Essential oil Composition, Antioxidant and Antibacterial Activities of Jatropha tanjorensis (Euphorbiaceae)
Oluwakayode Olubunmi Odeja1, Ejike Onwudiegwu Okpala2*, Michael Gabriel Ibok3*, Emeka Emea Okoro4, Joel Ojogbane Onoja5 and Chukwuemeka Clinton Ndukaji1
1Department of Chemistry, Federal University of Petroleum Resources, Effurun, Delta State, Nigeria
2Department of Chemistry, Faculty of Science, Federal University Lokoja, Kogi State, Nigeria
3Department of Chemistry, University of Ibadan, Ibadan, Oyo State-Nigeria and Organic Chemistry Section, Chemical Science division, National Institute for Interdisciplinary Science & Technology (CSIR-NIIST), CSIR - Council of Scientific & Industrial Research, Thiruvananthapuram, India
4Biochemistry Department, Adventist University of West Africa, Monrovia, Liberia
5Department of Pharmacognosy and Environmental Medicine, Faculty of Pharmaceutical Sciences, University of Nigeria, Nsukka, Nigeria
Submission: April 23, 2023; Published: May 01, 2023
*Corresponding author: Ejike Onwudiegwu Okpala, Department of Chemistry, Faculty of Science, Federal University Lokoja, Kogi State, Nigeria, email: ejike.okpala@fulokoja.edu.ng
Michael Gabriel Ibok, Department of Chemistry, University of Ibadan, Ibadan, Oyo State-Nigeria and Organic Chemistry Section, Chemical Science division, National Institute for Interdisciplinary Science & Technology (CSIR-NIIST), CSIR - Council of Scientific & Industrial Research, Thiruvananthapuram, India, Email: michaelibok@ymail.com
How to cite this article: Oluwakayode O, Ejike Onwudiegwu O, Michael G I, Emeka Emea O, Joel O O. Essential oil Composition, Antioxidant and Antibacterial Activities of Jatropha tanjorensis (Euphorbiaceae). Ann Rev Resear. 2023; 9(1): 555751. DOI: 10.19080/ARR.2023.09.555751
Abstract
Background: Jatropha tanjorensis (Euphorbiaceae) leaves are traditionally used for the treatment of diabetes and also, possess antibacterial potential. The chemical composition, antioxidant and antibacterial activities of the oil were investigated. The essential oil (EO) was obtained by hydro-distillation method and characterised using gas chromatography-mass spectrometry (GC-MS). The antioxidant activity was investigated using 2, 2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging technique and antibacterial activity of the volatile oil was evaluated against 6 strains of bacteria: Escherichia coli, Pseudomonas aeruginosa, Klebsiella pneumonia, Staphylococcus aureus, Bacillus subtilis and Salmonella typhi, using broth dilution method.
Results: The EO produced a percentage yield of 0.3% (w/w) and a total of 48 compounds were identified in J. tanjorensis leaf EO accounting for 75.16% of the total oil composition. The dominant compounds identified in the EO were phytol (8.92%), n-Nonacosane (7.35%), and β-Cis-Ocimene (7.29%). The EO showed a potent antioxidant activity when compared to the Butylated hydroxyanisole used as a standard drug, with % inhibition varying from 93.21 ± 0.02 to 92.90 ± 0.01 at 1000-125 μg/mL. The leaf essential oil exhibited good antibacterial activity on all the tested organisms at 1000-125 μg/mL.
Conclusion: The chemical composition and biological properties of J. tanjorensis leaf EO was determined for the first time and findings support the rationale for its medicinal applications.
