Three Pharmacological Models: Inducing Inflammatory Bowel Disease and Colitis-Associated Cancer
Sha Zeng A*, Ping Li and Juan Wang A
Department of TCM Pharmacy, Chengdu Integrated TCM and Western Medicine Hospital, China
Submission:July 26, 2023; Published:July 31, 2023
*Corresponding author: Sha Zeng, Department of TCM Pharmacy, Chengdu Integrated TCM and Western Medicine Hospital, Gaoxin, Chengdu 61000, China, Email: 1562970575@qq.com
How to cite this article: Sha Zeng A, Ping Li, Juan Wang A. Three Pharmacological Models: Inducing Inflammatory Bowel Disease and Colitis-Associated Cancer. Adv Res Gastroentero Hepatol, 2023; 19(5): 556025. DOI: 10.19080/ARGH.2023.19.556025.
Abstract
Colorectal cancer (CRC) is a kind of incurable cancer with high morbidity and mortality. In recent years, mounting evidence has revealed that inflammatory bowel disease (IBD) was correlated with colitis-associated cancer (CAC). Signal transduction is an effective way for cells to respond to external stimuli and eventually trigger specific biological effects. The occurrence, growth and metastasis of cancer are usually associated with disorders of signaling pathways. Although modern research can elucidate the pathogenesis of CAC and provide enhanced screening strategies, the prevalence of CAC is still rising. Studies have shown that several cells signaling pathways in CAC are dysfunctional, leading to the occurrence of malignant phenotypes. Therefore, analyzing the signaling pathways involved in CAC metastasis is necessary to elucidate the potential mechanisms of CAC progression and drug treatment. Our understanding of CAC-related signaling pathways is largely due to a mouse model that faithfully reproduces human CAC. In particular, chemical models can quickly and effectively analyze the molecular mechanism of CAC without increasing time-intensive genetic hybridization. Therefore, this article summarizes the following three chemical-induced CAC animal models, azoxymethane/dextran sodium sulfate (AOM/DSS), 1, 2-dimethylhydrazine (DMH)/DSS and 2, 4, 6-trinitrobenzene sulfonic acid (TNBS)/AOM. Among them, DSS and TNBS were used as inflammatory agents to simulate the pathogenesis of IBD inflammatory models, while AOM and DMH were used to producing carcinogenic methylated DNA, combined with inflammatory agents, could better induce IBD to CAC in animals.
Keywords: Colitis-associated cancer; Inflammatory bowel disease; AOM/DSS; DMH/DSS; TNBS/AOM
Abbreviations: CRC: Colorectal Cancer; IBD: Inflammatory Bowel Disease; CAC: Colitis-Associated Cancer; AOM: Azoxymethane; DSS: Dextran Sodium Sulfate; DMH: 1, 2-Dimethylhydrazine; TNBS: 2, 4, 6-Trinitrobenzene Sulfonic Acid; ACF: Abnormal Crypt Foci; UC: Ulcerative Enteritis; CD: Crohn’s Disease; MAM: Methylazomethanol; ROS: Reactive Oxygen Species; MDSC: Myeloid-Derived Suppressor Cells; DC: Dendritic Cells; Th: T Lymphocytes; MyD88: Myeloid Differentiation Factor 88; CPB: CREB Binding Protein; GR: Glutathione Reductase; HO-1: Heme Oxygenase-1; TR: Thioredoxin Reductase; PEITC: Phenethyl Isothiocyanate; GSK3β: Glycogen Synthase Kinase 3β; CK1α: Casein Kinase 1α; DVL: Disheveled; TCF: T Cell-Specific Factor; LEF: Lymphoid Enhancer Binding Factor; MDF: Mucin-Depleted Foci; JAK2: Janus Kinase 2; STAT3: Signal Transducer and Activator of Transcription 3
Introduction
Colorectal cancer (CRC), as one of the common malignancies with high morbidity and mortality [1,2], usually occurs at the junction of the rectum and sigmoid colon. It is the result of healthy colonic epithelial cells transforming into cancer [3]. This process is called ‘adenoma-carcinoma sequence’, which is developed through a series of orderly events. The initial step is the transformation of normal colonic epithelium to abnormal crypt foci (ACF). ACF progresses to CRC within 10-15 years [4,5]. Accumulating evidence has shown that its occurrence and development were related to many factors, such as environment, eating habits, congenital heredity, personal physique and so on [6]. Over the past few decades, CRC, including colitis-associated cancer (CAC) has posed a serious threat to people’s health and life, which develops from long-term colitis in inflammatory bowel disease (IBD) patients [7] .
IBD, one of the key influential factors resulting in CAC, is a chronic and recurrent gastrointestinal inflammatory disease that can affect the body’s immune mechanism [8]. Generally speaking, the occurrence of IBD in humans may be related to environmental factors [9]. but in the current research hotspots, diet is also considered to be the cause of IBD to a certain extent [10]. especially the intake of allergenic substances in food [11]. Clinically, IBD is usually divided into two kinds of disease, ulcerative enteritis (UC) and Crohn’s disease (CD) [12,13]. Studies have found that long-term IBD may not only lead to the occurrence and development of CRC, but also increase the risk of prostate cancer [14]. gastric cancer and other cancers [15]. Morever, the risk of colorectal cancer is increased twofold in CD and in UC [16] while about 8% of UC patients develop CAC within 20 years and 18% within 30 years [17].
The rodent carcinogen-induced model recalls a long tradition but still retains its usefulness for some applications. They provide a platform for dietary research and insight into the pathways of food-borne carcinogens and colitis-related carcinogenic effects. Since the development of carcinogen-induced rodent models for CAC more than 80 years ago, these animal models have played a huge role in chemoprevention research and assessment of colitisrelated carcinogenic mechanisms [18]. In 1915, scientists proved the carcinogenic properties of coal tar by repeatedly applying coal tar to rabbit ears [19].
At about the same time, the first researchers studied the occurrence of colon cancer by applying chemical or radioactive substances [20]. In the 1960s, heme and its metabolite methylazooxymethanol have been shown to induce rodent cancer [21,22]. In the following years, the chemically more stable substance AOM and its precursor DMH were widely used to induce colon cancer in mice and rats. Both of the two compounds are metabolized to methylazoformaldehyde, which can alkylate DNA bases guanine and thymine [23]. After two-phase reaction treatment, it is secreted into the bile, and its carcinogenic effect on intestinal mucosa exceeds its carcinogenic effect [24].
The most typical AOM/DSS model needs to be combined with a single injection of AOM, a carcinogen that is metabolized in the liver to the active agent methylazomethanol (MAM), and then use inflammatory damage agent DSS [25]. The pathological manifestations of the AOM/DSS model were severe colitis, accompanied by weight loss and bloody diarrhea, and followed by multiple colon tumors. Another carcinogen DMH undergoes a series of metabolic reactions and eventually reaches the colon, producing the ultimate carcinogen and reactive oxygen species (ROS), which further alkylate DNA and cause colon cancer [26]. TNBS, a classical skin contact ant serves as a happen, which could induce a specifically inflammation model in the gastro-intestinal tract [27].
It was reported that the exact location of tumor in colon length depends on the mouse strain and background [28]. The carcinogenic process of the system has a pathological progression from normal intestinal crypts to crypt fission to lesions with abnormal crypts and eventually to microadenomas [29,30]. These steps outline the sequence of CAC formation in humans from inflammation to developmental abnormalities and to cancer.
In recent years, AOM/DSS [31-33] DMH/DSS [34,35] and TNBS/AOM [36,37] are three main methods to establish CAC animal models, as these three chemical pathways could better simulate the process of IBD to CAC in vivo. Therefore, this paper summarized the mechanism of these three modeling methods which can reflect IBD and CAC as follows.
AOM/DSS
Chemical properties
AOM is a class of chemical compound with the molecular formula of C2H6N2O (molecular weight: 74.08g/mol) and strong oxidative activity. DSS is a kind of polyanion derivative of dextran with the chemical formula of (C6H7Na3O14S3) n), which could be used for modeling CAC, separating lipoproteins and improving nucleic acid hybridization rate.
Modeling method
AOM/DSS model is the most widely used and reproducible method to induce CAC. Mice were intraperitoneally injected with AOM (7mg/kg or 12.5mg/kg) [38]. After 5 days, mice received 3 cycles of drinking 1%-3% DSS solution for 5 days and water for 14 days [39]. At the end of the experiment, the mice showed obvious intestinal polyps with intestinal mucosal injury.
Mechanism
As shown in figure 1, AOM reaches the liver through blood circulation. Under the action of cytochrome P4502E1 [40,41]. it is hydroxylated to form MAM [42]. which can spontaneously decompose into formaldehyde and highly active alkylate after entering the intestine [43]. In addition, some scholars have proposed that MAM may be metabolized through another pathway, that is, the metabolic reaction involving tissue-specific cytoplasmic enzyme ADH in the intestine.
ADH generates reactive methylases, alkylases, or such reactive methylases, which can promote DNA alkylation [44,45], and subsequently induce DNA to produce O6-methylguanine adduct [46], resulting in the conversion of G to A in the DNA base sequence. MAM through the liver, and the product ADH produced by intestinal metabolism of MAM leads to changes in DNA base sequence. And after free drinking of DSS, the balance of T lymphocytes in animals is destroyed, promoting the inflammatory response. Combined use of inflammatory and carcinogenic agents alters the genome sequence of cancer, eventually leading to tumorigenesis. AOM/ DSS is one of the most commonly used chemical reagents to induce CAC in rodents [47] which can better simulate the pathological process of the body from normal intestinal epithelial mucosa to abnormal crypt lesions [48] adenoma and colorectal cancer [49].
Among them, DSS reagent has the characteristics of convenient use and high efficiency [50], as a chemical reagent for inflammatory stimulation, it continuously stimulates the mucosa of intestinal epithelial cells during free drinking, causing repeated inflammation in model animals. In the DSS model, sulfated polysaccharides do not directly induce intestinal inflammation, but act as a direct chemical toxin of colonic epithelium, leading to epithelial cell damage. The mechanism is that oral administration of DSS leads to the destruction of the intestinal epithelial monolayer lining, the entry of intraluminal bacteria and associated antigens into the mucosa, and the transmission of pro-inflammatory intestinal contents to the lower tissues [51-53].
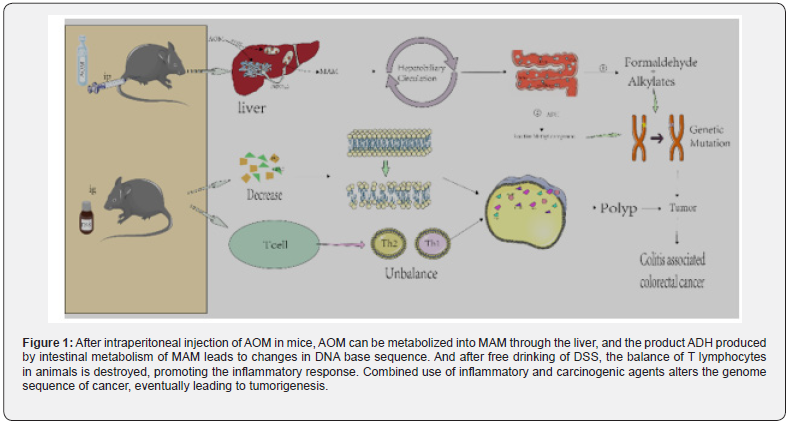
It is generally believed that in chronic inflammation, the immune system is characterized by intensive infiltration of innate immune cells (such as macrophages, neutrophils, myeloidderived suppressor cells (MDSC), dendritic cells (DC) and NK cells and adaptive immune cells (such as T and B lymphocytes), further suggesting that the changes in the intestinal inflammatory microenvironment promote tumor formation [29] In the internal environment of the body, helper T lymphocytes (Th) play an important role in the immune response. The dynamic balance of Th1 and Th2 is one of the necessary conditions to maintain the normal physiological function of the body [54]. Therefore, the increase or decrease of Th1 or Th2 could cause immune response [55] eventually resulting in the occurrence of inflammation [56,57].
After oral administration of DSS in mice, the dynamic balance between Th1 and Th2 was broken, which promoted the expression of inflammatory factors such as TNF-α, IL-1β, IL-10 and IFN-γ, and so on [58,59]. In AOM/DSS model animals, the continuous production of pro-inflammatory cytokines leads to mutations in proto-oncogenes and tumor suppressor genes APC, K-RAS and p53, as well as genomic sequence variations, generating tumorigenesis, progression and metastasis [60]. Myeloid differentiation factor 88 (MyD88), a molecule essential for TLR intracellular signaling, appears to have a protective effect against AOM/DSS-induced inflammation-associated CRC in mice [61]. Yassin M proposed that AOM/DSS-induced inflammation led to an increase in the total number of CD3+, TCRγδ and TCRαβ cells, including a significant increase in CD4+TCRαβ cells, but only moderate changes in CD8αβ TCRαβ and CD8αβ TCRαβ/TCRγδ cells. This suggests that CD4+T cell recruitment plays a key role in regulating various parts of the mucosal immune response during acute and chronic DSS-induced colitis [62].
