- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Improving the Problems of Writing Chemical Symbols, Formulae and Chemical Equations an Action Research
Dula DE*
*Department of Chemistry, Nekemte College of Teacher Education, Ethiopia
Submission: September 25, 2018; Published: November 13, 2018
*Corresponding author: Dula DE, Department of Chemistry, Nekemte College of Teacher Education, Post Box No. 88, Nekemte, Ethiopia
How to cite this article: Dula DE. Improving the Problems of Writing Chemical Symbols, Formulae and Chemical Equations an Action Research. Ann Rev Resear. 2018; 4(3): 555636. DOI: 10.19080/ARR.2018.04.555636
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Abstract
Objective: The researchers interested to assess and improve the general understanding of Generalist Focus Year II students in Nekemte College of Teacher education on chemical symbols, chemical formulae and chemical equations which are the fundamental elements of chemistry for anyone to begin learning this subject.
Methods: The study was both qualitative and quantitative type of research, because both numerical and non-numerical data were collected during the study through questionnaire, classroom observation, pre- and post- attitude tests and written tests. As type and source of data, the study used only primary. We used all the population as samples (totally 98 students in both sections) purposely. For the implementation of the action research, we first prepared action plan, applied the interventions, analyzed the changes and reported the results.
Results: Based on our null hypothesis, from both attitude tests (questionnaire) and written tests (for both pre-and post-) there is progressive change observed after interventions. From the base line data questionnaire of 15 items, almost 79.5 % of the respondents disagreed for items 1-11 (except item 5) which are assessing the students background on chemical symbols, chemical formulae and chemical equations and very high interest was raised from the students on items 12-15 (highest agreement for both item 12 & 13 by 93.88 % response) which were asking students’ interest to learn the basic elements of chemistry languages. After interventions, both post-tests showed correlatively progressive figure in addition to the observations conducted during all interventions. For the attitude test analysis almost, the reverse responses were recorded with agreement response (in average 71.93 %) and for the written test result analysis at p (0.05) the t-statistic is greater than t-critical in all the three cases i.e. chemical symbols, chemical formulae and chemical equations.
Keywords: Chemical Formulae; Chemical Equations; Chemical Symbols; Focus; Generalist; Qualitative Analysis and Quantitative Analysis
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Introduction
From our experience of teaching in the Nekemte College of Teachers Education (NCTE) since the introduction of linear and stream case in Educational colleges of Ethiopia, we have found that most of our students lack confidence in writing chemical symbols and Formulae. This problem was aggravating especially with the 2nd and 3rd year generalist students. It was what we have been confronted from student with different activities like class work, individual and group activities, and tests on different courses such as BNSc.101, BNSc. 102 and general chemistry (Chem. 201) in recent cases. When such difficulties occur in chemistry courses, it is difficult to measure whether the objectives of courses attained or not. In addition we have assessed that most of our students hesitate to choose Integrated and Generalist streams after completing bridge courses.
Literature Review
Studies of [1,2] argued that success in studying Chemistry depends upon the familiarity of students with a few basic ideas, conventions, and methods upon which later studies are built. When a student has achieved mastery of them, further studies can be pursued with greater confidence. One of the studies [1] further adds that without mastery of these concepts, it is difficult for students to find higher levels of study in Chemistry. Specially, the use of chemical symbols, Formulae, writing chemical equations, calculations involving moles (solids, gases, and solutions) etc. are areas where students of chemistry beginners face most challenges. Action research, according to [3] defines as “a systematic collection of data in order to answer a research question for the purpose of improvement”. For Lo Castro (1995) is “one form of classroom centered research which is seen as being small scale and situational focused on a particular problem, to try to understand and perhaps solve some concrete problem in an individual teacher’s classroom” [4].
Thus, action research solves the problem and brings improvement to the student nationwide. It gives the area to the teacher in solving the problem faced by the students. There are three methods of doing action research, quantitative, qualitative and mix method. This paper focuses on a mixed method where results are quantified and analyzed accordingly. Research has become primary elements in education system around the world. With the developmental stages in the country, research would help to face adverse competition. It will moreover help to understand the need of the society and will help accordingly tobring development. With stage changes must happen. Differentdisciplines of sciences Biology, Chemistry and Physics are ofimportance now a day. Coming to chemistry, student across thecountry finds it a difficult subject to learn. They even go to theextent of losing interest to study. Thus, our action research willmotivate and inculcate interest in Chemistry, especially balancingchemical equation where student finds it difficult.
