Investigation to Determine the Performance of Acid Leaching for Metal Recovery from Copper Tailing
Niaz Umera, Jahansher Qambranib, Fu Kaibina*, Feng Taifenga, Yahya Faqira*
aSouthwest University of Science and Technology, China
bMir Chakar Khan Rind University Sibi, Pakistan
Submission: July 18, 2022; Published: July 28, 2022
*Corresponding author: Fu Kaibin & Yahya Faqir, Southwest University of Science and Technology, China
How to cite this article: Niaz U, Jahansher Q, Fu K, Yahya F. Investigation to Determine the Performance of Acid Leaching for Metal Recovery from Copper Tailing. Ann Rev Resear. 2022; 7(4): 555719. DOI: 10.19080/ARR.2022.07.555719
Abstract
It has been reviewed that the Cu, Fe, and Mn are harmful to our environment, it needs our consideration to recover the water which has been contaminated by these metals and as well as when released without being treated and recovered, it will be harmful to the land and the soil and can be unfriendly for our environment. Toxic mining waste is a problem of more serious concern for both industries and the government. Several copper tailings dumps have been abandoned and not sufficiently stabilized. Once the tailings containing toxic minerals and residual metals contact diluents like rainwater, it forms acid mine water that can leach metals into groundwater, rivers, and streams. The effect of the different HCl addition was investigated under a fixed S: L of 1:2. The leaching temperature was set to 40°C, and leaching occurred for 2h. As presented in Tables 1 & 2, the leaching efficiency of the metals increased gradually with an increasing amount of HCl. The AAS method showed the leaching efficiency of 0.03±0.01% and 0.04±0.02% for Cu with the addition of 1.5 and 2 mL/g of HCl. For Fe, leaching efficiencies of 17.38±3.55% and 15.94±2.41% were obtained with the addition of 1.5 and 2mL/g of HCl. However, 0.39±0.02% and 0.38±0.01% for Mn were leached when adding 1.5 and 2mL/g of HCl. Statistically, there was a non-significant (p<0.05) difference in the leaching efficiency of Cu, Fe, and Mn among the different concentrations of HCl. Using the XRF method, the leaching efficiency of 12.09±2.40% and 9.5±1.84% for Cu was obtained by adding 1.5 and 2mL/g of HCl. For Fe, leaching efficiencies of 1.5±0.11% and 1.35±0.05% were obtained with the addition of 1.5 and 2mL/g of HCl. However, 16.54±5.21% and 15±2.49% for Mn were leached when adding 1.5 and 2mL/g of HCl. Statistically, there was a non-significant (p<0.05) difference in the leaching efficiency of Cu, Fe, and Mn among the different concentrations of HCl. It is concluded that Cu, Fe, and Mn from the low-grade copper tailing are successfully recycled via H2SO4 leaching and subsequent fractional precipitation technology. Based on the mineral composition analysis results, leaching temperature, the amount of H2SO4 added, leaching time, and S:L have been evaluated for leaching efficiencies. The amount of H2SO4 added, and S:L have strongly influenced the leaching efficiency.
Keywords: Copper tailing; Acid leaching; Heavy metals; AAS; XRF
Introduction
The mining of metals generates considerable amounts of waste materials. These generally have very little economic value, making their exploitation not profitable, though they often have the potential to pose a long-term threat and cause damage to the environment. Mine wastes vary depending on their physical and chemical composition, the type of mining, and how the mineral is processed. Millions of tons of ore are processed every year by the mining industry, >95% of which is disposed of in waste rocks and mine tailings. The latter is finely ground rock particles generated during the processing of ore materials and separation of target metal minerals and are highly reactive because of their small particle size and content of reactive minerals, such as pyrite (FeS2). Mine tailings may contain base transition metals, such as iron, copper, nickel, and zinc, in relatively high concentrations, and occasionally precious metals, such as gold and silver, in minerals (and native metals) that have not been separated by froth flotation [1, 2]. Toxic elements, such as arsenic, may also be present in elevated concentrations [3]. Exposure of mine tailings to oxygen and water facilitates the (microbially-catalyzed) oxidative dissolution of the sulfide minerals they contain, generating acidic wastewaters with elevated concentrations of dissolved metals and sulfate. Acid mine drainage (AMD) is a widely reported environmental pollutant with global distribution [4].
Copper tailing is the solid waste material left during the purification of the precious copper from the copper ores. The disposal of copper tailing is one of the significant critical environmental issues in a copper mine. To prevent the uncontrolled liberation of copper tailings material into the atmosphere, copper mines generally have a dumping facility in the form of a dam or a pond [5]. This is a convenient way of storage when tailings are usually in the form of slurry when they are discharged from the concentrator. However, these types of dumping also need more land in the future, and failure to provide more land results in environmental problems.
