, Egypt
3Department of Clinical Pathology, NCI-Cairo University, Cairo, Egypt
Submission: May 01, 2024; Published: May 10, 2024
*Corresponding author: Ahmed El-hemaly, Department of Pediatric Oncology, National Cancer Institute, Cairo University, and Children’s Cancer Hospital of Egypt (CCHE), Cairo, Egypt
How to cite this article: Mahmoud K, Samah E, Noha R, Ahmed E-h. Predictive Potential of Interleukin-6, Pentraxin-3, and Procalcitonin in Identifying Sepsis and Septic Shock Among High-Risk Fever and Neutropenia Pediatric Cancer Patients Running Head: Biomarkers Predictive Value for Septic Shock . Ann Rev Resear. 2024; 11(2): 555807. DOI: 10.19080/ARR.2024.11.555807
Abstract
Background: This study aimed to assess the role of interleukin-6(IL-6), Pentraxin-3(PTX-3), and procalcitonin (PCT) in predicting pediatric cancer patients who may develop sepsis or septic shock.
Aim: The study aimed to determine which biomarker has the highest diagnostic value for sepsis/septic shock and to analyze the correlation of the biomarkers with the severity of organ dysfunction represented by the pediatric sequential organ failure assessment (PSOFA) scores.
Methods: Serum levels of interleukin-6, Pentraxin-3, and procalcitonin were measured in 86 pediatric cancer patients who presented with FN, sepsis, and septic shock. Blood samples for IL-6, PTX3, and PCT were obtained within 6 hours of presentation. Patients were categorized into septic and non-septic groups and PSOFA scores at D1, D3, and D28 were calculated. Optimal cut-off values for sepsis and prognostic PSOFA scores were determined.
Results: IL-6, as a single biomarker had the highest ability to predict sepsis at a cut-off value of 89.8 pg/mL with 100% sensitivity and 60.3% specificity (P value <0.001). Combining biomarkers increased the sensitivity and specificity for predicting septic shock and mortality at D28. There was a positive relationship between delta PSOFA and in-hospital mortality. The median levels of IL-6, PTX3, and procalcitonin in gram-negative MDR septic shock patients were significantly higher than the medians in septic shock patients without gram-negative MDR (P value was < 0.05).
Conclusion: Novel biomarkers can help predict sepsis and guide protocols for managing high-risk fever and neutropenia.
Keywords: Predictive; Interleukin-6; Pentraxin-3; Procalcitonin; Septic Shock; Fever and Neutropenia; Cancer
Abbreviations: IL-6: Interleukin-6; PTX-3: Pentraxin-3; PCT: Procalcitonin; PSOFA: Pediatric Sequential Organ Failure Assessment; CNS: Central Nervous System; CVS: Cardiovascular; AEs: Adverse Events; CTC: Common Toxicity Criteria; ALL: Acute Lymphoblastic Leukemia; GIT: Gastrointestinal Infections; MDR: Multi-Drug Resistant; ICU: Intensive Care Unit; IRB: Institutional Review Board
Background
There is limited data on the incidence of sepsis and septic shock in neutropenic pediatric cancer patients. Studies suggest that there are common causes of critical illness in children with cancer, with mortality rates between 41% and 64% [1]. Initial manifestations of sepsis in children include fever, tachycardia, tachypnea, hypotension, and hypothermia. They are highly variable, nonspecific, and are often unnoticed. The most striking feature of pediatric sepsis is that children can sustain tachycardia for long periods, and hypotension may not occur until advanced sepsis compared to adults with sepsis [2,3]. Early administration of empiric intravenous broad-spectrum antibiotics successfully reduced the mortality rate associated with infection in pediatric patients to <1 % [4]. Sepsis is a complex condition involving various immune responses, making it challenging to improve outcomes with drugs targeting single events [5].