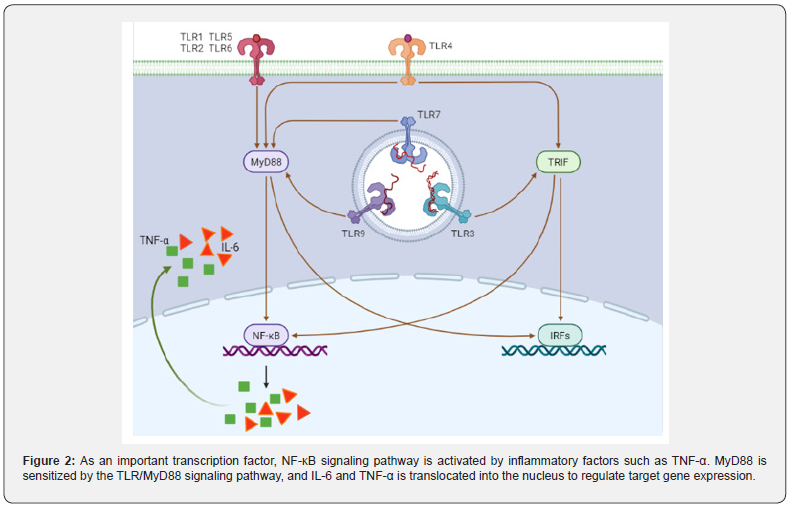
The NF-Κb signaling pathway, a central mediator between inflammation and cancer, promoting CAC development: It is well known that TNF-α can directly activate NF-κB signaling pathway and induce inflammatory response. Furthermore, MyD88 signaling molecules can also activate NF-κB-induced kinase and indirectly activate NF-κB signaling pathway through TLR/MyD88 signaling pathway [63,64] (Figure 2). Xie et al. [65] synthesized a MyD88 inhibitor TJ-M2010-5, which can bind to the TLR domain of MyD88, inhibit the production of its homodimer, and ultimately inhibit the TLR/MyD88 signaling pathway. TJM2010- 5 can significantly reduce the incidence of AOM/DSSinduced colitis, shorten colon length, reduce tumorigenesis, and inhibit the production of inflammatory factors. Moreover, when the TLR4-deficient mice and the wild-type mice were treated with AOM/DSS at the same time, it was observed that the TLR4- deficient mice had a lighter degree of colon cancer than the wild type mice [66]. After the MyD88-/- mice were injected with AOM, colonoscopy showed no tumor formation in the intestine, and the mRNA expression of pro-inflammatory cytokines TNF-α and IL12P40 was lower than that of the model group [67]. On the one hand, the production of TNF-α can activate the NF-κB signaling pathway, on the other hand, when the NF-κB signaling pathway is activated, it can also promote the transcription and translation of TNF-α. El-Daly et al. [68]. showed that in AOM/DSS-treated mice, the positive staining of p65 in the nucleus of the vagus nerve crypt of colorectal epithelial cells gradually increased with the increase of DSS administration cycle. In addition, AOM/DSS also induced the high expression of TLR4 and p65 proteins upstream of NF-κB signaling pathway. Furthermore, Song et al. [69]. found that the expression level of pro-inflammatory enzyme COX-2, which is mainly regulated by NF-κB signaling pathway, was significantly higher than that of the control, and the expression levels of the other two target genes TNF-α and IL-6 also tended to increase [70].
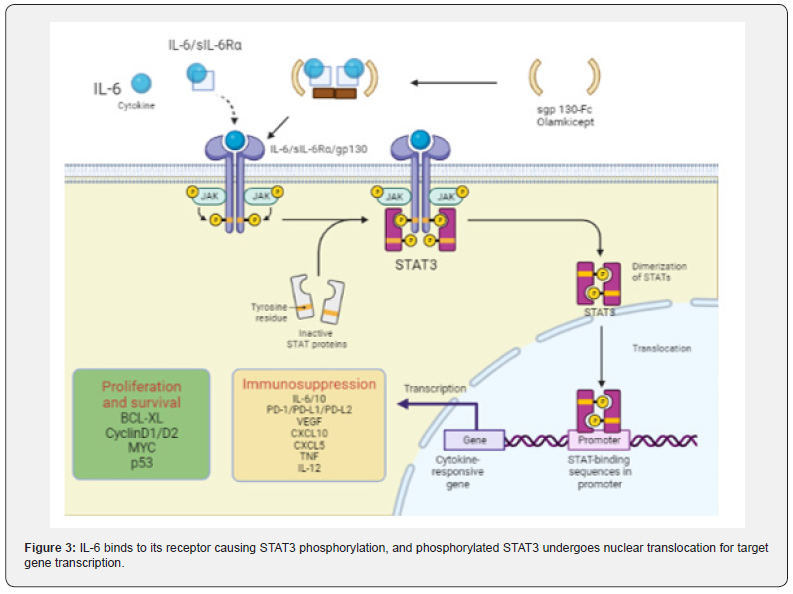
The IL-6/STAT3 signaling pathway is enhanced and accelerates carcinogenesis in CAC: As an inflammatory cytokine, IL-6 binds to soluble IL-6 receptors to form an IL-6/ sIL-6R complex [71,72]. which activates gp130 expression on the cell membrane, leading to signal transducer and activator of transcription 3 (STAT3) phosphorylation. STAT3 enters the nucleus in the form of dimers and activates the STAT3 signaling pathway (Figure 3) [73]. Studies have shown that p-STAT3 staining in the colorectal region of AOM/DSS-treated mice was deepened, and the deposition of brown granules in cytoplasm and nucleus was enhanced with the increase of the administration cycle [74]. MiR-18a is a transcription product in the STAT3 signaling pathway, in AOM/DSS-treated mice, it was found that the expression of MiR- 18a increased significantly with the extension of DSS treatment cycle [75]. but the expression of PIAS3 (an inhibitor of STAT family activation) decreased. In vitro studies have shown that the expression level of MiR-18a is negatively correlated with PIAS3. MiR-18a can negatively regulate PIAS3 [76]. while overexpression of PIAS3 leads to a decrease in IL6-activated STAT3 activity. Ma et al. reported that the proliferation and survival of precancerous intestinal epithelial cells (IECs) were regulated by IL-6/STAT3 signaling pathway, and CAC may be related to this signaling pathway [77].
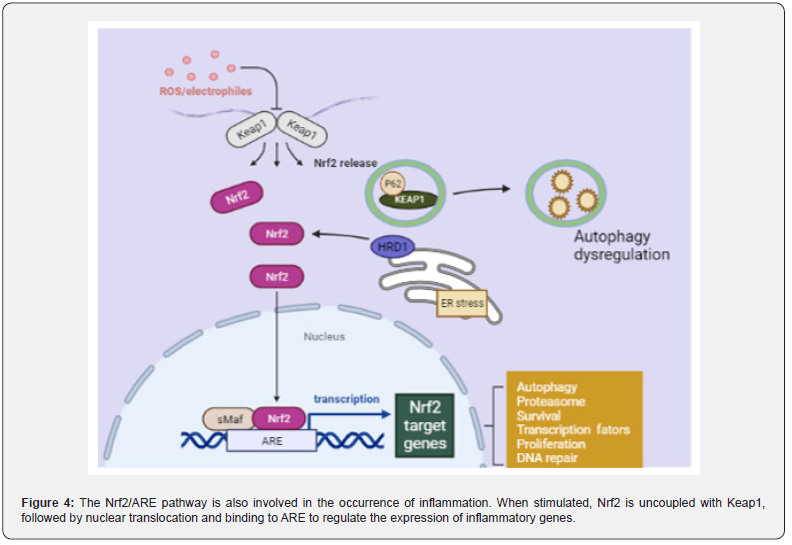
Nrf2/ARE signaling pathway plays an inflammatory role in CAC: In addition, the occurrence of inflammation is also related to the activation of the antioxidant Nrf2/ARE signaling pathway. When the Nrf2/ARE signaling pathway is closed, Keap1 in the cytoplasm specifically binds to the N-terminus of Nrf2, thereby maintaining the normal physiological function of the body. However, when cells receive external stimuli, the Nrf2/ARE signaling pathway is immediately activated, Nrf2 is uncoupled with Keap1, and then enters to the nucleus and binds to the small may protein to form a heterodimer. This process requires the participation of transcription cofactor CREB binding protein (CPB).
Heterodimers specifically recognize ARE and initiate transcription and translation of target genes, resulting in inhibition of the Nrf2/ARE signaling pathway (Figure 4). When the antioxidant process in the body is initiated, the expression of antioxidant proteins is significantly increased to protect cells from damage [78,79]. Wang et al. [80]. found that the positive expression of Nrf2 and the expression of glutathione reductase (GR), heme oxygenase-1 (HO-1) and thioredoxin reductase (TR) were significantly degraded in AOM/DSS treated mice. The researchers [81]. fed mice with 0.05% phenethyl isothiocyanate (PEITC) before and after AOM/DSS administration, found that the incidence of intestinal adenomas in mice decreased by 37.5% and 40%, the number of polyps reduced to 0.375 and 0.6, and the average size decreased to 1.5 and 2.6 mm, respectively. It can be inferred that PEITC can improve the development of intestinal tumors, as an activator of Nrf2, PEITC can activate Nrf2/ARE signaling pathway. In addition, some scholars performed Nrf2 gene knockout in wild AOM/DSS model mice, early model mice and 9-week model mice [82]. It was proved that Nrf2-/- mice showed the same phenotype as wild-type mice, but the incidence of tumors and the proportion of adenocarcinoma were higher.
DMH/DSS
Chemical properties
DMH is a class of compound whose molecular formula is C5H8N2O2 (molecular weight: 128.13g/mol) and provides raw materials for the synthesis of proteins in vivo.
Modeling method
Scientists [83] injected DMH (40mg/kg) into FMH rats three times in the first week, and then gave 1% DSS solution to drink freely for one week, repeating three cycles. DMH is a precursor of the carcinogen AOM, which is not carcinogenic in itself, but can be carcinogenic by oxidative dealkylation to form AOM [84]. The study confirmed that DMH-treated rats had traditional tubular or villous adenomas and serrated adenomas [85] in their colorectal regions, and it is generally believed that most colorectal cancers progress from adenomas.
Mechanism
As a proto-oncogene, DMH can be decomposed into azomethane and methylazomethanol under the catalysis of liver enzyme system. The latter substance flows to the colon tissue through the hepatobiliary circulation and is easily decomposed into a carcinogen methyl diazo ion [86] in the circulation. Methyl diazo ions can methylate DNA, RNA and proteins in normal intestinal epithelial cells. When they reach the colorectal site, they can produce activated carbon ions, leading to oxidative stress, over-expression of inducible nitric oxide synthase (i-NOS) and COX-2 [87], which in turn causes colorectal cancer.
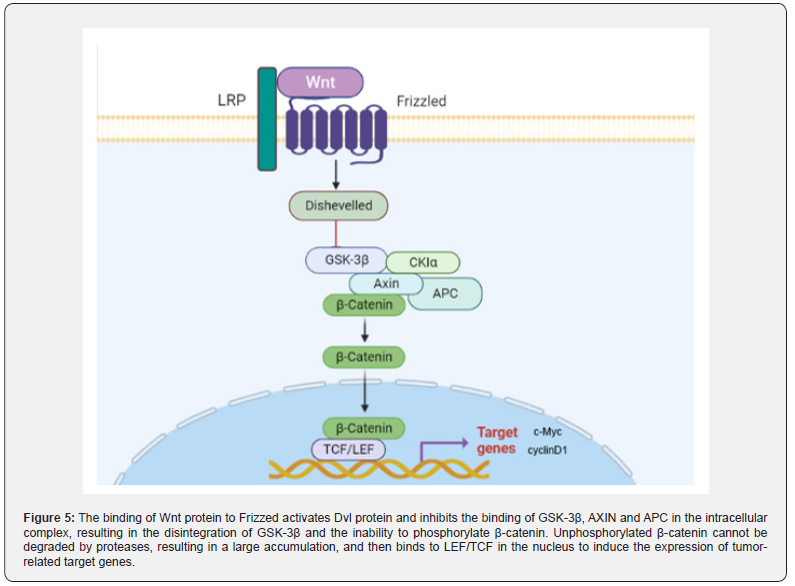
Up-regulated Wnt/ β -catenin signaling pathway promotes CAC progression: Wnt/β-catenin signaling pathway is a conserved signaling axis involved in a variety of physiological processes, such as proliferation, differentiation, apoptosis, migration, invasion and tissue homeostasis [88,89]. In the Wnt/β-catenin pathway, the abnormal regulation of the transcription factor β-catenin can lead to the occurrence of early cancer events [90,91]. In the degradation complex of this pathway, glycogen synthase kinase 3β (GSK3β) and casein kinase 1α (CK1α) mediate phosphorylation of β-catenin, promoting its ubiquitination and subsequent proteasome degradation [92]. The β-catenin dependent signaling pathway is triggered by the binding of secreted cysteine-rich glycoprotein ligands Wnts to LRP-5/6 receptors and FZD receptors. In the presence of Wnt ligands, the binding of Wnt ligands on the cell surface to the receptor induces disheveled (DVL), leading to the aggregation of complexes (AXIN, GSK3β, CK1, APC) to the receptor [93]. Subsequently, phosphorylation and inhibition of GSK3β ensured an increase in cytosolic β-catenin concentration. Unphosphorylated β-catenin in the cytosol migrates to the nucleus and accumulates, interacting with T cell-specific factor (TCF)/lymphoid enhancer binding factor (LEF) to trigger Wnt target genes such as c-Myc [94]. and cyclin D1 (95), resulting in up-regulation of TCF/LEF target genes [95].
