Molecular Modeling of Phenyl-Boronic Acids and Catechol-Borate Esters: A Mechanistic Rationale for Boron Binding to Melanin Catechols, Regarding the 10Boron Neutron-Capture Therapy of Melanoma
Juan Carlos Stockert1,2*
1Institute of Oncology “Angel H. Roffo”, Research Area, Faculty of Medicine, University of Buenos Aires, C1417DTB, Buenos Aires, Argentina
2Integrative Center of Biology and Applied Chemistry, University Bernardo O’Higgins, Santiago, 8370854, Chile
Submission: September 08, 2022; Published: September 19, 2022
*Corresponding author: Juan C Stockert, Institute of Oncology “Angel H. Roffo”, Research Area, Faculty of Medicine, University of Buenos Aires, C1417DTB, Argentina
Felipe Velasquez-Botero, CES University, Larkin Community Hospital, Miami, Florida, USA
How to cite this article: Juan C. Stockert. Molecular Modeling of Phenyl-Boronic Acids and Catechol-Borate Esters: A Mechanistic Rationale for Boron Binding to Melanin Catechols, Regarding the Boron Neutron-Capture Therapy of Melanoma. Ann Rev Resear. 2022; 8(1): 555726. DOI: 10.19080/ARR.2022.08.555726
Abstract
The 10Boron neutron-capture therapy (BNCT) is an emerging anti-tumor therapy based on the accumulation of a 10B isotope within the tumor followed by irradiation with low-energy neutrons. The nuclear fission process produces 7lithium, 4helium, and λ rays within the tumor tissue, inducing specific tumor cell damage and death. 10B can be delivered either as simple B-containing agents or as B clusters. In the first case, boronic acid derivatives such as 4-borono-phenylalanine (BPA) are currently applied for BNCT of melanoma and other tumors. Although it can be assumed that BPA would act as a precursor metabolite for melanin biosynthesis, the precise action mechanism is not yet clear. Direct binding of this boronic acid to a target substrate, namely the catechol groups from already synthesized melanin, could be an alternative mechanism. In keeping with this, high affinity for melanin would explain the action mode of boronic acids as melanoma seekers. DOPA- and dopamine-borate esters are also potential B-carrier candidates for BNCT, and they could be incorporated as melanin precursors or binding reagents. In this work, molecular modeling methods have been applied to analyze the structure of boronic acid and borate ester derivatives, as well as their interaction and binding mode with the recently proposed catechol-porphycene model for the melanin polymer. Interestingly, remarkable structural features drawn from chemical geometry, and also molecular orbital calculations, support the possibility that boronic acids and borate esters could be reactive agents for direct binding to melanin catechols.
Keywords: 10Boron; Borate esters; BNCT; Catechols; Boronic acids; Melanin; Melanoma
Abbreviations: BNCT: 10Boron neutron-capture therapy; BPA: 4-borono-phenylalanine; DA: dopamine; DHI: dihydroxy-indole; DHICA: DHI-carboxylic acid; CBE: Catechol-borate ester; LUMO: Lowest-unoccupied molecular orbital; CPBA: Catechol-phenylboronic acid
Introduction
The 10Boron neutron-capture therapy (BNCT) of cancer is a novel therapeutic modality that counts with an increasing biomedical potential and many interesting applications [1-8]. BNCT requires a 10B-containing agent that accumulates selectively in the tumor, which is then subjected to irradiation with low-energy thermal neutrons. When a neutron is captured by the 10B isotope, a nuclear fission occurs generating extremely energetic 4He (α particle), 7Li and γ radiation [4,9], which damages tumor cells because of the very short braking distance (~10 and ~5 μm) of the emitted α particles and recoiling 7Li nuclei in condensed matter, respectively [2].