This emphasizes the importance of early and precise sepsis detection. Effective biomarkers are crucial for the timely identification and management of sepsis, but no single laboratory test can accurately diagnose or assess its severity [6,7]. IL-6 production is stimulated by various factors, including IL-1, interferons, TNF, viruses, and pathogen-associated molecular patterns. High IL-6 level during inflammation is a poor prognostic factor [6,8]. The determination of IL-6 on the first day of FN was associated with high sensitivity (90%) and specificity (85%) in identifying patients who will develop sepsis or a long-lasting fever episode. Furthermore, IL-6 measurements had a positive predictive value of 94% with the cutoff value accepted at 42 pg/ml [9,10].
Many researchers identified that the extremely high level of IL6 in children with FN while on chemotherapy is strongly related to gram-negative bacteremia [9,11]. Other studies also provided evidence for the utility of IL-6 in the risk stratification of febrile cancer patients [9,12-14]. PTX 3 plays an important role in recognizing pathogens and damaged cells, activating the classical complement pathway, and stimulating phagocytosis [15-17]. Huttunen et al found that PTX 3 concentration exceeding 15 ng/ml is an independent risk factor in patients with bacteremia (sensitivity 72%, specificity 81%) [18]. Also, many researches demonstrated that high concentrations of PTX 3 lasting for the first five days of infection correlated inevitably with high mortality risk [17-21].
In a systematic review and meta-analysis, PCT was found to be more specific for differentiating bacterial infections among hospitalized patients [22]. Another meta-analysis showed that PCT is a useful marker for early diagnosis of sepsis in critically ill patients with sensitivity and specificity of 77% and 79%, respectively [23-25]. It was shown that sepsis caused by Gram-negative bacteria was associated with a significantly higher level of PCT than sepsis caused by Gram-positive bacteria [26-29]. PCT has drawn attention because it can be used for guidance of antibiotic stewardship to reduce inappropriate use of antibiotics [30]. Combining multiple biomarkers has been shown to compensate for the low prediction efficiency of a single marker [31].
Materials and Methods
The study was an observational prospective cross-sectional study conducted from June 2022 to December 2022 at the National Cancer Institute Cairo-Egypt. A total of 92 episodes of fever and neutropenia were analyzed in 86 pediatric cancer patients who presented to the ER with fever and neutropenia, sepsis, and septic shock after receiving chemotherapy. Patients who refused to be included in the study or to give blood samples within 24 hours from the initial presentation to the ER were excluded. The study followed the adapted pediatric version of Sequential Organ Failure Assessment (pSOFA scores) that was validated by Matics et al to facilitate the evaluation of the international sepsis-3 definitions in children [31]. The pSOFA score is a prompt bedside method that can identify patients with suspected infection who are at greater risk of a poor outcome outside the ICU. Each organ system has a score ranging from 0 to 4, the sum of the 6 sub scores for 6 organ systems (range, 0-24 points; higher scores indicate a worse outcome).
The studied organ systems in pSOFA scores were the central nervous system (CNS), cardiovascular (CVS), respiratory, hepatic, and renal systems. The coagulation system was excluded from our study as the assessment of the coagulation system in the score includes platelet count. All our patients were thrombocytopenic either disease or chemotherapy-related, so this will have a negative impact on the scoring system. The sum of the 5 sub scores for 5 organ systems ranges from 0-20 points. The patients were divided into septic and non-septic groups based on defined criteria. The pediatric SOFA score was calculated for each group at D1, D3, and D28. Delta pSOFA was calculated by subtracting pSOFA D3 from pSOFA D1. The study followed the standardized definitions for adverse events (AEs), known as the Common Terminology Criteria for Adverse Events (CTCAE, also called "common toxicity criteria" [CTC]), to describe the severity of organ toxicity for patients receiving cancer therapy [32].
Sampling for biomarkers and clinical data collection
All data were collected through medical records and conducting laboratory tests. Routine laboratory investigations were done, and blood gas analysis was performed as needed. Additional tests, such as CRP and blood cultures were carried out. Imaging tests were also done if fever persisted or specific infections were suspected. Bloodstream infection was confirmed through microbiological culture.
Blood sampling for biomarkers
All blood samples for initial IL-6, PTX3, and PCT measurements were obtained within 6 hours of initial presentation to the ER (or during admission into the department) with FN, sepsis, or septic shock. The samples were collected from all subjects as follows: venous blood samples were withdrawn by venipuncture using dry sterile vacutainers. All blood samples for IL-6 and PTX-3 were divided into five milliliters and distributed into 2 sterile vacutainers with gel for the ELISA technique. Procalcitonin was measured on cobas e411 (Electrochem luminescence immunoassay).
