Adynamic Ileus in Hospitalized Adults: Current Concepts and Practices
Gioacchino D De Sario Velasquez1, Miguel Eduardo Rodriguez Rodriguez1,2, Jesus A Cordova Guilarte1, Megha Bhushan Kalawar3, Víctor Bolívar1, Jhon Navarro Gonzalez4, Coralvia Yaroslangna Villanueva Perez5, Mariela Guerrero Rubio6, Samantha Redden1, Yeisson Rivero-Moreno1, Angeles Astrid Zamudio Fuentes7, Fernando Alfonso Galvez Coronel8 and Maria Isabel Gomez9*
1Universidad de Oriente, Venezuela
2Larkin Community Hospital, Miami, USA
3Liaquat University of Health and Medical Sciences, Pakistan
4Universidad del Zulia, Venezuela
5Universidad Nacional Experimental Francisco de Miranda, Venezuela
6Universidad Autónoma de Tamaulipas, México
7Universidad de Monterrey, México
8Universidad autónoma de Sinaloa, México
9Universidad del Valle, México
Submission:July 11, 2023; Published:July 18, 2023
*Corresponding author: Maria Isabel Gomez, Universidad Del Valle De México, México, Email: mariaisagcoral@gmail.com
How to cite this article: Gioacchino D D S V, Miguel E R R, Jesus A Cordova G, Megha BK, Víctor B, et al. Adynamic Ileus in Hospitalized Adults: Current Concepts and Practices. Adv Res Gastroentero Hepatol, 2023; 19(5): 556023. DOI: 10.19080/ARGH.2023.19.556023.
Abstract
Adynamic ileus is a common cause of obstruction of intestinal transit, hindering the passage of digested food and fluids. Many scenarios like compromised blood supply of the intestines, Ogilivie’s syndrome, surgery, and medications like opioids can trigger this condition, which results in a significant concern that may increase the chance of patient morbidity. Multiple strategies, including optimizing pain control, minimizing medications that can slow intestinal motility, early mobilization after surgery, and prompt treatment of underlying conditions, have demonstrated decreased risk in hospitalized patients. Close monitoring allows the opportunity to identify and treat the symptoms as a surgical emergency.
Keywords: Adynamic ileus; Postoperative ileus; Paraityc ileus; Ogilivie’s syndrome; Mesenteric vascular occlusion
Abbreviations: NG: Nasogastric; POI: Postoperative Ileus; GI: Gastrointestinal; NSAIDs: Non-Steroidal Anti-Inflammatory Drugs; ERAS: Enhanced Recovery After Surgery; ACPO: Acute Colonic Pseudo-Obstruction; AMI: Acute Mesenteric Ischemia; NOMI: Non-Occlusive Mesenteric Ischemia; SMA: Superior Mesenteric Artery; CT: Computed Tomography; CDT: Catheter-Directed Thrombolysis; PTA: Percutaneous Transluminal Angioplasty; ROMS: Retrograde Open Mesenteric Stenting
Introduction
Intestinal obstruction is defined as a partial or complete blockage of the intestinal transit, hindering the passage of digested food and fluids. There are two main types of obstruction: dynamic obstruction, caused by physical blockage or constriction, and adynamic obstruction, resulting from the failure of normal bowel motility [1]. Adynamic ileus encompasses various conditions such as paralytic ileus, characterized by a lack of intestinal peristalsis; mesenteric vascular obstruction, caused by compromised blood supply to the intestines; and Ogilvie’s syndrome, a pseudo-obstruction typically affecting the colon. Patients with certain risk factors, such as those undergoing surgery, those critically ill, and those taking certain medications, such as opioids, may experience paralytic ileus [1,2]. The incidence and prevalence of this condition can vary depending on the underlying cause and patient population. However, it is widespread in healthcare settings, especially among surgical patients and critically ill individuals. Prevention of paralytic ileus involves identifying and managing risk factors. Strategies may include optimizing pain control, minimizing medications that can slow intestinal motility, early mobilization after surgery, and prompt treatment of underlying conditions that may lead to ileus. Close monitoring and appropriate postoperative care can also help prevent complications and aid in the timely management of paralytic ileus [3,4].