Boron content in the tumor must be high enough (around 109 10B atoms/cell) to allow for enough capture reactions to occur for confined cell lethality [5,9]. So, the chemistry of boron-containing metabolites and boron clusters has been reviewed [9,10,11]. 10B-containing agents are mainly BPA (4-borono-phenylalanine), and deca- or dodeca-clusters of B atoms such as GB-10 and BSH [1,5,8,12], which are the only ones approved for use in Oncology, especially for BNCT of skin melanomas [13-18]. As an amino acid analogue, non-specific binding of BPA to proteins of cells and tissues are expected to occur. Therefore, BPA would be highly unlikely to be a specific melanin precursor such as catechol amines and oxidation products, namely 3,4-dihydroxy-L- phenylalanine (L-DOPA), dopamine (DA), dihydroxy-indole (DHI), and DHI-carboxylic acid (DHICA). In any case, BPA will be taken up by cancer cells because of enhanced amino acid transport displayed by proliferating cells. In the case of melanoma, response to BPA after BNCT has been proposed to be due to the formation of complexes with L-DOPA, DHI, or DHICA monomers, and thus the drug would be later incorporated into melanin [7,19]. In an early attempt to carry 10B into melanoma cells L-DOPA-borate was synthesized [20], but no further results have been reported by applying this catechol-borate compound.
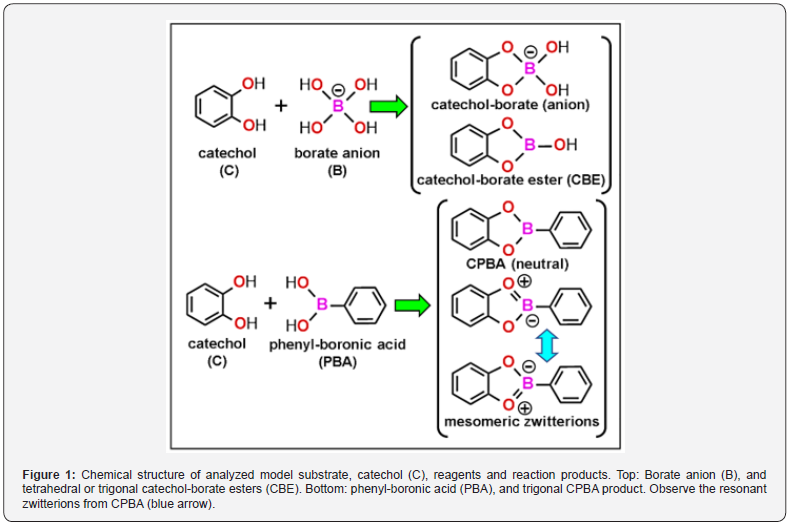
Vicinal aliphatic and aromatic cis-diols such as catechols, flavonols, β-diketones, anthraquinones, etc., form stable borate complexes [20-25]. Boronic acid (C-B[OH]2) derivatives are applied for detection of glycols and catechols [23]. Interestingly enough, the binding affinity of PBA for catechol is about 6000 times higher than that for ethylene-glycol [26]. Borate complexes formed at alkaline pH (higher than ~8), lose the hydroxyls at acid pH (lower than ~7) and become neutral (non-ionized) trigonal borate (BO3) esters [20]. Boric acid, borates, and boronic acids react easily with catechols [25-27]. Therefore, simple compounds such as BPA and catechol-borate ester (CBE) could form specific complexes directly with catechols from an already formed melanin, and work then as melanin-reactive drugs, and not as melanin precursors. This somewhat intriguing, but expected feature, seems to have been overlooked. Nevertheless, a mechanistic rationale for the possible binding of boronic acids and catechol-borate esters to melanin catechols is here described using a chemical and molecular modeling approach. The chemical structures of model compounds that will be analyzed, namely catechol-borate and phenyl-boronic acid, are shown in Figure 1. Concerning the chemistry of melanin and its precursors, several reviews have been published on melanin structure and its biosynthesis [28-30], being the most representative structure the porphyrin-like graphitic model from Olivieri and Nicolaus [31], and also the recent poly-catecholporphycene (poly-CPo) model [32,33], based on the Olivieri and Nicolaus structure [31].