In the analysis of 11 DMH/DSS-induced adenocarcinoma mice, Tanaka observed markedly enhanced expression of the β-catenin gene in 10 of 11 mice [96]. Furthermore, mutations of β-catenin, Apc and K-ras genes and cytoplasmic β-catenin expression were found in DMH-induced mucin-depleted foci (MDF) [97,98]. MDF is a microscopic dysplasia induced by specific colon carcinogens in the colon of rodents. Most MDF show Wnt pathway activation, while only one subset shows Ctnnb1 gene mutation, encoding β-catenin [99]. It is well known that β-catenin is a key upstream regulator of the Wnt signaling pathway. The activation of Wnt signaling pathway can lead to the increase of β-catenin into the nucleus (Figure 5), which leads to the increase of transcription of tumor-related factors and induces the occurrence of tumors in vivo.
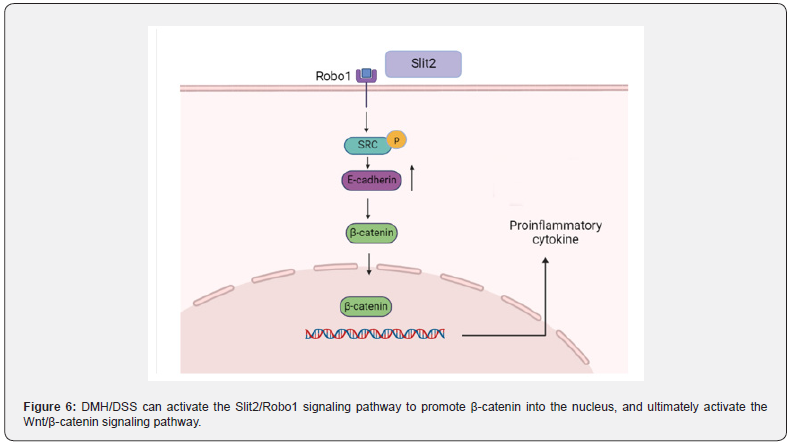
The Slit2/Robo1 signaling pathway induces the Wnt/β -catenin signaling pathway dysregulation to promote CAC progression: Slit/Robo signaling pathway includes Slit secreted protein family (Slit1, Slit2, Slit3) and specific receptors (Robo1, Robo2, Robo3, Robo4). The Slit2/Robo1 signaling pathway was initially found to be involved in the development of the central nervous system. In recent years, more and more studies have reported that it can induce tumorigenesis and participate in the proliferation and migration of tumor cells [100]. The activation of Slit2/Robo1 signaling pathway can be caused by ectopic expression of Slit2 protein (Figure 6). The Slit2 and Robo1 genes on chromosomes are located respectively at 4P15.2 and 3P12.3, Robo1 is a specific receptor of Slit2 [101]. The extracellular IgG region of Robo1 is an important position for binding to Slit2, and the intracellular region is a site for interaction with related signal molecules, which is involved in activating the downstream of Slit2/Robo1 signaling pathway and promoting the occurrence and development of tumors [102].
DMH/DSS can not only directly activate the Wnt signaling pathway, but also indirectly activate the Wnt signaling pathway by activating the Slit2/Robo1 signaling pathway Qian et al. [103]. found that the expression of Slit2 and Robo1 in DMH/DSS mice gradually increased with time. Morever, seven pairs of CRC tissues and non-cancerous colon tissues were randomly selected from patients with N0 CRC (no metastasis). The expression profiles of Slit2 and Robo1 were evaluated by immunohistochemical staining. It can be seen that the expression of Slit2 and Robo1 in CRC tissues is significantly higher than that in non-cancerous colon tissues. As long as the Slit2/Robo1 signaling pathway is activated, it can promote the activation of Src phosphorylation, resulting in increased expression of E-cadherin, thereby activating the Wnt/β-catenin signaling pathway, promoting β-catenin into the nucleus, leading to an increased expression of tumor-associated proteins.
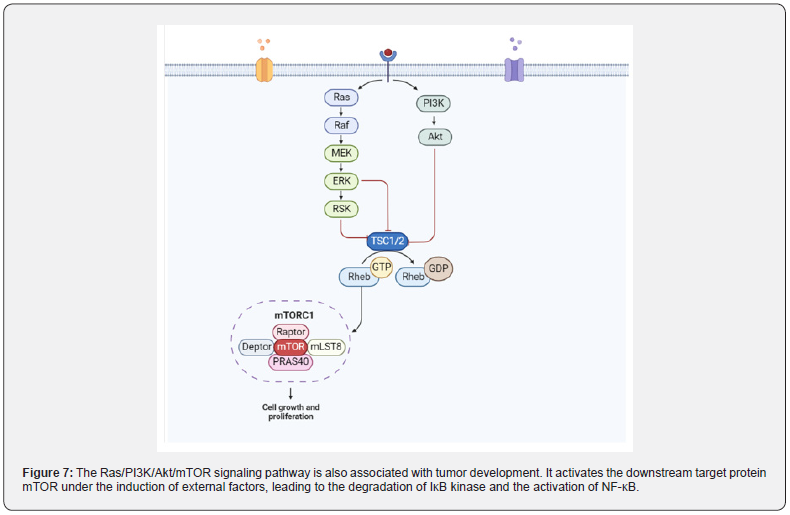
The Ras/PI3K/Akt/M TOR signaling pathway promotes the development of CAC by activating NF-κB protein: The carcinogenic activation of Ras protein caused by missense mutations is a well-known marker of tumor cell proliferation. Balb/c mice treated with DMH/DSS resulted in a significant increase in Ras protein levels. Activated Ras protein targets MEK to trigger ERK phosphorylation and nuclear translocation to express genes involved in cell proliferation. The levels of activated MEK1/2 and phosphorylated-ERK1/2 was significantly increased in DMH/ DSS-treated animals. In addition, Ras oncoprotein also interacts with PI3K and triggers Akt phosphorylation and subsequent activation. DMH/DSS-treated animals showed significantly increased levels of PI3K and phosphorylated Akt. The expression of NF-κB, a downstream effector of Akt, was also significantly increased. The authors believe that the increased expression of NF-κB can be attributed to chronic inflammation followed by circulatory treatment with the inflammatory agent DSS [104]. The mutation of RAS protein is induced by some external factors [105], and mutant RAS protein interacts with PI3K and causes autophosphorylation of PI3K. Type I PI3K is a heterodimer, the corresponding domain of the P85 subunit on the dimer can interact with the linker protein to recruit the corresponding activation receptor for PI3K, thereby converting PI3K into PIP2 and then into PIP3. PIP3 binds to Akt and PDK1 containing the PH domain, leading to the activation of Akt, which in turn regulates the expression of its downstream target protein mTOR. On the other hand, this binding can also activate IK-B, thereby relieving the inhibition of NF-κB, transferring NF-κB from the cytoplasm to the nucleus, and finally regulating the expression of inflammatory factors in the epidermis (Figure 7).
TNBS/AOM
Chemical properties
TNBS is a kind of dangerous chemical compound with a molecular formula of C6H3N3O9S (molecular weight: 293.17). In bioengineering, TNBS can be carried out determining aminoterminal groups, hydrophilic modification reagents, etc.
Modeling method
Xiao et al. [37] injected AOM (10mg/kg) intraperitoneally into C57BL/6 mice, and then 2.5mg TNBS was dissolved in 150μl 50% ethanol and injected into the mice through the rectum. The changes of intestinal tumors in mice were observed under rectal endoscopy. TNBS-induced colitis is very similar to CD, which can induce transmural colitis (Th1-mediated immune response) with severe diarrhea, weight loss, rectal prolapse [106,107], tissue destructive and recurrent [108].
Mechanism
Normally, TNBS was used in combination with anhydrous ethanol. By enema administration, the intestinal mucosa of the model animals was damaged, so that TNBS penetrated the intestinal wall, combined with the related proteins in the intestine, and caused the intestinal epithelial mucosaltenization. Therefore, intestinal cells are more likely to expose target proteins, stimulate immune cells above the intestinal lamina propria [109], and make target proteins more easily recognized by immune cells [110]. resulting in acute necrosis.
TNBS promotes the expression of the same T cytokines as CD [111]. which is an immune inflammatory disease mediated by Th1 [112]. However, the Th1-mediated cycle is short and mainly mediated by Th17, which is characterized by infiltration of CD4+T cells in the intestine [113]. and severe fibrosis of intestinal wall cells in advanced patients [114]. Alrafas Haider [115] treated TNBS-induced colitis with resveratrol and found that the induced inflammatory T cells (Th17 and Th1), cytokines and transcription factors (Foxp3 and TGF-β) were significantly decreased, and the severity of colitis in mice was significantly increased. In general, TNBS-induced colitis also involves Th17-related cytokines [116].
More and more evidence suggests that the growth and proliferation of Th17 cells depend on IL-23. Therefore, some scholars have proposed that the IL-17/IL-23 axis may also be one of the causes of IBD [117,118]. Cubes [119]. found that I-NOS is associated with TNBS-induced colitis and can cause intestinal mucosal damage in model animals, which is similar to some features of DSS-induced colitis. The pathogenesis of CD induced by TNBS is not clear, but the mutation of NOD2 gene was found in C57 mice treated with TNBS, and the mutation of NOD2 gene is related to the pathogenesis of CD. Hence, it is concluded that TNBS can induce CD-related enteritis in model animals [120].
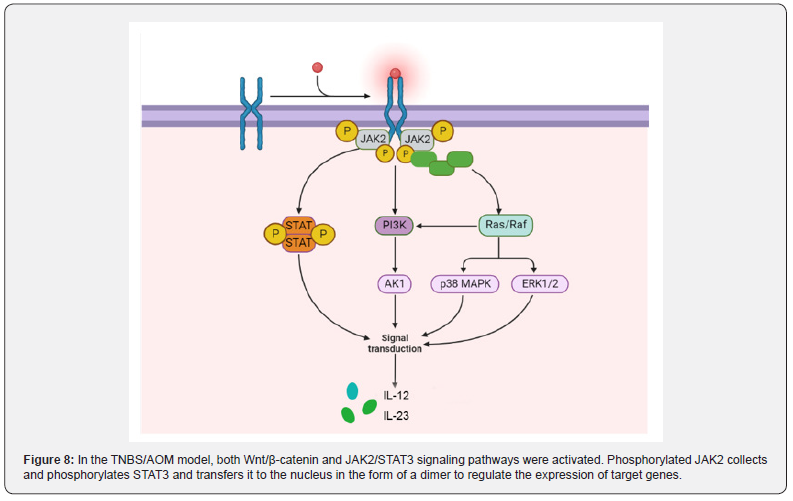
The JAK2/STAT3 signaling pathway plays an oncogenous role in the development and progression of CAC: JAK contains four tyrosine kinases (JAK1, JAK2, JAK3 and TYK2), while STAT contains seven transcription factors (STAT1, STAT2, STAT3, STAT4, STAT5A, STAT5B and STAT6). JAK2(Janus kinase 2)/STAT3 pathway is an important signal transduction pathway in the body, which plays a role in many physiological and pathological processes such as immunity, cell proliferation, differentiation, apoptosis and inflammatory response [121]. Under the stimulation of certain cytokines, JAK2 is activated, and then STAT3 is activated, which can transmit extracellular signal into the nucleus and regulate the expression of related inflammatory factors [122]. In the TNBS/ AOM animal model, the activation of Wnt/β-catenin and JAK2/ STAT3 pathways occurred with the development of colitis to adenoma.