The clinical presentation of paralytic ileus typically involves diffuse abdominal distension, pain, nausea, vomiting, and decreased or absent bowel sounds. There may be signs of dehydration, electrolyte imbalance, and systemic complications in severe cases. Diagnosis of paralytic ileus involves a combination of clinical evaluation, medical history review, physical examination, and diagnostic tests. Imaging techniques may show bowel dilation, air-fluid levels, or the absence of bowel gas [1,5]. Blood tests can help assess electrolyte imbalances and identify underlying causes. Lastly, the treatment of paralytic ileus focuses on addressing the underlying cause, relieving symptoms, and promoting bowel function. Management may involve supportive measures such as bowel rest, fluid and electrolyte replacement, pain control, and the discontinuation or adjustment of medications contributing to the ileus [2,5]. In severe cases, surgical intervention may be necessary to remove the physical obstruction or address any underlying structural abnormalities. This narrative review aims to provide an overview of the current concepts concerning early diagnosis and multidisciplinary management of adynamic ileus.
Epidemiology & Risk Factors
The incidence of ileus, a condition characterized by the impairment or cessation of bowel motility, can vary significantly depending on various factors. One crucial determinant is the type of surgery performed, the extent of bowel manipulation, and preexisting medical conditions. Generally, lower abdominal surgeries that involve larger open incisions and more extensive manipulation of the intestines are associated with a higher risk of developing ileus. On the other hand, laparoscopic procedures, which involve minimal bowel manipulation, such as cholecystectomy (gallbladder removal), tend to have a lower risk of ileus [6,7].
Despite extensive research, the precise cause of ileus remains unclear. However, several risk factors have been identified that can increase the likelihood and duration of this condition. Prolonged abdominal or pelvic surgeries, particularly those involving the lower gastrointestinal tract, open surgeries, and retroperitoneal spinal surgeries, have been associated with a higher risk of developing ileus. Additionally, the use of opioids for pain management, intra-abdominal inflammation resulting from conditions like sepsis or peritonitis, peritoneal carcinomatosis (the presence of cancerous cells in the peritoneal cavity), and perioperative complications such as pneumonia or abscess formation can all contribute to the development of ileus [6]. Other factors, such as intraoperative or postoperative bleeding, low levels of potassium in the blood (hypokalemia), and delayed initiation of enteral nutrition or placement of a nasogastric (NG) tube, can also increase the risk [6,7].
Many factors influence the risk of developing ileus, each affecting different aspects of the complex neuroimmune system. Certain medications, including opioids, antihypertensives, and antiemetics, have increased the risk. Furthermore, various medical conditions such as pneumonia, stroke, and electrolyte abnormalities can also contribute [6]. Understanding these risk factors is crucial for healthcare providers to identify patients at a higher risk of developing ileus and implement appropriate preventive measures and management strategies.
Physiopathology
Bowel motility is regulated by a complex interaction between the intrinsic and autonomic nerves and the individual and coordinated activity of the smooth muscle cells. Because ileus is a complex disorder involving many systems and factors, its exact mechanism and cause are unknown. Ileus is a neuroimmune interaction consisting of the early neurogenic and inflammatory phases. This interplay involves two-way communication between the autonomic nerve (afferent, efferent, enteric) and the immune (mast cells, macrophages) outside and inside the intestine [8].
Furthermore, considerable evidence from both experimental and clinical studies suggests that the inflammation cascade is involved in the development of ileus and is associated with the increased release of several cytokines in both human and animal models [9,10].
Also, macrophages in the muscle membrane outside the intestinal wall release cytokines that activate additional inflammatory cells and other pro-inflammatory cytokines (including interleukin-6 and TNF-alpha), neuropeptides, and nitric oxide [11]. Surgical stress and intestinal maneuvers activate a local molecular inflammatory reaction, and hormonal and neurohormonal releases, leading to persistent inhibitory sympathetic activity and neuromuscular depression with subsequent cessation of bowel mobility and stopped intestinal transit [10-12].