Keywords: Jatropha tanjorensis; Antioxidant; Antibacterial activity; Gas chromatography-Mass spectrometry; Essential oil
Abbreviations: EO: Essential oil; GC-MS: Gas chromatography-mass spectrometry; DPPH: 2, 2-diphenyl-1-picrylhydrazyl; J. tanjorensis: Jatropha tanjorensis; RT: Retention time; RI: Kovats retention index; JTE: Leaf essential oil of Jatropha tanjorensis; BHA: Butylated hydroxyanisole; ZOI: zone of inhibitory
Background
A medicinal plant chemical constituent contains substances that have therapeutic effects, or which are precursors for the synthesis of useful drugs. Various parts of plants such as the leaf, stem, bark, root, etc. are being used to prevent and cure various like malaria, diarrhea, tuberculosis, pneumonia, and asthma. Most of the potent medicinal plants have relatively no toxic or adverse effects when used by humans, their usage presently is on the increase due to easy availability, affordability, accessibility, and promising efficacy comparable to the often-high cost of the standard synthetic drugs Bakkali [1]; Bruneton [2] Jatropha tanjorensis belongs to the Euphorbiaceae family. It is a gregarious shrub and commonly called “Hospital too far” in Nigeria to portray its medicinal potential. J. tanjorensis leaf is edible and serves as a food in southern Nigeria. The leaf extracts have been reported to be used in the treatment of diabetes or hypoglycaemia Olayiwola [3]. All parts of the plant including seeds, leaves, and fresh stem bark or decoction of the plants’ parts are used in traditional and folk medicine. The leaves of J. tanjorensis also have been reported to have the potential for the treatment of cardiovascular diseases Oyewole & Akingbala [4]. The fresh leaves of J. tanjorensis are reported to possess blood-replenishing properties and are used in treating blisters, dry skin, and bruises. It is also having an antibacterial activity Iwalewa [5]; Arsari [6]. In continuation of our search for new potent antioxidants and antibacterial from plants with ethno-medicinal reported uses Odeja [7]; Odeja [8]; Okpala [9]; Okpala [10], we report the chemical composition, antioxidant and antibacterial activities of leaves essential oil of J. tanjorensis which has not been reported in the literature.
Methods
Plant material
The fresh leaves of J. tanjorensis were collected from the Nsukka, Enugu State of Nigeria (6o51’ 39’’N; 7o 24’21’’E). The plant was identified by Dr. S. K. Odewo of the Forestry Research Institute of Nigeria (FRIN) Ibadan, Oyo State, Nigeria.
Isolation of essential oil
The fresh leaves were air-dried and pulverized. The essential oil was extracted using the Hydro distillation method with all glass Clevenger-type apparatus for 3 hours according to the British Pharmacopoeia specifications (British Pharmacopoeia [11]). The percentage yields (w/w) of volatile oils obtained were calculated and the oil was preserved at 4oC using a refrigerator before being subjected to GC-MS analysis and biological assay.
Gas chromatography-mass spectrometry of the volatile oil
The essential oil was subjected to GC-MS analysis on an Agilent 7809A Gas Chromatography hyphenated with an Agilent mass detector having split/split less injector interfaced with a mass selective detector operating at 70 eV. The ion source temperature was set to 200°C over a mass spectral range of m/z 50–700 at a scan rate of 1428 amu/sec. The column of the GC used was HP- 5MS with a length of 30 m, an internal diameter of 0.25 mm and a film thickness of 0.25μm. The oven temperature was programmed at an initial temperature of 80 °C for 2 min, increased at 10 °C/min to the final temperature of 240 °C for 6 mins. Helium was used as the carrier gas at a flow rate of 1 mL/min. Injection volume, linear velocity, and pressure were adjusted at 1.0μl, 362 cm/s, and 56.2 KPa, respectively. The relative percentages of the essential oil components were obtained by FID peak-area normalization. Identification of the essential oil components was based on a comparison of their mass spectral fragmentation patterns with a pre-installed NIST MS search 2.0 database NIST data and with the data previously reported data in the literature.
