Prospective Study of Traumatic Optic Neuropathy among Active Military and Veterans: An Underestimated Pattern of Prevalence
Charles S Zwerling1* and Lea Carter2
1Associate Professor of Surgery, Campbell University School of Osteopathic Medicine, North Carolina, USA
2Campbell University School of Osteopathic Medicine, Class of 2023, North Carolina, USA
Submission: January 01, 2022; Published: January 11, 2022
*Corresponding author: Charles S Zwerling MD, Associate Professor of Surgery, Campbell University School of Osteopathic Medicine, North Carolina, USA
How to cite this article: Zwerling CS, Carter L. Prospective Study of Traumatic Optic Neuropathy among Active Military and Veterans: An Underestimated Pattern of Prevalence. Ann Rev Resear. 2022; 7(1): 555702. DOI: 10.19080/ARR.2022.07.555702
Abstract
A prospective study for traumatic optic neuropathy has been performed to review and revise management proto col for active military personnel and veterans who have sustained either traumatic brain injury (TBI) or mild traumatic brain injury (mTBI aka head concussions). A new protocol for visual field testing to include both kinetic and static testing was utilized to uncover occult cases of traumatic optic neuropathy (TON) either from traumatic brain injury or from mild head concussions as well as confirm suspected cases of TBI and/or mTBI. Furthermore, we have identified that military parachute jumpers (paratroopers) represent a high-risk group for undiagnosed mild TBI and traumatic optic neuropathy. We have urged that collaboration and development of safer helmet design would help reduce and maybe prevent further TBI, mTBI, and traumatic optic neuropathy for the military personnel.
Keywords: Traumatic optic neuropathy; Static visual field; Kinetic visual field; Traumatic brain injury; TBI; Head concussion; Mild TBI; mTBI; Paratrooper; Helmet; Electrophysiology; PERG; VEP; Veteran disability
Abbreviations: TBI: Traumatic brain injury; TON: Traumatic optic neuropathy; TBI: Traumatic brain injury; MVA: Motor vehicle accidents; SNFL: Superficial nerve fiber of ganglion cells; IOP: Intraocular space with higher pressure; CSFP: Cerebrospinal fluid pressure; RNFL: Retinal nerve fiber layer; LGN: Lateral geniculate nucleus; OERG: Pattern electroretinogram; RGC: Retinal ganglion cells; VEP: Visual Evoked Potential; CTE: Chronic Traumatic Encephalopathy; ACH: Army Combat Helmets; SD-OCT: Spectral Optical Coherent Topography
Introduction
In September 2021, we reported a case of a healthy 25-year-old male who presented with distorted central vision of his left eye, due to previously undiagnosed unilateral traumatic optic neuropathy. The patient was a recently discharged Army veteran who served from 2015-to 2020. In 2017 he sustained a mild head injury from a motorcycle accident. All head X-rays, MRI, and CT scans were normal. He required no medical treatment. His follow up neurological evaluations for TBI were considered normal. Despite his complaints of distorted central vision only in his left eye, his multiple eye exams after the incident were considered normal with 20/20 vision bilaterally, normal kinetic visual fields, and no pathology present on anterior and posterior segment examination. He was seen for veteran disability evaluation at our clinic which uncovered a unilateral traumatic optic neuropathy of his left eye. The diagnosis was confirmed with static visual field testing which had not been previously performed [1]. We were concerned that this case may have represented a potential pattern of disease for traumatic optic neuropathy (TON) among active military and veterans. The purpose of this paper is to report undiagnosed cases of traumatic optic neuropathy that were not detected with standard kinetic visual field testing and/or misinterpretation of the kinetic fields. All cases of active military service personnel on terminal leave and veterans with known diagnosis of either TBI or a history of mild TBI were placed into our new protocol for kinetic and static visual field testing and interpretation.
Traumatic optic neuropathy (TON) is a form of optic nerve damage caused by either direct or indirect trauma to the head/orbit [2,3]. Indirect TON is caused by a transmission of force through the skull to the optic nerve, propagating and shearing of the retinal ganglion cells [2-4]. Injury in the anterior optic nerve can cause disruption of the central retinal vessels, causing retinal hemorrhages and swelling on examination [3,4]. Injury to the posterior optic nerve commonly presents with a normal fundus examination because the retinal vessels re-main intact [2,3]. Typical TON presentation includes some combination of decreased visual acuity, visual field defects, relative afferent pupillary defect in unilateral cases, and chronic optic disc atrophy [2,3,5]. TON has been classified as a relatively rare cause of trauma induced visual impairment. While only seen in 1 per million in the general population, TON has been reported to occur in 0.5-5% of closed head injuries, and up to 40-72% of traumatic brain injuries with loss of consciousness [2,4-7].
Epidemiology TBI
Since World War I, traumatic brain injury (TBI) remains a devastating injury for our soldiers and veterans, with rising rates due to advancements in explosives and weaponry [8]. In the current time frame from 2000-2020, there were a total 430,720 cases of TBI reported within the military worldwide [8]. More than 185,000 Veterans who use VA for their health care have been diagnosed with at least one TBI [9]. The majority of those TBIs were classified as mild. TBI and its associated co-morbidities are also a significant cause of disability out-side of military settings [9]. Advanced weaponry plays a part in the high incidence of TBI, but also the advancement in medical care contributes to the rising diagnosis of TBI and observation of its chronic effects [8]. There is a direct well-established relationship between TBI and TON. In a ten-year longitudinal study, there was a 3-time greater risk for developing TON in those with TBI when compared to a population control group [6].