Prevention
Preventive measures for paralytic ileus aim to minimize the risk of its occurrence and promote normal intestinal motility. First and foremost, careful surgical techniques can help reduce the incidence of postoperative paralytic ileus. Surgeons should employ careful bowel management, gentle tissue manipulation, and avoiding excessive tissue trauma to minimize the risk of postoperative ileus. Additionally, laparoscopic surgery and minimally invasive techniques can help reduce surgical trauma and accelerate postoperative recovery, potentially decreasing the likelihood of paralytic ileus [13-15].
Optimal pain management plays a crucial role in preventing paralytic ileus. Adequate pain control can minimize opioid analgesics, as these medications are associated with a higher risk of ileus development. Employing multimodal analgesia techniques, including nonsteroidal anti-inflammatory drugs (NSAIDs), local anesthetics, and epidural analgesia, can effectively manage pain while reducing the need for opioids and the subsequent risk of paralytic ileus [14,15]. Early postoperative ambulation and physical activity are important preventive measures. Encouraging patients to move as soon as possible after surgery helps stimulate bowel motility and prevent the occurrence of ileus. Early mobilization, including walking and active range-of-motion exercises, promotes peristalsis and facilitates the return of normal bowel function [15]. Furthermore, carefully managing electrolyte imbalances, particularly potassium, and magnesium, are essential for preventing paralytic ileus. These electrolytes play a critical role in maintaining smooth muscle contractility, and abnormalities in their levels can disrupt intestinal motility. Regular monitoring and correcting electrolyte imbalances and administering supplements, as needed, can help prevent ileus development. Lastly, adequate hydration and appropriate nutritional support are vital preventive measures. Maintaining fluid and electrolyte balance through intravenous hydration, oral intake, or enteral nutrition promotes optimal intestinal function and reduces the risk of paralytic ileus [16,17].
Treatment Strategies
The management of paralytic ileus, also known as adynamic ileus, requires a multimodal approach to promote the restoration of normal bowel function. Several critical elements of treatment based on previous studies have been identified. Perioperative strategies play a crucial role in preventing paralytic ileus. The use of mechanical bowel preparation before surgery, a common practice in the past, is now evaluated on a case-by-case basis due to recent evidence suggesting its potential harm to bowel function. Specific lactobacillus preparations taken orally in the pre-and postoperative period have shown promise in maintaining gastrointestinal motility and preventing postoperative ileus. Additionally, consuming carbohydrate-rich liquids before surgery has demonstrated the potential to shorten the recovery time of bowel function postoperatively [18-21]. Pharmacological interventions can be employed to prevent paralytic ileus. Drugs such as alvimopan and methylnaltrexone, which are opioid antagonists, have shown promise in preventing postoperative ileus, particularly in high-risk patients. These medications counteract the adverse effects of opioids on bowel motility and facilitate gastrointestinal recovery. Anti-inflammatory medications, including non-steroidal anti-inflammatory drugs (NSAIDs) and COX-2 inhibitors, may also enhance gastrointestinal motility after abdominal surgery [20,22,23].
Multimodal approaches are essential to controlling this condition. Effective pain management is crucial in minimizing the adverse effects of pain and opioids on bowel function. Regional anesthesia techniques, epidural analgesia, or non-opioid analgesics can reduce opioid requirements and promote early recovery of bowel function [23-25]. Enhanced Recovery After Surgery (ERAS) protocols, involving preoperative counseling, optimized anesthesia techniques, early mobilization, and oral intake, can minimize the duration of postoperative ileus and facilitate faster recovery. The use of nasogastric decompression should be evaluated individually, as recent studies have questioned its efficacy in upper gastrointestinal surgery. Early enteral nutrition, if tolerated, can stimulate bowel function and promote recovery. Encouraging ambulation and physical activity early on can stimulate bowel motility and reduce the duration of paralytic ileus [22-25]. Overall, the management of paralytic ileus should be tailored to the individual patient, incorporating a combination of strategies with demonstrated effectiveness. This multimodal approach, including early feeding, epidural analgesia, laparoscopic surgery, and peripherally acting mu-opioid-receptor antagonists, targets the diverse factors contributing to paralytic ileus, resulting in faster return of normal bowel function and shorter hospital stays.
