Stable Disease on Targeted oral Therapy, A Rare Case Report of Carney’s Triad
Blackmore C1,2*, Leach M3,4 and Rutovitz J3,4
1Southwestern Sydney Clinical School, University of New South Wales, Australia
2School of Medicine Notre Dame Australia
3Department of Haematology and Oncology, Sydney Adventist Hospital, Sydney, Australia
4Northern Haematology and Oncology Group, Integrated Cancer Centre, Wahroonga, Australia
Submission:June 05, 2023; Published:June 14, 2023
*Corresponding author: Conner Blackmore, Southwestern Sydney Clinical School, University of New South Wales, School of Medicine Notre Dame Australia, Australia, Email: connerblackmore@gmail.com
How to cite this article: Blackmore C, Leach M, Rutovitz J. Stable Disease on Targeted oral Therapy, A Rare Case Report of Carney’s Triad. Adv Res Gastroentero Hepatol, 2023; 19(4): 556020. DOI: 10.19080/ARGH.2023.19.556020.
Abstract
Carney’s triad is a rare non-hereditary condition of unknown origin characterized by the presence of gastrointestinal stromal tumors, pulmonary chondromas and extra-adrenal paragangliomas or phaeochromocytomas. Surgical intervention is the current mainstay of treatment, with inconclusive evidence for the success of targeted oral therapy. We present a case of a young female who presented to the hospital with unilateral leg swelling, who upon further investigation was diagnosed a c-kit positive gastrointestinal stromal tumor and later diagnosed with carney’s triad. She underwent a partial complete surgical resection and commenced oral imatinib therapy, which was up titrated until her disease stabilized. This case demonstrates a further rare variant of Carney’s Triad with a c-kit positive GIST stable on oral targeted therapy.
Keywords: Carney’s triad; Oral therapy; Gastrointestinal stromal tumors; Upper gastro-intestinal bleeding
Abbreviations: CT: Carney’s Triad; GIST: Gastrointestinal Stromal Tumors; PHEO: Phaeochromocytomas; PGL: Extra-Adrenal Paragangliomas; CHO: Pulmonary Chondromas; SDH: Succinate Dehydrogenase; US: Ultrasound; CT: Computerized Tomography
Introduction
Carney’s triad (CT) is a rare non-hereditary sporadic condition of unknown origin characterized by the presence of tumors with low incidence, which include gastrointestinal stromal tumors (GIST), pulmonary chondromas (CHO) and extra-adrenal paragangliomas (PGL)/phaeochromocytomas (PHEO) [1-4]. CT is primarily seen in young women and most patient’s present with GIST and pulmonary CHO; PGL/PHEO are rarely the presenting tumor and are often an incidental finding at screening [5]. A marked reduction in enzymatic activity of succinate dehydrogenase (SDH) has been detected in tumors associated with CT, however the presence of gene mutations coding for the four SDH subunits (SDHA, SDHB, SDHC and SDHD, collectively known as the ‘SDHx’ genes) are very rare, and only recently described in the literature [4]. Current treatment for CT is surgical management of GIST, PGL and PHEO; with metastatic disease unresponsive to chemotherapy and radiotherapy [4]. Targeted therapy (e.g., imatinib and sunitinib) which has been shown to be successful in the treatment of sporadic GIST, has been predominantly ineffective or inconclusive positive results in the management of CT [3,4,6]. Here, we present a case of CT in a young female with an atypical presentation and histological confirmation of c-kit (CD 117), SDH-deficient wild-type pediatric GIST whose condition is currently stable on oral targeted therapy.