He et al. [123]. confirmed that in TNBS-treated mice, Ang II was significantly elevated and the JAK2/STAT3 signaling pathway was activated to induce colitis by stimulating phosphorylation of JAK2 and STAT3. The colitis of TNBS mice treated with JAK2/ STAT3 pathway inhibitor tofacitinib was significantly improved. Lu et al. [124] found that TNBS can not only induce colitis, but also accompanied by visceral allergies. Subsequently, Wan [125] found that the expressions of JAK2, STAT3, p-JAK2 and p-STAT3 was upregulated in TNBS-treated goats. JAK2 is one of the four members of the JAK family located on the cell membrane, specifically binds to some transmembrane receptors and is phosphorylated by internal conformational changes. Phosphorylated JAK2 recruits a large number of STAT3, leading to STAT3 phosphorylation and nuclear translocation in the form of dimers, ultimately mediating the expression of downstream target genes (Figure 8).
The Wnt/β-catenin signaling pathway plays a tumorigenic role in CAC: Zhao et al. [126]. showed that the expression of TCF, β-catenin and Wnt upstream proteins in the TNBS group was significantly higher than that in the blank group, and the expression of tumor-associated proteins c-Myc and cyclinD1 was also significantly enhanced. In the Wnt/β-catenin signaling pathway, Wnt protein binds to the coiled protein (coiled gene) on the cell membrane. After seven trans membranes [127]. the signal is transmitted to the Dvl protein. Subsequently, the activation of Dvl protein inhibited the binding of intracellular complexes GSK-3β, AXIN and APC, resulting in the dissociation of GSK-3β, and then GSK-3β could not phosphorylate β-catenin [128,129]. Unphosphorylated β-catenin cannot be degraded by proteases, resulting in a large accumulation of β-catenin. This process promotes the entry of β-catenin into the nucleus, where it binds to nuclear LEF/TCF and induces the expression of tumor-associated target genes. Therefore, TNBS-induced colitis can complete the process from CD to CAC under the combined action of AOM.
The Other Mechanism of Three Pharmacological Models
Inflammation, endoplasmic reticulum stress and autophagy
It is confirmed that infection and a large number of inflammatory stimuli could cause endoplasmic reticulum stress and activate the UPR signaling pathway, mainly by promoting Ca2+ release from the endoplasmic reticulum and ROS accumulation. For example, pro-inflammatory cytokines IL-1β and IFN-γ can directly down-regulate the expression of Ca2+-ATPase (SERCA2) on the endoplasmic reticulum to depleted ER Ca2+ stores, thereby causing ERS [130].
In addition, ROS, as an important mediator of inflammatory response, can cause disorders of endoplasmic reticulum folding protein-related functions, leading to the accumulation of unfolded/ misfolded proteins in the endoplasmic reticulum and the release of Ca2+ in the endoplasmic reticulum into the cytoplasm. High concentration of CA2+ leads to mitochondrial dysfunction and produces more ROS [131]. Due to the long-term existence of this feedback mechanism in the body, intracellular ROS is continuously generated and accumulated, and abnormally accumulated ROS can aggravate endoplasmic reticulum stress.
Endoplasmic is the main site for protein folding and quality control. Any disturbance that alters endoplasmic reticulum stress homeostasis may lead to the accumulation of misfolded proteins, the activation of unfolded protein response, autophagy, or apoptosis [132] (Figure 9). Zhang et al. [133] . treated mice with AOM/DSS and found that the endoplasmic reticulum lumen was enlarged, with swelling, vacuoles, and a significant reduction in ribosomes. When the endoplasmic reticulum integrity was destroyed, the expression of GRP78, IRE1α, p-ERK, ATF6 and p-eIF-2α related to endoplasmic reticulum stress was significantly enhanced. Similarly, in TNBS -induced rats [134], the expression of endoplasmic reticulum stress-related proteins ATF6, ATF4, CHOP, BIP and XBP-1s was enhanced. Sharma et al.confirmed that the increased expression of p-PERK, p-eIF2α and chop proteins in rats [135], induced by DMH can lead to endoplasmic reticulum stress.
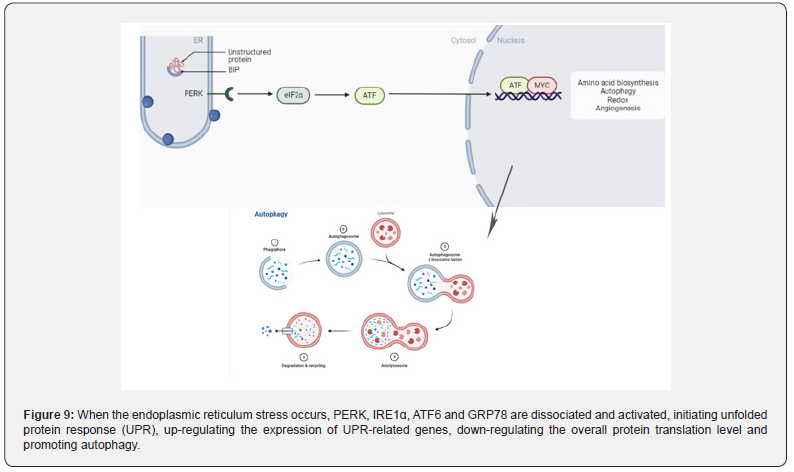
From the mechanism of action, studies have found that IBD-related endoplasmic reticulum stress was associated with autophagy dysfunction [136,137]. Autophagy is a conserved process in eukaryotic evolution through which cytoplasmic materials are degraded in lysosomes [138]. Zhang et al. [133] found that AOM combined with DSS could induce endoplasmic reticulum stress in the intestine of mice and observed a large number of double layers autophagic vesicles in the intestinal epithelial mucosal cells of mice by electron microscopy [138]. The expression of LC3II, an autophagy marker, was also significantly increased. In the DMH/DSS model mice [139]. the expression of P62 and the ratio of LC3II/LC3I were decreased. In TNBS model mice [140], the expression of autophagy related LC3II and autophagy flux were enhanced, and the autophagy ability of intestinal epithelial cells was enhanced.
When endoplasmic reticulum stress occurs in the body, autophagy function is activated [141], which in turn destroys the intestinal mucosal barrier and affects the physiological function of normal intestinal epithelial cells. Subsequently, the diversity of intestinal flora decreased, flora dysfunction [142]. and inflammatory cell infiltration [143] induced IBD, then developed into CAC. AOM/DSS, DMH/DSS and TNBS/AOM can well induce autophagy and endoplasmic reticulum stress during the progression of IBD to CAC.
Intestinal microbial imbalance
The intestinal epithelium is a monolayer of cells that serves as a physical barrier separating the mucosal immune system from symbiotic and pathogenic microorganisms [144]. Epithelial damage or increased epithelial permeability can destroy this barrier, thereby activating the mucosal immune system. Increased intestinal permeability is observed in patients with Crohn ‘s disease before clinical onset or recurrence, suggesting that barrier dysfunction may trigger disease progression [122,145]. The mechanism of IBD caused by intestinal flora imbalance is mainly divided into the following three aspects: the increased number of pathogenic bacteria, the decreased number of probiotics, and the imbalance of flora to break the normal immune tolerance of the body.
At present, the common human pathogenic bacteria are mostly found in Proteobacteria, and the bacteria belonging to Proteobacteria play an important role in the occurrence and development of IBD, especially Escherichia coli. When the intestinal flora is out of balance, the number of mutual pathogenic bacteria increases (such as adhesive invasive Escherichia coli, etc.). Pathogenic bacteria can destroy the normal internal mechanical barrier and immune barrier through invasion and secretion of toxins, increase the permeability of intestinal mucosa, and create conditions for the shift of flora. On the other hand, transferred bacteria over activate the immune response, causing indirect tissues to be attacked and damaged by the immune system [146]. which further increases the intestinal inflammatory response.
Relevant studies have found that the intestinal mucosal permeability of CD patients is significantly higher than that of normal people, resulting in a large number of bacteria and toxins in the intestine entering the intestinal mucosa and activating immune cells, which will trigger a strong immune response and destroy the mucosa [147]. This may be related to a series of reactions caused by the increase in the number of pathogenic bacteria in CD patients (Figure 9).
There are abundant probiotics in human body, such as Lactobacillus in Firmicutes, Bifidobacterium in Actinobacteria and Bacteroides in Bacteroidetes. When the intestinal flora is imbalanced, the number and richness of Firmicutes and Bacteroidetes in the normal body are reduced, and the number of probiotics such as Lactobacillus and Bifidobacterium is significantly reduced. The above functions are weakened, resulting in impaired intestinal mucosal barrier and proliferation and translocation of pathogenic bacteria. Studies have shown that the bacterial genus in the built-in bacteria can activate Paneth cells to secrete antimicrobial peptides to produce normal antibacterial effects. When the disease causes a significant increase in pathogenic bacteria and a decrease in bacterial genus, the production of antimicrobial peptides is reduced, which further causes excessive immune activation and damage to the intestinal mucosa [148].
Physiologically, there is a symbiotic relationship between intestinal microorganisms and hosts, which depends on a variety of immune mechanisms, such as intestinal mucus secreting immunoglobulin IgA and releasing antimicrobial peptides [149]. In IBD patients, the intestinal microecology is not regulated, and the imbalance of intestinal flora increases the expression and release of inflammatory factors. The normal immune balance of the intestine is broken [150], and the abnormal immune response is activated, resulting in intestinal mucosa and tissue damage.
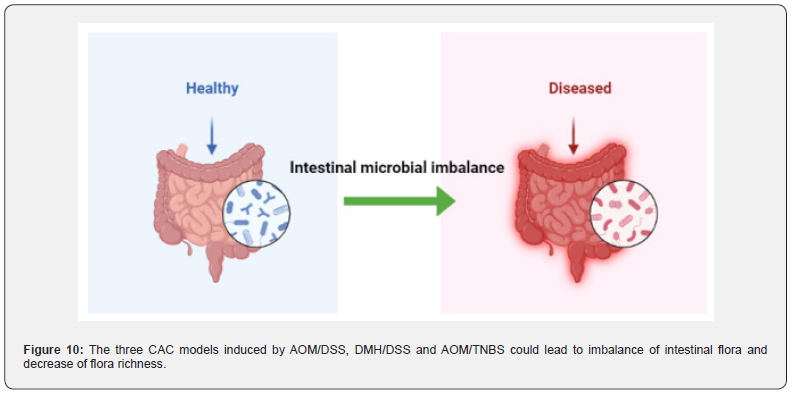
In AOM/DSS [151] and DMH/DSS [152] treated model animals, the abundance of Firmicutes increased and the abundance of Bacteroidetes decreased, resulting in a decrease in microbial diversity in vivo. In TNBS-induced model animals [153], the intestinal flora composition, species richness and diversity of the model group were significantly different from those of the control group, and the Proteobacteria and Bacteroidetes were significantly increased. After transplantation of fecal microbiota (FMT) in mice, the abundance of these two floras returned to normal levels and microbial diversity increased significantly by observing Shannon curve, PD forest index and CHAO1 index [154] (Figure 10).
Comparison of Three Animal Models (Table 1)
Advantage and disadvantage
AOM/DSS is the most widely used chemical reagent among the three models [155]. It is easy to use, easy to operate, and has a high tumor formation rate that can target tumor formation in animal colorectal regions. At the same time, it has the histological order change of ‘inflammation-dysplasia-carcinoma’. However, there are still some shortcomings in this model. For example, there is a lack of research on cancer stem cells in this model, and the mutations of tumor suppressor genes APC and P53 are rarely seen [156]. CRC has a low tendency of distant metastasis, and the life span of mice is shortened after illness, so it is not suitable for tumor invasion related research [28].
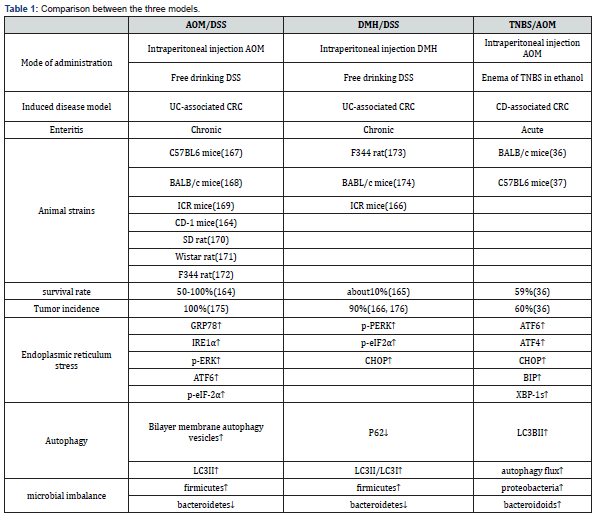
DMH/DSS-induced colon tumors in rodents are very close to human colon cancer in morphology, growth pattern and clinical manifestations [157]. And DMH-induced colorectal adenocarcinoma in mice often invades the submucosa and muscular layer, while AOM-induced colorectal adenocarcinoma does not exhibit such biological and histological properties [158]. However, the main disadvantage of the model using DMH is that multiple injections of DMH and long experimental periods are required to induce colon tumors in experimental animals. Secondly, after DMH is injected into the body, there are many uncontrollable factors in the process of producing AOM by oxidation, and its feasibility is not as high as AOM.
