Oil Spills Weathering
Yuriy Dachev* and Dobrin Milev
Nikola Vaptsarov Naval Academy, Varna, Bulgaria
Submission: November 12, 2022; Published: December 22, 2022
*Corresponding author: Yuriy Dachev, Nikola Vaptsarov Naval Academy, Varna, Bulgaria
How to cite this article: Yuriy D, Dobrin M. Oil Spills Weathering. Ann Rev Resear. 2022; 8(1): 555730. DOI: 10.19080/ARR.2022.08.555730
Abstract
Oil spills often generate a great deal of concern about their potential effects on environment. The environment that may be affected by spilled oil consists of several compartments: air, shoreline, sea surface, water column and sediments. Each of these compartments, except air, may contain a variety of different habitats, each with its own populations of species and individuals. Natural processes force spilled oils, or its chemical components, to be transferred from one environmental compartment to another.
Keywords: Oil spill; Environment; Weathering; Biodegradation; Spreading; Water column
Physical and Chemical Properties of Oil
A wide range of oil products is of concern to oil spill responders. These are mainly crude oils and heavy fuel oils. However, under certain circumstances, some other refined products such as gasoline and diesel will be of concern for responders. Occasionally spills of vegetable or animal oils occur. The content and proportion of various hydrocarbon compounds give a particular product its properties.
Crude Oil
Crude oil contains a wide range of compounds, from dissolved gases (or light ends) such as methane and propane at one extreme to heavy compounds such as waxes and asphaltenes. The variable content of these compounds in crude oils means that their behavior in an oil spill is also very different. The content of these compounds will also change as the oil “weathers” causing the oils properties to change.
1.1.1. Refined Products: Typical spilled refined products:
a) Gasoline, from the lighter end of the crude oil.
b) Diesel, a slightly heavier cut from the crude but still fairly light.
c) Residual fuel oils such as heavy fuel.
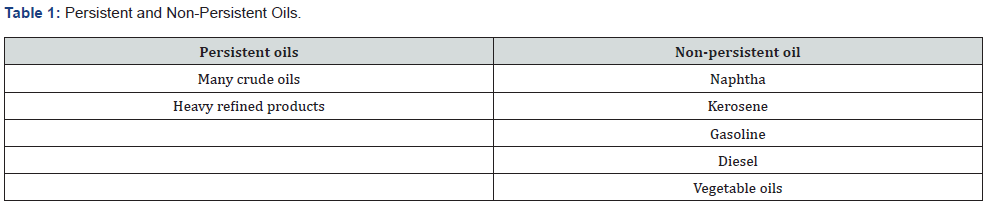
1.1.2. Persistent and Non-Persistent Oils: Non-persistent oils tend to disappear rapidly from the sea surface whereas persistent oils dissipate more slowly and usually require a clean-up response when spilled or threatening coastal waters. Examples of persistent and non-persistent oils are listed below.
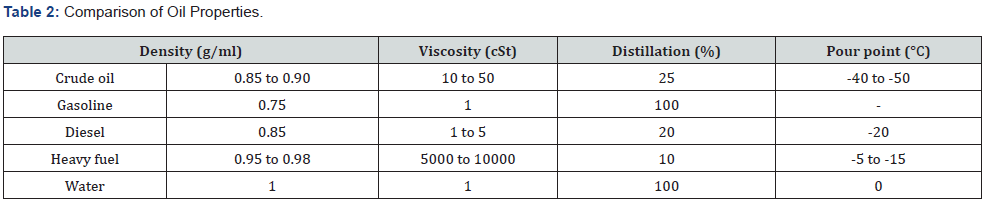
Oil has two important properties which both change during an oil spill:
a) Density (specific gravity/buoyancy) - oil’s ability to float
b) Viscosity - how much it flows - how thick it is?
As the light ends of the spill evaporate, the oil becomes heavier (more dense) and may then sink below the surface. As oil weathers it becomes thicker (viscosity increases) and more difficult to pump. It also spreads more slowly. Under warm conditions, viscosity reduces, and the oil becomes thinner and spreads more quickly. The viscosity is a physical property which has influence on the choice of skimmers used in oil spill response.
Other properties are:
i. Volatility (distillation characteristics): This describes oil tendency to evaporate. As the temperature of oil is raised different components reach their boiling point in turn and are distilled (evaporate).
ii. Pour Point: Pour point is the temperature below which oil will not flow. It represents the point at which the oil starts to solidify or gel.
iii. Asphaltene Content: These complex cyclic hydrocarbon compounds tend to enhance formation of stable water-in-oil emulsion (“chocolate mousse”), which may have higher viscosity and persistence than the parent oil [1].
iv. Flash Point: This is the lowest temperature at which vapors above a volatile substance will ignite in air when exposed to a flame.
v. Surface Tension: The surface tension is an indicator of oil tendency to spread and disperse.
Spreading and Movement of Oil
Spreading
When spilled in open water, oil rapidly spreads to form a thin layer that covers a large area. The rate of spreading is particularly high during the early stages of the spill, after which the rate gradually decreases.