Ogilvie’s Syndrome
Acute colonic pseudo-obstruction, also known as Ogilvie’s syndrome, was first described in 1948 and refers to massive dilation of the colon without underlying mechanical obstruction or other organic cause; it is confined to the cecum and ascending colon with transition near the splenic flexure [26,27]. The incidence of this disease is frequently cited as approximately 100 cases per 100,000 hospital admissions every year, although some underreporting is suspected. Prevalence appears to be slightly increased in males, but the reason for this is unknown. The average age at the presentation is approximately 60 years. Almost all patients have multiple underlying comorbidities, and those who are functionally dependent at baseline develop the disease with the highest frequency. Surgical patients will most likely be diagnosed on postoperative days 3 to 5 [27,28].
Ogilvie’s syndrome is typically found in hospitalized patients, who often have severe comorbid conditions, such as severe musculoskeletal abnormalities, trauma, surgery, or sepsis, and is associated with increased morbidity and mortality. Other conditions that appear to increase the risk of Ogilvie’s syndrome include electrolyte imbalances, medications (i.e., narcotics, anticholinergics), and debilitation [28,29]. Appropriate imaging is essential to the diagnosis and management of ACPO. The plan includes radiography consisting of upright and supine abdominal films, commonly performed but limited in their utility. Expected findings include varying degrees of colonic dilatation, normal haustral markings, and lack of signs of mechanical obstruction such as volvulus. Perhaps the most important use of plain radiography is to measure the degree of initial colonic dilatation and for serial imaging during observation after diagnosis. Abdominopelvic CT with intravenous contrast is the standard diagnostic test, with a sensitivity of 96% and a specificity of 93%. It confirms the presence of proximal colonic dilatation and excludes the presence of intrinsic or extrinsic mechanical obstruction [30].
Many different approaches have been used to manage Ogilvie’s syndrome. It may be managed by addressing underlying conditions (i.e., discontinuation of narcotics, correction of electrolyte abnormalities) and decompressing the gastrointestinal tract via nasogastric (NG) tube or rectal tube insertion. Additionally, neostigmine has gained popularity due to a small, randomized study suggesting its benefit [31]. Although not established in randomized clinical trials, Decompressive colonoscopy is also often used, as it can provide immediate colonic decompression [32,33]. Ogilvie’s syndrome is one of the few indications of cecostomy. Surgical cecostomy is the definitive intervention for patients with ACPO unresponsive to other therapies without evidence of ischemia or perforation [34].
Mesenteric Vascular Occlusion
Acute mesenteric ischemia (AMI) is characterized by the sudden interruption of blood supply to the intestine, resulting in cellular damage and intestinal necrosis and potentially leading to death if left untreated. AMI can be occlusive or non-occlusive (NOMI), with mesenteric arterial embolism, mesenteric arterial thrombosis, or mesenteric venous thrombosis as primary etiologies [35]. The overall incidence of AMI is low, accounting for 0.09-0.2% of all acute admissions to emergency departments. While it is an infrequent cause of abdominal pain, it is a significant cause of emergent intestinal resection. Prompt diagnosis and intervention are crucial to reduce the mortality rates associated with AMI, which can exceed 50% [36].
Patients with AMI typically present in their 60s or 70s, and women are up to three times more susceptible than men. Abdominal pain is the main complaint, and its characterization may vary depending on the cause of arterial obstruction. Symptoms are often present for more than 24 hours at the time of presentation, and AMI can be misdiagnosed as other conditions such as pancreatitis, hepatobiliary disease, diverticulitis, appendicitis, or bowel obstruction. A high level of suspicion is necessary for accurate diagnosis [35-37]. The classic description of AMI includes severe, constant pain disproportionate to physical examination findings. Patients may experience emesis along with the onset of pain. Physical examination findings such as rebound tenderness and guarding may be delayed due to the progression of ischemia. Other symptoms that may be present include nausea, diarrhea, hypotension, and blood per rectum [37,38].