Antioxidant assay
The antioxidant investigation of the J. tanjorensis leaves essential oil was studied using the 2, 2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging ability method, with some modifications Brand-Williams [12]. Five concentrations of the volatile oils and standard antioxidant (Butylated hydroxyl anisole) (1000-125) μg/mL were prepared. 2.0 mL of 100 μM methanolic DPPH solution (used as control) was added to the five concentrations of volatile oils and incubated at room temperature for 30 minutes. The absorbance of the treated essential oil samples at different concentrations and the blank 100 μM methanolic DPPH solution (control) were then measured at 517 nm with a GS UV-12, UV-V is spectrophotometer. The analyses were carried out in triplicates and the average values were obtained. The activities of all the analysed samples were calculated as a function of their % inhibition using the equation.
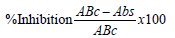
Where ABc = absorbance of control; Abs = absorbance of samples
Antibacterial assay
Test organisms
Six strains of bacteria; four Gram-negative: Escherichia coli, Salmonella typhi, Klebsiella pneumonia, and Pseudomonas aeruginosa and two Gram-positive: Staphylococcus aureus and Bacillus subtilis were used in this study. All bacteria microbes were clinical isolates obtained from the Department of Pharmaceutical Microbiology, University of Ibadan, Nigeria.
Preparation of Sample Solution
Oil samples were prepared such that 1 mL of the oil was regarded as 1000 μg/mL; 0.5 mL of this essential oil was taken into 0.5 mL of methanol to give 500 μg/mL. More serial dilutions gave different concentrations such as 250 μg/mL, 125 μg/mL and 62.50 μg/mL. The 6th and 7th test tubes were negative control (DMSO) and positive controls (Gentamycin) respectively [13].
Antibacterial Assay – Broth Dilution Method
Overnight cultures of microorganisms were prepared by taking two loopful of already grown organisms from the stock and inoculating each into the sterile nutrient broth of 5 mL each for 18-24 hrs at 37 ºC. The inoculation of six strains, from standardised suspensions, was made using a Sterr multi- inoculator using suspensions standardised at 0.5 MacFarland, with a second dilution performed onto BHI to obtain inoculum of an approximate concentration of 105–106 CFU/mL according to the method described by [14]. From the overnight culture, 0.1 mL of each organism was added to 9.9 mL of sterile saline (0.85 %) to get 10-2 dilutions of each organism. From the diluted organism, 0.2 mL was taken into the prepared sterile nutrient agar, which was at 45 ºC. This was aseptically poured into the concentrations of each essential oil and the control. The plates were incubated for 18-24 hrs at 37 ºC. Each experiment was carried in duplicates and the antimicrobial activity was evaluated by measuring the diameter of the inhibition growth zone in millimeters (mm) for the test organisms compared with Gentamicin (10 μg/mL) which was used as a positive control.
Statistical Analysis
All data represent the means of three replicates ± standard deviation. Results were subjected to analysis of variance (ANOVA) and the mean comparisons were performed by Bonferroni’s Multiple Comparison Test using the statistical analysis software package (GraphPadPrism 5).
