Advances and Challenges in Diagnosing Behçet’s Disease: A Comprehensive Review
Nadia Nishat1, Nune Azaryan Dermenjian2, Yohana Haddish Mogos3, Jubran Al Balushi4, Marcellina Nwosu5, Juan C Pantoja6, Chinyere Pamugo7, Marcela Garzon Olave8, Elba Lorena Moreno Ortiz9, Adolfo Izaguirre Membreño10, and Maria Isabel Gomez Coral11*
1Mandya Institute of Medical Sciences, India
2Yerevan State Medical University, USA
3Orotta School of Medicine, Canada
4University College Dublin, Ireland
5American University of Integrative Sciences, Barbados, and El Paso Interventional Pain Center, USA
6Universidad del Norte, Barranquilla, Colombia
7University of Texas Houston, Health Sciences Center, USA
8Universidad de los Andes, Colombia
9Instituto Salvadoreño del Seguro Social, El Salvador
10Universidad Nacional Autónoma de Honduras (UNAH), Honduras
11Universidad del Valle de México, México
Submission: April 06, 2024; Published: April 15, 2024
*Corresponding author:Maria Isabel Gomez Coral, Universidad del Valle de México, México
How to cite this article: Maria Isabel Gomez Coral. Advances and Challenges in Diagnosing Behçet’s Disease: A Comprehensive Review. Ann Rev Resear. 2024; 11(1): 555803. DOI: 10.19080/ARR.2024.11.555803
Abstract
Behçet's Disease (BD) presents significant diagnostic and management challenges due to its complex and multifaceted nature. Initially described in 1937, BD primarily affects populations along the Silk Road, with a prevalence of 14.6 per 100,000 globally and 420 per 100,000 in Turkey. Its autoimmune nature and lack of definitive etiology make diagnosis reliant on clinical manifestations, with recurrent oral ulcers being a hallmark symptom. Recent research has shed light on the genetic underpinnings of BD, with HLA-B*51 being a significant susceptibility locus. Immunological dysregulation involving T and B lymphocytes, dendritic cells, and various cytokines contribute to the pathogenesis. Clinical manifestations vary widely, affecting multiple organ systems, with mucocutaneous and ocular symptoms being the most common. Diagnosis is primarily clinical, aided by classification criteria such as the ISG and ICBD criteria, although challenges persist due to overlapping features with other autoimmune disorders. Multidisciplinary collaboration among specialists is crucial for accurate diagnosis and optimal management.
Keywords: Behçet's Disease; Vasculitis; Autoimmune Disease; Diagnosis in Behçet's Disease
Abbreviations: BD: Behçet's Disease; ISG: International Study Group; ICBD: International Criteria for Behçet's Disease; HLA: Human Leukocyte Antigen; MICA: MHC Class I Polypeptide-Related Sequence A; IL: Interleukin; TNF-α - Tumor Necrosis Factor Alpha; CRP: C-Reactive Protein; ESR: Erythrocyte Sedimentation Rate; ANA: Antinuclear Antibodies; ANCA: Antineutrophil Cytoplasmic Antibodies; EEG: Electroencephalogram; EMG: Electromyogram; NCS: Nerve Conduction Studies; CSF: Cerebrospinal Fluid; MRI: Magnetic Resonance Imaging; MRA: Magnetic Resonance Angiography; MRV: Magnetic Resonance Venography; CTV: Computerized Tomographic Venography; GI: Gastrointestinal; CNS: Central Nervous System; SLE: Systemic Lupus Erythematosus; ReA: Reactive Arthritis; AECA: Anti-Endothelial Cell Antibodies; HAP: Haplotype; SPR: Skin Pathergy Reaction; MS: Multiple Sclerosis
Introduction
First detailed by Hulusi Behçet, Behçet's Disease was introduced in the vasculitides class in 1937 [1,2]. It was initially discovered in patients with the same triad of symptoms, between relapsing uveitis, genital ulcers, and erythema nodosum [3]. It is an autoimmune disease that causes systemic vasculitis in vessels of all sizes and many other mucocutaneous and ocular manifestations [1]. Even in the modern era, there are no confirmed etiologies. However, it is believed that due to its autoimmune nature, there is a genetic factor that increases susceptibility and environmental factors that may be linked to geography [1]. There are no definitive tests for Behçet's disease, so diagnosis primarily relies on clinical manifestations. Modernly, Behçet's is diagnosed after at least 3 episodes of mouth sores over a period of 12 months, in addition to 2 other symptoms, like genital sores, uveitis, skin lesions, or a positive pathergy test [4,5]. A positive pathergy test is confirmed if two days after pricking the forearm with a sterile tiny needle forms a red bump, which proves there is an overreaction by the immune system. It is also recommended that blood tests be taken to rule out other potential causes of symptoms if they are looked at individually [4].