Diagnosis of AMI can be challenging, as some markers typically associated with the condition, such as leukocytosis, lactic acidosis, elevated creatinine, and elevated pancreatic enzymes, are nonspecific and may occur late in the acute process. Diagnostic imaging plays a crucial role in confirming the suspected diagnosis. Computed tomography (CT) imaging, including CT angiography, has become the gold standard for imaging mesenteric vessels. CT angiography provides immediate results and enables evaluation of compromised bowel, the cause of AMI, and aids in planning revascularization. However, patients with hemodynamic compromise, gross evidence of peritonitis, or severe acidosis do not require imaging and should proceed directly to exploratory laparotomy [38,39].
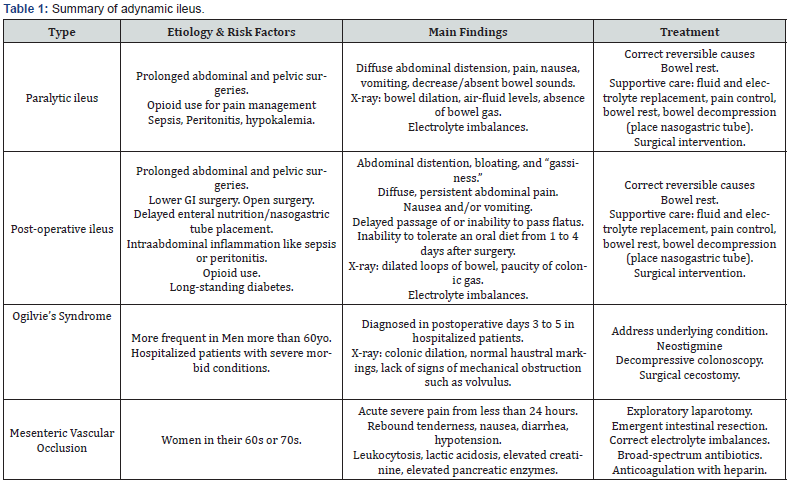
AMI is considered a surgical emergency, and immediate treatment should be initiated following confirmation of the diagnosis while awaiting operative intervention. Patients should be resuscitated with isotonic crystalloid infusion, correction of electrolyte imbalances, and broad-spectrum antibiotics [37-39]. Therapeutic anticoagulation with heparin should be obtained unless contraindicated. Vasopressors may be necessary for patients in distributive shock but should be initiated only after appropriate volume resuscitation to avoid exacerbating bowel ischemia. Surgical management aims to respect the infarcted bowel and revascularize the superior mesenteric artery (SMA). Revascularization should ideally be performed before resection to assess the viability of the affected bowel. In frank bowel necrosis with perforation and peritoneal contamination, source control is obtained through resection of the affected bowel. Revascularization options include thromboembolectomy, mesenteric bypass, endovascular therapies such as catheterdirected thrombolysis, percutaneous transluminal angioplasty, stent placement, or a hybrid approach involving laparotomy with retrograde open mesenteric stenting [38-41]. Table 1 provides a summary of the different types of adynamic ileus.
Conclusion
Several factors contribute to the development of adynamic ileus, including surgical procedures, medications, electrolyte imbalances, inflammation, and immobility. These factors disrupt the intricate balance of neural, hormonal, and muscular mechanisms responsible for gastrointestinal motility. By examining the underlying pathophysiological processes, the article sheds light on potential targets for intervention and prevention of adynamic ileus. Various diagnostic modalities, such as clinical assessment, abdominal imaging, and laboratory tests, are discussed in the review to aid in accurately identifying and differentiating adynamic ileus from other conditions. Prompt and accurate diagnosis is crucial to prevent management delays and minimize complications associated with adynamic ileus, such as bowel perforation, bacterial overgrowth, and prolonged hospital stays.