Results
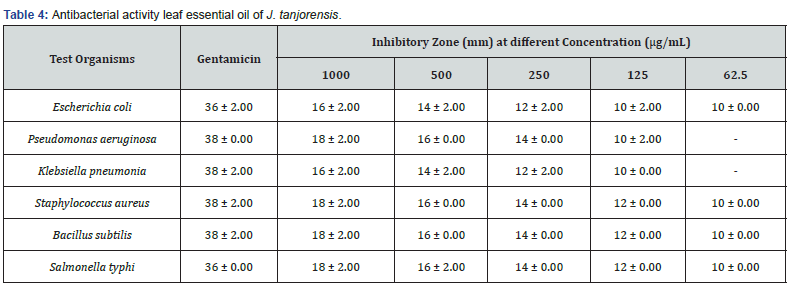
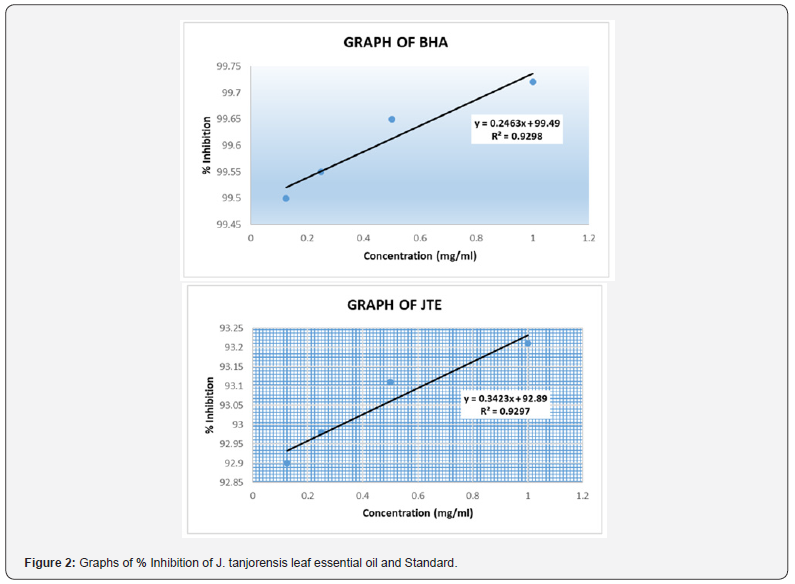
Fresh leaves of J. tanjorensis (300 g) were used in hydrodistillation extraction to produce 0.9 g of colourless volatile oil with a yield of 0.3% (w/w) (Table 1). The GC-MS chromatogram of the essential oil is given in Figure 1. The chemical compositions of the essential oil were presented in Table 2. Forty-eight compounds were identified from leaf essential oil of J. tanjorensis accounting for 75.16% of the total composition of the oil. The classes of compounds identified in the oil are sesquiterpenes and oxygenated sesquiterpenes (31.63%), n-alkanes (13.73%), diterpene (8.92%), monoterpene (7.29%), alkanoic acids (4.02%), esters (3.87%), alkanols (2.37%), alkynes (2.06%) and alkenes (1.27%) (Table 2). The DPPH radical scavenging activity of the essential oil of the leaves of J. tanjorensis is presented in Table 3, and the antibacterial activity of the volatile oil was determined based on inhibition zones observed against the four gram-negative and two gram-positive bacteria strain at various concentrations (1000-62.5 μg/mL) as compared to Gentamycin (standard antibacterial drug) (Table 4) & (Figure 2).

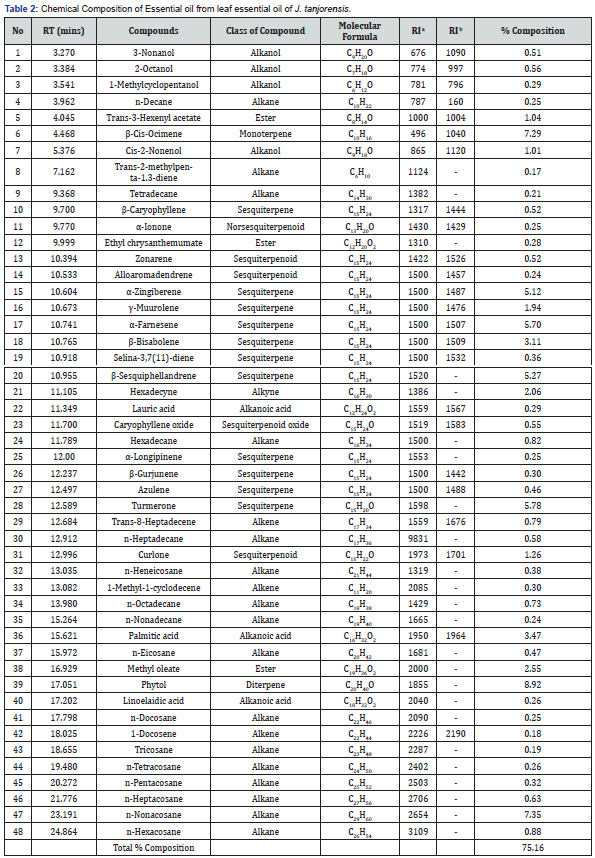
RT = Retention time; RIa = Kovats retention index (calculated); RIb = Kovats retention index (Literature).