According to Johnstone [5] “Chemistry is a difficult subjectfor students. The difficulties may lie in the capabilities of humanlearning as well as in the intrinsic nature of the subject.” Chiu[6] believes that “Chemistry is a world filled with interestingphenomena, appealing experimental activities, and fruitfulknowledge for understanding the natural and manufacturedworld. However, it is complex.” As a result of the difficult andcomplex nature of chemistry and also the fact that it is one ofthe most conceptually difficult subjects on the school and higherinstitution curricula, it is of major importance that anyoneteaching chemistry is aware of the areas of difficulty in the subject.The concepts and principles in chemistry range from concreteto abstract. Many students of chemistry find certain conceptsdifficult to comprehend. The root of many of these difficulties thatstudents have in learning chemistry is traceable to inadequateunderstanding of the underlying concepts of the atomic model,and how these are used to explain macroscopic properties andlaws of chemistry [7].
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Difficulties Faced in Writing Chemical Symbols and Formulae
Difficulties in the learning of chemistry can be precipitatedby a lack of chemistry language skills. [8] found that studentsexperienced greater problem in interpreting symbols than wordscorrectly (as cited in research of [9]. Danili & Reid [10] studiedthe effects of working memory space and field-dependency onthe learning of chemistry by Greek students. Learning not onlyof chemistry, but of all new information will fail if the workingmemory space is overloaded. This could occur if students aregiven too much information at once. Moreover, if studentsstudy chemistry in a language other than their mother tongue,difficulties experienced in chemical language could be linguistic,contextual or cultural in nature.
The understanding of valency, appreciation of concepts ofpolyatomic ions and molecules and ultimately the production ofcorrect chemical formulae will depend on student’s knowledge ofbonding. Unfortunately, concepts in chemical bonding are highlyabstract and it appears that only the most able students will bein a position to apply their knowledge of bonding effectively toscaffold the writing of chemical formulae [11].
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Difficulties Faced in Writing Chemical Equations
Chemical equations can be defined as symbolic andquantitative representations of the changes that occur in theprocess of chemical reactions, based on the principle that matteris neither created nor destroyed during chemical reactions. Forexample, the chemical equation: shows that A and B are thereactants while C and D are the products. The subscripts x, y, p andq are the stoichiometric coefficients which represent the relativeamount of substance of the reactants and products. The singleheadedarrow indicates the direction of the reaction and showsthat the reaction is an irreversible one. The arrow means “gives”,“yields” or “forms” and the plus (+) sign means “and”.
However, studies have shown that the ability to writechemical equations correctly is not a simple one [11,13]. It isone that requires a functional understanding of the requisitesubordinate concepts of atoms and atomicity, molecules andmolecular formula, atomic structure and bonding, valency,use of brackets, radicals, subscripts and coefficient and molarratio [11]. Studies conducted by Savoy [11] & Hines [14] havereported that chemistry students often have great difficulties inboth acquiring and using the skills required to balance chemicalequations. A similar study conducted by Johnstone, et al. [15] inScotland revealed that students in senior high schools are rarelyconfident about writing chemical equations and then carrying outcalculations based on them. A study by Anamuah-Mensah & Apafo[16] likewise revealed that students in Ghanaian senior highschools have difficulties in learning certain chemical concepts,including chemical combination. Approximately two-thirds ofthe students who took part in the study indicated that the topicchemical combination was either difficult to grasp or nevergrasped. Findings from research conducted by Lazonby, et al.[17]; Bello [18] have shown that students’ persistent difficulties insolving stoichiometric problems are partly associated with theirinability to represent chemical equations correctly.