TNBS/AOM, in the construction process, due to TNBS is a semi-antigenic substance, it needs to be combined with ethanol to form a complete antigen, so some researchers use rectal perfusion construction. This model is based on the increased membrane permeability that occurs in IBD, which contributes to the entry of the luminal antigen that immune system cannot fully eliminate, namely haptenization [159]. The main advantages of the model include simple and low-cost protocol and repeatable colonic injury, short duration of the experiment, persistent injury and ulcers with inflammatory cell infiltration. Because of the lack of practice of this technique, the presence of feces in the colon, the anatomical localization of the descending colon, and the volume of the injection rate administered may cause rectal reflux of TNBS, leading to defects in the induction method or increased variability in animal disease.
Pathology
Clinically, IBD patients are also divided into UC and CD. According to Lin et al. [160], UC patients typically present with abdominal pain, increased frequency of diarrhea, and bloody mucopurulent diarrhea. Radiological examination showed obvious edema and hemorrhage of colonic mucosa, diffuse thickening of intestinal wall, occasional cryptitis and deformation of crypt dissection. In CD patients, intestinal wall thickening often occurred in the ileum [161], mucosal thickening and adjacent mesenteric fluid with severe submucosal fibrosis [162]. Saade C et al. [163] reported that approximately 91% of CD patients develop an inflammatory response in the ileum.
In the AOM/DSS-induced mouse UC model, Zhang et al. [133] found that the mice in the model group had pathological symptoms such as diarrhea, vertical hair and anal prolapse, and bloody stools appeared on the 6th day of modeling. Adenoma was diagnosed by histological examination. In Li ‘s experiment, mice in the AOM/ DSS treatment group showed glandular structure disorder and inflammatory cell infiltration. In addition, researchers found that about 80% of mice [164] in the AOM/DSS model group developed adenocarcinoma. In DMH/DSS-induced mice, polyploid adenomatous hyperplasia with malignant transformation of colon, polypoid adenoma [165], thickening of intestinal mucosa, inflammatory cell infiltration [83] bloody stool and rectal prolapse [166].
In TNBS/AOM-treated mice [167], lesions occurred mostly in the distal colon, with colonic crypt abnormalities, accompanied by colorless or polypoidal adenomas, and eventually adenocarcinoma [36]. In addition, some scholars found that AOM/TNBS-induced mice often appear diarrhea [168], bloody stools, colonic mucosal ulcers, submucosal edema and other obvious symptoms of enteritis [37]. Broadly speaking, UC and CD patients will first appear in the body’ s clinical inflammatory response, and then accompanied by abnormal growth of the crypt. However [169], in the three models of AOM/DSS, DMH/DSS and TNBS/AOM, the atypical hyperplasia of intestinal crypt usually appears first, followed by inflammatory cell infiltration and high expression of proinflammatory factors [170].
Conclusion
Various available animal models provide important tools for studying the complex development and pathogenesis of diseases [171]. These models could be used to provide new insights into etiology [172], pathophysiological mechanisms and treatment of human cancers [173]. For example, animal models help us understand the sequential acquisition of genetic and epigenetic changes observed in humans, and the consequent changes in cell behavior and tumor biology [3,159]. Many models are also provided to show the transfer process and the sensitivity to treatment characteristics [174].
Animal studies cannot replace human clinical trials [175], but they must be used in preclinical studies in time so that human diagnosis and treatment-oriented trials can become more concentrated and have a greater chance of success [176]. In fact, these models are not only valuable tools to reveal new mechanisms of CAC pathophysiology [177], but also promising tools to advance our understanding of tumor responses to novel chemoprevention and treatment strategies [178].
Among the three models, AOM/DSS animal model is the most widely used [179,180] the most in-depth studied and the most reductive chemical reagent, which can well simulate the pathogenesis of IBD to CAC [181]. Its operation is simple and convenient [28, 30,182,183] and the success rate and tumor formation rate are higher than the other two modeling methods, and the mechanism of AOM/DSS in vivo is relatively clearer than the other two chemical reagents. Therefore, most of the animal models used for the IBD to CAC process are mainly AOM/DSS [28].
However, the process from IBD to CAC is a complex pathological process induced by multiple factors. Although the model of AOM/DSS-induced IBD to CAC is relatively mature, the effects of diet and environmental factors on the pathogenesis are still not involved [184]. Secondly, the occurrence of cancer is inevitably accompanied by the process of tumor metastasis, but this feature has not been found in AOM/DSS-induced model animals. Thirdly, like any model, the CAC animal model induced by AOM/DSS has its limitations. For example, Kras or p53 mutations are typical in humans, but have not been detected in this mouse model [185]. In contrast, Kras mutations have been observed in CAC rats model using AOM reagent alone [186,187]. In addition, few literatures have compared the pathological features of AOM/DSS, DMH/DSS and TNBS/AOM with clinical related diseases.
If the future technology can combine dietary and environmental factors with the three models of CAC, the clinical tumor metastasis will be modeled in animals, and the clinicopathological features will be compared with the pathological features in the pathogenesis of model animals. It is believed that the pathogenesis of IBD to CAC induced by these three models will be closer to the process of human IBD to CAC, and its pathogenesis will be more comprehensive, providing more reasonable treatment for such patients in the direction of clinical treatment.
References
- Asanka Sanjeewa KK, Jayawardena TU, Kim HS, Kim SY, Shanura Fernando IP, et al. (2019) Fucoidan isolated from Padina commersonii inhibit LPS-induced inflammation in macrophages blocking TLR/NF-κB signal pathway. Carbohydrate polymers 224: 115195.
- El-Daly SM, Omara EA, Hussein J, Youness ER, El-Khayat Z (2019) Differential expression of miRNAs regulating NF-κB and STAT3 crosstalk during colitis-associated tumorigenesis. Molecular and cellular probes 47: 101442.
- Zeng S, Tan L, Sun Q, Chen L, Zhao H, et al. (2022) Suppression of colitis-associated colorectal cancer by scutellarin through inhibiting Hedgehog signaling pathway activity. Phytomedicine 98: 153972.
- Aviello G, Romano B, Borrelli F, Capasso R, Gallo L, et al. (2012) Chemo preventive effect of the non-psychotropic Phyto cannabinoid cannabidiol on experimental colon cancer. Journal of molecular medicine (Berlin, Germany) 90(8): 925-934.
- Pino MS, Chung DC (2010) The chromosomal instability pathway in colon cancer. Gastroenterology. 138(6): 2059-2072.
- Song M, Chan AT, Sun J (2010) Influence of the Gut Microbiome, Diet, and Environment on Risk of Colorectal Cancer. Gastroenterology 158(2): 322-340.
- Rutter M, Saunders B, Wilkinson K, Rumbles S, Schofield G, et al. (2004) Severity of inflammation is a risk factor for colorectal neoplasia in ulcerative colitis. Gastroenterology 126(2): 451-509.
- Alekhya Sita GJ, Gowthami M, Srikanth G, Krishna MM, Rama Sireesha K, et al. (2019) Protective role of luteolin against bisphenol A-induced renal toxicity through suppressing oxidative stress, inflammation, and upregulating Nrf2/ARE/ HO-1 pathway. IUBMB life 71(7): 1041-1047.
- Pigneur B, Ruemmele FM (2019) Nutritional interventions for the treatment of IBD: current evidence and controversies. Therapeutic advances in gastroenterology 12: 1756284819890534.
- Gu P, Feagins LA (2020) Dining with Inflammatory Bowel Disease: A Review of the Literature on Diet in the Pathogenesis and Management of IBD. Inflammatory bowel diseases 26(2): 181-191.
- Johnson E, Therkelsen SP, Nentwich I, Nissen-Meyer LSH, Hetland G (2019) IgE-sensitization to food and inhalant allergens in IBD patients compared with normal blood donors at Oslo University Hospital, Norway. Scandinavian journal of gastroenterology 54(9): 1107-1110.
- Scarozza P, Schmitt H, Monteleone G, Neurath F M, Atreya R (2019) Oligonucleotides-A Novel Promising Therapeutic Option for IBD. Frontiers in pharmacology 10: 314.
- Kanneganti M, Kenudson M, Mizoguchi E (2011) Animal models of colitis-associated carcinogenesis. Journal of biomedicine & biotechnology 2011: 342637.
- Feagins LA, Kim J, Chandrakumaran A, Gandle G, Naik KH, et al. (2020) Rates of Adverse IBD-Related Outcomes for Patients with IBD and Concomitant Prostate Cancer Treated with Radiation Therapy. Inflammatory bowel diseases 26(5): 728-733.
- Biancone L , Armuzzi A , Scribano ML , Castiglione F , D'Incà R , et al. (2020) Cancer Risk in Inflammatory Bowel Disease: A 6-Year Prospective Multicenter Nested Case-Control IG-IBD Study. Inflammatory bowel diseases 26(3): 450-459.
- Bernstein CN, Benchimol EI , Bitton A , Murthy SK , Nguyen GC, et al. (2019) The Impact of Inflammatory Bowel Disease in Canada 2018: Extra-intestinal Diseases in IBD. Journal of the Canadian Association of Gastroenterology 2(Suppl 1): S73-S80.
- Lakatos PL, Lakatos L (2008) Risk for colorectal cancer in ulcerative colitis: changes, causes and management strategies. World journal of gastroenterology 14(25): 3937-3947.
- Bürtin F, Mullins CS, Linnebacher M (2020) Mouse models of colorectal cancer: Past, present and future perspectives. World journal of gastroenterology 26(13): 1394-1426.
- Yamagiwa K, Ichikawa K (1977) Experimental study of the pathogenesis of carcinoma. CA: a cancer journal for clinicians 27(3): 174-181.
- Lisco H, Finkel MP, Brues AM (1947) Carcinogenic properties of radioactive fission products and of plutonium. Radiology 49(3): 361-363.
- Laqueur GL (1964) Carcinogenic Effects of Cycad Meal and Cycasin, Methylazoxymethanol Glycoside, in Rats and Effects of Cycasin in Germfree Rats. Federation proceedings 23: 1386-1388.
- Druckrey H, Preussmann R, Ivanković S, Schmidt CH, So BT, et al. (1965) Carcinogenic effect of azoethane and azoxyethane on rats. Zeitschrift fur Krebsforschung 67(1): 31-45.
- Rosenberg DW, Giardina C, Tanaka T (2009) Mouse models for the study of colon carcinogenesis. Carcinogenesis 30(2): 183-196.
- Fiala ES, Stathopoulos C (1984) Metabolism of methylazoxymethanol acetate in the F344 rat and strain-2 guinea pig and its inhibition by pyrazole and disulfiram. Journal of cancer research and clinical oncology 108(1): 129-134.
- Tanaka T (2012) Development of an inflammation-associated colorectal cancer model and its application for research on carcinogenesis and chemoprevention. International journal of inflammation 2012: 658786.
- Venkatachalam K, Vinayagam R, Arokia Vijaya Anand M, Isa NM, et al. (2020) Biochemical and molecular aspects of 1,2-dimethylhydrazine (DMH)-induced colon carcinogenesis: a review. Toxicology research 9(1): 2-18.
- Antoniou E, Margonis GA, Angelou A, Pikouli A, Argiri P, et al. (2016) The TNBS-induced colitis animal model: An overview. Annals of medicine and surgery 11: 9-15.
- De Robertis M, Massi E, Poeta ML, Carotti S, Morini S, et al. (2011) The AOM/DSS murine model for the study of colon carcinogenesis: From pathways to diagnosis and therapy studies. Journal of carcinogenesis 10: 9.
- Okayasu I, Ohkusa T, Kajiura K, Kanno J, Sakamoto S (1996) Promotion of colorectal neoplasia in experimental murine ulcerative colitis. Gut 39(1): 87-92.
- Tanaka T, Kohno H, Suzuki R, Yamada Y, Sugie S (2003) A novel inflammation-related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer science 94(11): 965-973.
- Ashley J Snider, Agnieszka B Bialkowska, Amr M Ghaleb, Vincent W Yang, Lina M Obeid, et al. (2016) Murine Model for Colitis-Associated Cancer of the Colon. Methods mol bio 1438: 245-254.
- Ziliang Ke, Chaohui Wang, Ting Wu, Weihong Wang, Yong Yang (2020) PAR2 deficiency enhances myeloid cell-mediated immunosuppression and promotes colitis-associated tumorigenesis. Cancer letters 469: 437-446.