Behçet's disease is of growing concern because it primarily affects the working class. There is highly significant as the highest incidence of Behçet's disease is along the "Silk Road" populations, where the majority of the workforce, primarily individuals between 20 and 40 years old, are the most affected [2,6]. Although most cases are sporadic, which makes diagnosing and predicting them a lot more complex, familial clusters are reported [7]. Furthermore, in these cases, genetic anticipation has been observed [8]. The global prevalence 2017 reached 14.6 per 100,000, with even higher prevalence rates along the Silk Route, like Turkey, with 420 per 100,000 [9,2]. Moreover, it is reported that every patient with Behçet's disease has a health cost of roughly 21,600€ per year, which may lead to financial problems [10]. Severe morbidities are associated with Behçet's due to the wide range of affected organs and systems, although most of the lasting complications are focused on the ocular and mucocutaneous regions. In addition, high mortality rates have been observed due to the involvement of large vessels as well as neurological and cardiac systems [11]. The wide range of complications and their severities are of great concern, urging the medical world to establish early diagnostic assessments and suitable management plans for these patients.
Etiology and Pathogenesis
Recent advancements in the research of Behcet's disease pathophysiology provided insights into its complex genetics. Hughes et al. identified multiple susceptible loci in the HLA region associated with Behcet's disease—the association with HLA-B*51 being particularly significant. Consequently, this was explained by the variant being positioned between HLA-B and MICA genes. Other independent genetic associations were identified within genes PSORS1C1, upstream of HLA-F-AS1, and HLA-Cw*1602 [12]. Matteo Piga and Alessandro Mathieu conducted a systematic review that confirmed the crucial role of HLA-B*51 in the genetic susceptibility of Behcet's disease. The study also emphasized that other HLA or HLA-related genes may affect disease susceptibility [13]. Ahmet Gul discussed meta-analysis findings that highlighted the predominance of HLA-B*51 among different ethnicities. Epistatic interaction between HLA-B51 and ERAP1 variants was discovered, hinting at the possible pathophysiology mechanism of Behcet's disease. The investigations into the HLA Class I region uncovered weaker but independent associations around HLA-A and HLA-C regions. genomewide studies identified associations with various genes such as IL10, IL23R, CCR1, STAT4, KLRC4, GIMAP2/GIMAP4, and UBAC2. Deep resequencing of targeted genes also added additional associations with rare variants in TLR4, MEFV, and NOD2 genes [14]. Mahmoudi et al. reviewed the specific HLA alleles, such as HLA-B51, HLA-B15, HLA-B27, HLA-B57, and HLA-A26, and found them to be significant risk factors for Behçet's disease as well as protective alleles like HLA-B49 and HLA-A03 [15].
Complex and dysregulated immunological mechanisms characterize Behcet's disease [16,17]. Genetic and environmental factors play a role in the dysfunction of T and B lymphocytes and dendritic cells, contributing to the pathogenesis of this complex condition. Recent studies highlighted the involvement of polymorphisms at the DNA level, Treg Cells, Th17 cells, and vital cellular populations that drive the immune responses [16]. Tong et al. suggested that infection-related triggers such as antigens and autoantigens promote immune dysregulation in the genetically susceptible individual to Behcet's disease. This response includes the activation of innate and adaptive immune systems, resulting in the production of various cytokines and chemokines [17]. Epidemiological evidence suggests that infectious agents, microbial dysbiosis, and other environmental factors trigger autoimmune dysregulation of Behcet's disease. Infectious agents, such as Streptococcus Sanguinis, Saccharomyces cerevisiae, and mycobacteria, have been implicated in the pathogenesis. Viral agents like herpes simplex virus-1, hepatitis C virus, and varicella zoster virus may also play a role. Oral ulcers precede the disease onset, suggesting the potential involvement of oral microbial flora [18,19]. Furthermore, factors such as stress, fatigue, food consumption, and hormonal influences can also influence the clinical manifestations of Behçet's disease [19].
Clinical Manifestations
Behçet's disease, classified as a systemic vasculitis condition, is often referred to as Behçet's syndrome due to its multi-system involvement [20]. It exhibits a relatively equal distribution among genders and is predominantly prevalent along the Silk Road, encompassing regions across the Mediterranean, Middle East, and Far East. The manifestations of Behçet's syndrome vary widely and may present independently or concurrently. When significant symptoms occur concurrently, they have been referred to as disease phenotypes [21]. The syndrome manifests diverse symptoms across mucocutaneous, ocular, vascular, dermatologic, musculoskeletal, gastrointestinal, and neurological domains, each occurring with varying frequencies.
Mucocutaneous and ocular symptoms are the most common manifestations and typically appear earliest, often serving as hallmark indicators [22,23]. In approximately 95% of cases, oral ulcers typically occur on the lips, gingiva, cheeks, and tongue and are often recurrent. Several factors have been proposed as exacerbating or triggering factors for the recurrence of oral ulcers, including fatigue, stress, histamine-containing food, smoking cessation, and, in women, menstruation [24]. Genital ulcers, seen in 80-90% of cases, often occur on the scrotum or the labia majora and minora. While both types of ulcers are typically self-limiting and heal spontaneously, genital ulcers tend to scar formation.