Chief Examiners’ (CE) reports available through the WestAfrican Examinations Council (WAEC) confirm that senior highschool students experience difficulty when writing chemicalequations. The 1994 CE report showed that most candidateswere unable to write balanced chemical equations for the SeniorSecondary School Certificate Examination (SSSCE) chemistrypaper. The 1995 CE report followed suit and reiterated thatmany candidates demonstrated problems when writing chemicalequations. In 1999, the CE report indicated that students wereunable to write equations for reactions between Bronsted-Lowrybases and concentrated HCl. In 2001, the CE reported that thewriting of ionic equations was poorly handled by candidates [19].The 2004 chemistry theory paper required candidates to write abalanced chemical equation for the production of oxygen whenKClO is heated and then calculate the volume of the dry oxygengas evolved [20]. The examiners’ CE report for the above questionnoted that candidates had problems writing the equation correctlyand hence could not get the correct mole ratio [21].
Based on the above, it is clear that over the years, studentshave experienced serious problems when writing chemicalequations even though this is a basic requirement in chemistry.Without the proper writing of the chemical equation, studentscannot subsequently solve or analyze equations.
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Methods to Practice Writing Chemical Symbols and Formulae
Marais & Jordaan [8] recommended that students’understanding of symbols should be tested by including meaningitems in content related tests and students should be discouragedfrom regarding chemical symbols as merely short-hand notations.Providing with a glossary of symbols and assigning group orindividual exercises to supply correct symbolic notation is anothertechnique [9]. Chunking or grouping pieces of information isimportant to reduce the demands on the amount of informationto be held in the working memory. It will be affected by students’prior knowledge, experience and skills in a particular subject.Since chunking is highly individualized, students should be giventhe opportunity to develop their own chunking techniques [10].The second language science students require the opportunity topractice science in the presence of more capable peers and theyneed to be introduced overtly to the language requirements of theparticular discipline [22].
According to Bradley & Steenberg [9] the difficulty of lack ofchemistry language skill can be solved by maximizing exposureto chemical language. Teachers should not assume that studentsare familiar with chemical terms; rather they should introducethe terms carefully. The symbols of common elements should bememorized thoroughly, and as well the less common ones mightalso need to be memorizing as they come to attention (Clark,1982). Chemical formula writing worksheet solutions is another.It is a tabulate form consisting of boxes. We have to write chemicalFormulae for the compounds in each box. The names are found byfinding the intersection between the cations and anions writtenin the boxes. We can also write formulae using valencies. To writechemical formula using valencies, we need to know how to use thezero-sum rule; “for neutral chemical formulae containing ions, thesum of positive valencies plus negative valencies of the atoms in acompound must equal to zero”.
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Methods to Practice Writing and Balancing Chemical Equations Understanding the Language of Chemistry
Student should be familiar with the language of chemistryso that they can easily balance chemical equation. Knowingsymbols, knowing the difference between ions, atoms, moleculesand compound will invariably play important role. And not onlyknowing formula of compound but also the teacher should helpstudents to think why chemical equation is balanced; will alsohelp to easily balance the chemical equation. According to Beek& Louters [23], Marais & Jordaan [8] and Danili & Reid [10] alsostates “Difficulties in the learning of chemistry can be precipitatedby a lack of chemistry language skills”. They provide followingrecommendation to maximize students learning:
a. Students exposure to chemical language should be maximized.
b. Teachers should not assume that students are familiarwith chemical terms and terms should be introduced carefully.
Marais & Jordaan [8] found that students experienced greaterproblems in interpreting symbols than words correctly. Based ontheir findings, they recommended that:
a. Students understanding of symbols should be tested byincluding meaning items in content related tests,
b. Students should be discouraged from regarding chemicalsymbols as merely short-hand notations which could be adaptedto suit the individual user,
c. Students should be provided with a glossary of symbols,and
d. Students should be given group or individual exercisesto supply correct symbolic notation [9].
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Motivation
According to Johnstone and Kellett there is no doubt thatmotivation to learn is an important factor controlling the successof learning and teachers face problems when their students donot all have the motivation to seek to understand [24]. Motivatingstudents in learning chemistry play key role when teachingstudents balancing chemical equation. Whenever they get theirequation correctly balanced one should reinforce them andthey overtly motivated for future learning. Through motivationstudents participate more and more and this makes the teacherto be highly interested to work hard at the same time to helpstudents furthermore.