Within the military, parachute jumpers or paratroopers are prone to specific injuries including lower extremity, axial skeleton, and closed head injuries [10,11]. Parachute jumpers specifically have a higher risk for TBI, as it is reported to be the second most common injury experienced during paratroop procedures [10]. In a 2002 study at Fort Bragg, TBI prevalence and character was examined in 2,337 active-duty service members by a selfreported survey [10]. Of the surveyed soldiers, approximately 30% of Army parachute jumpers reported sustaining TBI’s during active duty compared to 13.5% in non-parachute soldiers [10]. TBI among paratroopers was more commonly due to parachute related activities (18.4%) versus non-parachuting activities like falls, vehicles crashes, or explosions, accounting for 8.4% within paratroopers [10]. This data demonstrated that the proportion of parachute related TBI was 1.4 times greater than non-parachute related TBI [10]. Invis’s 2002 data also demonstrated that in soldiers who had a history of TBI before the Army, paratroopers were at increased risk for sustaining a second TBI while in active service compared to non-paratroopers [10]. Various ocular injuries can occur from mTBI/TBI resulting in potential for vision loss (Table 1). Since active military and veterans are a subpopulation where TBI, and therefore TON may be more prevalent, proactive assessment and proper diagnostic measures should be emphasized in all post-TBI evaluation.
Veteran Disability Exams
The current Veteran Disability Exams require a comprehensive eye evaluation of the veteran. Visual acuity with and without best correction, intra-ocular eye pressures, and evaluation of the anterior and posterior segments of the eye with a dilated exam. Pupil reaction to light and accommodation as well as muscle balance are observed and recorded. Color vision and depth perception are also evaluated. In addition, kinetic visual field testing and interpretation are required. After recording all normal and abnormal findings a nexus is established to determine any causal relationship between military service and any documented and observed eye pathology. Any claimed condition by the veteran, because of his/her military service, is addressed by the physician. The nexus can be classified as follows: eye pathology that is Direct Service Connection, Secondary Service Connection, Aggravation of a condition that existed prior to service, or Aggravation of a nonservice connected condition by a service-connected condition. In order to obtain veteran disability benefits, the examiner only has to demonstrate that the diagnosed medical condition is as likely as not caused by an in-service incident or service-connected condition.
Malingering is always a concern when evaluating for disability benefits, but accurate assessment to identify true impairments should be actively pursued in all veteran disability evaluations. Veteran disability examinations require kinetic field testing but lack an indication or coverage for static field testing. Visual field defects can cause serious deterioration in quality of life, including the status of a patient’s independence. Visual disability may affect the ability of an individual to take care of themselves (hygiene, cooking, cleaning) and can cause safety issues such as ability to drive a vehicle. Disability payment for eligible veterans results in a tax-free monthly payment, which defers depending on type of claim, dependent status, and percent disability rating [12]. Table 2 shows the monthly payment veterans could receive with 70-100% disability rating [12].

Patients and Methods
In our prospective study to identify TON, we performed a comprehensive eye exam including both kinetic and static visual fields using the Humphrey Field Analyzer on all active military personnel on terminal leave and retired veterans who had a history of TBI and/or mTBI (head concussions) from any source, while in active military service. Veterans who had a history of any type of head injury before or after military service were eliminated from the study; however, these veterans were still evaluated for TON but are not included in our results. Veterans who had other eye pathology that could affect visual field testing such as glaucoma, diabetic retinopathy, MS etc. were also removed from the study group. Important variables that were examined included age, sex, cause of TBI and mTBI, bilateral v. unilateral TON diagnosis, and type of visual field loss. The study was conducted from May 25, 2021 to November 25, 2021. A total of 413 active military personnel who were in the process of terminal leave (discharge from active service) and veterans who were already discharged and seeking disability evaluation with possible benefits were examined by referral from QTC (qtc.com), VES (vesserrvices.com) and LHI (logistics.com) as part of a disability evaluation/benefit exam.
Results
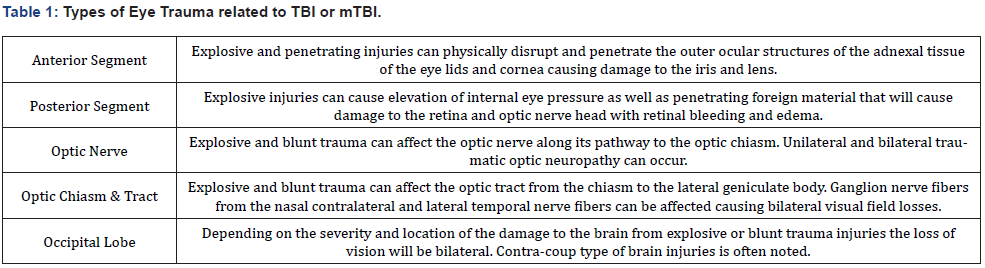
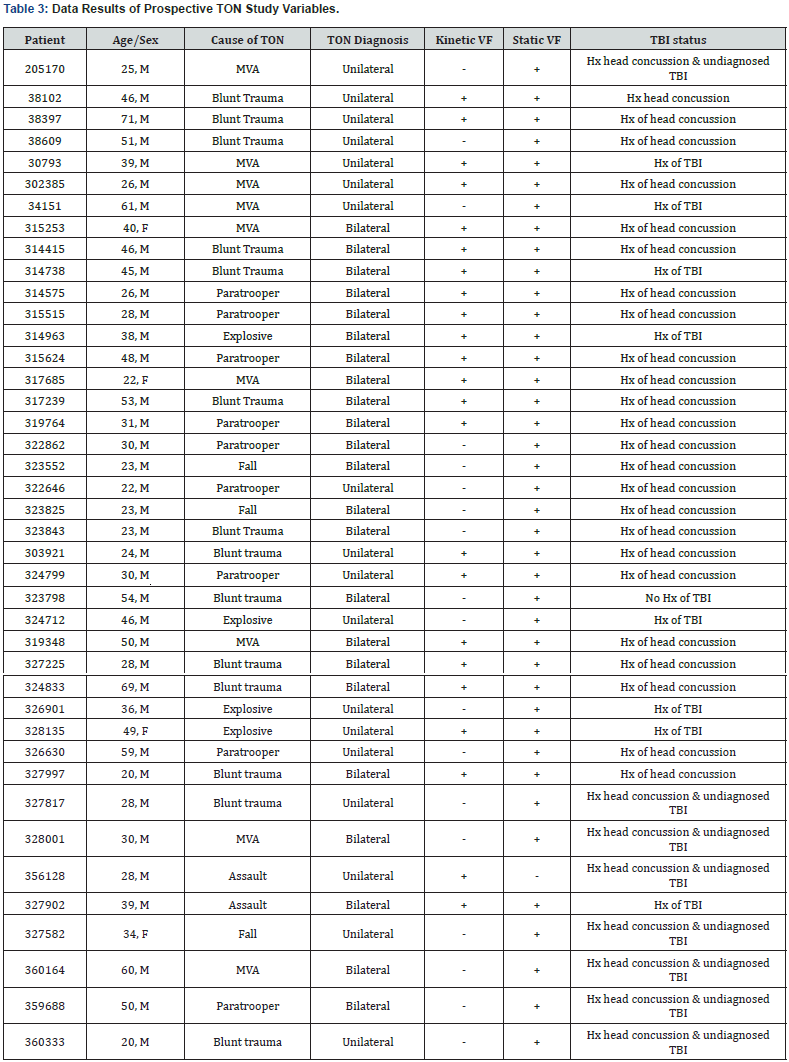