Dermatologic manifestations typically arise in around 85% of the patients and include erythema nodosum, predominantly in the lower extremities, papulopustular and acneiform nodules similar to adolescent acne but with a more robust distribution involving the arms, buttock, and chest in addition to the face. An intriguing finding in BD is Skin pathergy reaction (SPR). This involves inducing a hyperinflammatory response through needle pricks, with positivity determined by the presence of an erythematous papule ≥2 mm or a pustule after 24–48 h. The positivity rate is highly variable, ranging between 7.7% and 84%, and is affected by many factors, such as the type of needles used, the number of needle pricks, geographic area, gender, and disease activity [25]. Furthermore, SPR is a nonspecific test as it can be positive for other diseases such as Sweet's syndrome, Crohn's disease, pyoderma gangrenosum, and others.
Articular manifestations affect around 50–80% of BD patients [21]. Recurrent, self-limited arthritis and arthralgia are common rheumatologic findings, typically presenting as non-deforming, non-erosive, asymmetric mono-oligoarthritis affecting larger joints such as the knees, wrists, ankles, and elbows. Ocular involvement arises in up to 30-70% of patients, with uveitis (often bilateral and either pan or posterior) being the predominant manifestation and retinal vasculitis in around half of cases. The course of ocular symptoms typically alternates between exacerbation and remission phases, with chronic involvement potentially leading to complications like cataracts, glaucoma, and loss of visual acuity [26].
A distinguishing feature of Behçet's disease from other systemic vasculitides is the simultaneous involvement of venous and arterial vessels of different sizes with an additional propensity towards aneurysm formation. Behçet's disease has thus been coined as variable-vessel vasculitis. Vascular manifestations commonly include superficial and deep venous thrombosis, often leading to post-thrombotic syndrome [27]. Although these events frequently affect upper and lower extremity veins, occurrences in atypical sites such as the hepatic veins, portal vein, or cerebral venous sinus have been observed [28]. Gastrointestinal and neurological manifestations occur at a much lesser frequency of about 5%. Gastrointestinal manifestations resembling those of Crohn's disease occur at a lesser frequency. Like Crohn's disease, BD leads to segmental transmural inflammation and punched-out, fissuring, or aphthoid ulcers, most frequently localized to the ileocaecal region. Fistula and granuloma formation have occasionally been discovered as well. Neurological symptoms may include severe headache, cranial nerve palsies, dysarthria, ataxia, and hemiparesis. Around 80-90% of all neurologic symptoms are attributable to parenchymal neurological manifestations, also known as neuro-Behçet's, while the remaining cases are related to cerebral venous sinus thrombosis [29].
Diagnosis
Behçet's disease is primarily a clinical diagnosis, but several classification criteria have been introduced to aid diagnosis. The criteria in use for the diagnosis and classification of BD are the original International Study Group (ISG) criteria (established in 1990) and the later developed and adopted International Criteria for Behçet's Disease (ICBD) (established in 2014) [30,31]. According to ISG, the presence of oral ulcers and two of the following, genital ulceration, eye lesions, skin lesions, and positive pathergy test, is required to diagnose BD. This criterion has been shown to have 95% sensitivity and 98% specificity [31]. Conversely, the new International Criteria for Behçet's Disease (ICBD) also considers neurological and vascular manifestations. The ICBD criteria employ a scoring system, with a certain threshold (4 points or more) determining classification as Behçet's Disease. This scoring system allows 2 points each for oral ulcer, genital ulcer, and ocular lesions and 1 point each for positive pathergy test, neurologic, and vascular involvement. In one multinational study, the ICBD criteria were shown to be more sensitive but slightly less specific diagnostic criteria than the ISG criteria [32].
What makes BD diagnosis challenging is that specific biomarkers for BD still need to be identified, unlike other autoimmune diseases like SLE. Additionally, unlike other autoimmune diseases, autoantibodies such as rheumatoid factor, antinuclear, anticardiolipin, and antineutrophil cytoplasmic antibodies are generally absent. Inflammatory markers like CRP, ESR, and various cytokines are often elevated in Behçet's Disease but lack specificity for diagnosis. Recent advancements have identified CTDP1 as a potential autoantigen. Thus, the anti-CTDP1 antibody is a novel autoantibody for Behcet disease and is expected to help in more accurate clinical diagnosis [33].
Imaging studies that play a role in diagnosing BD-related neurologic symptoms include MRI and MR angiography. Most common MRI findings include parenchymal changes and heterogeneous contrast enhancement after gadolinium injection involving the brainstem, cerebral white matter, basal ganglia, internal capsule, thalamus, and spinal cord [34]. They are usually extensive, confluent, and distributed over white matter without predilection for the periventricular regions in opposition to multiple sclerosis (MS) [35-37]. Atypical MRI presentations such as periventricular and white matter nonspecific lesions tend to occur with comorbid vascular conditions like hypertension. MR angiography is a diagnostic tool in patients with an extraparenchymal form of the disease.