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Methods
The Subjects of the Study
The target population of the study as aforementioned, areall Generalist Focus of Year-II regular and evening students ofNekemte CTE in semester two and 2017 academic year. Thereare two sections (one Regular-IIA and one Evening-IIA) in whichtotally 98 students were enrolled and attending their education.
The Sample and Sampling Techniques of the Study
All of the populations are included in the study purposely.
Research Approach and types & Sources of Data
The study was both qualitative and quantitative type ofresearch, because both numerical and non-numerical data werecollected during the study. As type and source of data, the studyused only primary.
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Instruments for Gathering Data
Questionnaire
The researchers employed questionnaire as a primary tool forgathering data. The questionnaire contains both open-ended andclose-ended questions. This is because it is the most appropriatetool to obtain qualitative as well as quantitative informationrelative to the other methods. Besides, questionnaire is easy forthe researcher to manage the construction of the questions andanalysis of the responses. Moreover, it
Minimizes respondents’ difficulties of anonymity and reduceseffect of biased conclusion and misinterpretation of the data.
Classroom Observation
The other instrument used was classroom observation. Theresearchers decided to use this instrument to obtain data aboutthe actual classroom situation during instructions. This wasexpected to enable the researchers collect information regardingthe participation of students, the way the teacher implements theactive learning methods and other related classroom conditions.
Written Test
Pre- and post-tests were carefully prepared by the team andconducted to assess performance of students in all the targeteditems in the research i.e. chemical symbols, chemical formulae andchemical equations.
a. Baseline Data collection
1. Checking accuracy in writing chemical symbols and formulae,
2. Common mistakes everybody makes,
3. Barrier in writing chemical symbols and formulae,
4. Pre-test (including attitude test).
b. Methods of Data Analysis
The findings of the study were put into three categories inrelation to our specific research questions that we found fromthe data collected (i.e. checking accuracy of students in writingchemical symbols, formulas and equations; barriers in writingthese basic elements and the ways of teaching that enhancestudents’ learning of chemistry). Therefore, the data collectiontools used were to answer the specific research questions as muchas possible.
The data gathered from the respondents were organizedand analyzed by the use of tables and percentages. This wasconducted in such a way that the data were tabulated in tables andgraphically indicated to illustrate more quantitatively describedfor the test results which were interpreted by using T-test methodfor the pre-test and post-test data. Responses gathered fromtargeted students in the Attitude tests were tabulated one by onefor each item where necessary and converted in to percentageswhich enabled the researcher to describe in relation to the datacollected through observation. Moreover, the responses that arecollected by the open-ended questions collected from studentsspecially, Attitude test questions, were organized and analyzedin accordance with the responses gathered through close-endedquestions and classroom observations.
c. Implementation of the Action Plan
The researchers were implemented the proposed actionstrategies by interventions to improve student’s performancethrough designing student-centered approach that help easily toavoid errors in writing the basic elements of Chemistry Languagethereby developing confidence in how easily learn chemistryconcepts. The interventions were implemented after the pretestresult analysis including other data results. Generally, theinterventions implemented step-by-step are indicated in the tablebelow (Table 1).

Note: Methods implemented to practice writing chemical symbols and chemical formulae were in accordance with Jordaan (2000); Bradley and Steenberg (2006); Danieli and Reid (2004); Rollnick (2000); Clark (1982); Johnstone & Kellett (1980). * Methods implemented to practice writing chemical equations were additionally used as Beek and Louters (1991).
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Findings and Discussions
Attitude Test Results (Questionnaire)
Baseline data results and analysis
Students were made to tick against their degree of acceptanceagainst each item. After collecting the baseline data we countedthe total number of students in each item against [SD-stronglyagree (4), A-Agree (3), DA- Disagree (2) and SD-Strongly disagree(1)]. Then we have taken the percentage of each item and analyzedit (Table 2 & Figure 1). From both Table 2 and graph 1 above morethan 79.5 % of the respondents (on average) disagreed indicatinglack of knowledge and understanding of the basic elementsof chemistry languages i.e. chemical symbols, formulae andchemical equations except in item 5 (which is relatively odd whencompared to others, only 27.56 % of respondents showed theirdisagreement). The disagreement responses were figurativelyhigh (more than 79 %) out of 10 (1-11) items asking their levelof knowledge assessment, the highest response of disagreementwere observed in item 11 (91.84 %) and items 9, 7, 3 and 1 (83.67%, 82.65 %, 79.59 % and 79.59 % respectively) showing that thestudents were very poor background in having knowledge andskill in concepts of radicals, writing chemical formulae and aboutbalancing chemical equations which is the worst of all. The reasonthat about 70 students showed agreement in item 5 is that they seem to know and have better understanding of the symbols ofthe common most 20 elements of the periodic table. This is alsosupported by chemistry that of all the items forwarded to therespondents, item 5 seems the common and easiest one.