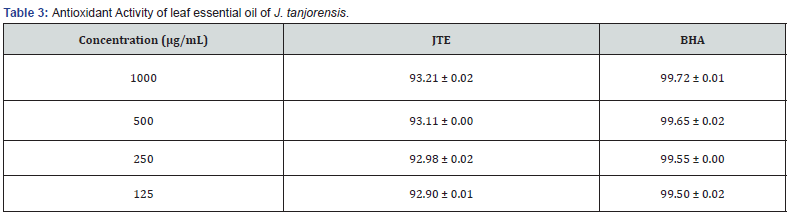
JTE: Leaf essential oil of Jatropha tanjorensis, BHA: Butylated hydroxyanisole, ± = Standard deviation.
Discussion
A total of 48 compounds were characterized in the leaf essential oil of J. tanjorensis which comprises mainly of terpene compounds (47.84%), n-alkanes (13.73%), and other non-terpene compounds (13.59%). The most abundant compound identified in the oil was phytol (8.92%) and other dominant compounds in the leaf essential oil are n-Nonacosane (7.35%), β-Cis-Ocimene (7.29%), Turmerone (5.78%), α-Farnesene (5.70%), β-Sesquiphellandrene (5.27%) and α-Zingiberene (5.12%). The identified chemical constituents from essential oil J. tanjorensis are similar to previously reported compositions of essential oils from plants in the Jatropha genus. A previous study on Jatroph intigrimma, Jatroph gossypifolia and Jatroph roseae Gamal El-Din [15] showed Heneicosane, phytol and nonacosame as the major constituents which were also identified in our study. Previous study on essential oils of Jatropha curcas Adeosun [16], Jatropha mutabilis Costa [17] and Jatropha gossypifolia Aboaba [18], reported phytol as the major compound; the dominance of sesquiterpenes and oxygenated sesquiterpenes in their essential oils, which is almost relative to our study.
The calculated percentage inhibition shows that the antioxidant activity of leaves essential oils of J. tanjorensis and standard (Butylated hydroxyanisole) is concentration dependant.
The essential oil at all tested concentrations (1000-6.25 μg/mL) exhibited good scavenging ability on DPPH radical which was comparable to Butylated hydroxyanisole. The leaf essential oil exhibited good antibacterial activity at 1000-125 μg/mL with the zone of inhibitory (ZOI) 18 ± 2.00 - 10±2.00 mm, against all the tested organisms as compared to 38±2.00 - 36±0.00 ZOI recorded for Gentamycin; a known antibacterial drug at the same concentrations. The leaf essential oils of J. tanjorensis showed sensitivity for Escherichia coli, staphylococcus aureus, Bacillus subtilis and salmonella typhi at all tested concentrations. The leaves and stem essential oils of Jatropha gossypifolia have been reported to have shown potent antibacterial and antioxidant activities Okoh [19].
Conclusions
Our study on the chemical composition, antioxidant and antibacterial properties of the essential oil from J. tanjorensis leaf is presented here to the best of our knowledge for the first time. The biological properties of J. tanjorensis leaf EO serve as the foundation for its pharmaceutical applications.
References
- Bakkali F, Averbeck S, Averbeck D, Idaomar M (2008) Biological effects of essential oils- a review. Food and Chemical Toxicology 46(2): 446-475.
- Bruneton J (1995) Pharmacognosy, Phytochemistry and Medicinal Plants. Lavoister publishing Paris pp. 538-544.