Results
Eighty-six patients with 92 episodes of fever and neutropenia were included. Patients were divided by age into four groups: <= 2 years, 2-6 years, 6-12 years, and 12-18 years. The majority of patients (38%) belonged to the group of >6-12 years old. Sixty-two patients had hematological malignancies, while thirty patients had solid malignancies. The most common hematological malignancy was acute lymphoblastic leukemia (ALL) (33.7%). The majority of patients (86%) were denovo cases of malignancy. High-risk FN was observed in 90% of patients. Most patients had severe infections (88%) and exhibited apparent clinical foci (78.3%). Gastrointestinal infections (GIT) were the most common type of infections (31.5%), followed by respiratory tract infections. Necrotizing enterocolitis was documented in 23 (25%)episodes. Positive blood cultures were documented in 47.8% of the episodes, while forty-eight (52.2%) episodes did not yield positive blood culture. Gram-negative E-coli was the most commonly documented organism (21.7%). Thirty (32.6%) episodes had documented a multi-drug resistant (MDR) spectrum and susceptibility. Sepsis was documented in fifty-five episodes (61.7%), however in 24 (26.1%) episodes, patients presented with septic shock, and finally in 7 (7.6%) episodes, sepsis was not documented.
Finally, Sixty-five (65/86) (75.5%) patients improved and were discharged while twenty-one (21/86) (24.4%) patients died. Severe infection was reported in 96.7% and 70.8% of episodes with sepsis and septic shock, respectively. There was a statistically significant correlation between the occurrence of sepsis or septic shock and the degree of infection (P value <0.001). There was a statistically significant correlation between the occurrence of sepsis or septic shock and the gram-negative infection (p < 0.001). The majority (97.2%) of septic shock patients showed an MDR gram-negative spectrum and sensitivity. Median and mean levels of IL6, pentraxin 3, and procalcitonin were significantly higher in the septic shock group than in the non-septic group (Table 1). The medians of IL-6, PTX3, and procalcitonin in gram-negative MDR septic shock patients were significantly higher than the medians of the same biomarkers in septic shock patients without gram-negative MDR (Table 2).
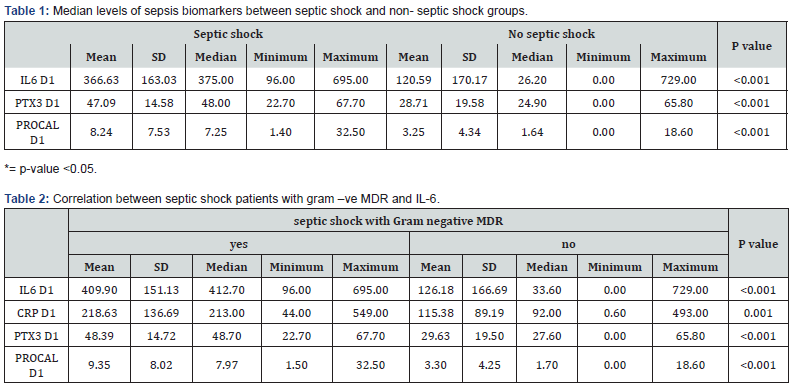
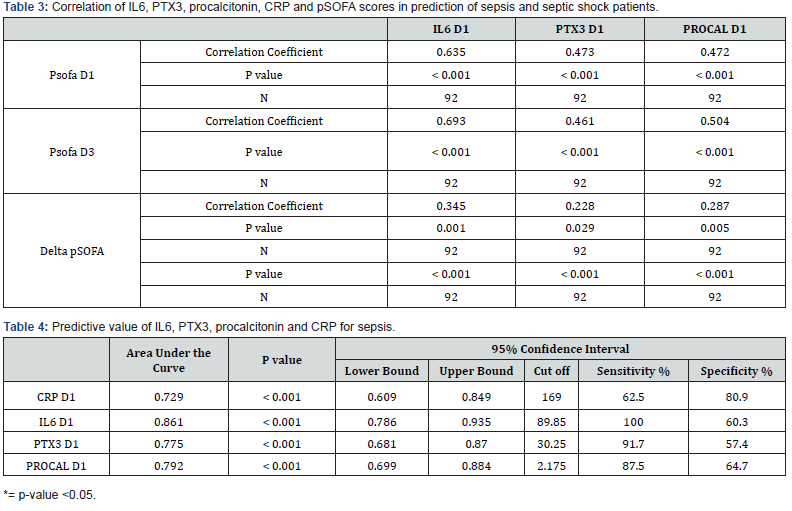
The AUCs for the prediction of sepsis were 0.86 for IL6, 0.775 for PTX3, and 0.792 for procalcitonin. IL-6 as a single biomarker showed the highest ability for prediction of sepsis with a cut-off value of 89.8 pg/mL with 100% sensitivity and 60.3% specificity (P value <0.001) (Figure 1). Delta pSOFA showed the highest ability for prediction of sepsis when using a cut-off value increasing by more than one point with 95.8 % sensitivity and 94.1% specificity (P value <0.001) (Figure 1). IL 6 was significantly and positively correlated with pSOFA D1, pSOFA D3, and delta pSOFA with correlation coefficients 0.635,0.693 and 0.345, respectively (P value was < 0.001) (Table 3).