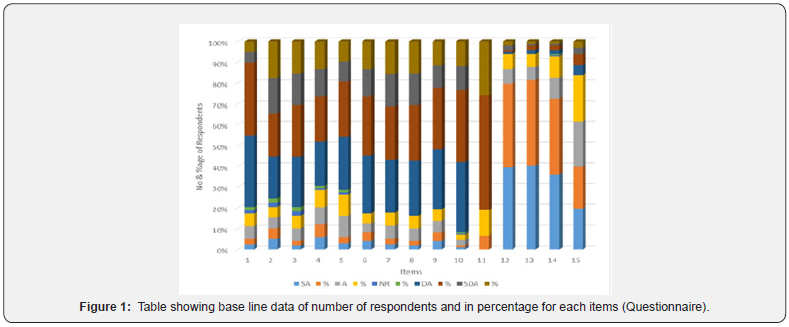
Key: - SA-Strongly Agree; A-Agree; NR-No Response; DA-Disagree; SDA-Strongly Disagree; TNR-Total Number of Respondents for eachitems.
When we come to their interest pre-test assessment to learnabout the basic elements of chemistry languages, responses wererecorded in items 12 and 13 (almost 93.88 % for both) withhighest agreement but least interest in item 15 (83.68 %) whencompared to the rest three items. This might indicate that in onecase the difficulty level of balancing chemical equations and on theother hand, it needs various methods and investment of time toget students’ understanding at all levels (Table 2) (Figure 1).
Post data results and analysis
After intervention the researchers started collecting postdata’s. We used the same tools to collect data for the Questionnaire.Different strategies were used considering the learning difficultiesof the students when implementing the interventions according tothe designed strategies of interventions. After our interventionswe analyzed the following data’s based on questionnaire. Theresponses made by the students in each items (questionnaire)were compared in (Table 3) (Figure 2). During pre-data collectiononly less respondents have accepted (agreed) that they know theconcepts stated in each the items 1 through 11 (except item 5)of the questionnaire. In post data collection almost the reverseresponses were recorded (in average 71.93 %). respondents foritems 1 through 7 have accepted that they can explain and applythe knowledge they acquired by interventions as stated in eachitems). Highest respondents in item 5 (85 respondents or 86.73%) and lowest respondents in item 9 (52 respondents or 53.06%) indicating that the difficulty level and investment time fordifferent level of students’ learning which is similarly interpreted for the low in number of respondents on agreement in items11 through 8 (57 and 59 respondents or 58.17 % and 60.21 %respectively). After implementation of intervention strategies, wecould see commendable improvement about understanding theconcept stated in each item of questionnaire which were clearlyindicated in (Figure 2).
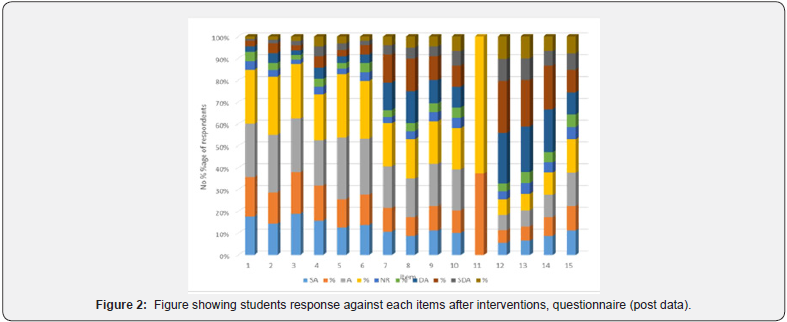
Student’s exposure to the language of chemistry wasmaximized through concept teaching. The chemical terms usedwere explained in detail by applying various methodologies.Differences between atoms, molecules and compound were madeclear. The only way student could learn Symbol and formula ofdifferent compound was through rote learning, memorization butbackground based concept analysis. Thus the above strategieshave given area to learn and improvement was seen at the sametime in our students after our interventions.