- Olayiwola G, Iwalewa EO, Omobuwajo OR, Adeniyi AA, Verspohl EJ (2004) The antidiabetic potential of Jatropha tanjorensis Nigerian Journal of Natural Products and Medicine 8: 55-58.
- Oyewole IQ, Akingbala PF (2011) Phytochemical analysis and Hypolpidemic properties of Jatropha Tanjorensis leaf extract. European Journal of Medicinal Plants 1(4): 180-185.
- Iwalewa EO, Adewumi CO, Omisore NO, Adebanji OA, Azike CK (2005) Pro- and antioxidant effects and cytoprotective potentials of nine edible vegetables in Southwest Nigeria. J Med Food 8(4): 539-544.
- Arsari RA, Rabiu KM, Ayuba V, Omolabake OO (2020) Review on Jatropha Tanjorensis (Hospital too far); significance as an anti-anaemia plant. International Blood Research & Reviews 11(4): 1-7.
- Odeja OO, Ibok MG, Okpala EO (2020) Chemical Composition, Antimicrobial and Antioxidant Activities of Root Essential Oil of Nigerian Specie of Asparagus Flagellaris (Kunth) Baker, International Journal of Innovative Research & Development 9(5): 2278-0211.
- Odeja OO, Ibok MG, Okpala EO (2021) Composition and biological assays of the leaf essential oil of Asparagus Flagellaris (Kunth) Bak. Clinical Phytoscience 7: 12.
- Okpala EO, Onocha PA, Ali MS, Zikr-Ur-Rehmen S, Lateef M (2021) Zenkeramide: a new iso-benzofuranone propanamide and urease inhibitory constituents of Celtis zenkeri Engl stem bark (Ulmaceae). Nat Prod Res 37(1): 93-98.
- Okpala E, Onocha P, Ali M (2022) Antioxidant activity of phytol dominated stem bark and leaf essential oils of Celtis zenkeri Engl., Trends in Phytochemical Research 6(2): 137-144.
- (1981) British Pharmacopoeia, London, HM Station p. 109.
- Brand-Williams W, Cuvelier ME, Berset C (1995) Use of a free-radical method to evaluate antioxidant activity. Food Science and Technology 28(1): 25-30.
- Kalemba D, Kunicka A (2003) Antibacterial and antifungal properties of essential oils. Curr Med Chem 10(10): 813-829.
- Andrade BFMT, Barbosa LN, Probst ID, Júnior AF (2014) Antimicrobial activity of Essential Oils. Journal Essential Oil Research 26(1): 34-40.
- Gamal El-Din MI, Youssef FS, Altyar AE, Ashour ML (2022) GC/MS Analyses of the Essential Oils Obtained from Different Jatropha Species, Their Discrimination Using Chemometric Analysis and Assessment of Their Antibacterial and Anti-Biofilm Activities. Plants 11(9): 1268.
- Adeosun TE, Ogunwande I.A, Avoseh ON, Raji IP, Lawal OA (2017) Composition and Anti-inflammatory Activity of Essential Oil of Jatropha curcas, Natural Product Communication 12(3): 439-440.
- Costa E, Oliveira AP, Almeida J, Filho JS, Araujo E (2014) Chemical Composition of Essential Oil from the Leaves of Jatropha mutabilis (Pohl) Baill (Euphorbiaceae). Journal of Essential Oil-Bearing Plants 17(6): 1156-1160.
- Aboaba SA, Adebayo MA., Ogunwande IA, Olayiwola TO (2015) Volatile constituents of Jatropha gossypifolia L. grown in Nigeria. American Journal Essential Oils and Natural Products 2(4): 8-11.
- Okoh SO, Iweriebor BC, Okoh OO, Nwodo UU, Okoh AI (2016) Antibacterial and antioxidant properties of the leaves and stem essential oils of Jatropha gossypifolia L. BioMed Research International 2016: 2716.






