Independent Factors for prediction of septic shock
Univariate logistic analysis of PCT, IL-6, PTX-3, CRP, and pSOFA D1 score was performed within the sepsis and non-sepsis groups. Binary logistic regression to predict septic shock in patients with sepsis showed that among PCT (B = -0.036, odds ratio (OR) = 0.964), IL-6 (B =0.05, OR= 1.005, P value <0.05), CRP (B =0.002, OR = 1.002, PTX-3 (B = 0.020, OR = 1.020), and pSOFA D1 score (B= 0.997, OR = 0.2710, P = 0.001), IL6 and pSOFA D1 score were strong and independent predictors of septic shock in patients with sepsis (Table 4). The Hosmer–Lemeshow test for goodness of fit was applied to the multivariate logistic regression related to septic shock and pSOFA D1 and indicated good calibration with a P value estimated as 0.646 for pSOFA D1. Binary logistic regression was conducted to examine whether PCT, IL-6, PTX-3, and pSOFA D1 scores were significantly associated with the odds of non-survival. The findings suggested that IL-6 and pSOFA D1 score were strongly correlated to mortality at day 28 (P value <0.05) .
Combinations of PCT, IL-6, and PTX-3 for prediction of septic shock and D28 mortality
Combinations of these biomarkers resulted in the following: PCT + IL-6 (AUC = 0.866); PCT + PTX-3(AUC = 0.824); IL-6 + PTX-3 (AUC = 0.888) and PCT + IL-6 + PTX-3 (AUC = 0.895). The sensitivity and specificity of prediction of the septic shock of the combination between IL-6 and PTX-3 were 100% and 77.9%, respectively. There were no significant differences in the sensitivity and specificity of the former combination and other markers combinations (either 2 or 3 combinations). However, there was a significant increase in the sensitivity and specificity of the prediction of septic shock with any combinations of biomarkers than any single marker alone. The same previous results were observed in the prediction of D28 mortality.
Discussion
This study aimed to assess the value of IL-6, PTX-3, and PCT levels in predicting septic complications in pediatric cancer patients with high-risk FN after chemotherapy. The study found that patients with septic shock had higher levels of IL6, PTX3, and procalcitonin compared to those without septic shock. Additionally, the levels of pSOFA D1, D3, and delta pSOFA were significantly higher in the septic shock group. These findings are similar to a previous study conducted by Zhang Y. et al who compared the diagnostic and predictive value of different biomarkers in sepsis patients in an intensive care unit (ICU). He concluded that although the SOFA score is considered the gold standard, analyzing multiple biomarkers can enhance the diagnostic and prognostic capabilities of sepsis in ICU patients [33].
