Dabigatran Induced severe Esophagitis in a Patient with Budd–Chiari Syndrome: A Case Report
Pukar Thapa1* and Binod Karki2
1Norvic International Hospital, Nepal
2KIST Hospital, Nepal
Submission:January 23, 2026;Published:February 03, 2026
*Corresponding author:Pukar Thapa, Norvic International Hospital, Nepal
How to cite this article:Pukar T, Binod K. Dabigatran Induced severe Esophagitis in a Patient with Budd–Chiari Syndrome: A Case Report. Adv Res Gastroentero Hepatol, 2025; 22(2): 556083.DOI: 10.19080/ARGH.2026.22.556083.
Abstract
Dabigatran is a direct oral anticoagulant commonly prescribed for stroke prevention in patients with non-valvular atrial fibrillation. Although gastrointestinal adverse effects are frequently reported, dabigatran-induced esophagitis remains an under-recognized complication. The tartaric acid core of the dabigatran capsule may cause localized esophageal mucosal injury when prolonged contact with the esophageal lining occurs. We report a case of severe dabigatran-induced esophagitis in a patient with Budd–Chiari syndrome, confirmed by upper gastrointestinal endoscopy. This case highlights the clinical presentation, endoscopic findings, and pathophysiological mechanisms of dabigatran-related esophageal injury. Management strategies, including supportive therapy, and preventive measures such as appropriate pill-swallowing techniques, are discussed. Early recognition of this condition is essential to prevent complications, reduce patient morbidity, and avoid unnecessary diagnostic investigations.
Keywords:Dabigatran; Esophagitis; Pill-induced esophagitis; Direct oral anticoagulants; Dysphagia
Abbreviations:DIE: Dabigatran-Induced Esophagitis; HVOTO: Hepatic Venous Outflow Tract Obstruction
Introduction
Dabigatran etexilate is a direct thrombin inhibitor widely used for the prevention of thromboembolic events in patients with nonvalvular atrial fibrillation and venous thromboembolism. While dyspepsia and gastrointestinal discomfort are common adverse effects, dabigatran-induced esophagitis (DIE) has only recently been described in the literature [1]. The capsule contains tartaric acid to enhance absorption, which can cause chemical mucosal injury if the capsule lodges or dissolves in the esophagus [2]. We report a case of severe DIE and review the relevant literature to increase awareness among clinicians.
Case Presentation
A 37-year-old woman with a history of Hepatic Venous outflow tract obstruction (HVOTO) presented with progressive retrosternal pain, dysphagia, and odynophagia for 2 weeks. She was diagnosed with Budd Chiari Syndrome 3 months back and had been receiving dabigatran 110 mg twice daily since then. There was no history of gastroesophageal reflux disease, alcohol intake, smoking, caustic ingestion, or immunosuppression. She provided a history of having swallowed a dabigatran tablet hurriedly without water multiple times. Physical examination was unremarkable. Laboratory investigations, including Complete Blood Count and Inflammatory markers which were within normal limits. Esophagogastroduodenoscopy revealed extensive longitudinal sloughing of the esophageal mucosa with erosions in the distal esophagus, consistent with severe chemical esophagitis (Figure 1). The stomach and duodenum appeared normal. Histopathological examination demonstrated epithelial degeneration with inflammatory cell infiltration, without evidence of malignancy or infection. Based on the temporal association with dabigatran use and characteristic endoscopic findings, a diagnosis of dabigatraninduced esophagitis was established [1,2]. Proton pump inhibitor therapy was initiated, and the patient was counseled to ingest dabigatran with sufficient water and remain upright for at least 30 minutes after administration [3]. Her symptoms improved significantly within two weeks. Follow-up endoscopy showed marked mucosal healing (Figure 2).
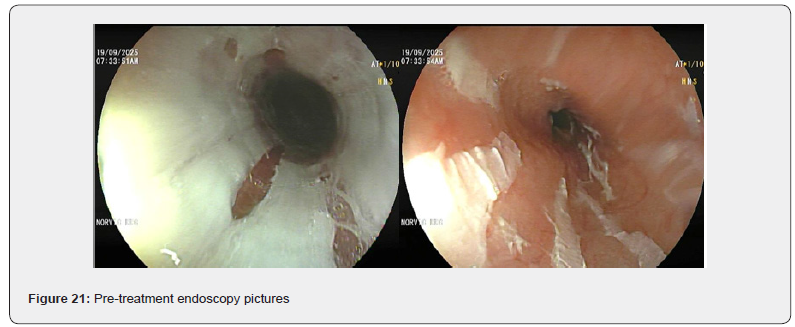
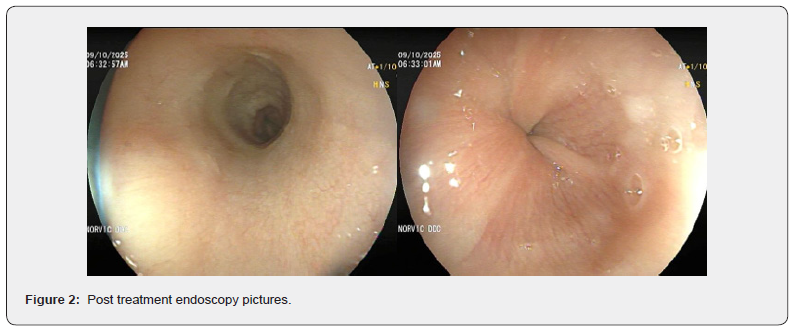
Discussion
Dabigatran-induced esophagitis is believed to result from prolonged contact between the esophageal mucosa and the tartaric acid core of the capsule [1,4]. Characteristic endoscopic findings include longitudinal sloughing epithelial casts, erosions, and ulcerations, most commonly in the mid to distal esophagus. Reported risk factors include advanced age, inadequate water intake, recumbency after pill ingestion, esophageal dysmotility, and anatomical compression of the esophagus [1,3]. Management primarily involves acid suppression therapy, patient education on proper medication ingestion, and consideration of switching to an alternative direct oral anticoagulant in recurrent or severe cases [5]. Recognition of DIE is crucial, as symptoms may mimic cardiac or malignant conditions, leading to unnecessary investigations. Prompt diagnosis and intervention usually result in rapid symptom resolution.
Conclusion
Dabigatran-induced severe esophagitis is an important and preventable adverse drug reaction. Clinicians should maintain a high index of suspicion in patients presenting with dysphagia or chest pain while receiving dabigatran therapy. Early diagnosis, patient education, and appropriate management can lead to rapid symptom resolution and prevent complications.
References
- Toya Y, Nakamura S, Tomita K (2016) Dabigatran-induced esophagitis: the prevalence and endoscopic characteristics. J Gastroenterol Hepatol 31: 610-614.
- Zhou Y, Dai Y, Lu L, Fu Z (2020) Dabigatran-induced esophagitis: a case report. Medicine (Baltimore) 99: e19890.
- Kajihara Y (2019) Dabigatran-induced esophagitis. Cleve Clin J Med 86: 303-304.
- Kubo K, Ishida H, Matsumoto T (2015) Pill-induced esophagitis related to dabigatran. World J Gastroenterol 21: 7682-7685.
- Kosuke I, Daiki Y, Takahiro M, Masatomi I (2022) Dabigatran-induced esophagitis improved by switching to apixaban. BMJ Case Rep 14(8): e245443.






