This review also explores the management strategies for adynamic ileus, which primarily focus on supportive measures. These include bowel rest, fluid and electrolyte management, pain control, early ambulation, and the judicious use of prokinetic agents. While these interventions often alleviate symptoms and promote recovery, the evidence base supporting their efficacy is limited, and there needs to be more consensus on the optimal approach to managing adynamic ileus. Therefore, the importance of further prospective research studies must be considered. Future investigations should aim to elucidate the underlying mechanisms of adynamic ileus and identify novel therapeutic targets. Largescale, multicenter trials are needed to evaluate the effectiveness of various management strategies and determine the optimal timing and duration of interventions. Additionally, studies exploring the role of emerging therapies, such as neuromodulation techniques or targeted pharmacological agents, may provide new avenues for improving outcomes in patients with adynamic ileus.
References
- Weinberg L, Bader A, Clark JM (2017) Diagnosis and treatment of intestinal obstruction. Aust Fam Physician 46(12): 918-922.
- Chang SS, Chuang SC, Kuo WH (2016) Prognostic factors associated with postoperative ileus after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. World J Surg Oncol 14(1): 42.
- Richards WO, Williams LF (1992) Obstruction and pseudo-obstruction of the large bowel. Surg Clin North Am 72(1): 85-105.
- Gadelkareem RA, Elgaddal AA, Elhanafy E (2021) Acute colonic pseudo-obstruction (Ogilvie's syndrome) in general surgical practice: a systematic review. World J Emerg Surg 16(1): 36.
- Vather R, Bissett IP, Fenwick SW (2018) Paralytic ileus affecting the surgical patient. BJS Open 2(1): 9-16.
- Beach EC, De Jesus O Ileus (2023) In: StatPearls [Internet]. StatPearls Publishing, Treasure Island (FL), Florida, USA.
- Liang W, Li J, Zhang W, Liu J, Li M (2019) Prolonged postoperative ileus in gastric surgery: Is there any difference between laparoscopic and open surgery? Cancer medicine 8(12): 5515-5523.
- Wehner S, Vilz TO, Stoffels B, Kalff JC (2012) Immune mediators of postoperative ileus. Langenbecks Arch Surg 397(4): 591-601.
- Wehner S, Behrendt FF, Lyutenski BN, Lysson M, Bauer AJ, et al. (2007) Inhibition of macrophage function prevents intestinal inflammation and postoperative ileus in rodents. Gut 56(2): 176-185.
- Hupa KJ, Stein K, Schneider R, Lysson M, Schneiker B, et al. (2019) AIM2 inflammasome-derived IL-1β induces postoperative ileus in mice. Sci Rep 9(1): 10602.
- Vilz TO, Wehner S, Pantelis D, Kalff JC (2014) Immunomodulatory aspects in the development, prophylaxis and therapy for postoperative ileus. Zentralbl Chir 139(4): 434-444.
- Gero D, Gié O, Hübner M, Demartines N, Hahnloser D (2017) Postoperative ileus: in search of an international consensus on definition, diagnosis, and treatment. Langenbecks Arch Surg 402(1): 149-158.
- Asgeirsson T, El-Badawi KI, Mahmood A, Barletta J, Luchtefeld M (2010) Postoperative ileus: it costs more than you expect. J Am Coll Surg 210(2): 228-231.
- Kalff JC, Schraut WH, Simmons RL, Bauer AJ (1998) Surgical manipulation of the gut elicits an intestinal muscularis inflammatory response resulting in postsurgical ileus. Ann Surg 228(5): 652-663.
- Senagore AJ (2007) Pathogenesis and clinical and economic consequences of postoperative ileus. Am J Health Syst Pharm 64(20 Suppl 13): S3-S7.
- Steinbrook RA, Alther JC, Ding S, Kreis ME (2012) The role of epidural anesthesia and analgesia in postoperative outcome. Anesthesiol Clin North Am 18(2): 407-428.
- Vather R, Trivedi S, Bissett I (2013) Defining postoperative ileus: results of a systematic review and global survey. J Gastrointest Surg 17(5): 962-972.
- Saclarides TJ (2006) Current choices--good or bad--for the proactive management of postoperative ileus: A surgeon's view. J Perianesth Nurs 21(2A Suppl): S7-S15.