In our study, IL-6 as a single biomarker, showed the highest ability for prediction of sepsis and septic shock when using a cut-off value of 89.8 pg/Ml, the sensitivity was 100% but the specificity was 60.3%. There was a significant correlation between septic shock patients with gram-negative MDR and levels of IL-6, PTX3, and procalcitonin. The medians of IL-6, PTX3, and procalcitonin in gram-negative MDR septic shock patients were significantly higher than the medians of the same biomarkers in septic shock patients without gram-negative MDR. These findings were consistent with previous research conducted by Xu et al. He aimed to compare the efficacy of serum IL-6, procalcitonin, and C-reactive protein in identifying pediatric cancer patients at high risk for infection. The results showed that IL-6 had a higher AUC of 0.89 compared to CRP and procalcitonin. This indicated that IL-6 is a more reliable marker for identifying high-risk patients, distinguishing between Gram-positive and negative bacteremia, assessing the severity of infection, and predicting the outcome for those patients [34]. In another research conducted by Song et al in Korea, he found that septic shock could be distinguished by serum IL-6 levels (AUC, 0.71 to 0.89, 76.1% sensitivity, 78.4% specificity) [35].
In our study, PTX-3 showed a moderate predictive ability for sepsis in comparison to IL-6, when using a cut-off value of 30.25 ng/mL, the sensitivity was 91.7% but the specificity was 57.4% (P value <0.001). These findings were similar to another study conducted by Hamed et al who aimed to evaluate the diagnostic value of PTX-3 in patients with sepsis and septic shock. The research demonstrated that PTX-3 can serve as a useful diagnostic tool in differentiating between sepsis and septic shock, and its levels in the plasma can be indicative of the severity of the condition [36]. Several prior studies provided significant and beneficial AUCs for PTX-3 for distinguishing sepsis or septic shock from healthy controls [37-39].
Our study found that the combination of IL-6 and PTX-3 had a sensitivity of 100% in predicting septic shock and mortality at D28. This combination also had the highest specificity (77.9%) in predicting septic shock. There were no significant differences in the sensitivity and specificity between the three combined biomarkers (IL-6, PTX-3, and procalcitonin) and any combination of two biomarkers. However, there was a significant increase in sensitivity and specificity for predicting septic shock and mortality at D28 when using any combination of sepsis biomarkers compared to using a single marker [40].
Conclusion
IL-6 as a single biomarker showed the highest ability for prediction of sepsis and septic shock. The combination of IL-6+PTX-3 showed the highest ability for predicting septic shock and D28 mortality.IL 6 was significantly and positively correlated with pSOFA D1, pSOFA D3, and delta pSOFA. This study provides valuable information about the inflammatory response in pediatric cancer patients with high-risk fever and neutropenia following chemotherapy which can aid clinicians in identifying children who are at a higher risk of developing sepsis or septic shock, allowing for earlier intervention and appropriate management. Finally, this study provides a foundation for further research and development in the field of sepsis biomarkers. By identifying IL-6, PTX3, and procalcitonin as potential biomarkers, this study opens avenues for future investigations exploring their mechanisms of action, refining cutoff values, and assessing their performance in different populations.
References
- Agulnik A (2023) Management of septic shock in children with cancer- Common challenges and research priorities. J Pediatr (Rio J) 99(2): 101-104.
- Kleinman ME, Chameides L, Schexnayder SM, Ricardo AS, Mary FH, et al. (2010) Pediatric advanced life support: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 122(18 Suppl): S876-S908.
- Weiss SL, Parker B, Bullock ME, Sheila S, Carolynn P, et al. (2012) Defining pediatric sepsis by different criteria: discrepancies in populations and implications for clinical practice. Pediatr Crit Care 13(4): e219-e226.
- Evans L, Rhodes A, Alhazzani W, Massimo A, Craig MC, et al. (2021) Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med 47(11): 1181-1247.
- Vincent JL, Sakr Y, Sprung CL, Marco RV, Konard R, et al. (2006) Sepsis in European intensive care units: results of the SOAP study. J Crit Care Med 34(2): 344-353.
- Reinhart K, Bauer M, Riedemann NC, Christiane SH (2012) New approaches to sepsis: molecular diagnostics and biomarkers. J Clin Microbiol Rev 25(4): 609-634.