Case Presentation
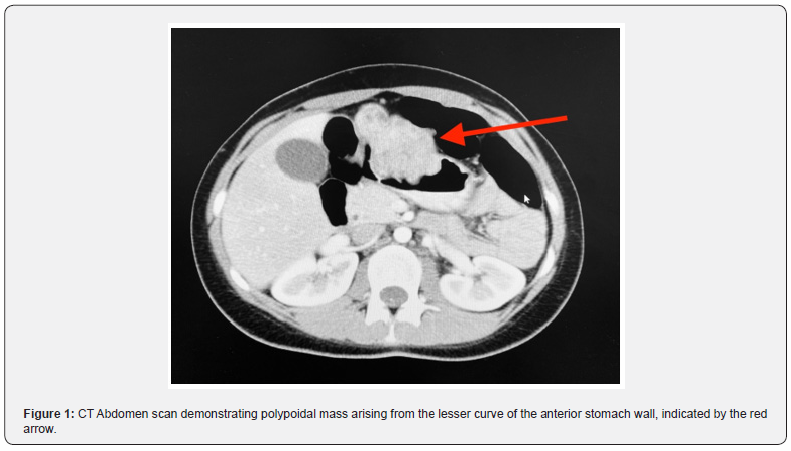
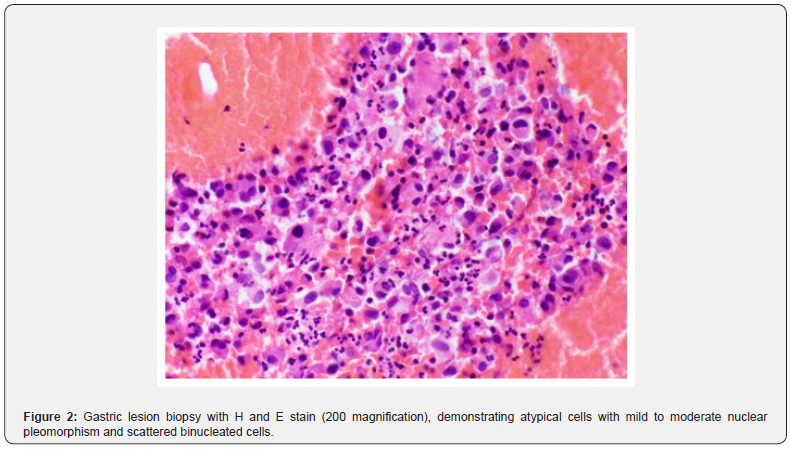
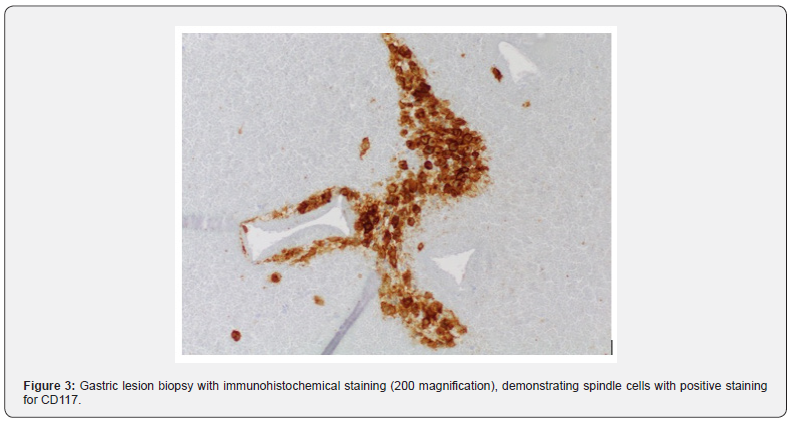
An 18-year-old woman presented to the emergency department with acute unilateral leg swelling, which following an ultrasound was diagnosed as a deep vein thrombosis. Otherwise, she was asymptomatic for any other pathology, the rest of her examination was normal, and she had no previous medical history prior to this presentation. She was followed up by her community general practitioner who performed an Ultrasound (US) and Computerized Tomography (CT) scan which demonstrated an 11.5cm polypoidal lesion arising from the lesser curve and anterior wall of the stomach with multiple liver lesions (Figure 1). Upper Gastrointestinal endoscopic examination was performed, and three biopsies of the gastric mass were taken which demonstrated atypical cells (Figure 2) and following immunoperoxidase staining these cells were strongly positive for CD 117 (Figure 3), consistent with a diagnosis of GIST, with suspicion for a diagnosis of CT due to the patient’s demographics. At the request of a clinical geneticist, additional immunohistochemistry was performed which confirmed a diagnosis of SDHB deficient GIST, further adding to the suspicion of a diagnosis of CT. Complete resection was achieved with a partial gastrectomy, hepatectomy and pancreatectomy, and she was placed under the care of a medical oncologist and commenced on oral Imatinib 400mg daily.