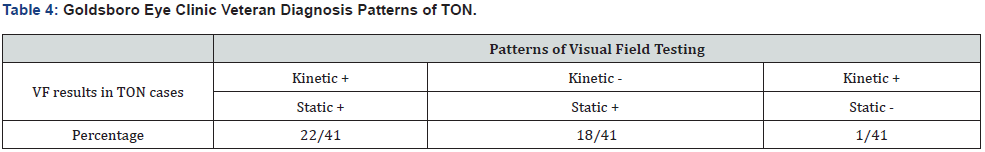
In this six-month prospective study, 41 cases of traumatic optic neuropathy were diagnosed out of the 413 veteran disability exams performed at the Goldsboro Eye Clinic (Table 3). Of these 41 cases, 56.1% of patients presented with bilateral TON (n = 23), versus 43.9% with unilateral TON (n= 18). The average age of TON patients was 38.3 years, with the vast majority of patients being male. Of the 41 cases of diagnosed TON, 24 were still active military and 17 were veterans with an average time of 15.8 years since discharge. Out of the 413 ex-ams, 51 patients had a known diagnosed history of TBI/mTBI. 41 cases out of the 51 patients (80.3%) with known diagnosis of TBI/mTBI had some evidence of TON. The causes of TON were divided into blunt trauma (any direct blow to the head being struck by/against), falls, motor vehicle accidents (MVA), explosions, and paratrooper-related activities (falls and/or blunt trauma). The most common cause of TBI/mTBI resulting in TON was due to blunt trauma (34%), with paratrooper-related activities as the second most common activity (22%) (Figure 1). Multiple patterns of visual field testing were observed between these cases, which are outlined in Table 4. Most importantly, it was recognized that neither individual static nor kinetic visual field testing confirmed every visual field deficit seen in these TON presentations. 18 VF defects were missed during kinetic field examination, while 1 VF defect was missed if individual static testing were used (Figure 2). The sensitivity of kinetic testing for TON in this study is 0.56, while sensitivity for static visual field testing in TON is 0.98.

Discussion
Visual dysfunction is a common presentation after traumatic head injury. In a 2019 MSMR report among military service members, the most common visual symptoms were subjective central vision loss, convergence insufficiency, visual field loss, and accommodative dysfunction [13]. A total of 430,000 TBI cases have been reported by the Defense and Veterans Brain Injury Center, since the year 2000, making TBI a considerable injury within the military population [13]. There is a clear, direct relationship between TBI/head injuries and TON. A longitudinal ten year follow up study established a 3-time increased risk of developing TON after a traumatic brain injury [6]. Even mild TBI can cause subtle damage to the optic nerve without widespread cortical neurodegeneration [12]. Although optic nerve injury is one of the most common events after TBI, nerve damage can be difficult to evaluate clinically [9]. This points to the importance of obtaining a medical history of concussion or head trauma in the evaluation of patients with unspecified visual field loss.
The variability in TON presentation makes subtle cases easy to overlook. TON often presents with sudden temporary visual loss, but routine neurological assessment can appear normal, and may not be a sensitive tool for evaluating potential optic neuropathy. Importance should be placed on a patient’s history and symptoms when evaluating for suspected TON, even in the context of cleared neurological status. When TON occurs from damage to the posterior optic nerve, retinal examination will not display any gross abnormalities, due to the lack of retinal vasculature disruption [2,3,14]. Damage to the retinal ganglion cells is also delayed, occurring 3-6 weeks post trauma, when signs of possible optic disc pallor and atrophy would first be appreciated on fundus examination [6]. Sometimes the only clinical sign of unilateral TON is the presence of a rapid afferent pupillary defect [2]. Due to the unpredictability in TON presentation, the protocol for TON workup includes visual acuity, color vision, visual field, RAPD testing, and fundus examination [3,15].
However, there is no pathognomonic visual field loss for TON. Visual field loss following partial avulsion of the optic nerve from the globe tends to correspond with the lesion (Figure 3) [14,16]. In Hughes’ series, 24 patients with intracanalicular TON recovered enough vision to record a visual field. Half of these cases demonstrated an inferior altitudinal defect with macular and upper field sparing. Nerve fiber bundle defects generalized constriction and depression, as well as central and paracentral scotomas were also reported [17]. The most common site of injury of the optic nerve is the intracanalicular portion of the nerve, followed by the intracranial portion, and intra-ocular area. Deceleration injuries from motor vehicle accidents account for primary cause (17 to 63%) of TON cases, followed by blunt trauma/ falls as the second most common cause [14]. Optic neuropathy is associated with traumatic brain injury and is more likely to occur when there is a loss of consciousness. The role of high-dose steroids and surgical orbital decompression in treating TON is controversial and no strong evidence for the use of these treatments have been expressed in the literature [2].