- Mickiewicz B, Thompson G, Blackwood J (2015) Development of metabolic and inflammatory mediator biomarker phenotyping for early diagnosis and triage of pediatric sepsis. J Crit Care 19(1): 320.
- Lippitz BE, Harris RA (2016) Cytokine patterns in cancer patients: A review of the correlation between interleukin 6 and prognosis. Oncoimmunology 5(5): e1093722.
- Chaudhary N, Kosaraju K, Bhat K, Indira B, Anupama B (2012) Significance of interleukin-6 (IL-6) and C-reactive protein (CRP) in children and young adults with febrile neutropenia during chemotherapy for cancer: a prospective study. J Pediatr Hematol Oncol 34(8): 617-623.
- Hilde van der Galiën T, Loeffen EA, Miedema KG, Wim JE Tissing (2018) Predictive value of PCT and IL-6 for bacterial infection in children with cancer and febrile neutropenia. Support Care in Cancer 26(11): 3819-3826.
- Rao L, Zhang K, Luo H, Shuya H, Yuping L, et al. (2022) The ability of inflammatory markers to recognize infection in cancer patients with fever at admission. J Immunol Res 70(5): 667-677.
- Phillips RS, Wade R, Lehrnbecher T, Lesley AS, Alex JS (2012) Systematic review and meta-analysis of the value of initial biomarkers in predicting adverse outcome in febrile neutropenic episodes in children and young people with cancer. BMC Med 10(1): 1-13.
- Plesko M, uvada J, Makohusova M, Waczulikova I, Behulova D, et al. (2016) The role of CRP, PCT, IL-6, and presepsin in early diagnosis of bacterial infectious complications in pediatric haemato-oncological patients. Neoplasma 63(5): 752-760.
- Oikonomakou Z, Gkentzi D, Gogos C, Karolina AK (2020) Biomarkers in pediatric sepsis: a review of recent literature. Biomark Med 14(10): 895-917.
- Diepold M, Noellke P, Duffner U, Kontny U, Reinhard B (2008) Performance of Interleukin-6 and Interleukin-8 serum levels in pediatric oncology patients with neutropenia and fever for the assessment of low-risk. BMC Infect Dis 8(1): 28-28.
- Bottazzi B, Doni A, Garlanda C, Alberto M (2009) An integrated view of humoral innate immunity: pentraxins as a paradigm. Annu Rev Immunol 28: 157-183.
- Kunes P, Holubcova Z, Kolackova M (2012) Pentraxin 3(PTX 3): an endogenous modulator of the inflammatory response. Mediators Inflammat 2012: 920517-920517.
- Huttunen KM, Raunio H, Rautio J (2011) Prodrugs-from serendipity to rational design. Pharmacol Rev 63(3): 750-771.
- Al-Ramadi BK, Ellis M, Pasqualini F, Alberto M (2004) Selective induction of pentraxin 3, a soluble innate immune pattern recognition receptor, in infectious episodes in patients with hematological malignancy. Clin Immunol 112(3): 221-224.
- Biagi E, Col M, Migliavacca M, Mariagrazia D, Daniela S, et al. (2008) PTX3 is a potential novel tool for the diagnosis and monitoring of pulmonary fungal infections in immuno-compromised pediatric patients. J Pediatr Hematol Oncol 30(12): 881-885.
- Juutilainen A, Vänskä M, Pulkki K, Sari H, Nousiainen T, et al. (2011) Pentraxin 3 predicts a complicated course of febrile neutropenia in hematological patients, but the decision level depends on the underlying malignancy. Eur J Haematol 87(5): 441-447.
- Simon L, Gauvin F, Amre DK, Patrick SL, Jacques L (2004) Serum procalcitonin and C-reactive protein levels as markers of bacterial infection: a systematic review and meta-analysis. Clin Infect Dis 39(2): 206-217.
- Wacker C, Prkno A, Brunkhorst FM, Peter S (2013) Procalcitonin as a diagnostic marker for sepsis: a systematic review and meta-analysis. Lancet Infect Dis 13(5): 426-435.