- Chunsaier Wang, Wenbin Li, Hongying Wang, Yiming Ma, Xinhua Zhao, et al. (2019) Saccharomyces boulardii alleviates ulcerative colitis carcinogenesis in mice by reducing TNF-α and IL-6 levels and functions and by rebalancing intestinal microbiota. BMC microbiology 19(1): 246.
- Sing-Chung Li, Han-Pei Lin, Jung-Su Chang, Chun-Kuang Shih (2019) Lactobacillus Acidophilus-Fermented Germinated Brown Rice Suppresses Preneoplastic Lesions of the Colon in Rats. Nutrients 11(11): 2718.
- Hua Ye, Qiong Wu, Meng Guo, Kefeng Wu, Yingnian Lv, et al. (2016) Growth inhibition effects of ent-11α-hydroxy-15-oxo-kaur-16-en-19-oic-acid on colorectal carcinoma cells and colon carcinoma-bearing mice. Mol Med Rep 13(4): 3525-3532.
- Emi Osawa, Atsushi Nakajima, Toshio Fujisawa, Yuki I Kawamura, Noriko Toyama-Sorimachi, et al. (2006) Predominant T helper type 2-inflammatory responses promote murine colon cancers. Int J Cancer 118(9): 2232-2236.
- Y Xiao, X Dai, K Li, G Gui, J Liu, et al. (2017) Clostridium butyricum partially regulates the development of colitis-associated cancer through miR-200c. Cell Mol Biol (Noisy-le-grand) 63(4): 59-66.
- Yu-Lin Xu, Cui-Ling Ding, Chun-Lin Qian, Zhong-Tian Qi, Wen Wang (2019) Retinoid acid induced 16 deficiencies aggravates colitis and colitis-associated tumorigenesis in mice. Cell death dis 10(12): 958.
- Schepelmann M, Kupper N, Gushchina V, Mesteri I, Manhardt T, et al. (2022) AOM/DSS Induced Colitis-Associated Colorectal Cancer in 14-Month-Old Female Balb/C and C57/Bl6 Mice-A Pilot Study. International journal of molecular sciences 23(9): 5278.
- Helmut K Seitz (2019) The role of cytochrome P4502E1 in the pathogenesis of alcoholic liver disease and carcinogenesis. Chem Biol Interact 316: 108918.
- Teresa Peccerella, Tatjana Arslic-Schmitt, Sebastian Mueller, Kirstin-Berit Linhart, Devanshi Seth, et al. (2018) Chronic Ethanol Consumption and Generation of Etheno-DNA Adducts in Cancer-Prone Tissues. Adv Exp Med Biol 1032: 81-92.
- T Chewonarin, T Kinouchi, K Kataoka, H Arimochi, T Kuwahara, et al. (1999) Effects of roselle (Hibiscus sabdariffa ), a Thai medicinal plant, on the mutagenicity of various known mutagens in Salmonella typhimurium and on formation of aberrant crypt foci induced by the colon carcinogens azoxymethane and 2-amino-1-methyl-6-phenylimidazo[4,5-b] pyridine in F344 rats. Food Chem Toxicol 37(6): 591-601.
- O S Sohn 1, H Ishizaki, C S Yang, E S Fiala (1991) Metabolism of azoxymethane, methylazoxymethanol and N-nitrosodimethylamine by cytochrome P450IIE1. Carcinogenesis 12(1): 127-131.
- E S Fiala 1, C Joseph, O S Sohn, K el-Bayoumy, B S Reddy (1991) Mechanism of benzylselenocyanate inhibition of azoxymethane-induced colon carcinogenesis in F344 rats. Cancer Res 51(11): 2826-2830.
- H Matsumoto, H H Higa (1966) Studies on methylazoxymethanol, the aglycone of cycasin: methylation of nucleic acids in vitro. Biochem J 98(2): 20C-22C.
- O S Sohn, E S Fiala, S P Requeijo, J H Weisburger, F J Gonzalez (2001) Differential effects of CYP2E1 status on the metabolic activation of the colon carcinogens azoxymethane and methylazoxymethanol. Cancer Res 61(23): 8435-8440.
- Huayue Zhang, Dengcheng Hui, Yuan Li, Guangsu Xiong, Xiaoling Fu (2019) Canmei Formula Reduces Colitis-Associated Colorectal Carcinogenesis in Mice by Modulating the Composition of Gut Microbiota. Front Oncol 9: 1149.
- Masumi Suzui, Takamitsu Morioka, Naoki Yoshimi (2013) Colon preneoplastic lesions in animal models. Journal of toxicologic pathology 26(4): 335-341.
- Anastasios Angelou, Nikolaos Andreatos, Efstathios Antoniou, Argiro Zacharioudaki, George Theodoropoulos, et al. (2018) A Novel Modification of the AOM/DSS Model for Inducing Intestinal Adenomas in Mice. Anticancer Res 38(6): 3467-3470.
- Michael Bramhall, Oscar Flórez-Vargas, Robert Stevens, Andy Brass, Sheena Cruickshank (2015) Quality of methods reporting in animal models of colitis. Inflamm Bowel Dis 21(6): 1248-1259.
- Stefan Wirtz, Clemens Neufert, Benno Weigmann, Markus F Neurath (2007) Chemically induced mouse models of intestinal inflammation. Nat Protoc 2(3): 541-546.
- Samak G, Chaudhry KK, Gangwar R, Narayanan D, Jaggar JH, et al. (2015) Calcium/Ask1/MKK7/JNK2/c-Src signalling cascade mediates disruption of intestinal epithelial tight junctions by dextran sulfate sodium. The Biochemical journal 465(3): 503-515.
- Okayasu I, Hatakeyama S, Yamada M, Ohkusa T, Inagaki Y, et al. (1990) A novel method in the induction of reliable experimental acute and chronic ulcerative colitis in mice. Gastroenterology 98(3): 694-702.
- José Romeo Villarreal-Calderón, Ricardo X. Cuéllar, Martín R. Ramos-González, Nestor Rubio-Infante, et al. (2019) Interplay between the Adaptive Immune System and Insulin Resistance in Weight Loss Induced by Bariatric Surgery. Oxidative medicine and cellular longevity 2019: 3940739.
- Farhana Muhammad Yusoff, Kah Keng Wong, Norhanani Mohd Redzwan (2020) Th1, Th2, and Th17 cytokines in systemic lupus erythematosus. Autoimmunity 53(1): 8-20.
- Michelle Carvalho de Rezende, João Marcelo Peixoto Moreira, Laura Liana Maggi Fernandes, Vanessa Fernandes Rodrigues, Deborah Negrão-Corrêa (2020) Strongyloides venezuelensis-infection alters the profile of cytokines and liver inflammation in mice co-infected with Schistosoma mansoni. Cytokine 127: 154931.
- Letizia Venturini, Sara Bacchi, Enrica Capelli, Lorenzo Lorusso, Giovanni Ricevuti (2019) Modification of Immunological Parameters, Oxidative Stress Markers, Mood Symptoms, and Well-Being Status in CFS Patients after Probiotic Intake: Observations from a Pilot Study. Oxidative medicine and cellular longevity 2019: 1684198.
- Daiana K Frade Silva, Sâmia S Duarte, Thaís M H Lisboa, Rafael C Ferreira, Ana Luíza de O Lopes, et al. (2019) Antitumor Effect of a Novel Spiro-Acridine Compound is Associated with Up-Regulation of Th1-Type Responses and Antiangiogenic Action. Molecules 25(1): 29.
- Soon-Young Lee, Bossng Kang, So-Hyeon Bok, Seung Sik Cho, Dae-Hun Park (2019) Macmoondongtang modulates Th1-/Th2-related cytokines and alleviates asthma in a murine model. PloS one 14(12): e0224517.
- Janos Terzić, Sergei Grivennikov, Eliad Karin, Michael Karin (2010) Inflammation and colon cancer. Gastroenterology 138(6): 2101-14. e5.
- Salcedo R, Worschech A, Cardone M, Jones Y, Gyulai Z, et al. (2010) MyD88-mediated signaling prevents development of adenocarcinomas of the colon: role of interleukin 18. The Journal of experimental medicine 207(8): 1625-1636.
- Yassin M, Sadowska Z, Djurhuus D, Nielsen B, Tougaard P, et al. (2019) Upregulation of PD-1 follows tumour development in the AOM/DSS model of inflammation-induced colorectal cancer in mice. Immunology 158(1): 35-46.
- KK AS, TU J, HS K, SY K, IP SF, et al. (2019) Fucoidan isolated from Padina commersonii inhibit LPS-induced inflammation in macrophages blocking TLR/NF-κB signal pathway. Carbohydrate polymers 224: 115195.
- A W, R Z, J Y, GD B, CC DS, et al. (2020) Potential Therapeutic Targets for Lung Repair During Human Ex Vivo Lung Perfusion. The European respiratory journal 55(4): 1902222.
- L X, FC J, LM Z, WT H, JH L, et al. (2016) Targeting of MyD88 Homodimerization by Novel Synthetic Inhibitor TJ-M2010-5 in Preventing Colitis-Associated Colorectal Cancer. Journal of the National Cancer Institute 108(4).
- M F, A C, AS V, J C, K B, et al. (2007) Toll-like receptor-4 promotes the development of colitis-associated colorectal tumors. Gastroenterology 133(6): 1869-1881.
- JM U, M M, HH H, TC R, GS J, et al. (2009) Modulation of the intestinal microbiota alters colitis-associated colorectal cancer susceptibility. PloS one 4(6): 6026.
- SM E-D, EA O, J H, ER Y, Z E-K (2019) Differential expression of miRNAs regulating NF-κB and STAT3 crosstalk during colitis-associated tumorigenesis. Molecular and cellular probes 47: 101442.
- CH S, N K, SM L, RH N, SI C, et al. (2019) Effects of 17β-estradiol on colorectal cancer development after azoxymethane/dextran sulfate sodium treatment of ovariectomized mice. Biochemical pharmacology 164: 139-151.
- KS C, SY C, SS R, M L, HJ A. et al. (2018) Chemopreventive Effect of Aster glehni on Inflammation-Induced Colorectal Carcinogenesis in Mice. Nutrients 10(2): 202.
- XP W, M S, KJ K, C N, C T, et al. (2004) The interleukin-6 cytokine system regulates epidermal permeability barrier homeostasis. The Journal of investigative dermatology 123(1): 124-131.
- P P, L C, P G, MB H, M M, et al. (2003) High circulating levels of biologically inactive IL-6/SIL-6 receptor complexes in systemic juvenile idiopathic arthritis: evidence for serum factors interfering with the binding to gp130. Clinical and experimental immunology 13(2): 355-363.
- X P, Y Z, X S, B L, F M, et al. (2019) Hydrostatin-SN10 Ameliorates Pancreatitis-Induced Lung Injury by Affecting IL-6-Induced JAK2/STAT3-Associated Inflammation and Oxidative Stress. Oxidative medicine and cellular longevity 2019: 9659757.
- BR J, KS C, M L, HJ A (2020) Aster glehni High-Fat Diet Propelled AOM/DSS-Induced Colitis-Associated Colon Cancer Alleviated by Administration of via STAT3 Signaling Pathway. Biology 9(2): 24.
- J M, Y Y, Y F, F G, X Z, et al. (2018) PIAS3-mediated feedback loops promote chronic colitis-associated malignant transformation. Theranostics 8(11): 3022-3037.
- T H, K M, N S, Y C, G W, et al. (2018) Post-transcriptional regulation of PIAS3 expression by miR-18a in malignant mesothelioma. Molecular oncology 12(12): 2124-2135.
- S G, E K, J T, D M, GY Y, et al. (2009) IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer cell 15(2): 103-113.
- Cai Y, Liu W, Lin Y, Zhang S, Zou B, et al. (2019) Compound polysaccharides ameliorate experimental colitis by modulating gut microbiota composition and function. Journal of gastroenterology and hepatology 34(9): 1554-1562.
- GJ AS, M G, G S, MM K, K RS, et al. (2019) Protective role of luteolin against bisphenol A-induced renal toxicity through suppressing oxidative stress, inflammation, and upregulating Nrf2/ARE/ HO-1 pathway. IUBMB life 71(7): 1041-1047.
- X W, SM S, F W, S H, X Z, et al. (2020) Protective effect of Shao Yao decoction on colitis-associated colorectal cancer by inducing Nrf2 signaling pathway. Journal of ethnopharmacology 252: 112600.
- Cheung KL, Khor TO, Huang MT, (2010) Kong AN. Differential in vivo mechanism of chemoprevention of tumor formation in azoxymethane/dextran sodium sulfate mice by PEITC and DBM. Carcinogenesis 31(5): 880-885.