Laboratory Investigations
Behcet's disease (BD) is a polygenic disorder, and its mode of inheritance is not yet clear [38]. The association between BD and human leukocyte antigen 5 (HL-A5) antigen was first detected four decades ago by a Japanese professor, Shigaeki Ohno, and it was later renamed human leukocyte antigen B5 (HLA-B5) [39]. Evidence has shown that BD has unusual geographic distribution and close association with the major histocompatibility complex allele, HLA-B51, specifically the HLA-B51:01 allele [38-40]. The endoplasmic reticulum aminopeptidase 1 (ERAP1) haplotype 10 (Hap10) variant contributes to the development of BD by producing specific HLA-B51 peptides. The interaction between HLA-B51 and ERAP1 disrupts the function of regulatory T cells (Tregs), enhancing type 1 T helper (Th1) and Th17 effector cells [41,42]. The rs116799036 locus, situated between HLA-B and MHC Class I Polypeptide-Related Sequence A (MICA) within the HLA region, has been identified as the most significant risk loci strongly associated with BD [41,42]. HLA-B51 significantly elevates the susceptibility to Behçet's disease in various ethnic groups by a factor of 1.5 to 16. Individuals possessing this genotype exhibit a greater prevalence of posterior uveitis than those lacking it [43].
In patients with BD, pro- and anti-inflammatory cytokines have been demonstrated to be involved, and high levels of cytokines have been documented in clinical studies [39]. Studies have shown higher levels of interleukin-6 (IL-6) in patients with positive HLA B51 and increased levels of soluble intracellular adhesion molecules (sICAM-1) in patients with negative HLA-B51 than in those with HLA-B51 favorable allele. However, increased levels of leptin are observed in both groups of patients [44]. BD does not exhibit any pathological or distinctive laboratory findings [45]. Erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) are two non-specific indicators of systemic inflammation. High-sensitivity CRP is synthesized in the vascular endothelium, and it plays a role in the development of atherosclerosis. So, high serum levels of CRP may indicate cardiovascular involvement [46]. Primarily in patients with vascular disease, arthritis, or erythema nodosum-like lesions, the erythrocyte sedimentation rate and C-reactive protein levels are marginally elevated [45]. In patients with BD, no significant differences were found in ESR levels in patients with vascular involvement when compared to those without vascular involvement [46]. Substantial serum amyloid A (SAA) levels are associated with aphthosis and neurological and ocular diseases [44]. The absence of autoantibodies is generally observed in BD patients, including antinuclear antibodies (ANA), antineutrophil cytoplasmic antibodies (ANCA), anticardiolipin, and rheumatoid factor. In contrast, antibodies against Saccharomyces cerevisiae were more prevalent among BD patients with gastrointestinal (GI) involvement than those without GI involvement [45]. Anti-endothelial cell antibodies (AECA) have been identified in a subset of patients with BD and numerous vasculitic disorders. Research has established a significant correlation between AECA and central nervous system (CNS) involvement in BD patients with aseptic meningitis [45].
Imaging Modalities
In BD patients, radiological examinations, such as computerized tomography (CT), magnetic resonance imaging (MRI), magnetic resonance angiography (MRA), magnetic resonance venography (MRV), computerized tomographic venography (CTV), electroencephalogram (EEG), electromyogram (EMG), nerve conduction studies (NCS), and cerebrospinal fluid (CSF) analysis are used in different organ manifestations of the disease [47-50]. Using magnetic resonance diffusion imaging with magnetic resonance spectroscopy in diagnosing and managing BD is still under investigation [47]. BD affects all sizes of both arterial and venous vasculature, and it is a significant cause of morbidity and mortality in these patients. As a result, it is critical to utilize appropriate imaging modalities in diagnosing vascular and neurological involvements, such as MRA, MRI, CTV, CT, CSF, EEG, EMG, NCS, MRV, and Doppler ultrasound studies [47-51]. CT is vital in providing good anatomical details in these premises, while MRA is used to evaluate the structural changes in blood vessels, especially in an aneurysm [47-50]. The clinical disease activity can be assessed with F-fludeoxyglucose positron emission tomography with CT or magnetic resonance [47,49].
Vision impairment is frequently attributed to ocular diseases, specifically affecting adolescent male patients with BD. Ophthalmological examination, fluorescein angiogram, optical coherence tomography, visual charting, Schirmer’s test, and visual evoked potentials can assess eye disease in BD to diagnose ocular BD and monitor therapeutic response. Intraocular fluid culture can be performed to rule out infections [47]. Nasal endoscopy is used to evaluate ear, nose, and throat involvement, and fecal calprotectin, endoscopy, and biopsy are used for GI assessment. In the case of musculoskeletal involvement, ultrasound, plain x-ray, MRI, and synovial fluid analysis can be used to evaluate disease activity [47,50]. Immunofluorescence and skin biopsy can diagnose skin involvement, while electrocardiogram and echocardiogram assess cardiac disease [47,51]. CT of the chest can be used to evaluate mediastinal and chest involvement, and CT and cystoscopy can be used to determine urological BD [47,49].