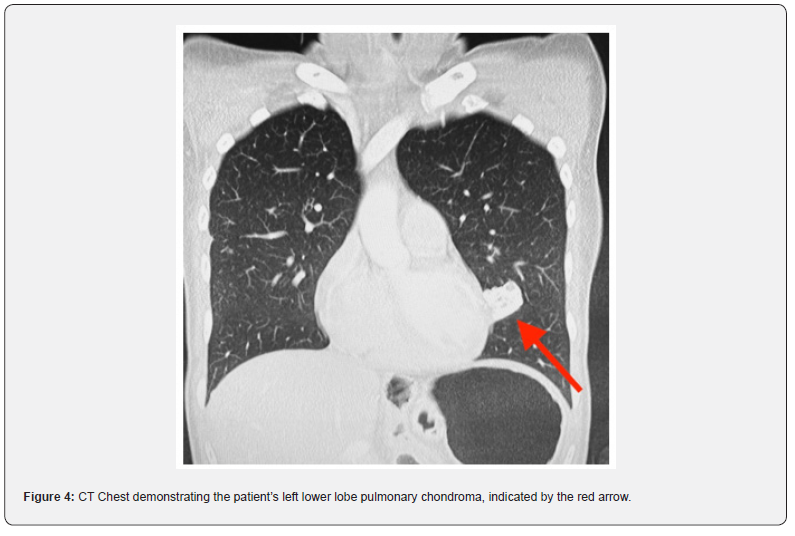
One year following initial diagnosis she was found to have new suspicious liver lesions, which following a primavist MRI were determined to represent atypical haemangiomata, not requiring intervention and to be monitored on 12-monthly CT scans. Six years following her initial diagnosis her liver lesions remained stable. However, she was found to have a calcified left lower lobe pulmonary lesion (Figure 4), which did not demonstrate any uptake on a following PET scan. This lesion was deemed to represent a pulmonary chondroma following a hospital lung multi-disciplinary meeting, not requiring surgical intervention. Following the identification of the additional lesion her oncologist increased her dose of oral Imatinib to 600mg daily. On a routine yearly monitoring PET scan 12 years following her initial diagnosis a left jugular paraganglioma was suggested, which was confirmed on MRI. The patient did not demonstrate radiological evidence or symptoms suggestive of a functional paraganglioma. As this paraganglioma was non-functional and only 1.3cm the decision, in conjunction with advice from an endocrinologist, was to continue current management and monitor on 12-monthly MRI scans. On routine follow up her lung and liver lesions remain stable on the increased dose of oral Imatinib and following the identification of a paraganglioma this confirmed the diagnosis of Carney’s Triad in this patient.
Discussion
First described in 1977 at the Mayo clinic by the pathologist J. Carney, Carney’s triad describes the association of three rare tumors: Gastrointestinal stromal tumor, pulmonary chondroma and extra-intestinal paraganglioma [1-3]. The presence of the triad at diagnosis is uncommon only found in 22% of patients [2,4]. The most common combination is gastric and pulmonary tumours [8]. Due to the rarity of the triad at presentation, Carney claimed that a person with only 2 tumors can be diagnosed with CT, which later coined the term ‘incomplete Carney triad’ [8]. CT has a predominance for young females, with a review from 2000 highlighting of the 104 cases in the literature 80% were identified in women, and most under the age of 30-years-old [8]. The most common clinical manifestation of CT is upper gastro-intestinal bleeding with associated signs and symptoms (E.g., anaemia, haematemesis and melaena), and is occasionally associated with abdominal pain [4,6,7]. As in this case, an asymptomatic abdominal mass discovered following investigation of another pathology has also been recorded in literature [4].