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
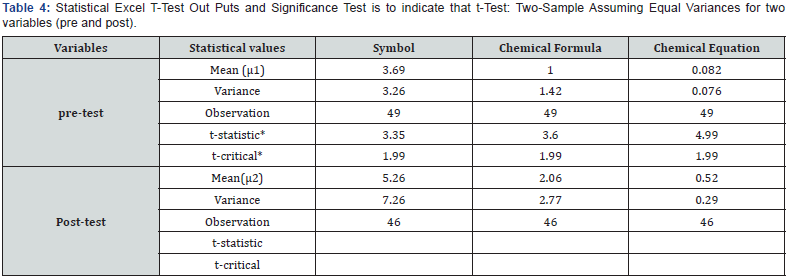
Results and Data Analysis on Pre- and Post-tests
Significance test of pre-test and post-test results
The researchers’ analysis is that the significance differencebetween the mean scores in case of chemical symbol, chemicalformulae and chemical equations based on our null hypothesis inall three cases is μ1= μ2 and the alternative hypothesis is μ1 < μ2.At p (0.05) the t-statistic (3.35) is greater than t-critical (1.99); asa result, the null hypothesis is rejected for the chemical symbolsi.e. there is significance difference between the post and pretestresult. Students who participated in the tutoring program,scored statistically significantly higher (p<0.05) on the post-testthan on the pre-test in chemical symbols questions. Similarly, atp (0.05) the t-statistic (3.6) is greater than t-critical (1.99); as aresult, the null hypothesis is rejected for the chemical formulaei.e. there is significance difference between the post and pre-testresult. Students who participated in the tutoring program, scoredstatistically significantly higher (p<0.05) on the post-test than onthe pre-test in chemical formulae questions (Table 4).
And also at p (0.05) the t-statistic (4.99) is greater than t-critical(1.99); as a result, the null hypothesis is rejected for the chemicalequation as well i.e. there is significance difference between thepost and pre-test result. Students who participated in the tutoringprogram, scored statistically significantly higher (p<0.05) on thepost-test than on the pre-test in chemical equations questions.
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Observation Assessment Discussions
Both the base data and pre-data (observation) was collectedonly after our interventions. First class work questions wereforwarded in the class during interventions and checking andcorrecting their errors were conducted. Most of the studentswere successful on matching the chemical symbols and theirword written on flip chart. However, there was still difficulties onbalancing chemical equations for most students. But convincingimprovements were observed through repeated practiceand correcting for few students. In case of writing chemicalformulae, average students shown interesting improvementsafter interventions specially, for monoatomic ions. Then throughanother intervention, the concepts were clear for most studentson how to write chemical formulae including for polyatomic ions.After our overall interventions and student’s continuous practices,there were admirable improvements in maintaining the retentionof the concepts of basic elements of chemistry languages. Theseimprovements were brought about by the researcher’s moststrategic interventions. When students do their work correctlyon time they were reinforced and motivated at the same time.Sirhan [24] in his article pronounce the key to successful learningof the students are through motivations. When reinforced andmotivated, students do their work on time and do it correctly [25-27].
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Conclusion
Assessment through all of our tools (questionnaire, testsand observation) indicated us that almost all of the students lackgood background on basic concepts of chemistry, especially, incase of our target (the basic elements of chemistry languages). Asevidences shown us, our action research was successful. Therewere improvements in terms of writing chemical symbols (highachievement), writing chemical formulae (more than average) andbalancing chemical equation (average to low). The success was dueto the quality of intervention strategies implemented that wereput forward by different authors. Following were the strategies;understanding the language of chemistry, motivation, steps anddifferent method in doing so Getting student exposed to languageof chemistry was the best way to enhance student understanding.When student were taught with different chemical languages, likedifferences between ions, atoms, molecules, symbol and formulaof the compound, student learn with ease. Motivation on otherhand brings students interest and participation into teaching andlearning process. Steps and methods helped student accuratelyto write chemical formulae and balance chemical equations.Additionally, action research helped us boom our professionalqualities. Moreover this gives us area to learn ourselves and bringforth best improvement in our teaching. Research participantsare benefitted as researchers put forward best learnt strategiesin process of answering research issues. Thus, action research isvery helpful especially to benefit oneself and community at large.