The disposal of copper tailings has long been recognized as an environmental problem due to earth and aquatic pollution hazards. The method of growing plants over the tailings appears to be an effective method of decreasing these effects [6], and dumping the frequently produced, massive mass of tailing material after the processing operations is one of the common environmental problems related to the mineral industries. As the mining industries discharge their waste materials to the surrounding location, it generates many problems. The pollution-creating waste can be disposed of through dams (which comprise factory sediments, mine tailings, and deposited materials; and are generally considered the most significant human-made structures in the world) that have played a significant role in protecting valuable soil and water resources from dirty slurries [5]. Slags are a leftover from the pyrometallurgical stages of recovery of non-ferrous metals from sulfide materials. Production of 1t of copper results in average information of 2.2t of slag; the annual global output of copper-smelting slags is ~25Mt. Significant areas required for stockpiling of the slags and the products of their processing are unavailable for land utilization. Leakage fluxes of toxic metals developing around the sites of slag stockpiling have a long-term adverse effect on the biosphere [7].
However, mine tailings may be recycled and reused, e.g., manganese- rich tailings could be used in agro-forestry, building and construction materials, coatings, cast resin products, glass, ceramics, and glazes [8]. Increasing demands for metals, coupled with depleting reserves of high-grade primary ores, have meant that waste materials from historic mining operations may have more excellent metal contents than primary ores currently being excavated. Since mine tailings have already been part-processed (by comminution to produce fine-grain particles), the cost of extracting residual metals from them is often economically more attractive than mining a deep-buried primary ore body. In addition, mine wastes may contain metals (such as rare earth elements) that were not considered worth extracting when the ores were initially processed but have since increased in value and use. This was the case with cobalt-rich tailings produced at a former copper mine in Kasese, Uganda, which was reprocessed by bioleaching decades after mining at the site ended to extract cobalt and remove the environmental threat posed by the tailings deposits [9,10].
Comprehensive reuse of the tailing would improve economic efficiency and lessen environmental damage as well. The leaching process could promote such comprehensive reuse. This work aims to recover Cu, Fe, and Mn from low-grade copper tailing effectively using an environment-friendly and economical leaching method.
Materials and Methods
This present research was laid out to study the metal recovery from copper tailing through acid leaching. For this purpose, the solid material samples were brought into the Environmental resources laboratory. The solid material samples were crushed into the pestle and mortar. H2SO4, HCL, and H2O were added to prepare the samples. The samples were placed into a shaker to get them homogeneous for about different times. The homogenized samples were filtrated with the help of a Water-Circulation multifunction vacuum pump machine in a way to get liquid filtrate samples. After the liquid filtrate was tested by AAS analysis, reduced solid residue samples were tested under XRF for their elemental analysis. The expected result showed us the percentage of leached material, the determination of heavy metals, and their total contents studied. At first, the pH of the leaching filtrate was adjusted with the addition of Ca(OH)2 to recover Fe. After that, Cu was recovered through the addition of Na2S solution. Finally, Mn was recovered by adjusting the pH in the leaching filtrate.
The solid material samples were brought into the environmental resources laboratory. The solid material samples were crushed into the pestle and mortar. H2SO4, HCL, and H2O were added to prepare the samples. The samples were placed into shakers to get them homogeneous for about different times. The homogenized samples were filtrated with the help of a Water-Circulation multifunction vacuum pump machine in a way to get liquid filtrate samples. After the liquid filtrate was tested by AAS analysis, reduced solid residue samples were tested under SEM & EDS for their elemental analysis. The expected result showed us the percentage of leached material, and the determination of heavy metals and their total contents were studied. At first, the pH of the leaching filtrate was adjusted with the addition of Ca(OH)2 to recover Fe. After that, Cu was recovered through the addition of Na2S solution. Finally, Mn was recovered by adjusting the pH in the leaching filtrate.
The solid samples were digested by microwave digestion equipment (WX-8000, EU Microwave Chemistry Technology Co., Ltd.) according to method 3052. In this section, the major metals were determined using an AAS (Shimadzu AA6300, made in Japan), whereas minor metals at significantly low concentrations were determined through ICP-MS (Agilent 7500, made in America). The mineral composition was investigated by SEM–EDX and analyzed through the SIP system of QEMSCAN. The bound moisture content was determined through the method of specific gravity. The parameters affecting the recovery efficiency, such as leaching temperature, amount of added leaching reagent, leaching time, and leaching solid-to-liquid ratio (S:L, volume weight), were investigated. The fractional precipitation was used for the metal separation recovery from the leachate, whose principle is the solubility discrepancy of different metals. The metal recovery from the leaching filtrate was preceded by fractional precipitation by adding different chemical agents.