Methodology
Following previous studies on modeled chemical structures [30,34-38], the use of chemical drawn, and molecular modeling software allowed to analyze and illustrate the structure of some boron-containing agents. Chemical structures with formal double bonds were drawn with ChemDraw Ultra v12.0 software. Molecular modeling of melanin precursors, drugs and model compound was carried out with the HyperChem v7 and v7.5 software, using geometry optimization by Molecular Mechanics Force Field (MM+), converged at energy E = 0.1, 0.05, or 0.01 kcal/(Å mol). Energy minimization and calculation of molecular orbitals (MOs) were performed using either the semi-empirical PM3 method (Polak-Riviere algorithm, conjugate gradient) converged at E = 0.1 or 0.05 kcal/(Å mol), or the extended-Hückel method. To illustrate molecular structures and processes, MOs were represented as Jorgensen-Salem surface or Gouraud shaded 3D isosurfaces, using adequate orbital contours (1/orbital radius). Electrostatic potentials were recorded by plotting molecular graphics tool (contour value: 0.05, grid mesh size: fine).
It must be noted that the ground singlet (S0) state. and the first photo-excited singlet (S1) state of small aromatic molecules, are also represented by different MOs. The highest-occupied molecular orbital (HOMO), and lowest-unoccupied molecular orbital (LUMO) states represent the energy levels of ground and excited states of the molecule, respectively [39]. In HOMO-d and LUMO+d, d is a value from 0 to the maximum energy level, and the separation between HOMO-0 and LUMO+0 corresponds to the forbidden Fermi’s energy gap (Eg). Orbital lobes corresponding to phases of π-electron conjugation are denoted by different colors. Fused lobes with the same color are in-phase, and those with isolated (unfused) lobes are out-of-phase.
Results and Discussion
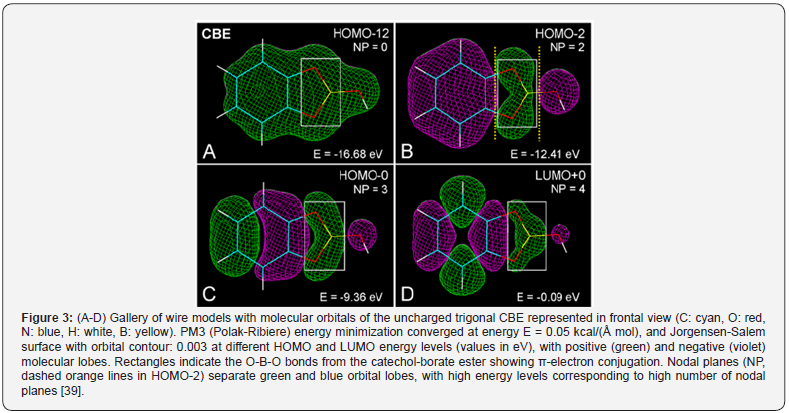
In the presence of borate anions or boronic acids, the catechol group forms in vitro a catechol-borate ester (CBE) or the boronic compound, catechol-phenylboronic acid (CPBA). The geometry of PBA and CBE, shown as space-filling (atomic volume) models, offers a more realistic representation of the molecular structure, as is clearly observed in Figure 2. Considering the stereochemistry of boronic acids and the formation of catechol-borate esters [25,26,40,41], molecular modeling methods are very suitable to illustrate their molecular shape and orbitals. The characteristic trigonal or tetrahedral configuration of neutral borate or borate anion in PBA or CBE, respectively, was found in atomic volume models (Figure 2), in agreement with chemical features of boric acid and anionic esters. Borate and boronic compounds are expected to occur as charged tetrahedral anions, neutral trigonal forms, or trigonal zwitterions, depending on the pH and type of solvent [20,26], but the stabilizing π-electron resonance between O and B atoms of the borate ester in the zwitterion mesomers of trigonal esters seems to have been rather overlooked. Interestingly, molecular modeling shows a clear π-conjugation involving the O-B-O atoms of the borate ring, as is revealed in the corresponding HOMO and LUMO patterns of trigonal CBE (Figure 3).