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
Recommendation
This study suggests that students should be made aware ofchemistry learning tips of the following nature [23]:
To the Teacher
a. Ensure that students’ exposure to chemical languageshould be maximized.
b. Teachers should not assume that students are familiarwith chemical terms and terms should be introduced carefully[9].
c. In the similar way of our case i.e. recommendationhas helped to the maximum and is necessary when teachingstudent the skill of balancing.
d. Moreover, when teaching, students must be providedwith a glossary of symbols of different elements and formulasof different compounds and make them to rote learn withnormal pacing to help students at all levels.
e. Teaching students clear rules and steps and makingthem follow strictly is another way to enhance learning.
f. Using video clips on how to write chemical formulae (therules) and balancing will also enhance student’s understanding aswe take them to different mode of teaching [28-30].
To the Students
a. Read the text before class
At least, the students should skim it. If they know what isgoing to be covered in class, they will be in a better position toidentify their troubles and ask questions that will help them tounderstand the material. It is possible to learn chemistry on theirown, but if they attempt this, they are going to need some sort ofwritten material as a reference.
b. Work Problems
Studying problems until you understand them is not thesame as being able to work them. If you can›t work problems, youdon›t understand chemistry. It›s that simple! Start with exampleproblems. When you think you understand an example, cover itup and work it on paper yourself. Once you have mastered theexamples, try other problems. This is potentially the hardest partof chemistry, because it requires time and effort. However, this isthe best way to truly learn chemistry.
c. Do Chemistry Daily
If you want to be good at something, you have to practice it.This is true of music, sports, video games, science, everything! Ifyou review chemistry every day and work problems every day,you›ll find a rhythm that will make it easier to retain the materialand learn new concepts. Don›t wait until the weekend to reviewchemistry or allow several days to pass between study sessions.Don›t assume class time is sufficient, because it isn›t. Make time topractice chemistry outside of class.
Finally, it is obvious that students are introduced to formalscience and science concepts after they joined Colleges. Hence, itwould be easy for them to learn things starting from what theyknow previously. This is preferred especially in the case wherenames of elements and compounds with vast properties arechunked to one or more symbols of letters which remain mysteryknowing properties underlying. In this case students associatingthe concepts to ideas in their own language could be helpful[31,32].
- Research Article
- Abstract
- Introduction
- Difficulties Faced in Writing Chemical Symbols andFormulae
- Difficulties Faced in Writing Chemical Equations
- Methods to Practice Writing Chemical Symbols and Formulae
- Methods to Practice Writing and Balancing ChemicalEquations Understanding the Language of Chemistry
- Motivation
- Methods
- Instruments for Gathering Data
- Results and Data Analysis on Pre- and Post-tests
- Observation Assessment Discussions
- Conclusion
- Recommendation
- References
References
- Modic AL (2011) Student Misconceptions: Identifying andreformulating what they bring to the Chemistry Table. Montana StateUniversity, USA.
- Baah R, Anthony-Krueger C (2012) An investigation into senior highschool student’s understanding and difficulties in writing chemicalformulae of inorganic compounds. International Journal of ResearchStudies in Educational Technology 1(2): 31-39.
- Maxwell TW (2003) Action Research for Bhutan Rabsel. The CERDEducational Journal 3:1-2.
- Rinchen (2009) Developing reflective thinking: Encouraging preserviceteachers to be responsible for their own learning. Paperpresented at Teacher education crossing borders: Cultures, contexts,communities and curriculum’ the annual conference of the AustralianTeacher Education Association (ATEA), Albury.
- Johnstone AH (2006) Chemical education research in Glasgow inperspective. Chem Educ Res Pract 7(2): 49-63.