CT is accepted in the current literature as a genetic disorder rather than familial disease, as it is not passed on from generation to generation. Mosaicism and post-zygotic mechanisms have been implicated [8]. A marked reduction in enzymatic activity of SDH has been detected in tumours associated with CT, however the presence of gene mutations coding for the four SDH subunits (collectively known as the ‘SDHx’ genes) are very rare, and only recently described in the literature [4,8]. GIST is the most commonly observed component of Carney triad seen in 99% of cases, and responsible for the majority of initial presentations [4,8]. Gastric GISTs in CT occur at an early age, have intramural origin presenting typically in the antrum and lesser curvature, and have slow progression of disease [8]. All of these GIST characteristics were observed in this case. Traditional GISTs express specific activating mutations in c-kit (c-kit positive (CD117)) or platelet-derived growth factor receptor-A (PDGFA) receptor tyrosine kinases, which promotes susceptibility to targeted pharmacological inhibitors such as imatinib mesylate and sunitinib malate [4].
Non-surgical therapies for metastatic GISTs such as chemotherapy, radiation and thermo/cryo-ablation are usually ineffective. The role of imatinib mesylate in metastatic GISTs as a tyrosine kinase inhibitor is well known, however until recently its use in the management of gastric GISTs in CT was rarely reported. It has been postulated the poor response of gastric GISTs in CT to imatinib mesylate was due to the absence of c-kit and platelet derived growth factor receptor mutations, either of which occur in most sporadic GISTs [9]. In contrast our case of CT was strongly positive for c-kit (CD 117) and has been stable for 6 years on an increased dose of imatinib mesylate following identification of metastatic lesions. Gulavani et al. [9], like this case, has also identified a case of c-kit positive CT stabilised on daily imatinib mesylate.
Conclusion
Carney’s triad is a very rare condition, with a paucity of literature, so the presentation of the syndrome may differ from previous presentations described in the literature. This case demonstrates a further rare variant of Carney’s Triad with a c-kit positive GIST stable on oral targeted therapy with annual screening for progression of disease. This case highlights the importance of additional testing, such as lesion immunohistochemical analysis, for an accurate diagnosis when suspicious of a rare disease to guide the appropriate clinical course, especially when no clear treatment guidelines are available.
Ethical Approval
This study is exempt from ethical approval in my institution.
Consent
Authors have written and signed consent to publish a case report from this patient.
Ethical Approval
This study is exempt from ethical approval in my institution.
References
- Carney J, Sheps S, Go V, H Gordon (1977) The triad of gastric leiomyosarcoma, functioning extraadrenal paraganglioma and pulmonary chondroma. New Engl J Med 296(26): 1517-1518.
- Carney J (1999) Gastric stromal sarcoma, pulmonary chondroma, and extra-adrenal paraganglioma (Carney triad): natural history, adrenocortical component, and possible familial occurrence. Mayo Clin Proc 74: 543-552.
- Carney J (2013) Carney triad. Front Horm Res 41: 92-110.
- Alonso I, Pardo R (2021) Carney triad: A case report, characteristics, and literature review of this rare entity. International Journal of Surgery Case Reports 79: 14-17.
- Boikos SA, Xekouki P, Fumagalli E, Fabio R Faucz, Margarita Raygada, et al. (2016) Carney triad can be (rarely) associated with germline succinate dehydrogenase defects. European Journal of Human Genetics 24(4): 569-573.
- Chen C, Chuang C, Liu M, et al. (2010) Clinical, radiologic and pathologic characteristics of Carney triad: a case report and literature review. Kaohsiung J Med Sci 24(8): 428-434.
- Byrne C, Daneshjoo R, Wyatt J (2007) Gastrointestinal haemorrhage as a presentation of Carney’s triad. ANZ J Surg 77(1-2): 88-90.
- Shi G, Cui Y, He Y, Min Gong (2016) An unusual case of incomplete Carney triad: an 18-year-old girl suffering from multiple benign tumors. J Thorac Dis 8(10): E1202-E1206.
- Gulavani N, Patil A, Polavarupu K (2017) Carney’s triad. Journal of Case Reports 7(2): 192-195.






