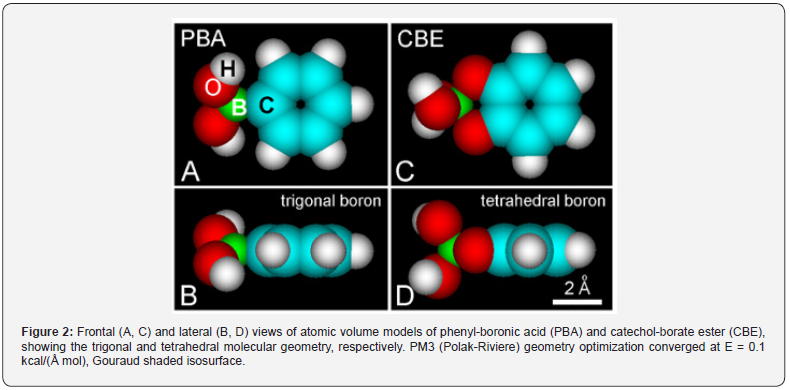
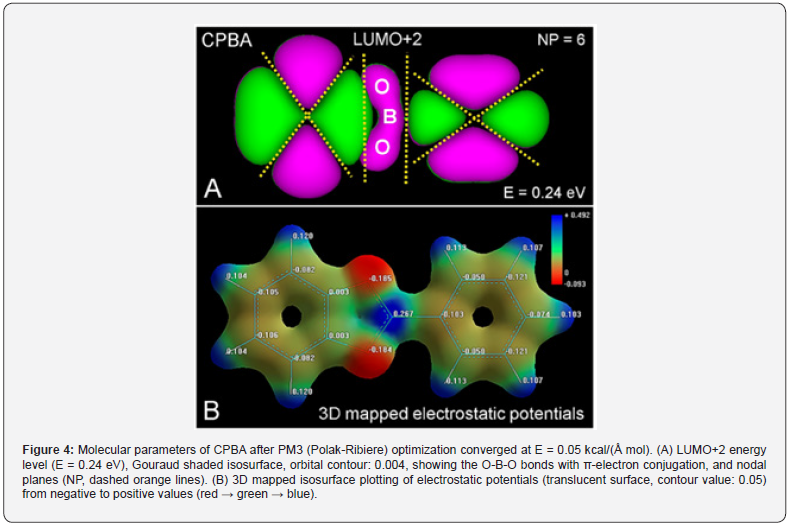
Regarding zwitterions, the B-containing pentagonal ring shows high stability due to 6π-electron resonance [40], which also occurs for other heteroaromatic pentagonal rings stabilized by π-electron delocalization [42,43]. In its valence state with three available electrons, the B atom has one 2s-electron and two 2p-electrons, and therefore, trigonal (3q) sp2-hybrid orbitals determine the binding behavior of boron [44, 45]. Likewise, the O atom is characterized by tetrahedral (4q) sp3 hybrid orbitals, and then π-electron conjugation is allowed for O-B-O bonding. In keeping with this, the O-B bond length in tetrahedral borates is 1.49 Å, whereas it is only 1.40 Å in trigonal esters. In the case of phenyl-boronic acid, in addition to the O-B-O conjugation revealed in MOs image (Figure 4, A), a differential distribution of partial charges in B and O atoms is clearly observed. Figure 4, B shows the localization of the highest negative and positive charges in the trigonal CPBA complex (see Figure 1). This reveals that the B atom is positive (δ = + 0.26 eV), contrary to the commonly claimed negative charge of B represented in chemical formulations, which instead, appears distributed on each O atom (δ = -0.18 eV). Comparing trigonal and tetrahedral structures, the differential negative charge for BO3 and BO4 groups is -0.18 and -0.75 eV, respectively.
Eumelanin contains a great amount of catechol groups [28,33,46-48], which can react with borate anions and boronic acids (see Figure 1). Borate-ligand 1:1 and 1:2 complexes are the most and less usual, respectively [25,27], which could difficult the occurrence of borate 1:2 complexes using melanin catechols. However, a variety of easily synthesized arylboronic acids [49] would allow a more direct reaction with catechol groups. Boroncontaining metabolite precursors could be incorporated into the polymer during biosynthesis. As each catechol group can bind a single borate anion or boronic acid, numerous borate esters are introduced into melanin. Binding of metal ions can account for up to 6 x 1020 sites per gram of dried melanin [50], and thus, catechol sites would account for a large amount of melanin-bound B atoms. At pH > 4 catechols from synthetic DOPA-melanin are ionized [51] and thus, it is a basophilic polyanion. At pH < 8 most borates esters are uncharged [20,26], and then melanin-borates complexes must be neutral and remain unstained.