- A H, ZH Z, Y G, A N, HF S, et al. (2019) Identification of novel Nrf2 target genes as prognostic biomarkers in colitis-associated colorectal cancer in Nrf2-deficient mice. Life sciences 238: 116968.
- K A, G S (2015) Carvacrol exhibits antioxidant and anti-inflammatory effects against 1, 2-dimethyl hydrazine plus dextran sodium sulfate induced inflammation associated carcinogenicity in the colon of Fischer 344 rats. Biochemical and biophysical research communications 461(2): 314-320.
- VE S, RC M, RA L, CJ G, BS R, et al. (1994) Preclinical efficacy evaluation of potential chemopreventive agents in animal carcinogenesis models: methods and results from the NCI Chemoprevention Drug Development Program. Journal of cellular biochemistry Supplement 20: 32-54.
- CA R (2017) Three Pathways of Colonic Carcinogenesis in Rats. Anticancer research 37(1): 15-20.
- N G, Z R, Q Q, Y H, BJ J-C. et al. (2011) In vitro and in vivo biodistribution of ZRS1, a stabilized type I N-acetoxymethyl carbamate-containing combi-molecule. Drug metabolism letters 5(2): 141-149.
- OO H, MU R, M T, R K, AQ K, et al. (2012) Amelioration of 1,2 Dimethylhydrazine (DMH) induced colon oxidative stress, inflammation and tumor promotion response by tannic acid in Wistar rats. Asian Pacific journal of cancer prevention: APJCP 13(9): 4393-4402.
- Salik B, Yi H, Hassan N, Santiappillai N, Vick B, et al. (2020) Connerty P, et al. Targeting RSPO3-LGR4 Signaling for Leukemia Stem Cell Eradication in Acute Myeloid Leukemia. Cancer cell 38(2): 263-278.e6.
- Choi BR, Cave C, Na CH, Sockanathan S (2020) GDE2-Dependent Activation of Canonical Wnt Signaling in Neurons Regulates Oligodendrocyte Maturation. Cell reports 31(5): 107540.
- Zhou J, Toh SH, Chan ZL, Quah JY, Chooi JY, et al. (2018) A loss-of-function genetic screening reveals synergistic targeting of AKT/mTOR and WTN/β-catenin pathways for treatment of AML with high PRL-3 phosphatase. Journal of hematology & oncology 11(1): 36.
- Wei CY, Zhu MX, Yang YW, Zhang PF, Yang X, et al. (2019) Downregulation of RNF128 activates Wnt/β-catenin signaling to induce cellular EMT and stemness via CD44 and CTTN ubiquitination in melanoma. Journal of hematology & oncology 12(1): 21.
- Roel Nusse R, Clevers H (2017) Wnt/β-Catenin Signaling, Disease and Emerging Therapeutic Modalities. Cell 169(6): 985-999.
- Bilic J, Huang YL, Davidson G, Zimmermann T, Cruciat CM, et al. (2007) Wnt induces LRP6 signalosomes and promotes dishevelled-dependent LRP6 phosphorylation 316(5831): 1619-1622.
- He TC, Sparks AB, Rago C, Hermeking H, Zawel L, et al. (1998) Identification of c-MYC as a target of the APC pathway 281(5382): 1509-1512.
- Tetsu O, McCormick F (1999) Beta-catenin regulates expression of cyclin D1 in colon carcinoma cells 398(6726): 422-426.
- Tanaka T, Kohno H, Murakami M, Shimada R, Kagami S, et al. (2000) Colitis-related rat colon carcinogenesis induced by 1-hydroxy-anthraquinone and methylazoxymethanol acetate 7(3): 501-508.
- Femia AP, Bendinelli B, Giannini A, Salvadori M, Pinzani P, et al. (2005) Mucin-depleted foci have beta-catenin gene mutations, altered expression of its protein, and are dose- and time-dependent in the colon of 1,2-dimethylhydrazine-treated rats. International journal of cancer 116(1): 9-15.
- Femia AP, Tarquini E, Salvadori M, Ferri S, Giannini A, et al. (2008) K-ras mutations and mucin profile in preneoplastic lesions and colon tumors induced in rats by 1,2-dimethylhydrazine. International journal of cancer 122(1): 117-123.
- Femia AP, Dolara P, Giannini A, Salvadori M, Biggeri A, et al. (2007) Frequent mutation of Apc gene in rat colon tumors and mucin-depleted foci, preneoplastic lesions in experimental colon carcinogenesis. Cancer research 67(2): 445-449.
- Nones K, Waddell N, Song S, Patch AM, Miller D, et al. (2014) Genome-wide DNA methylation patterns in pancreatic ductal adenocarcinoma reveal epigenetic deregulation of SLIT-ROBO, ITGA2 and MET signaling. International journal of cancer 135(5): 1110-1118.
- Yuying Yao, Zijun Zhou, Liuyou Li, Junchen Li, Lixun Huang, et al. (2019) Activation of Slit2/Robo1 Signaling Promotes Tumor Metastasis in Colorectal Carcinoma through Activation of the TGF-β/Smads Pathway 8(6): 635.
- Barak R, Lahmi R, Gevorkyan Airapetov L, Levy E, Tzur A, et al. (2014) Crystal structure of the extracellular juxta membrane region of Robo1. Journal of structural biology 186(2): 283-291.
- Qian Qian Zhang, Da Lei Zhou, Yan Lei, Li Zheng, Sheng Xia Chen, et al. (2015) Slit2/Robo1 signaling promotes intestinal tumorigenesis through Src-mediated activation of the Wnt/β-catenin pathway. Oncotarget 6(5): 3123-3135.
- Kumar S, Agnihotri N (2019) Piperlongumine a piper alkaloid targets Ras/PI3K/Akt/mTOR signaling axis to inhibit tumor cell growth and proliferation in DMH/DSS induced experimental colon cancer. Biomedicine & pharmacotherapy Biomedecine & pharmacotherapie 109: 1462-1477.
- Gi Shih Lien, Chien Huang Lin, You Lan Yang, Ming Shun Wu, Bing Chang Chen, et al. (2016) Ghrelin induces colon cancer cell proliferation through the GHS-R, Ras, PI3K, Akt, and mTOR signaling pathways. European journal of pharmacology 776: 124-131.
- Inês Silva, Rui Pinto, Vanessa Mateus (2019) Preclinical Study in Vivo for New Pharmacological Approaches in Inflammatory Bowel Disease: A Systematic Review of Chronic Model of TNBS-Induced Colitis. Journal of clinical medicine 8(10): 1574.
- Motavallian Naeini A, Andalib S, Rabbani M, Mahzouni P, Afsharipour M, et al. (2012) Validation and optimization of experimental colitis induction in rats using 2, 4, 6-trinitrobenzene sulfonic acid. Research in pharmaceutical sciences 7(3): 159-169.
- Udai P Singh, Chandrasekar Venkataraman, Rajesh Singh, James W Lillard (2007) CXCR3 axis: role in inflammatory bowel disease and its therapeutic implication. Endocrine, metabolic & immune disorders drug targets 7(2): 111-123.
- W Strober, B Kelsall, I Fuss, T Marth, B Ludviksson, et al. (1997) Reciprocal IFN-gamma and TGF-beta responses regulate the occurrence of mucosal inflammation. Immunology today 18(2): 61-64.
- Stefan Wirtz, Clemens Neufert, Benno Weigmann, Markus F Neurath (2007) Chemically induced mouse models of intestinal inflammation. Nature protocols 2(3): 541-546.
- Sophia B Zeeff, Cindy Kunne, Gerd Bouma, Rob B de Vries, Anje A Te Velde, et al. (2016) Actual Usage and Quality of Experimental Colitis Models in Preclinical Efficacy Testing: A Scoping Review. Inflammatory bowel diseases 22(6): 1296-1305.
- CO E, RB S, GS T, RH R (1995) Experimental models of inflammatory bowel disease. Gastroenterology 109(4): 1344-1367.
- Puneet Kaur Randhawa, Kavinder Singh, Nirmal Singh, Amteshwar Singh Jaggi (2014) A review on chemical-induced inflammatory bowel disease models in rodents. The Korean journal of physiology & pharmacology: official journal of the Korean Physiological Society and the Korean Society of Pharmacology 18(4): 279-288.
- Warren Strober, Atsushi Kitani, Stefan Fichtner Feigl, Ivan J Fuss (2009) The signaling function of the IL-13Ralpha2 receptor in the development of gastrointestinal fibrosis and cancer surveillance. Current molecular medicine 9(6): 740-750.
- HR A, PB B, M N, PS N (2019) Resveratrol Downregulates miR-31 to Promote T Regulatory Cells during Prevention of TNBS-Induced Colitis. Molecular nutrition & food research e1900633.
- Efstathios Antoniou, Georgios Antonios Margonis, Anastasios Angelou, Anastasia Pikouli, Paraskevi Argiri, et al. (2016) The TNBS-induced colitis animal model: An overview. Annals of medicine and surgery 11: 9-15.
- Ewa Kontny, Joanna Dmowska Chalaba, Brygida Kwiatkowska, Włodzimierz Maśliński (2017) Cytokines and integrins related to inflammation of joint and gut in patients with spondyloarthritis and inflammatory bowel disease. Reumatologi 55(6): 276-283.
- Stasa Kosler, Borut Strukelj , Ales Berlec (2017) Lactic Acid Bacteria with Concomitant IL-17, IL-23 and TNFα- Binding Ability for the Treatment of Inflammatory Bowel Disease. Current pharmaceutical biotechnology 18(4): 318-326.
- P Kubes (2000) Inducible nitric oxide synthase: a little bit of good in all of us Gut 47(1): 6-9.
- Tomohiro Watanabe, Naoki Asano, Peter J Murray, Keiko Ozato, Prafullakumar Tailor, et al. (2008) Muramyl dipeptide activation of nucleotide-binding oligomerization domain 2 protects mice from experimental colitis. The Journal of clinical investigation 118(2): 545-559.
- Sugimoto K (2008) Role of STAT3 in inflammatory bowel disease. World journal of gastroenterology 14(33): 5110-5114.
- Aggeletopoulou I, Assimakopoulos SF, Konstantakis C, Triantos C (2018) Interleukin 12/interleukin 23 pathway: Biological basis and therapeutic effect in patients with Crohn's disease. World journal of gastroenterology 24(36): 4093-4103.
- L H, J D, Y C, C L, M Z, S A, et al. (2019) The Renin-angiotensin system promotes colonic inflammation by inducing T17 activation via JAK2/STAT pathway American journal of physiology Gastrointestinal and liver physiology 316(6): G774-G784.
- Y L, BC J, DL C, LX Z, YL Z (2017) Chemokine CCL8 and its receptor CCR5 in the spinal cord are involved in visceral pain induced by experimental colitis in mice. Brain research bulletin 135: 170-178.
- J W, Y D, AH T, MK S, H J, et al. (2017) Electroacupuncture Attenuates Visceral Hypersensitivity by Inhibiting JAK2/STAT3 Signaling Pathway in the Descending Pain Modulation System. Frontiers in neuroscience 11: 644.
- HM Z, Y L, XY H, XK L, F C, et al. (2019) Pharmacological mechanism of Sishen Wan attenuated experimental chronic colitis by inhibiting Wnt/β-catenin pathway. Journal of ethnopharmacology 240: 111936.
- O S-G, JJ V-A, VM B-A (2019) Wnt/β-Catenin Signaling as a Molecular Target by Pathogenic Bacteria. Frontiers in immunology 10: 2135.
- Zhao H, Tang S, Tao Q, Ming T, Lei J, et al. (2023) Ursolic Acid Suppresses Colorectal Cancer by Down-Regulation of Wnt/β-Catenin Signaling Pathway Activity. Journal of agricultural and food chemistry 71(9): 3981-3993.
- Zeng S, Chen L, Sun Q, Zhao H, Yang H, et al. (2021) Scutellarin ameliorates colitis-associated colorectal cancer by suppressing Wnt/β-catenin signaling cascade. European journal of pharmacology 906: 174253.
- Cardozo AK, Ortis F, Storling J, Feng YM, Rasschaert J, et al. (2005) Cytokines downregulate the sarcoendoplasmic reticulum pump Ca2+ ATPase 2b and deplete endoplasmic reticulum Ca2+, leading to induction of endoplasmic reticulum stress in pancreatic beta-cells Diabetes 54(2): 452-461.
- Malhotra JD, Miao H, Zhang K, Wolfson A, Pennathur S, et al. (2008) Antioxidants reduce endoplasmic reticulum stress and improve protein secretion. Proceedings of the National Academy of Sciences of the United States of America105(47): 18525-18530.
- Senft D, Ronai ZA (2015) UPR, autophagy, and mitochondria crosstalk underlies the ER stress response. Trends in biochemical sciences 40(3): 141-148.