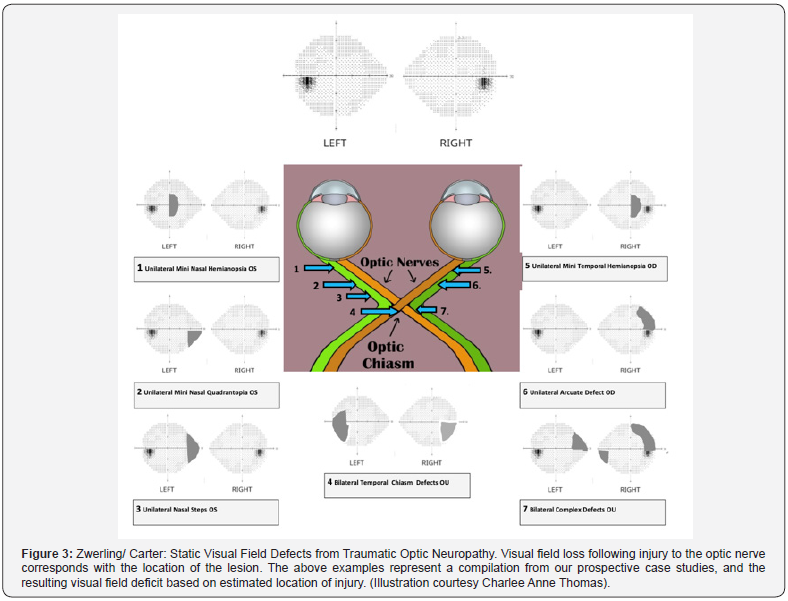
Anatomy & physiology of optic nerve
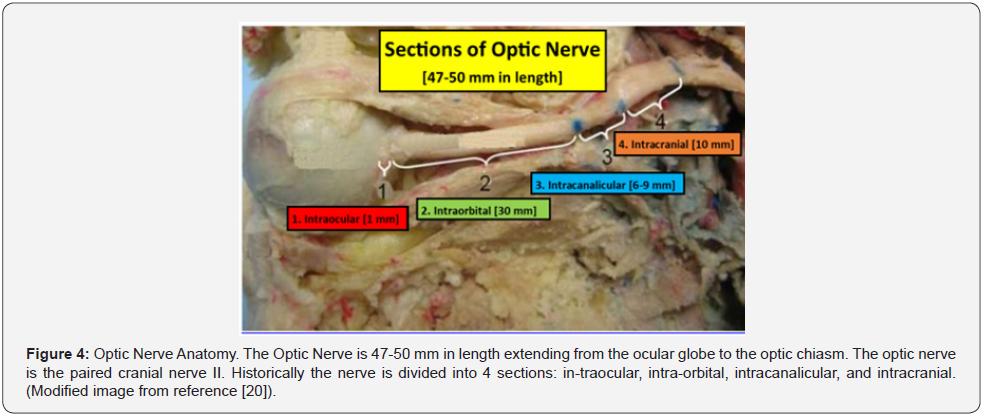
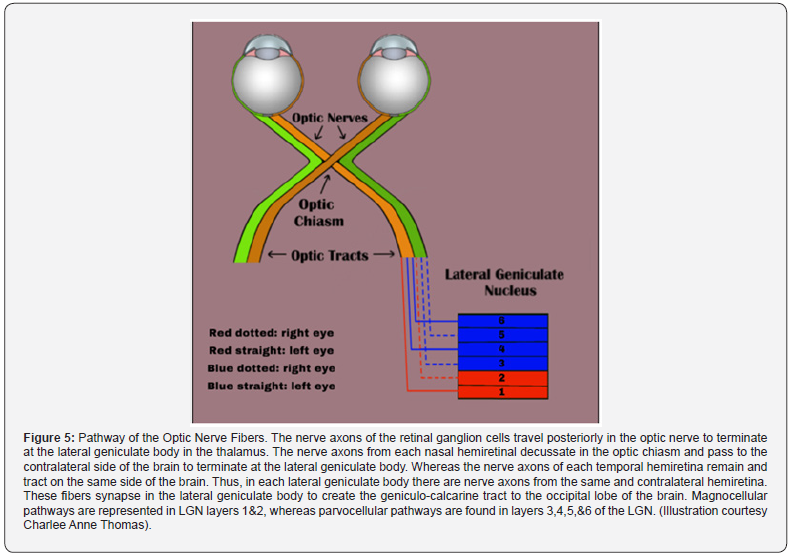
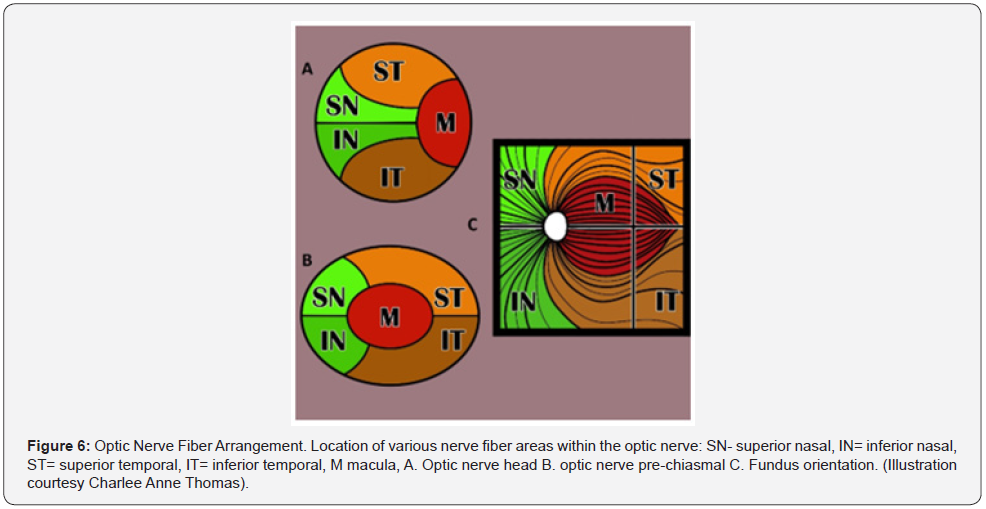
Before discussing the pathophysiology of TON, a brief review of the optic nerve anatomy would be appropriate. The optic nerve is 3-4 mm in diameter and measures 35-50 mm from the retina to the optic chiasm. The nerve is composed of intraocular (~1 mm), intra-orbital (30 mm) intracanalicular (6-9 mm), and intracranial (10 mm) segments (Figure 4) [18-20]. The axons comprising the nerve have their origin in the superficial nerve fiber layer of the retina (SNFL) and extend beyond the chiasm and optic tracts before synapsing within the lateral geniculate body [19]. From the synapse in the lateral geniculate body the 2nd order neuron extends to the occipital area of the brain where visual interpretation occurs (Figure 5). The topographic organization of the axons, as arranged by the retina, is preserved within the optic nerve. Except for its intraocular segment, the axons of the optic nerve are myelinated [18,19,21]. The optic nerve begins its formation at the optic nerve head or disc by the congruence of the superficial nerve fiber of ganglion cells (SNFL) [19]. The optic disc is 1.5mm in diameter. As these neurons pass through the Lamina Cribosa, myelin sheaths are added to the exterior of the individual nerve fibers, causing the size of the optic nerve to expand to 3mm [19]. The intraocular portion of the optic nerve is divided into 4 sections: I. SNFL, II. Pre-Laminar, III. Lamina Cribrosa, and IV. Retrolaminar sections [19]. The arrangement of these fibers change as the optic nerve continues its course to the optic chiasm (Figure 6) [19].