Studies on the localization of boron-containing drugs for BNCT have shown rather poor results. BPA was found in the cytoplasm and nuclei of human glioblastoma and murine sarcoma cells [52-54]. In the case of melanocytes, PBA should be accumulated specifically within melanosomes. As they are acidic organelles with a clear lysosome lineage [55] and intramelanosomal pH range of 3-5 [56], the occurrence of negatively charged borate esters of eumelanin is unlikely to occur in living melanocytes. Regarding the recently proposed catechol-porphycene (CPo) model [32,33], it must be noted that this structure fulfills all characteristic features of natural and synthetic eumelanins, appearing as a multilayer graphitic structure. Modeling of a planar tetra-CPo molecule with catechol-attached PBA residues illustrates the possible organization of the polymer (Figure 5). Long continuous LUMO+4 lobes showing trigonal OBO groups, and fused CC and N:HN orbitals are clearly visible, accounting for the massive aromatic conjugation of this graphitic material. It is conceivable that the great amount of catechol groups makes melanin a suitable substrate to form borate esters, but the simple binding mechanism of boronic acids to melanin catechols have been mostly overlooked. Although a role as melanin precursor for BPA cannot be excluded, the main action mechanism could be a direct chemical reaction with suitable substrates such as aromatic diols, as seems to occur in the case of some melanoma seekers [37].
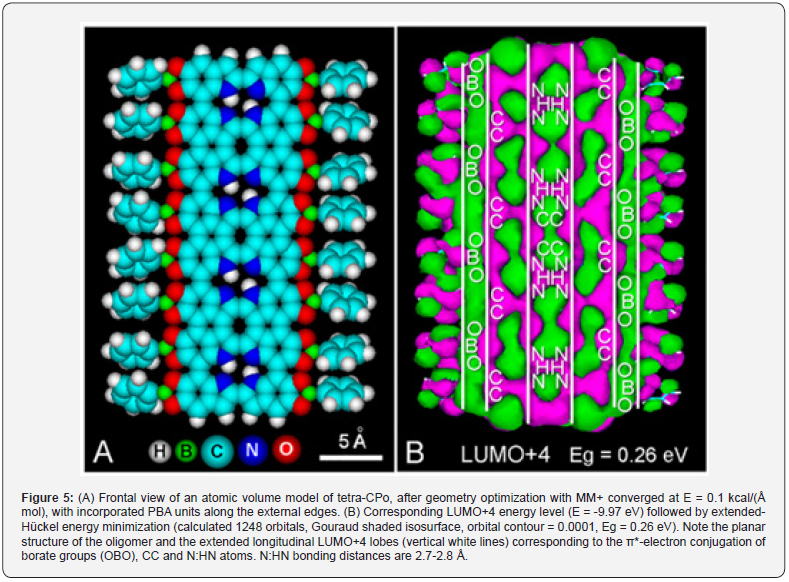
Conclusion
In spite of the increasing biomedical interest in using BNCT against melanoma, the reaction mechanism of 10B-carriers with melanin remains unclear, but nevertheless some new interpretations and predictions are put forward in this work. It may be assumed that catechol-borate precursors could be either incorporated during eumelanin biosynthesis or directly bound to catechol groups of already formed polymers. The application of molecular modeling methods for analyzing the incorporation mechanism of 10B-carriers into melanin revealed interesting structural features of both reagents and reaction products. In keeping with this, OBO π-conjugation in trigonal boronic and borate groups is clearly demonstrated, as well as its participation in the extended longitudinal LUMO+4 energy level of a melanin model based on a poly-catechol-porphycene structure containing PBA units. Geometric and energetic considerations suggest that this proposal of a direct reaction between a boronic acid and melanin catechols may be right, resulting on a rather simple way to attach 10B to the melanin content of melanoma cells for BNCT
References
- Feakes DA (2001) Chemistry and pharmacology of agents for BNCT. In: Frontiers in Neutron Capture Therapy (Hawthorne MF, Shelley K, Wiersema RJ, Eds.), Vol. 1. New York: Kluver Academic/Plenum Publisher p. 23-34.