Histopathological Evaluation
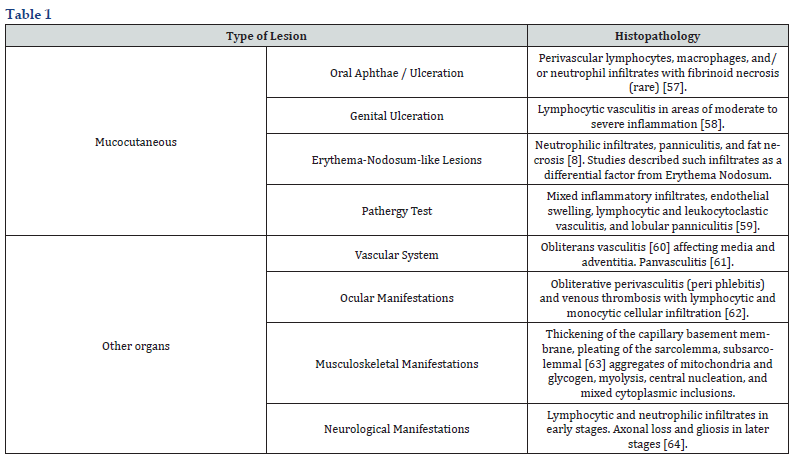
Behçet's disease is a rare condition prevalent in the Mediterranean and Japan and in countries alongside the ancient Silk Road north of the Equator. It is most commonly seen in the Turkish population (80-140/100000) [52,53]. Even though there are immune (γδ T lymphocytes) and inflammatory (IL-1, IL-4, IL-6, TNF-α, etc.) mechanisms involved in the pathogenesis of BD, it is not considered an autoimmune disease [54]. Due to the multisystemic involvement in BD, clinical manifestations vary from one patient to another. In general, symptoms manifest between 20 - 30 years of age, having more severity when it starts earlier in life. Overall, men have a higher risk of significant organ involvement even if they only have cutaneous manifestations [55]. The diagnostic criteria of BD are clinically based, including the presence of oral ulceration plus any two of the following: genital ulceration, typically defined eye lesions, typically defined skin lesions, or a positive pathergy test [56]. Such findings are not pathognomonic nor specific since they could manifest multiple diseases; hence, histopathological studies would benefit from a definite diagnosis. The following table summarizes the most relevant findings of BD's mucocutaneous and other organ lesions [57-64] (Table 1).
Differential Diagnosis and Challenges
There are several challenges in diagnosing Behçet's disease due to its overlapping features with other autoimmune diseases, atypical presentations, and the importance of multidisciplinary expertise—Behçet's disease shares clinical manifestations with various autoimmune disorders, complicating the diagnostic process. For instance, both Behçet's disease, Systemic lupus erythematosus (SLE), and Reactive Arthritis (ReA) may present with symptoms such as oral ulcers, skin lesions, uveitis, arthritis, and joint pain, making it challenging to differentiate between the two conditions (Seyahi et al., 2021) [65,66]. Studies demonstrate that positive HLA B51 is not diagnostic of BD but may affect clinical phenotypes, whereby oral and genital ulcerations, thrombophlebitis, and positive family history of BD are found in patients with HLA-B51 positivity [67]. These overlapping features can complicate the diagnostic process, leading to misdiagnosis or delayed diagnosis. Atypical presentations of Behçet's disease can mimic symptoms of other medical conditions, further complicating diagnosis. The disease's variable course may result in manifestations resembling unrelated disorders, such as infections or malignancies [68,69]. For example, ocular involvement in Behçet's disease can mimic uveitis associated with other autoimmune diseases, further complicating diagnosis and treatment decisions [70]. The study conducted by reported that " forty patients (16.8%) of 238 patients referred to a national Behçet's clinic were finally diagnosed with Behçet's disease whereby ocular and genital lesions, with a positive pathergy test and HLA-B51 were significantly more prevalent in those who are finally diagnosed with Behçet's disease" [66,69].
Again, accurate diagnosis of Behçet's disease necessitates a multidisciplinary approach involving specialists from various fields. Collaboration among rheumatologists, dermatologists, ophthalmologists, and gastroenterologists is crucial for recognizing the distinct clinical patterns of Behçet's disease, interpreting diagnostic tests accurately, and implementing appropriate management strategies [69]. Access to healthcare providers with experience in diagnosing and managing Behçet's disease is essential for improving diagnostic accuracy and ensuring optimal patient outcomes [68,69,71]. Hence, recognizing these challenges underscores the need for increased awareness among healthcare providers, utilization of advanced diagnostic tools, and collaboration among specialists to achieve accurate diagnosis and optimal management of Behçet's disease.