- Chiu H (2005) A national survey of students’ conceptions in chemistryin Taiwan. Chem Educ Int 6: 1-8.
- Upahi JE, Olorundare AS (2012) Difficulties Faced by Nigerian SeniorSchool Chemistry Students in Solving Stoichiometric Problems. Journalof Education and Practice 3(12): 181-189.
- Marais Jordaan F (2000) Are we taking symbolic language for granted?Journal of Chemical Education 77(10): 1355- 1357.
- Steenberg (2006) Investigation of the development of bridgingstudents’ cognitive skills relevant to the use and understanding ofchemical formulae and equations. Unpublished MSc Thesis, UNISA,Pretoria, RSA.
- Danili E, Reid N (2004) Some strategies to improve performance inschool chemistry, based on two cognitive factors. Research in Scienceand Technology 22(2): 203-226.
- Savoy LG (1988) Balancing chemical equations. School Science Review69(249): 713-720.
- Gower M, Daniels DJ, Lioyd G (1977) Hierarchies among the conceptswhich underlie the mole. School Science Review 59 (201): 285-297.
- Suderji B (1983) Chemical equations conquered. School ScienceReview 65 (230): 101-102.
- Savoy LG, Hines C (1990) Student’s understanding of chemicalequations in secondary schools in Botswana. School Science Review72(285): 138-140.
- Johnstone H, Morrison TI, Sharp DWA (1976) Topic difficulties inchemistry. Education in Chemistry 20: 212-218.
- Anamuah Mensah, Apafo NT (1986) Students perceived difficulties withordinary level chemistry topics. Chemistry and Industry Proceedings,1(1): 38-39.
- Lazonby N, Morris JE, Waddington DJ (1982) The muddle some mole.Education in Chemistry 19: 109-111.
- Bello O (1988) An analysis of students’ error in stoichiometricproblems. Nigerian Education Forum 1(2): 181-186.
- Barke HD, Temechegn E (2001) “Structural Chemistry and SpatialAbility in Different Cultures.” Chemistry Education: Research andPractice in Europe 2(3): 227-239.
- Ababio OY (2004) New School Chemistry for SSS, Onitsha, Nigeria:African First Publishers Ltd, Africa.
- Baah R, Ampiah JG (2012) Senior high school students’ understandingand difficulties in writing chemical equations. International Journal ofScientific Research in Education 5(3): 162-170.
- Rollnick (2000) Current issues and perspectives on Second Languagelearning of science. Studies in Science Education, 35: 93-122.
- Ver Beek, Louters L (1991) Chemical language skills. Journal ofChemical Education 68(5): 389- 394.
- Sirhan G (2007) “Learning Difficulties in Chemistry. Journal of TurkishScience Education 4(2): 2-20.
- Bradley JD, Steenberg E (2010) Symbolic Language in Chemistry- ANew Look at an Old Problem.
- Katz DA (2002) Symbols and Formulae of chemical reaction, adoptedfor classroom teaching purpose. Royal University of Bhutan SamtseCollege of education, Bhutan.
- Temechegn E (2002) Students alternative Conceptions in Chemistry: Challenging the Challenge. Solutions, Chemical Society of Ethiopia10(1-2): 53-61.
- Onasanya FF, Orowale TO (2005) Chemistry for Senior SecondarySchools (Macmillan Mastering Series). Ibadan, Macmillan NigeriaPublishers Ltd, Nigeria.
- Taye H (2005) New Systematic Book Series of Chemistry, G7-8, (2ndedn). Addis Ababa, Ethiopia: Aster Nega PLC, pp. 5-6.
- IUPAC. Compendium of Chemical Terminology, (2nd edn).
- Crosland MP (1959) The use of diagrams as chemical ‘equations’ in thelectures of William Cullen and Joseph Black. Annals of Science 15(2):75-90.
- Bajah ST, Teibo BO, Onwu G, Obikwere A, Geleta T (2014) How Can IImprove N12 Students’ Ability to Write Simple Chemical Entities UsingChemical Symbols and Formulas on Introductory General ChemistryCourse-I (Chem. 101)? AJCE 4(1): 56-83.






