- Wittig A, Michel J, Moss RL, Stecher-Rasmussen F, Arlinghaus HF (2008) Boron analysis and boron imaging in biological materials for boron neutron capture therapy (BNCT). Crit Rev Oncol Hematol 68(1): 66-90.
- Barth RF (2009) Boron neutron capture therapy at the crossroads: challenges and opportunities. Appl Rad Isot 67(7-8 Suppl): 3-6.
- Hopewell JW, Morris GM, Schwint A, Coderre JA (2011) The radiobiological principles of boron neutron capture therapy: A critical review. Appl Rad Isot 69(12): 1756-1759.
- Schwint AE, Trivillin VA (2015) “Close-to-ideal” tumor boron targeting for boron neutron capture therapy is possible with “less-than-ideal” boron carriers approved for use in humans. Ther Deliv 6(3): 269-272.
- Farhood B, Samadian H, Ghorbanic M, Zakariaeed SS, Knaup C (2018) Physical, dosimetric and clinical aspects and delivery systems in neutron capture therapy. Rep Pract Oncol Radiother 23(5): 462-473.
- Fukuda H (2021) Boron neutron capture therapy (BNCT) for cutaneous malignant melanoma using 10B-p-boronophenylalanine (BPA) with special reference to the radiobiological basis and clinical results. Cells 10(11): 2881.
- Sauerwein WAG, Sancey L, Hey-Hawkins E, Kellert M, Panza L, et al. (2021) Theranostics in boron neutron capture therapy. Life 11(14): 330.
- Hu K, Yang Z, Zhang L, Xie L, Wang L, Xu H, et al. (2020) Boron agents for neutron capture therapy. Coord Chem Rev 405: 213139.
- Kabalka GW, Yao ML (2006) The synthesis and use of boronated amino acids for boron neutron capture therapy. Anti-Cancer Agents Med Chem 6(2): 111-125.
- Pietrangeli D, Rosa A, Ristori S, Salvati A, Altieri S (2013) Carboranyl-porphyrazines and derivatives for boron neutron capture therapy: From synthesis to in vitro tests. Coord Chem Rev 257(15-16): 2213-2231.
- Schwint AE, Monti Hughes A, Garabalino MA, Santa Cruz GA, González SJ, et al. (2020) Clinical veterinary boron neutron capture therapy (BNCT). Studies in dogs with head and neck cancer: Bridging the gap between translational and clinical studies. Biology 9(10): 327.
- Mishima Y, Ichihashi M, Hatta S, Honda C, Yamamura K (1989) New thermal neutron capture therapy for malignant melanoma: melanogenesis-seeking 10B molecule–melanoma cell interaction from in vitro to first clinical trial. Pigm Cell Res 2(4): 226-234.
- Fukuda H, Honda C, Wadabayashi N, Kobayashi T, Yoshino K, et al. (1999) Pharmacokinetics of 10B-p-boronophenylalanine in tumors, skin and blood of melanoma patients: a study of boron neutron capture therapy for malignant melanoma. Melan Res 9(1): 75-83.
- González SJ, Bonomi MR, Santa Cruz GA, Blaumann HR, Calzetta Larrieua OA (2004) First BNCT treatment of a skin melanoma in Argentina: dosimetric analysis and clinical outcome. Appl Rad Isot 61(15): 1101-1105.
- Menéndez PR, Roth BMC, Pereira MD, Casal MR, González SJ, et al. (2009) BNCT for skin melanoma in extremities: Updated Argentine clinical results. Appl Rad Isto 67(7-8Suppl): S50-S53.
- Carpano M, Perona M, Rodriguez C, Nievas S, Olivera M, et al. (2015) Experimental studies of boronophenylalanine ((10)BPA) biodistribution for the individual application of boron neutron capture therapy (BNCT) for malignant melanoma treatment. Int J Rad Oncol Biol Phys 93(2): 344-352.
- Hiratsuka J, Kamitani N, Tanaka R, Tokiya R, Yoden E, et al. (2020) Long-term outcome of cutaneous melanoma patients treated with boron neutron capture therapy (BNCT). J Rad Res 61(6): 945-951.