Most of the Intraocular optic nerve axons take a direct course as they exit through the meshwork of the Lamina Cribosa [20,21]. However, about 10% of the axons can be diverted to pass through the cribriform pores in the central and peripheral areas of the disc. Consequently, these axons are more vulnerable to alterations of the Lamina Cribosa in diseases like glaucoma and traumatic optic neuropathy [19]. The Lamina Cribosa forms a barrier between two differentially pressurized compartments: the intraocular space with higher pressure (IOP) and the retrobulbar space with a lower pressure retrobulbar cerebrospinal fluid pressure (CSFP) [11]. From a biomechanical point of view, the Lamina Cribosa constitutes a weak point in the mechanical load systems, and therefore, this location is where the stress can be concentrated either from long term issues with glaucoma or short-term event from the shearing and stretching forces generated by the displacement of the Lamina Cribosa from trauma (Figure 7) [19].

Pathophysiology of TON
Direct and indirect traumatic brain injuries can cause TON by both mechanical and/or ischemic damage to the optic nerve. Generally, direct injuries have a worse prognosis than indirect injuries, but sometimes TON injuries are so subtle there may be no external evidence [3,14,22]. Two mechanisms, primary and secondary, result in damage to the optic nerve. Primary injury occurs when mechanical shearing forces directly damage the nerve and vasculature, resulting in immediate tissue damage and irreversible loss [14-16]. Neuronal damage in TBI can be caused by either focal injury such as penetrating trauma, or diffuse axonal injury from blast wave propagation i.e. IED [8]. The energy from an explosive wave is capable of dramatically increasing the pressure within the orbit, thereby subjecting the optic nerve to damage [8]. Secondary injury occurs because of swelling and vasospasm, causing increased pressure that impinges axon flow within the nerve. The combined result is ischemia of the optic nerve that can lead to permanent or temporary loss of nerve function [14].
Evanson’s mice model of TBI demonstrated that optic nerve response to closed head injury was definitive axonal degeneration, showing cellular signs characteristic of astrocytosis and microglial inflammation, even in the absence of gross damage on neuroimaging [9]. They also demonstrated neurodegeneration in structure directly innervated by the optic tract, including the superior colliculus and lateral geniculate nucleus [9]. Shearing forces can acutely cause vascular and axonal injury, but additionally, increased intracranial pressure. Post-TBI has been shown to cause chronic thinning of the optic nerve sheath diameter [8,9,12]. Gilmore’s 2020 longitudinal study followed 139 veterans with mild TBI compared to veterans without a history of head trauma [23]. This study revealed a significant difference in the thickness of the retinal nerve fiber in veterans with chronic mild TBI [23]. Retinal nerve fiber layer (RNFL) thinning was associated with progressive visual field loss and was correlated with the severity of TBI. Interestingly, thinning of ganglion cells within the macula seemed to be relatively spared, which is suggestive to why patients do not always perceive subjective visual field changes over the course of time [23]. Post-traumatically, retinal hemorrhages, optic pallor and/or atrophy may or may not be visible on direct fundus exam depending on the location of the injury, as well as its extent [14]. Evaluation of the optic nerve function has been evaluated historically by visual field testing: kinetic and static visual field analysis.
Kinetic and Static Perimetry
Visual field testing is an important tool in providing information about location of optic nerve damage, monitoring for disease progression, and revealing visual impairments possibly unknown to patients [3,24]. Historically, visual field testing was performed with manual kinetic (Goldmann) technique and then later with an automated static (Humphrey) [25,26]. Besides the difference in stimulus delivery, Goldmann perimetry was distinct from Humphrey due to the need of a trained technician. In 2016, the Humphrey field analyzer 3 was developed to include both static and kinetic modes [25]. The new device permitted automated modes for both static and kinetic perimetry. The Humphrey field analyzer 3 was used for the assessment of the patients in this study (Figure 8).

Automation of perimetry has provided standardization and reduction in examiner bias, but the difference in testing mechanism still results in delivery of different outcomes. Kinetic perimetry uses various isomers of light that move into the patient’s periphery, thereby outlining the distinct shape of the visual field [26]. Static perimetry measures sensitivity to a stationary stimulus presented for 200ms, while the patient keeps their gaze centrally fixated (Figure 8) [26]. While both kinetic and static perimetry have been shown to reliably detect visual field loss, the two tests are not equal [25]. The fundamental mechanism of each test is stimulating different retinal ganglia cells [19,27]. Information about large, fast moving objects (low spatial frequency; high temporal frequency) with lack of color is detected primarily through M cells in the retina ganglia layer and is projected through the magnocellular pathway to layers 1 & 2 of the lateral geniculate nucleus (LGN). Meanwhile information about small, slow stationary, colorful objects (high spatial frequency information; low temporal frequency information), is largely detected by P cells, which projects through the parvocellular pathway to layers 3, 4, 5, & 6 of the LGN [19,28].