- YS Z, F W, SX C, XJ Q (2018) Natural dietary compound naringin prevents azoxymethane/dextran sodium sulfate-induced chronic colorectal inflammation and carcinogenesis in mice. Cancer biology & therapy 19(8): 735-744.
- I C, B S-M, C P, N M, MJ C, et al. (2012) Glutamine treatment attenuates endoplasmic reticulum stress and apoptosis in TNBS-induced colitis. PloS one7(11): e50407.
- SH S, J SK, S M, S N (2020) Morin supplementation modulates PERK branch of UPR and mitigates 1,2-dimethylhydrazine-induced angiogenesis and oxidative stress in the colon of experimental rats. Toxicology mechanisms and methods 30(4): 306-315.
- A L, N B, HTT N (2020) New insights into the interplay between autophagy, gut microbiota and inflammatory responses in IBD. Autophagy16(1): 38-51.
- Rubio CA (2017) Three Pathways of Colonic Carcinogenesis in Rats. Anticancer research 37(1): 15-20.
- Nguyen HT, Lapaquette P, Bringer MA, Darfeuille-Michaud A (2013) Autophagy and Crohn's disease. Journal of innate immunity 5(5): 434-443.
- PP T, GB J, KB T, V K (2016) Melatonin modulated autophagy and Nrf2 signaling pathways in mice with colitis-associated colon carcinogenesis. Molecular carcinogenesis 55(3): 255-267.
- Z W, L S, S H, C Q, M F (2019) IL-33 ameliorates experimental colitis involving regulation of autophagy of macrophages in mice. Cell & bioscience 9: 10.
- A T, S S, A T, ET C, P P (2020) Targeting endoplasmic reticulum stress and autophagy as therapeutic approaches for neurological diseases. International review of cell and molecular biology 350: 285-325.
- G N, F B, CL B, S G, F L, et al. (2019) TREM Receptors Connecting Bowel Inflammation to Neurodegenerative Disorders. Cells 8(10).
- Z L, F L, W W, C S, D G, et al. (2020) Study of the alleviation effects of a combination of Lactobacillus rhamnosus and inulin on mice with colitis. Food function 11(5): 3823-3837.
- Sun Q, He M, Zhang M, Zeng S, Chen L, et al. (2020) Ursolic acid: A systematic review of its pharmacology, toxicity and rethink on its pharmacokinetics based on PK-PD model. Fitoterapia 147: 104735.
- Wyatt J, Vogelsang H, Hübl W, Waldhöer T, Lochs H (1993) Intestinal permeability and the prediction of relapse in Crohn's disease. Lancet (London, England) 341(8858): 1437-1439.
- Pickard JM, Zeng MY, Caruso R, Núñez G (2017) Gut microbiota: Role in pathogen colonization, immune responses, and inflammatory disease. Immunological reviews 279(1): 70-89.
- Buhner S, Buning C, Genschel J, Kling K, Herrmann D, et al. (2006) Genetic basis for increased intestinal permeability in families with Crohn's disease: role of CARD15 3020insC mutation? Gut 55(3): 342-347.
- Vaishnava S, Behrendt CL, Ismail AS, Eckmann L, Hooper LV (2008) Paneth cells directly sense gut commensals and maintain homeostasis at the intestinal host-microbial interface. Proceedings of the National Academy of Sciences of the United States of America 105(52): 20858-20863.
- Goethel A, Turpin W, Rouquier S, Zanello G, Robertson SJ, et al. (2019) Nod2 influences microbial resilience and susceptibility to colitis following antibiotic exposure. Mucosal immunology 12(3): 720-732.
- Glassner KL, Abraham BP, Quigley EMM (2020) The microbiome and inflammatory bowel disease. The Journal of allergy and clinical immunology145(1): 16-27.
- Y L, S W, Y S, W X, H Z, Y W, et al. (2020) Apple polysaccharide protects ICR mice against colitis associated colorectal cancer through the regulation of microbial dysbiosis. Carbohydrate polymers 230: 115726.
- Qingchao Zhu, Zhiming Jin, Wen Wu, Renyuan Gao, Bomin Guo, et al. (2014) Analysis of the intestinal lumen microbiota in an animal model of colorectal cancer. PloS one 9(6): e90849.
- Ying Cai, Wen Liu, Yixuan Lin, Shaobao Zhang, Baorong Zou, et al. (2019) Compound polysaccharides ameliorate experimental colitis by modulating gut microbiota composition and function. J Gastroenterol Hepatol 34(9): 1554-1562.
- Zitao Wang, Wenjie Hua, Chen Li, Hao Chang, Ran Liu, et al. (2019) Protective Role of Fecal Microbiota Transplantation on Colitis and Colitis-Associated Colon Cancer in Mice Is Associated with Treg Cells. Fron microbiol 10: 2498.
- Sun Q, Yang H, Liu M, Ren S, Zhao H, et al. (2022) Berberine suppresses colorectal cancer by regulation of Hedgehog signaling pathway activity and gut microbiota. Phytomedicine 103: 154227.
- Kobaek-Larsen M, Thorup I, Diederichsen A, Fenger C, Hoitinga MR (2000) Review of colorectal cancer and its metastases in rodent models: comparative aspects with those in humans. Comparative medicine 50(1): 16-26.
- Rogers AE, Nauss KM (1985) Rodent models for carcinoma of the colon. Digestive diseases and sciences 30(12 Suppl): 87s-102s.
- Pozharisski KM, Likhachev AJ, Klimashevski VF, Shaposhnikov JD (1979) Experimental intestinal cancer research with special reference to human pathology. Advances in cancer research 30: 165-237.
- Jurjus AR, Khoury NN, Reimund JM (2004) Animal models of inflammatory bowel disease. Journal of pharmacological and toxicological methods 50(2): 81-92.
- YY L, JM Z, YJ J, Z M, HD Z, Y H, et al. (2020) Typical ulcerative colitis treated by herbs-partitioned moxibustion: A case report. World journal of clinical cases 8(8): 1515-1524.
- J D, O E, P L, S M, B O (2020) Volumetric analysis of small bowel motility in an unselected cohort of patients with Crohn's disease. Neurogastroenterol Motil 2020: e13909.
- Nina Zidar, Cord Langner, Miha Jerala, Emanuela Boštjančič, David Drobne (2020) Pathology of Fibrosis in Crohn's Disease-Contribution to Understanding Its Pathogenesis. Frontiers in medicine 7: 167.
- C Saade, L Nasr, A Sharara, K Barada, A Soweid, et al. (2019) Crohn's disease: A retrospective analysis between computed tomography enterography, colonoscopy, and histopathology. Radiography 25(4): 349-358.
- Margarita Damazo-Lima, Guadalupe Rosas-Pérez, Rosalía Reynoso-Camacho, Iza F Pérez-Ramírez, Nuria E Rocha-Guzmán, et al. (2020) Chemopreventive Effect of the Germinated Oat and its Phenolic-AVA Extract in Azoxymethane/Dextran Sulfate Sodium (AOM/DSS) Model of Colon Carcinogenesis in Mice. Foods 9(2).
- Isha Rani, Kim Vaiphei, Navneet Agnihotri (2014) Supplementation of fish oil augments efficacy and attenuates toxicity of 5-fluorouracil in 1,2-dimethylhydrazine dihydrochloride/dextran sulfate sodium-induced colon carcinogenesis. Cancer chemotherapy and pharmacology. 74(2): 309-322.
- Yuhua Li, Changxu Liu, Dan Xiao, Jing Han, Zhenggang Yue, et al. (2015) Trillium tschonoskii steroidal saponins suppress the growth of colorectal Cancer cells in vitro and in vivo. Journal of ethnopharmacology 168: 136-145.
- Nina Seiwert, Jörg Fahrer, Georg Nagel, Jan Frank, Dariush Behnam (2021) Curcumin Administered as Micellar Solution Suppresses Intestinal Inflammation and Colorectal Carcinogenesis. Nutrition and cancer 73(4): 1-8.
- R G, A A, F M, P A, A J, M T, et al. (2020) Lactobacillus species inhibitory effect on colorectal cancer progression through modulating the Wnt/β-catenin signaling pathway. Molecular and cellular biochemistry 470: 1-13.
- YC C, PY H, WJ C, SH W, MH P (2020) Lactobacillus fermentum V3 ameliorates colitis-associated tumorigenesis by modulating the gut microbiome. American journal of cancer research 10(4): 1170-1181.
- He L, Du J, Chen Y, Liu C, Zhou M, Adhikari S, et al. (2019) Renin-angiotensin system promotes colonic inflammation by inducing T(H)17 activation via JAK2/STAT pathway. Am J Physiol Gastrointest Liver Physiol 316(6): G774-g784.
- Burhannudin, N M, S W, D P (2018) Chemopreventive Effects of Edible Canna (Canna edulis Kerr.) Against Colorectal Carcinogenesis: Effects on Expression of Adenomatous Polyposis Coli and Inducible Nitric Oxide Synthase in Rat Inflammatory Model. A Asian Pac J Cancer Prev 19(3): 839-844.
- T T, Y Y, M T, T T, T O, KM R (2009) Melatonin suppresses AOM/DSS-induced large bowel oncogenesis in rats. Chem Biol Interact 177(2): 128-136.
- SC L, HP L, JS C, CK S (2019) Lactobacillus Acidophilus-Fermented Germinated Brown Rice Suppresses Preneoplastic Lesions of the Colon in Rats. Nutrients 11(11).
- Sandeep Kumar, Navneet Agnihotri (2019) Piperlongumine, a piper alkaloid targets Ras/PI3K/Akt/mTOR signaling axis to inhibit tumor cell growth and proliferation in DMH/DSS induced experimental colon cancer. Biomed Pharmacother 109: 1462-1477.
- M T, M I, H K, T T, H M, K M, et al. (2019) Dietary Fucoxanthin Induces Anoikis in Colorectal Adenocarcinoma by Suppressing Integrin Signaling in a Murine Colorectal Cancer Model. J Clin Med 9(1).
- Y L, L L, Y N, J F, Y S, X K, et al. (2012) Modified apple polysaccharide prevents tumorigenesis in a mouse model of colitis-associated colon cancer: role of galectin-3 and apoptosis in cancer prevention. European journal of nutrition. 51(1): 107-117.
- Zhao H, Ren S, Yang H, Tang S, Guo C, Liu M, et al. (2022) Peppermint essential oil: its phytochemistry, biological activity, pharmacological effect and application. Biomed Pharmacother 154: 113559.
- Marian B (2004) Colorectal cancer: modeling causes, prevention and therapy. Drug Discovery Today: Disease Models 1(1): 1-7.
- Parang B, Barrett CW, Williams CS (2016) AOM/DSS Model of Colitis-Associated Cancer. Methods Mol Biol 1422: 297-307.
- Lin R, Piao M, Song Y, Liu C (2020) Quercetin Suppresses AOM/DSS-Induced Colon Carcinogenesis through Its Anti-Inflammation Effects in Mice. Journal of immunology research 2020: 9242601.
- Angelou A, Andreatos N, Antoniou E, Zacharioudaki A, Theodoropoulos G, et al. (2018) A Novel Modification of the AOM/DSS Model for Inducing Intestinal Adenomas in Mice. Anticancer research. 38(6): 3467-3470.
- Suzuki R, Kohno H, Sugie S, Nakagama H, Tanaka T (2006) Strain differences in the susceptibility to azoxymethane and dextran sodium sulfate-induced colon carcinogenesis in mice. Carcinogenesis 27(1): 162-169.
- Neufert C, Becker C, Neurath MF (2007) An inducible mouse model of colon carcinogenesis for the analysis of sporadic and inflammation-driven tumor progression. Nature protocols 2(8): 1998-2004.
- Snider AJ, Bialkowska AB, Ghaleb AM, Yang VW, Obeid LM, et al. (2016) Murine Model for Colitis-Associated Cancer of the Colon. Methods in molecular biology 1438: 245-254.
- Suzui M, Yoshimi N, Ushijima T, Hirose Y, Makita H, et al. (1995) No involvement of Ki-ras or p53 gene mutations in colitis-associated rat colon tumors induced by 1-hydroxyanthraquinone and methylazoxymethanol acetate. Molecular carcinogenesis 12(4): 193-197.
- Erdman SH, Wu HD, Hixson LJ, Ahnen DJ, Gerner EW (1997) Assessment of mutations in Ki-ras and p53 in colon cancers from azoxymethane- and dimethylhydrazine-treated rats. Molecular carcinogenesis 19(2): 137-144.
- Takahashi M, Mutoh M, Kawamori T, Sugimura T, Wakabayashi K (2000) Altered expression of beta-catenin, inducible nitric oxide synthase and cyclooxygenase-2 in azoxymethane-induced rat colon carcinogenesis. Carcinogenesis 21(7): 1319-1327.






