The spreading rate is dependent upon oil properties (i.e., specific gravity, surface tension, and viscosity), volume, temperature, wind velocity, waves and current. Although many factors influence the spreading rate and the ultimate size of the slick, approximations can be made using spreading tables if the spill volume and general oil type are known. For example, after 2 hours, a 5000 t spill of crude oil with a density of 0.875 and a viscosity of 10 cSt will cover an area of 0.5 km2. After 24 hours it will cover an area of 1.7 km2 [2]. Note that there is rapid initial spreading of the slick, and that for a specific spill volume, there is an equilibrium area beyond which the oil will not spread. The average slick thickness can be estimated by dividing the spill volume by the spill area. However, actual sea conditions result in variations in thickness inside a spill area – thick or thin portions of slick, windrows (streams of thick oil separated by open water, tar balls). Typically, oil spilled on water will form a thin lens with the inner portion thicker than the edges and with a major part of the total volume in the up-wind part of the slick.
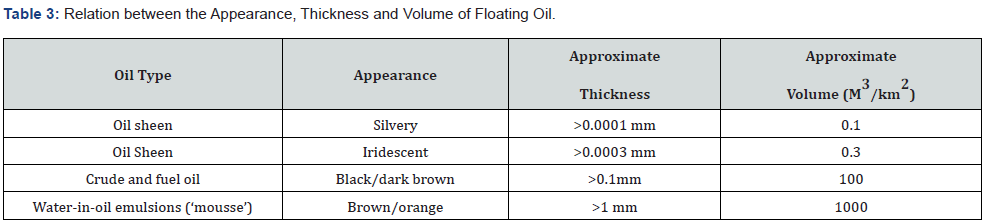
The bulk of the spill is thus contained in a relatively small area of thick oil surrounded by a thin sheen covering a much greater area. A rule of-thumb for this breakdown is that 90 % of the oil is contained in 10% of the area, with remaining 10% of the oil covering 90% of the total slick area [3]. Most crude oils spread to a thickness of approximately 0.3 mm within 12 hours. However, some crude oils and heavy fuel oils are exceptionally viscous and tend not to spread much but remain in rounded patches. The volume of the oil layer in a given area can to a certain extent be estimated by visual means as the color of the oil on the sea surface is normally directly related to the thickness of the layer. Time and the spreading process both detract from the efficiency of any oil spill response. Over time, oil slicks will spread to cover unmanageable areas, with slick thicknesses that are too thin for effective removal by skimmers. Therefore, if we want to be effective at cleaning up the spill, a rapid response is essential.
Movement
An oil slick is carried by the surface layer on the upper few centimeters of water. The driving forces for this surface layer include wind, the local components of large-scale circulation patterns, tidal influences and freshwater inflows. It is important to note that each of these forces will vary through time and as the slick moves from one area to another. It may be difficult to measure one or more of them at the time of a spill. An approximation of slick movement can be made using some very simple methods. Basic rule-of-thumb: the slick will move at about 3% of the wind speed in a direction 10° to 15° to the right of the wind direction, with the speed and direction modified by the addition of other water currents. In open water (away from the effects of near shore currents, tidal currents, and freshwater influences), a good approximation is to consider only the effect of the wind. Example using the 3% rule: a 30 km/h (16 knots) wind will move a slick at about 0.9 km/h (0.5 knots). Other currents - tidal, freshwater influences - are added to the wind effects using vector addition. This graphical technique can be done in units of either distance or speed but must be consistent through the calculation and must be for the same time period. Represent each of the wind and current components by an arrow, the length of each one scaled according to the speed (or distance) and drawn in the appropriate direction. If the two arrows or vectors are drawn with one starting from the point where the other ends, then the resulting vector - the vector that completes the triangle - is the predicted slick movement vector [3].
Key Spills Processes
When oil is spreading and moving over the sea surface, a series of natural processes change the physical and chemical properties of the oil. This includes spreading, evaporation, dispersion, emulsification, dissolution, biodegradation, oxidation and sedimentation [4].
Evaporation
Light volatile components of a slick evaporate which leads to changes in the properties of the remaining oil. The residue remaining on the sea surface has a higher density and viscosity than the original oil. Evaporation of light crude oils and light refined products also leads to a significant reduction in total spill volume. This evaporation can cause fire and explosion.
The factors which control evaporation rate are:
a) Chemical composition of the oil
b) Slick Thickness: the evaporation rate increases as the slick thickness declines
c) Temperature: oil will evaporate faster with higher temperatures (note that although evaporation rates will be lower in cold temperatures, gasoline and most crude oils will still evaporate at freezing temperatures)
d) Wind speed: the greater the wind speed, the greater the potential evaporation rate
e) Sea state: the higher the sea state, the greater the potential evaporation rate.
Under certain external circumstances, volume loss for light refined products and crude oils could be in the range of 25 to 30% within 12 hours and up to 50% within one day. Gasoline can evaporate 100%. Heavy crude oil and heavy fuel oil lose very little volume [2].