The moving stimulus in kinetic perimetry utilizes the corresponding magnocellular pathways, while the stationary stimulus in static perimetry uses the parvocellular pathways [19]. Differences in visual field profiles between static and kinetic testing has long been observed and is referred to as static-kinetic dissociation [27,28]. The occurrence of static-kinetic dissociation demonstrates that these perimetry techniques are not equivalent, but instead are complimentary to each other. It has been noted in the literature that static perimetry is generally superior to kinetic in evaluating the central field of vision, while kinetic perimetry is better at evaluating the extent of the peripheral field [24,29-32]. A comparison study of varying neuro-ophthalmic disease found that kinetic perimetry tended to underestimate both the depth and spread of central/paracentral scotomas [33]. Another 2009 comparison showed static perimetry was able to detect small paracentral scotomas while the relative kinetic perimetry remained normal [32]. Static perimetry was 19% more sensitive for paracentral scotoma defects in this study [32]. In our prospective study, static perimetry was more sensitive for VF defects in TON cases, mainly in revealing para-central type scotomas. Kinetic perimetry proved useful in revealing peripheral defects that static perimetry missed. The degree of error in using these perimetry tests individually confirms the hypothesis of our earlier case report: a single visual field analysis is not adequate in the examination of visual problems resulting from TBI.
Evaluation and Recommendations
To our knowledge, this study demonstrates a missed TON diagnosis due to the lack of static field visual field testing, as well as missed interpretation of kinetic visual fields in some of the other cases. A pattern of undiagnosed TON cases is present in the military population where mild TBI (mTBI) with TON are more prevalent and only kinetic testing is used for the visual field examination. We would recommend that all active service military personal and all veteran eye screening exams need to be amended to include both static and kinetic visual field testing with a history of TBI and/or mild TBI head concussions (Table 5). This recommendation allows the strengths of both tests to be utilized in the evaluation of highly variable outcomes. Static perimetry has a higher sensitivity for detecting defects in the central 30 degrees of vision, while kinetic perimetry provides a better assessment of the periphery. Initiating routine use of both perimetry testing would not significantly change the cost, time, or personnel needed in workup, but could prevent a proportion of false negative results and unidentified visual disabilities. Furthermore, there is an objectively higher percentage of TON patients among military paratroopers as compared to all the other sectors of military service.

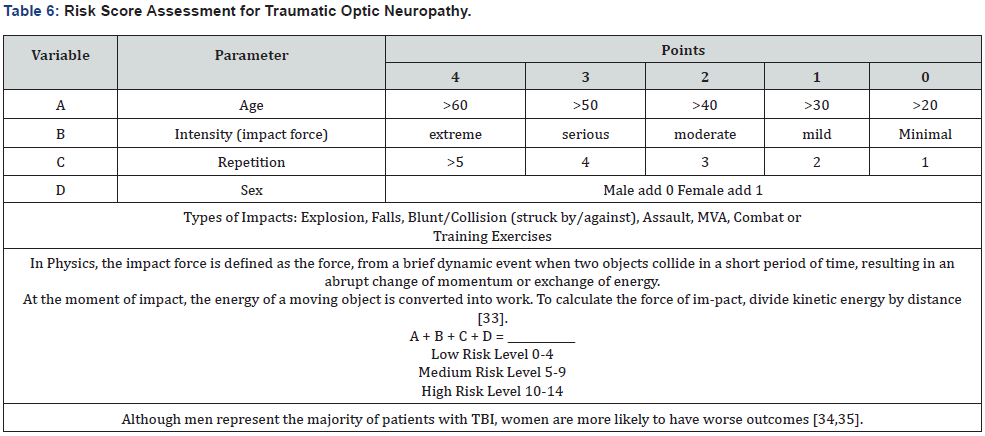
It is also important to anticipate that patients who have TBI and TON together, that there may be a complex presentation of visual loss in either the kinetic and/or static visual fields. Some of the loss is due to cortical brain injury and other losses may be due to damage to the optic nerve. Based on our accumulated data, we have created a Risk Score for Traumatic Optic Neuropathy [34,35,36] (Table 6). This TON Risk Score can be helpful in identifying those individuals that are at risk or might already have TON and are unaware. Finally, there needs to be an updated methodology in determining a veteran disability rating for confirmed diagnoses of traumatic optic neuropathy. Many of the study cases had problems with oculomotor issues like convergence, accommodation, and visual tracking of moving objects in addition to the confirmed visual field losses and/or visual acuity loss. Currently, disability ratings are limited to visual acuity and visual field calculations and do not have a clear rating system for oculomotor dysfunction.
Role of Electrophysiological Testing
With the development of electrophysiological testing, we will be studying these tests as a supplementary method to confirm optic nerve pathology in future prospective studies. Ophthalmic electrophysiology analyzes the electrical signal generated in vision [37], which involves the creation of an electrical signal generated in the retina and subsequently propagated through the optic nerve and optic tracts in the brain to the occipital lobe. This electrical signal is interrupted in the visual cortex. There are various electrophysiological testing methods to measure this electrical signal by isolating the signal production in the retina with precise measured amounts of flashes of light and comparison to the produced electrical signal to the brain [37].
i. Full field ERG (ffERG): measures the overall response in the retina specifically the rods and cones to specific flashes of light [38]. This test is very helpful in the management of diabetic retinopathy [38].
ii. Multi-Focal ERG (mfERG): measures specific areas of the retina to assist in the diagnosis of various types of retinal disease [39]. This test is especially useful for patients on Plaquenil therapy [39].
iii. Pattern ERG (PERG): The pattern electroretinogram calculates the electrical activity of the retina in response to a test stimulus, such as a reversing checkerboard stimulus, and measures the function specifically of retinal ganglion cells [13]. PERG can help in the determination of dysfunctional cell activity and is useful in detecting early disease abnormalities in glaucoma and diabetic retinopathy [23]. Pattern electroretinogram (PERG) has been useful in detecting and evaluating early damage to retinal ganglion cells (RGC) [13]. Simultaneous recording of the PERG and VEP have proven beneficial in detection and differential diagnosis of demyelinating optic neuropathies and separation from macular disease.