The statistical analysis was performed by SPSS and Origin software. The three replicates of each sample were analyzed for their statistical analysis. The results were expressed as the arithmetic means ± standard deviation. Statistical data were analyzed using Microsoft Excel 2007 and SPSS 19.0. One way analysis of variance (ANOVA) was used to detect the statistical significance of differences between data groups with a significance level of p < 0.05.
Results and Discussion
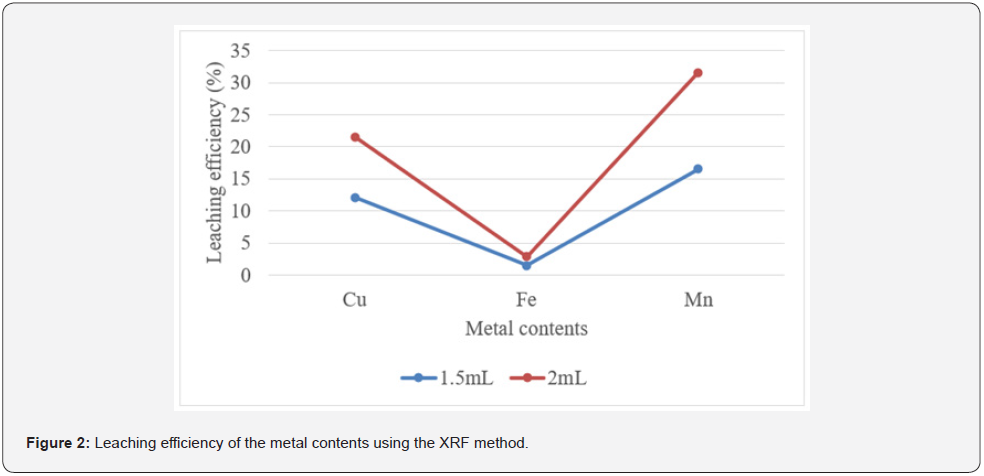
The effect of the different HCl addition was investigated under a fixed S: L of 1:2. The leaching temperature was set to 40°C, and leaching occurred for 2h. As presented in Tables 1 & 2, the leaching efficiency of the metals increased gradually with an increasing amount of HCl. The AAS method showed the leaching efficiency of 0.03±0.01% and 0.04±0.02% for Cu with the addition of 1.5 and 2 mL/g of HCl. For Fe, leaching efficiencies of 17.38±3.55% and 15.94±2.41% were obtained with the addition of 1.5 and 2mL/g of HCl. However, 0.39±0.02% and 0.38±0.01% for Mn were leached when adding 1.5 and 2mL/g of HCl. Statistically, there was a non-significant (p<0.05) difference in the leaching efficiency of Cu, Fe, and Mn among the different concentrations of HCl (Table 1 & Figure 1).

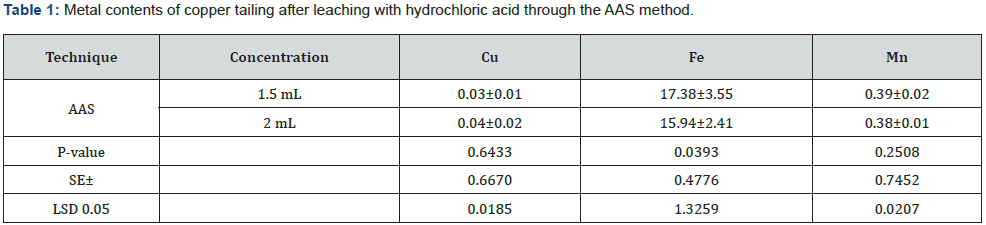
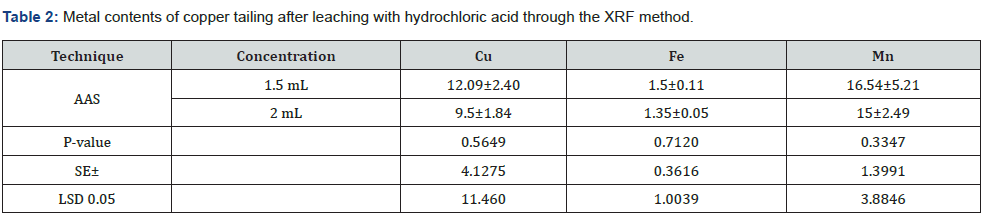
Using the XRF method, the leaching efficiency of 12.09±2.40% and 9.5±1.84% for Cu was obtained by adding 1.5 and 2mL/g of HCl. For Fe, leaching efficiencies of 1.5±0.11% and 1.35±0.05% were obtained with the addition of 1.5 and 2mL/g of HCl. However, 16.54±5.21% and 15±2.49% for Mn were leached when adding 1.5 and 2mL/g of HCl. Statistically, there was a non-significant (p<0.05) difference in the leaching efficiency of Cu, Fe, and Mn among the different concentrations of HCl (Table 2 & Figure 2). The metal recovery from the leaching filtrate was preceded by fractional precipitation by adding different chemical agents. At first, the pH of the leaching filtrate was adjusted to 3.0 with the addition of Ca(OH)2 to recover Fe as hydroxide. Thereafter, Cu and Zn were recovered as their metal sulfides through the sequence dropwise addition of a 0.5wt % aqueous Na2S solution. The pH value of the solution after Cu and Zn precipitations is 3.6 and 3.8, respectively. Finally, Mn was recovered by adjusting the pH to 9.0 in the leaching filtrate. With the UV conversion of the Fe sludge, magnetite particles could be obtained and applied to the paint industry, while the Fe2O3 content of the magnetite particles should be higher than 75%. It has been found that mostly Cu and Zn sludges and those generated in the sulfide precipitation process were covellite and sphalerite, which could be refined and sold to smelters to recover the metals. Generally, the Cu and Zn contents of the sludge are about 0.5% and 2% for their flotation refinement, respectively. At the same time, the Cu and Zn contents of the sludge were 19.5% and 33.5%, respectively. The metal contents of the sludge are higher than these specifications; therefore, the sludge could be refined with the flotation process. Furthermore, Cu and Zn could be obtained through the smelting process of the flotation concentrates. The Mn sludge can be used as cement material, and the leaching of heavy metals from the solidified blocks would be negligible.