Dispersion
The process of natural dispersion - as opposed to dispersion aided by the addition of chemical dispersion agents - can be an important process for the removal of oil from the water surface. Small droplets of oil will tend to be “permanently” suspended in the water column; their natural buoyancy unable to overcome the forces of large scale mixing currents through the water body. This process can be enhanced with chemical dispersants. Refined products such as gasoline, diesel and most crude oils are prone to dispersion given the right mixing conditions. The onset of weathering and emulsification will limit or stop natural dispersion. It tends to compete with other weathering processes, such as emulsification.
Emulsification
Emulsification is important because:
i. It is a process in which water is incorporated into the slick, leading to increases in the total volume of spilled product.
ii. It leads to tremendous increases in the viscosity of the slick, which makes it resistant to natural dispersion and makes it more difficult to recover with skimmers and transfer with pumps.
Emulsification tends to compete with the dispersion process in that dispersion will essentially cease once oil emulsifies. Compare the emulsification process with the dispersion process: at the same time as wave energy is mixing oil into the water, the same energy is mixing water into the oil. Emulsions may contain as much as 80 to 90% water, that is, four to nine parts waters for one part oil. This has huge implications for the total spill volume that has to be dealt with and for activities such as skimming, transfer, storage and disposal. To some degree, the evaporation, dispersion and emulsification processes interact. For example, as oil evaporates it becomes prone to emulsification and if oil emulsifies it will resist dispersion.
Dissolution
Losses from dissolution are relatively low since most petroleum hydrocarbons have a low solubility in seawater.
Biodegradation
This is the process whereby oil is broken down by microorganisms that occur naturally in the marine environment.
Biodegradation is a slow process which is very dependent on ambient temperatures, and which will do little to moderate the effects of a large spill during the duration of the oil spill combating operation. However, it is of some interest in terms of how the environment recovers from a spill over periods of months or years [5].
Oxidation
Some hydrocarbons can react with oxygen to form products which are soluble in water or to form persistent tars. Many of these oxidation reactions are promoted by sunlight (photo-oxidation). Although they occur throughout the lifetime of a slick, the effect on the overall dissipation is minor in relation to the weathering process [6].
Sedimentation
Sedimentation is sometimes classified as a weathering phenomenon as evaporation and emulsification increasing the density of the oil. Some heavy refined products have a specific gravity greater than 1, and so will sink in fresh or brackish water. However, very few crude oils are sufficiently dense, or weather to such an extent that their residues will sink in sea water. Sinking is usually brought about by the adhesion of particles of sediment or natural organic matter to the oil [7]. Shallow waters and river estuaries are often laden with suspended solids and thus provide favorable conditions for sedimentation. Temperature may also affect the behavior of neutrally buoyant oil; while it barely floats during a cold day, it may sink as the temperature falls during night and resurface later on a warmer day [8-10].
Conclusion
Evaporation transfers the more volatile components from the oil into the air. The limited proportion of oil components that are water soluble will dissolve into the water column. The action of breaking waves will transfer some oil from the sea surface into the water column as small oil droplets – this is known as natural dispersion. Spilled oil that is naturally dispersed into the water column in shallow waters by waves in the surf-zone may become incorporated into near-shore sediments. Spilled oil on the sea surface will drift under the influence of the wind and current and may stand ashore. Oil can eventually become negatively buoyant by loss of volatile components, emulsification, biodegradation and interaction with sediments suspended in the water column and sink to the seabed.
References
- Bobra M (1991) Water-in-Oil Emulsification: A Physiochemical Study. In International Oil Spill Conference Proceedings: March 1991(1): 483-488.
- (1989) ITOPF Technical Information Paper No. 11 Fate of oil spills.
- (2001) ITOPF Technical Information Paper No. 1 Aerial observation of oil spills.
- Payne JR, Mc Nabb GD (1984) Weathering of petroleum in the marine environment. In Marine Technology Society Journal 18(3): 24-40.
- Atlas RM, Cerniglia CE (1995) Bioremediation of Petrolium Pollutants. In Bioscience 45(5) 332-338.
- Atlas RM, Bartha R (1992) Hydrocarbon biodegradation and oil spill bioremediation. In Advances in Microbial Ecology 12: 287-338.
- Daling PS, Brandvik PJ, Mackay D, Johansen O (1990) Characterization of crude oils for environmental purposes. In Oil and chemical pollution 7: 199-224.
- Di Toro DM, Mc Grath JA, Stubblefield WA (2007) Predicting the toxicity of neat and weathered crude oil: Toxic potential and the toxicity of saturated mixtures. In Environmental Toxicology and Chemistry 26(1): 24-36.
- Pace CB, Clark JR, Bragin GE (1995) Comparing crude oil toxicity under standard and environmentally realistic exposures. In International Oil Spill Conference Proceedings 1: 1003-1004.
- Prince RC (1997) Bioremediation of marine oil spills. In Trends in Biotechnology 15(5): 158-160.






