iv. Visual Evoked Potential (VEP): measures the time for a visual stimulus to travel from the eye to the occipital cortex in the brain [37]. This test is designed to demonstrate alterations in optic nerve function and injuries in the anterior part of the visual pathway. Although not specific, VEP aids in detecting diseases that affect the optic nerve like optic neuropathies, glaucoma, and tumors compressing the optic nerve [37]. VEP has demonstrated more accuracy in the evaluation of pre-chiasmatic disorders. Another application of VEP is to quantify visual system function following trauma. It is not unusual that compression of optic pathways immediately after severe trauma results in no recordable VEPs [37]. However, VEPs may be recordable days later when inflammation subsides [37]. Interpretation of VEPs must be considered within the context of the patient’s clinical appearance and information available from other tests and examinations. VEP results can be predictive of visual recovery in traumatic optic neuropathy [2]. VEP can provide an objective assessment of visual field defects not yet present on automated perimetry in patients with glaucomatous and non-glaucomatous optic neuropathies [23,37]. Electrophysiological testing is an interesting area to utilize in the diagnosis of traumatic optic neuropathy. Additional comparative studies need to be performed to determine which particular method offers the best help in the overall diagnosis of traumatic optic neuropathy.
Prevention with Protection
Based upon the ground-breaking research in Chronic Traumatic Encephalopathy (CTE) among retired NFL players by Bennet I. Omalu, M.D., M.P.H.T, new helmet design and protocols for football player safety have been developed [40,41]. By improving military helmet design, we can reduce mild TBI and related TON. Not only will this approach better protect our military personnel it will also reduce costly medical care and disability payments after military discharge. Based on regular disability rating and the average age of TON in this study being 38.3 years, a veteran could be receiving disability payments for most of their life. Following average life expectancy, 100% disability payments could approximate 1.5 million US dollars per person. Parachute jumpers wear slightly modified Army Combat Helmets (ACH) [42,43] (Figure 9). Some para-chute jumpers are given CRYE Helmets which offer better protection [44] (Figure 10). According to verbal discussions with various military parachute jumpers, not everyone can get these helmets because “they are too expensive”. After each parachute jump, the paratrooper must report to an on-site medic if they suffered any bodily/ head injury. If the paratrooper reports any type head injury or abnormal symptom, then the paratrooper may not be allowed to continue as a paratrooper. Clearly, if these statements are true, this type of evaluation is too subjective and lacks any in-depth medical evaluation. It is appropriate that an onsite medic is present to respond to any immediate concern; however, a more objective analysis needs to be performed on all paratroopers on a regular basis.


Future Diagnostic Concerns
Every patient that was examined and diagnosed with TON was given an explanation of this medical condition. Our concern was to anticipate future diagnostic potential issues. Namely, some of the visual field losses from TON could easily be interpreted as a loss from another disease. For example, it is possible that another eye care professional, who is unaware of the TON history, might diagnosis and treat the patient for normotensive type glaucoma, or optic neuropathy from an unknown cause like multiple sclerosis, etc. By informing and educating the patient about TON, hopefully, a future incorrect diagnosis and/or treatment could be avoided.
Future Research Needed
Our Prospective Study represents only the beginning in understanding how traumatic optic neuropathy is involved with TBI and mTBI cases among our military personnel. We were limited in our study due to only one exam allowed for each veteran, who was then lost to our follow up. We did not have access to spectral Optical Coherent Topography (SD-OCT) domain analysis and had to rely on the older stratus OCT machine. With SD-OCT testing, a detailed study of the retinal nerve fiber layer, optic nerve head, and the ganglion cell complex could be performed and used as an objective comparison especially in cases of unilateral TON as well as additional confirmation of traumatic optic neuropathy.
Furthermore, we were not able to coordinate follow-up of the patient with neurology and neuro-ophthalmologists who could evaluate further with radiologic studies of CT scans and MRI testing, which would provide more evidenced based medicine in the diagnosis of traumatic optic neuropathy. None the less, we did make recommendations for neurologic referral and further evaluation to the veteran’s disability benefit. Currently we are performing another prospective study adding VEP analysis to our protocol.
Conclusion
We reported the results of a prospective study to detect occult cases of traumatic optic neuropathy among the active military and veteran population that was performed as part of a disability benefit evaluation. The research used our new protocol of kinetic and static visual field testing in all cases of suspected and/or confirmed cases of TBI and mild TBI. The results of our investigation demonstrated the value of this new protocol in order to diagnose cases of traumatic optic neuropathy as well as confirm suspected cases of TBI and/or mTBI. Moreover, the study clearly identified military paratroopers as a high-risk military group for under reported head injuries. We would recommend periodic kinetic and static field testing especially for this military group. Finally, based upon our research, we would recommend further investigation into traumatic optic neuropathy among military personnel as well as development of safer helmet design for the military paratroopers. Using the NFL research for safer helmets as a starting point, an improved helmet design could reduce concussion events and reduce permanent brain damage to our veterans.
References
- Zwerling C and Carter L (2021) Unilateral Traumatic Optic Neuropathy Case Report: a rarity or a pattern. Ann Rev Research 6(5): 555699.
- Karimi S, Arabi A, Ansari I, Shahaki T, Safi S (2021) A Systematic Literature Review on Traumatic Optic Neuropathy. J Ophthalmol 2021: 5553885.
- Srinivasan R, Chaitra S (2008) Traumatic Optic Neuropathy- a review. Kerala Journal of Ophthalmology 100 (1).
- Man PYW (2015) Traumatic optic neuropathy- clinical features and management issues. Taiwan Journal of Ophthalmology 5(1): 3-8.
- Lee V, Ford RL, Xing W, Bunce C, Foot B (2010) Surveillance of traumatic optic neuropathy in the UK. Eye 24(2): 240-250.
- Chen Y, Liang C, Tai M, Chang Y, Lin T, et al (2017) Longitudinal relationship between traumatic brain injury and the risk of incident optic neuropathy: A 10-year follow-up nationally representative Taiwan survey. Oncotarget 8: 86924-86933.