Conclusion
Behçet's Disease presents a complex clinical picture, necessitating a thorough understanding of its pathogenesis, clinical manifestations, and diagnostic challenges. Recent advancements in genetics and immunology have provided valuable insights, yet significant gaps in understanding remain. The lack of specific biomarkers and the overlapping features with other autoimmune diseases underscores the importance of a multidisciplinary approach to diagnosis and management. Enhanced awareness among healthcare providers, utilization of advanced diagnostic tools, and collaboration among specialists are essential for improving patient outcomes in Behçet's Disease. Efforts to address these challenges will facilitate early diagnosis, appropriate treatment, and improved quality of life for individuals with this debilitating condition.
References
- Adil A, Goyal A, Bansal P, Quint JM (2020) Behcet Disease. PubMed. Treasure Island (FL): Stat Pearls Publishing.
- Davatchi F, Chams Davatchi C, Shams H, Shahram F, Nadji A, et al. (2017) Behcet’s disease: epidemiology, clinical manifestations, and diagnosis. Expert Review of Clinical Immunology 13(1): 57-65.
- Feigenbaum A (1956) Description of Behcet’s Syndrome in the Hippocratic Third Book of Endemic Diseases. British Journal of Ophthalmology 40(6): 355-357.
- Behcet’s disease - Diagnosis and treatment - Mayo Clinic [Internet]. www.mayoclinic.org.
- Diagnosing Behcet’s Disease [Internet]. nyulangone.org.
- Karincaoglu Y, Borlu M, Toker SC, Akman A, Onder M, et al. (2008) Demographic and clinical properties of juvenile-onset Behçet’s disease: A controlled multicenter study. Journal of the American Academy of Dermatology 58(4): 579-584.
- Koné-Paut I, Geisler I, Wechsler B, Ozen S, Ozdogan H, et al. (1999) Familial aggregation in Behçet’s disease: high frequency in siblings and parents of pediatric probands. The Journal of Pediatrics 135(1): 89-93.
- Gülbay B, Acican T, Erçen Diken Ö, Pinar Önen Z (2012) Familial Behçet’s disease of adult age: a report of 4 cases from a Behçet family. Internal Medicine (Tokyo, Japan) 51(12):1609-1611.
- Thomas T, Chandan JS, Subramanian A, Gokhale K, Gkoutos G, et al. (2020) Epidemiology, morbidity and mortality in Behçet’s disease: a cohort study using The Health Improvement Network (THIN). Rheumatology 59(10): 2785-2795
- Lorenzoni V, Marinello D, Palla I, Mosca M, Turchetti G, et al. (2020) A cost-of-illness study of Behçet syndrome in Italy. The European journal of health economics: HEPAC: health economics in prevention and care 25(3): 411-422.
- Alpsoy E, Leccese P, Ergun T (2021) Editorial: Behçet’s Disease: Epidemiology, Etiopathogenesis, Diagnosis and Treatment. Front Med (Lausanne) 8: 794874.
- Hughes, Travis (2013) Identification of multiple independent susceptibility loci in the HLA region in Behcet's disease. Nature genetics 45(3): 319-324.
- Piga, Matteo, Alessandro Mathieu (2011) Genetic susceptibility to Behcet's disease: role of genes belonging to the MHC region. Rheumatology 50.2(2011): 299-310.
- Gül, Ahmet (2014) Genetics of Behçet's disease: lessons learned from genome wide association studies. Current opinion in rheumatology 26(1): 56-63.
- Mahmoudi, Mahdi (2022) A comprehensive overview on the genetics of Behcet's disease. International Reviews of Immunology 41(2): 84-106.
- Hamzaoui, Kamel, Agnès Hamzaoui (2012) Immunological responses in patients with Behçet’s disease: advances in understanding. Expert Review of Ophthalmology 7(3): 261-270.
- Tong, Bainan (2019) Immunopathogenesis of Behcet's disease. Frontiers in immunology 10: 665.
- Mumcu, Gonca, Haner Direskeneli (2019) Triggering agents and microbiome as environmental factors on Behçet’s syndrome. Internal and Emergency Medicine 14(5): 653-660.
- Galeone, Massimiliano, Massimiliano D'Erme, Moretti S, Lotti T (2012) Potential infectious etiology of Behcet's disease. Pathology research international 2012: 595380.
- Yazici H, UgurluS, Seyahi E. Behçet syndrome: is it one condition? Clin Rev Allergy Immunol 43: 275-280.
- Seyahi E (2019) Phenotypes in Behçet’s syndrome. Intern Emerg Med 14: 677-689
- Yazici H, Seyahi E, Hatemi G, Yazici Y (2018) Behçet syndrome: a contemporary view. Nat Rev Rheumatol 14: 107-119.