Conclusion
Based on the mineral composition analysis results, leaching temperature, the amount of HCl added, leaching time, and S: L have been evaluated for leaching efficiencies. The amount of HCl added and S: L has strongly influenced the leaching efficiencies. Under the optimum ambient temperature and atmospheric pressure conditions, 0.24mL/g of HCl, S: L of 1:2, stirring speed of 400rpm, and leaching time of 2h, leaching efficiencies were achieved for Cu, Fe, and Mn, respectively. Fe and Mn were enriched as hydroxides with the fractional precipitation process, whereas Cu was enriched as sulfides. The Cu, Fe, and Mn sludges could be further refined and sold to the paint industry or the smelters to recover the metals, and with the recycling disposal, the environmental problems caused by the tailing could be relieved. While the leaching residue reuse needs further consideration for its environmental impact.
References
- Bryan CG, Hallberg KB, Johnson DB (2015) Mobilisation of metals in mineral tailings at the abandoned São Domingos copper mine (Portugal) by indigenous acidophilic bacteria. Hydrometallurgy 83(1-4): 184-194.
- Ahmadi A, Khezri M, Abdollahzadeh AA, Askari M (2015) Bioleaching of copper, nickel and cobalt from the low-grade sulfidic tailing of Golgohar Iron Mine, Iran. Hydrometallurgy 154: 1-8.
- Lee MH, Park HJ, Lee JU (2015) Bioleaching of arsenic and heavy metals from mine tailings by pure and mixed cultures of Acidithiobacillus spp. Journal of Industrial and Engineering Chemistry 21: 451-458.
- Liao J, Ru X, Xie B, Zhang W, Wu H, et al. (2017) Multi-phase distribution and comprehensive ecological risk assessment of heavy metal pollutants in a river affected by acid mine drainage. Ecotoxicology and environmental safety 141: 75-84.
- Tabelin CB, Park I, Phengsaart T, Jeon S, Villacorte-Tabelin M, et al. (2021) Copper and critical metals production from porphyry ores and E-wastes: A review of resource availability, processing/recycling challenges, socio-environmental aspects, and sustainability issues. Resources, Conservation and Recycling 170: 105610.
- Rubinos DA, Jerez O, Forghani G, Edraki M, Kelm U (2021) Geochemical stability of potentially toxic elements in porphyry copper-mine tailings from Chile as linked to ecological and human health risks assessment, Environ Sci Pollut Res Int 28(41): 57499-57529.
- Spitz K, Trudinger J (2019) Mining and the environment: from ore to metal, CRC Press.
- Lottermoser BG (2011) Recycling, reuse and rehabilitation of mine wastes, Elements 7(6): 405-410.
- Falagán C, Grail BM, Johnson DB (2017) New approaches for extracting and recovering metals from mine tailings. Minerals Engineering 106: 71-78.
- Morin DHR, d' Hugues P (2007) Bioleaching of a cobalt-containing pyrite in stirred reactors: a case study from laboratory scale to industrial application, in: Biomining, Springer pp. 35-55.






