- Yoshino K, Mishima Y, Kimura M (1997) Capture of p-boronophenylalanine in malignant melanoma cells by complex formation with melanin monomers, DOPA, DHI and DHICA. In: Advances in Neutron Capture Therapy (Larsson B, Crawford J, Weinreich R, Eds). Vol. II. Amsterdam: Elsevier pp. 234-238.
- Yoshino K, Kotaka M, Okamoto M, Kakihana H (1979) 11B-NMR study of the complex formation of borate with catechol and L-DOPA. Bull Chem Soc Japan 52(10): 3005-3009.
- Feigl F, Anger V (1958) Spot Tests in Inorganic Analysis. 6th Amsterdam: Elsevier Publishing Co pp. 136-142.
- Frohne D (1974) Anatomisch-mikrochemische Drogenanalyse. Stuttgart: Georg Thieme Verlag pp. 123.
- Yang W, Yan J, Springsteen G, Deeter S, Wang B (2003) A novel type of fluorescent boronic acid that shows large fluorescence intensity changes upon binding with a carbohydrate in aqueous solution at physiological pH. Bioorg Med Chem Lett 13(6): 1019-1022.
- Frasconi M, Rea S, Matricardi P, Favero G, Mazzei F (2009) Scleroglucan-borax hydrogel: a flexible tool for redox protein immobilization. Langmuir 25(18):11097-11104.
- Lenskiy MA, Shul’ts EE, Korabel’nikov DV, Ozhogin AV, Novitskiy AN (2020) Pyrocatechol borates: Synthesis, reaction with formaldehyde, and solvent effect on polycondensation process. Polymer 188: 122162.
- Yan J, Fang H, Wang B (2005) Boronolectins and fluorescent boronolectins: An examination of the detailed chemistry issues important for the design. Med Res Rev 25(5): 490-520.
- Rebstöcková M, Bartusek M (1997) Reactions of boric acid with oxalic, glycolic and tartaric acids. Czechoslov Chem Commun 42: 627-636.
- d’Ischia M, Wakamatsu K, Cicoira F, di Mauro E, Garcia-Borron JC (2015) Melanins and melanogenesis: From pigment cells to human health and technological applications. Pigm Cell Melan Res 28(5): 520-544.
- Scognamiglio F, Travan A, Turco G, Borgogna M, Marsich E, et al. (2017) Adhesive coatings based on melanin-like nanoparticles for surgical membranes. Coll Surf B: Biointer 155: 553-559.
- Blázquez-Castro A, Stockert JC (2021) Biomedical overview of melanin. 1. Updating melanin biology and chemistry, physico-chemical properties, melanoma tumors, and photothermal therapy. Biocell 45: 849-862.
- Olivieri M, Nicolaus RA (1999) Sulla DHI-melanina. Rend Accad Sci Fis Matem Napoli 66: 85-96.
- Stockert JC (2021) Melanin and melanoma: Updating molecular structure and photothermal therapy. InVet 23(1):1-15.
- Stockert JC, Blázquez-Castro A (2022) Biomedical overview of melanin. 2. Updating molecular modeling, synthesis mechanism, and supramolecular properties regarding melanoma therapy. Biocell 46(6): 1391-1415.
- Stockert JC, Blázquez A, Galaz S, Juarranz A (2008) A mechanism for the fluorogenic reaction of amino groups with fluorescamine and 2-methoxy-2,4-diphenyl-3(2H)-furanone. Acta Histochem 110(4): 333-340.
- Stockert JC, Abasolo MI (2011) Inaccurate chemical structure of dyes and fluorochromes found in the literature can be problematic for teaching and research. Biotech Histochem 86(1): 52-60.
- Stockert JC (2020) Lipid peroxidation assay using BODIPY-phenylbutadiene probes: a methodological overview. Meth Mol Biol 2202: 199-214.
- Stockert JC, Espada J, Blázquez-Castro A (2022) Melanin-binding colorants: Updating molecular modeling, staining and labeling mechanisms, and biomedical perspectives. Colorants 1: 91-120.