- Rotondo C, Lopalco G, Iannone F (2015) Mucocutaneous involvement in Behçet’s disease: how systemic treatment has changed in the last decades and future perspectives. Mediators Inflame 2015: 451675.
- Volle G, Fraison JB, Gobert D, Goulenok T, Dhote R, et al. (2017) Dietary and nondietary triggers of oral ulcer recurrences in Behçet’s disease. Arthritis Care Res (Hoboken) 69: 1429-1436.
- Tugal-Tutkun I, Onal S, Altan-Yaycioglu R, Huseyin Altunbas H, et al. (2004) Uveitis in Behçet disease: an analysis of 880 patients. Am J Ophthalmol 138: 373-380.
- Nasr H, Scriven JM (2015) Superficial thrombophlebitis (superficial venous thrombosis). BMJ 350: h2039.
- Sarr SA, Fall PD, Mboup MC, Dia Khadidiatou, Bodian M, et al. (2015) Superior vena cava syndrome revealing a Behçet’s disease. Thromb J 13: 7.
- Saip S, Akman Demir G, Siva A (2014) Neuro-Behçet syndrome. Handb Clin Neurol 121: 1703-1723.
- Alibaz Oner F, Direskeneli H (2023) Update on the Diagnosis of Behçet’s Disease. Diagnostics 13(1): 41.
- (1990) Criteria for Diagnosis of Behçet’s Disease. International Study Group for Behçet’s Disease. Lancet 335: 1078-1080.
- Alibaz Oner F, Direskeneli H (2023) Update on the Diagnosis of Behçet’s Disease. Diagnostics 13(1): 41.
- (2014) International Team for the Revision of the International Criteria for Behçet's Disease (ITR-ICBD), Davatchi F, Assaad-Khalil S, Calamia KT et al. The International Criteria for Behçet’s Disease (ICBD): a collaborative study of 27 countries on the sensitivity and specificity of the new criteria. J Eur Acad Dermatol Venereol 28: 338-347.
- Hu CJ, Pan JB, Song G, Wen Xiao T, Yan Wu Z, et al. (2017) Identification of Novel Biomarkers for Behcet Disease Diagnosis Using Human Proteome Microarray Approach. Mol Cell Proteomics 16(2): 147-156.
- Mohamed C, Najib K, Essaadouni L (2015) Radiological findings in Behçet disease. Pan Afr Med J 20: 51.
- Essaadouni L, Jaafari H, Abouzaid CH, Kissani N (2010) Les manifestations neurologiques de la maladie de Behçet: étude de 67 patients. Revue Neurologique 166(8-9): 727-733.
- Afshin Borhani Haghighi, Sirous Sarhadi, Siamak Farahangiz (2011) MRI findings of neuro-Behcet's disease. Clin Rheumatol 30(6): 765-770.
- Vidaller A, Carratala J, Moreno R, Arbizu T, Rubio F, et al. (1988) Magnetic resonance imaging in neuro-Behset's disease. Br J Rheumatol 27(1): 79-80.
- Mendoza Pinto C, García-Carrasco M, Jiménez-Hernández M, Hernández CJ, Riebeling-Navarro C, et al. (2010) Etiopathogenesis of Behcet's disease. Autoimmunity reviews 9(4): 241-245.
- Adeeb F, Khan MU, Stack AG, Fraser AD (2017) Etiology, Immunopathogenesis and Biomarkers in Behçet’s disease [Internet]. Behcet’s Disease InTech.
- Kaya Tİ (2012) Genetics of Behçet's Disease. Patholog Res Int 2012: 912589.
- Gu F, Huang X, Huang W, Zhao M, Zheng H, et al. (2023) The role of miRNAs in Behçet's disease. Front Immunol 14: 1249826.
- Leccese P, Alpsoy E (2019) Behçet's Disease: An Overview of Etiopathogenesis. Front Immunol 10: 1067.
- Rokutanda R, Kishimoto M, Okada M (2014) Update on the diagnosis and management of Behçet's disease. Open Access Rheumatol 7: 1-8.
- Cantarini L, Pucino V, Vitale A, Talarico R, Lucherini OM, et al. (2016) Immunometabolic biomarkers of inflammation in Behçet's disease: relationship with epidemiological profile, disease activity and therapeutic regimens. Clin Exp Immunol 184(2): 197-207.
- Alibaz Oner F, Direskeneli H (2022) Update on the Diagnosis of Behçet's Disease. Diagnostics (Basel) 13(1): 41.
- Yücel Ç, Omma A, Sertoğlu E, Sezer S, Turhan T, et al. (2019) Evaluation of atherogenic laboratory markers in Behçet's disease patients with vascular involvement. Arch Med Sci 16(3): 531-537.
- Nair JR, Moots RJ (2017) Behcet's disease. Clin Med (Lond) 17(1): 71-77.