- Steinsapir KD, Goldberg RA (1994) Traumatic Optic Neuropathy. Survey of Ophthalmology 38(6): 487-518.
- Hussain SF, Raza Z, Cash ATG, Zampieri T, Mazzoli RA, et al (2021) Traumatic brain injury and sight loss in military and veteran populations– a review. Military Med Res 8(42).
- Traumatic Brain Injury (2020) Veterans Affairs office of Research and Development.
- Ivins BJ, Schwab KA, Warden D, Harvey S, Hoilien M, et al (2003) Traumatic Brain Injury in U.S. Army Paratroopers: Prevalence and Character. J Trauma: Injury, Infection, and Critical Care 55(4): 617-621.
- Craig SC, Morgan J (1997) Parachuting injury surveillance, Fort Bragg, North Carolina, May 1993 to December 1994. Journal of Military Medicine 162(3): 162-164.
- Veterans Disability Compensation rates (2021). Veterans Affairs Benefits and Healthcare.
- Junghardt A, Wildberger H, and Torok B (1995) Pattern electroretinogram, visual evoked potential and psychosocial functions in maculopathy. Doc Ophthalmol 90(3): 229-245.
- Hoyt WF, Miller N, Biousse V, Kerrison JB (2005) Traumatic Optic Neuropathies. In Walsh and Hoyts Clinical Neuro-Ophthalmology. Lippincott Williams & Wilkins, Philadelphia 431-447.
- Lin HL, Yen JC (2020) Acute monocular nasal hemianopia following a mild traumatic brain injury. Medicine 99(30).
- Gupta D (2021) Traumatic Optic Neuropathy. AAO Eyewiki.
- Steinsapir KD, Goldberg RA (2005) Traumatic Optic Neuropathy Update. Comprehensive Ophthalmology Update 6(1): 11-21.
- Crumbie L (2021) Optic Nerve.
- Salazar JJ, Ramirez AI, Hoz RD, Garcia ES, Rojas P, et al (2018) Anatomy of the Human Optic Nerve: Structure and Function. IntechOpen.
- Acharya B (2018) Anatomy of the Optic Nerve.
- Moraes CG (2013) Anatomy of the visual pathway. J Glaucoma 22 Suppl 5: 2-7.
- Guern A, Delesalle C, Borry L, Chekroun J, Gruchala C, et al. (2016) Traumatic optic neuropathy: report of 8 cases and review of the literature. J For Ophtalmol 39(7): 603-608.
- Mantravadi A (2017) Comparing sd-tVEP and PERG tests between patients with glaucoma, patients with high eye pressure, and normal patients. Clinical Trials Gov. Identifier: NCT02604953.
- Kedar S, Ghate D, Corbett JJ (2011) Visual fields in neuro-ophthalmology. Indian Journal of Ophthamol 59(2): 103-109.
- Rowe FJ, Hepworth LR, Hanna KL, Mistry M, Noonan CP (2019) Accuracy of kinetic perimetry assessment with the Humphrey 850; an exploratory comparative study. Eye 33(12): 1952-1960.
- Carroll JN (2013) Eye Rounds Ophthalmology and Visual Sciences: Visual Field Testing from one medical student to another.
- Rowe FJ, Noonan C, Manuel M (2013) Comparison of octopus semi-automated kinetic perimetry and Humphrey peripheral static perimetry in neuro-ophthalmic cases. Ophthalmol 2013: 753202.
- Phu J, Kalloniatis M, Wang H, Khuu SK (2018) Differences in Static and Kinetic Perimetry Results are Eliminated in Retinal Disease when Psychophyscial Procedures are Equated. Transl Vis Sci Technolo 7(5): 22.
- Pineles SL, Volpe NJ, Miller-Ellis E, Galetta SL, Sankar PS, et al. (2006) Automated Combined Kinetic and Static Perimetry: An Alternative to Standard Perimetry in Patients with Neuro-ophthalmic Disease and Glaucoma. Arch Ophthalmol 124(3): 363-369.
- Crabb DP (2015) Visual Fields. In: Glaucoma (second edition). B. Saunders, New York: 109-124.
- Beck RW, Bergstrom TJ, Lighter PR (1985) A clinical comparison of visual field testing with a new automated perimeter, the Humphrey Field Analyzer, and the Goldmann Perimeter. Ophthalmology 92(1): 77-82.
- Nowomiejska K, Rejdak R, Zagorski Z, Zarnowski T (2009) Comparison of static automated perimetry and static kinetic perimetry in patients with bilateral visible optic nerve drusen. Acta Ophthalmology 87(7): 801-805.
- Charlier JR, Defoort S, Rouland JF, Hache JC (1989) Comparison of automated kinetic and static visual fields in neuro-ophthalmology patient. Perimetry Update: 3-8.
- Georgiev GZ. (2021) Impact Force Calculator.
- Farace E, Alves WM (2000) Do women fare worse? A meta-analysis of gender differences in outcome after traumatic brain injury. Neurosurg Focus 8(1): e6.
- Bazarian JJ, Blyth B, Mookerjee S, He H, Mcdermott MP (2010) Sex differences in outcome after mild traumatic brain injury. J Neurotrauma 25(3): 527-539.
- Creel D (2015) Visually Evoked Potentials. WebVision.
- Asanad S, Karanjia R (2021) Full Field Electroretinogram. Statpearls Publishing. Treasure Island, Florida, USA.
- Asanad S, Karanjia R (2021) Multifocal Electroretinogram. Statpearls Publishing, Treasure Island, Florida, USA.
- Omalu BI, DeKosky ST, Hamilton RL, Minister RL, Kamboh MI, et al (2006) Chronic Traumatic Encephalopathy in a National Football League Player: Part II. Neurosurgery 59(5): 1086-1093.
- NFL Helmet Innovation (2021) NFL Player Health and Safety.
- Carr FJ (2017) Approved Use of Static Line Operations. United State Army Advanced Airborne School, Fort Bragg NC.
- Advanced Combat Helmet (2006).
- Crye Airframe helmet (2021) CRYE Precision.






