- Stockert JC, Felix-Pozzi MN (2022) Updating curvature of graphitic materials: Molecular modeling studies on the spiral structure of graphene and eumelanin models. Adv Biol.
- Stockert JC, Blázquez-Castro A (2017) Fluorescence Microscopy in Life Sciences. E-Book. Sharjah, United Arab Emirates: Bentham Science Publishers.
- Gerrard W, Lappert MF, Mountfield BA (1959) Interaction of boron trichloride with catechol, quinol, resorcinol, and pyrogallol. J Chem Soc 1529-1535.
- Lang A, Knizek J, Nöth H, Schur S, Thomann M (1997) Beiträge zur Chemie des Bors. 237. Bis(benzo-l,3,2-dioxaborolanyl)oxid und 2-(o-Hydroxyphenoxy)-benzo-l,3,2-dioxaborolan. Vorstufen zur Synthese von Catecholboran (Benzo-l,3,2-dioxaborolan). Zeitschr anorg allgem Chemie 623(1-6): 901-907.
- Dewar MJS, Kubba VP, Pettit R (1958) New heteroaromatic compounds. Part II. Boron compounds isoconjugate with indole, 2:3-benxofuran, and thionaphthen. J Chem Soc pp. 3076-3079.
- Schubert DM, Smith RA, Visi MZ (2003) Studies of crystalline nonmetal borates. Glass Technol 44(2): 63-70.
- Seel F (1965) Atomic structure and chemical bonding. London: Methuen & Co p. 42-47.
- Christen HR (1976) Atommodelle und chemische Bindung. 3. Auflage. Frankfurt: Umschau Verlag p. 34-36.
- Di Mauro E, Xu R, Soliveri G, Santato C (2017) Natural melanin pigments and their interfaces with metal ions and oxides: Emerging concepts and technologies. MRS Comm 7: 141-151.
- Ruiz-Molina D, Saiz Poseu J, Busque F, Nador F, Mancebo J (2018) The chemistry behind catechol-based adhesion. Angew Chem Int Ed 58(3): 696-714.
- Cavallini C, Vitiello G, Adinolfi B, Silvestri B, Armanetti P, et al. (2020) Melanin and melanin-like hybrid materials in regenerative medicine. Nanomater 10(8): 1518.
- Zhao CJ, Xue D, Jia ZH, Wang C, Xiao J (2014) Methanol-promoted borylation of arylamines: A simple and green synthetic method to arylboronic acids and arylboronates. Synlett 25: 1577-1584.
- Sarna T, Hyde J, Swartz H (1976) Ion-exchange in melanin: An electron spin resonance study with lanthanide probes. Science 192: 1132-1134.
- Ball V (2010) Impedance spectroscopy and zeta potential titration of dopa-melanin films produced by oxidation of dopamine. Coll Surf A 363(1-3): 92-97.
- Chandra S, Kabalka GW, Lorey DR, Smith DR, Coderre JA (2002) Imaging of fluorine and boron from fluorinated boronophenylalanine in the same cell at organelle resolution by correlative ion microscopy and confocal laser scanning microscopy. Clin Cancer Res 8(8): 2675-2683.
- Chandra S, Tjarks W, Lorey DR, Barth RF (2008) Quantitative subcellular imaging of boron compounds in individual mitotic and interphase human glioblastoma cells with imaging secondary ion mass spectrometry (SIMS). J Microsc 229(1): 92-103.
- Wittig A, Arlinghaus HF, Kriegeskotte C, Moss RL, Appelman K, et al. (2008) Laser postionization secondary neutral mass spectrometry in tissue: a powerful tool for elemental and molecular imaging in the development of targeted drugs. Mol Cancer Ther 7(7): 1763-1771.
- Raposo G, Marks MS (2007) Melanosomes-Dark organelles enlighten endosomal membrane transport. Nature Rev Mol Cell Biol 8(10):786-797.
- Orlow SJ (1995) Melanosomes are specialized members of the lysosomal lineage of organelles. J Invest Dermatol 105(1): 3-7.






