- Adeeb F, Khan MU, Stack AG, Fraser AD (2017) Etiology, Immunopathogenesis and Biomarkers in Behçet’s disease [Internet]. Behcet’s Disease. InTech.
- Qi L, Cai J, Mao D, Wang M, Ge X, et al. (2019) Use of contrast-enhanced computed tomographic imaging to diagnose and evaluate Behçet's disease with vascular complications. Exp Ther Med 18(6): 4265-4272.
- Alibaz-Oner F, Direskeneli H (2022) Update on the Diagnosis of Behçet's Disease. Diagnostics (Basel) 13(1): 41.
- Manzari RS, Mirfeizi Z, Poorzand H, Abbasi Shaye Z (2022) Echocardiography Parameters in Behcet's Disease, A Comparative Study. Curr Rheumatol Rev 19(2): 168-173.
- Scherrer MAR, Rocha VB, Garcia LC (2017) Behçet's disease: review with emphasis on dermatological aspects. A Bras Dermatol 92(4): 452-464.
- Gül A, Inanç M, Ocal L, Aral O, Koniçe M (2000) Familial aggregation of Behçet's disease in Turkey. Ann Rheum Dis 59(8): 622-625.
- Kapsimali VD, Kanakis MA, Vaiopoulos GA, Kaklamanis PG (2010) Etiopathogenesis of Behçet's disease with emphasis on the role of immunological aberrations. Clin Rheumatol 29(11): 1211-1216.
- Hatemi G, Seyahi E, Fresko I, Talarico R, Hamuryudan V (2015) Behçet's syndrome: a critical digest of the 2014-2015 literature. Clin Exp Rheumatol 33(6Suppl94): S3-14.
- (2022) Criteria for diagnosis of Behçet's disease. International Study Group for Behçet's Disease. Lancet 335(8697): 1078-80.
- Kaneko F, Takahashi Y, Muramatsu R, Adachi K, Miura Y, et al. (1985) Natural killer cell numbers and function in peripheral lymphoid cells in Behcet's disease. Br J Dermatol 113(3): 313-318.
- Chun SI, Su WP, Lee S (1990) Histopathologic study of cutaneous lesions in Behçet's syndrome. J Dermatol17(6): 333-341.
- Demirkesen C, Tüzüner N, Mat C, Senocak M, Büyükbabani N, et al. (2021) Clinicopathologic evaluation of nodular cutaneous lesions of Behçet syndrome. Am J Clin Pathol 116(3): 341-346.
- Ozluk E, Balta I, Akoguz O, Kalkan G, Astarci M, et al. (2014) Histopathologic Study of Pathergy Test in Behçet's Disease. Indian J Dermatol 59(6): 630.
- Ishibashi H (2018) What Is Vascular Behçet's Disease? Ann Vasc Dis 11(1): 52-56.
- Amahzoune B, Boulahya A, Selkane C, Houssa MA, Bekkali Y, et al. (2002) Manifestations artérielles de la maladie de Behç A propos de 5 cas opérés [Arterial manifestations of Behcet's disease. A report of five operated cases]. Arch Mal Coeur Vaiss 95(2): 109-116.
- Paovic J, Paovic P, Sredovic V (2013) Behcet's disease: systemic and ocular manifestations. Biomed Res Int 2013: 247345.
- Afifi AK, Frayha RA, Bahuth NB, Tekian A (2018) The myopathology of Behçet's disease--a histochemical, light-, and electron-microscopic study. J Neurol Sci 48(3): 333-342.
- Jawad ASM, Petrushkin H (2024) Challenges in the diagnosis of Behçet's disease. International journal of rheumatic diseases 27(1): e14914.
- Seyahi E, Melikoglu M (2021) Differentiating Behçet’s disease from other rheumatic diseases. Best Practice & Research Clinical Rheumatology 35(1): 101653.
- Erdem Sultanoğlu T, Eröz R, Ataoğlu S (2023) Evaluation of HLA-B51 frequency and its relationship with clinical findings in patients with Behçet’s disease: 4-year analysis in a single center. Egypt Rheumatol Rehabil p. 15.
- Tugal-Tutkun I, Onal S, Gül A (2021) Multidisciplinary management of Behçet's disease. Ocular immunology and inflammation 29(4): 652-662.
- Suhler EB, Martin TM, Mermelstein J (2023) Uveitis in Behçet's Disease. American Journal of Ophthalmology 234: 180-187.
- Kiafar M, Faezi ST, Kasaeian A, Baghdadi A, Kakaei S, et al. (2021) Diagnosis of Behçet's disease: Clinical characteristics, diagnostic criteria, and differential diagnoses. BMC rheumatology 5(1): 2.
- Tuğal-Tutkun İ, Onal S, Stanford MR, Akman M, Twisk JW, et al. (2020) An algorithm for the diagnosis of Behçet disease uveitis in adults. Ocular immunology and inflammation 29: 1154-1163.






