- Hoeboer SH, van der Geest PJ, Nieboer D, Groeneveld ABJ (2015) The diagnostic accuracy of procalcitonin for bacteremia: a systematic review and meta-analysis. Clin Microbiol Infect 21(5): 474-481.
- Downes KJ, Fitzgerald JC, Weiss SL (2020) Utility of procalcitonin as a biomarker for sepsis in children. J Clin Microbiol 58(7): e01851-e01859.
- Fleischhack G, Cipic D, Juettner J, Hasan C, Bode U (2000) Procalcitonin-a sensitive inflammation marker of febrile episodes in neutropenic children with cancer. Intensive care Med 26(Suppl 2): S202-S211.
- Sakr Y, Sponholz C, Tuche F, Brunkhorst F, Reinhart K (2008) The role of procalcitonin in febrile neutropenic patients: a review of the literature. Infection 36(5): 396-407.
- Martinez-Albarran M, de Jesus Perez MJ, Gallegos CS, Frenando SZ, Susana Del TA, et al. (2009) Procalcitonin and C-reactive protein serum levels as markers of infection in a pediatric population with febrile neutropenia and cancer. J Pediatr Hematol Oncol 26(6): 414-425.
- Memar MY, Varshochi M, Shokouhi B, Asgharzadeh M, Hossein SK (2017) Procalcitonin: the marker of pediatric bacterial infection. J Biomed Pharmacother 96: 936-943.
- Branche A, Neeser O, Mueller B, Philipp S (2019) Procalcitonin to guide antibiotic decision-making. Curr Opin Infectious Dis 32(2): 130-135.
- Yin J, Chen Y, Huang JL, Lei Y, Zhong SK, et al. (2021) Prognosis-related classification and dynamic monitoring of immune status in patients with sepsis: A prospective observational study. World J Emerg Med 12(3): 185.
- Matics TJ, Sanchez-Pinto LN (2017) Adaptation and validation of a pediatric sequential organ failure assessment score and evaluation of the sepsis-3 definitions in critically ill children. JAMA Pediatrics 171(10): e172352-e172352.
- (2017) Common Terminology Criteria for Adverse Events (CTCAE), Version 5.0. US. Department of Health and Human Services, National Institutes of Health, National Cancer Institute.
- Zhang Y, Khalid S, Jiang L (2019) Diagnostic and predictive performance of biomarkers in patients with sepsis in an intensive care unit. Int J Med Res 47(1): 44-58.
- Xu XJ, Luo ZB, Xia T, Hua S, Shi Long Y, et al. (2019) Comparison of interleukin-6, interleukin-10, procalcitonin and C-reactive protein in identifying high-risk febrile illness in pediatric cancer patients: a prospective observational study. Cytokine 116: 1-6.
- Song J, Park DW, Moon S, Han JC, Jong HP, et al. (2019) Diagnostic and prognostic value of interleukin-6, pentraxin 3, and procalcitonin levels among sepsis and septic shock patients: a prospective controlled study according to the Sepsis-3 definitions. BMC Infect Dis 19(1): 1-11.
- Hamed S, Behnes M, Pauly D, Dominic L, Max B, et al. (2017) Diagnostic value of Pentraxin-3 in patients with sepsis and septic shock by latest sepsis-3 definitions. BMC Infect Dis 17(1): 1-10.
- Muller B, Peri G, Doni A, Landmann R, Bottazzi B, et al. (2001) Circulating levels of the long pentraxin PTX3 correlate with the severity of infection in critically ill patients. Crit Care Med 29(7): 1404-1407.
- Uusitalo-Seppälä R, Huttunen R, Aittoniemi J, Pertti K, Aila L, et al. (2013) Pentraxin 3 (PTX3) is associated with severe sepsis and fatal disease in emergency room patients with suspected infection: a prospective cohort study. PloS One 8(1): e53661.
- Bastrup-Birk S, Skjoedt MO, Munthe-Fog L, Jens JS, Ying JM, et al. (2013) Pentraxin-3 serum levels are associated with disease severity and mortality in patients with systemic inflammatory response syndrome. PloS One 8(9): e73119.






