- Story SK, Chamberlain RS. (2009) A comprehensive review of evidence-based strategies to prevent and treat postoperative ileus. Dig Surg 26(4): 265-275.
- Augestad KM, Delaney CP (2010) Postoperative ileus: impact of pharmacological treatment, laparoscopic surgery and enhanced recovery pathways. World J Gastroenterol 16(17): 2067-2074.
- Weledji EP (2020) Perspectives on paralytic ileus. Acute Med Surg 7(1): e573.
- Asgeirsson T, El-Badawi KI, Mahmood A, Barletta J, Luchtefeld M (2010) Postoperative ileus: it costs more than you expect. J Am Coll Surg 210(2): 228-231.
- Holte K, Kehlet H (2000) Postoperative ileus: a preventable event. Br J Surg. 87(11): 1480-1493.
- Lobo DN. Fluid (2004) electrolytes and nutrition: physiological and clinical aspects. Proc Nutr Soc 63(3): 453-466.
- Watters JM, Kirkpatrick SM, Norris SB, Shamji FM (1999) Prevention of postoperative ileus in elective colorectal surgery: a multi-factorial analysis of "protective factors". Am J Surg 177(4): 273-277.
- Kaiser AM (2010) Ogilvie transition to colonic perforation. Am J Surg 200: e15-16.
- Haj M, Haj M, Rockey DC (2018) Ogilvie's syndrome: management and outcomes. Medicine (Baltimore) 97(27): e11187.
- Maloney N, Vargas HD (2005) Acute intestinal pseudo-obstruction (Ogilvie's syndrome). Clin Colon Rectal Surg 18(2): 96-101.
- Saunders MD, Kimmey MB (2005) Systematic review: acute colonic pseudo-obstruction. Aliment Pharmacol Ther 22: 917-925.
- P Pereira, F Djeudji, P Leduc, F Fanget, X Barth (2015) Ogilvie's syndrome-acute colonic pseudo-obstruction, Journal of Visceral Surgery, 152(2): 99-105.
- De Giorgio R, Barbara G, Stanghellini V, et al. (2001) Review article: the pharmacological treatment of acute colonic pseudo-obstruction. Aliment Pharmacol Ther 15(11): 1717-1727.
- Jetmore AB, Timmcke AE, Gathright JB, Jr, T C Hicks, J E Ray, et al. (1992) Ogilvie's syndrome: colonoscopic decompression and analysis of predisposing factors. Dis Colon Rectum 35(12): 1135-1142.
- Geller A, Petersen BT, Gostout CJ (1996) Endoscopic decompression for acute colonic pseudo-obstruction. Gastrointest Endosc 44: 144-150.
- Corman M L (1998) Colon and Rectal Surgery. (4th), Lippincott-Williams and Wilkins, Philadelphia, USA.
- Clair DG, Beach JM (2016) Mesenteric Ischemia. N Engl J Med 374(10): 959-968.
- Bala M, Catena F, Kashuk J, De Simone B, Gomes CA, et al. (2022) Acute mesenteric ischemia: updated guidelines of the World Society of Emergency Surgery. World J Emerg Surg 17(1): 54.
- Wyers MC (2010) Acute Mesenteric ischemia: diagnostic approach and surgical treatment. Semin Vasc Surg 23(1): 9-20.
- Sise MJ (2014) Acute mesenteric ischemia. Surg Clin North Am 94(1): 165-181.
- Oldenburg WA, Lau LL, Rodenberg TJ, Hope J Edmonds, Charles D Burger (2004) Acute mesenteric ischemia: a clinical review. Arch Intern Med 164(10): 1054-1062.
- Boley SJ, Feinstein FR, Sammartano R (1981) New concepts in the management of emboli of the superior mesenteric artery. Surg Gynecol Obstet 153(4): 561-569.
- Park WM, Gloviczki P, Cherry KJ, John W Hallett Jr, Thomas C Bower, et al. (2002) Contemporary management of acute mesenteric ischemia: Factors associated with survival. J Vasc Surg 35(3): 445-452.






























