Characteristics and Outcomes in Patients with Upper Gastrointestinal Symptoms: A Cross-sectional Study at a Tertiary Care Hospital
Khaled Matar1*, Nour Ismail2, Iyad Abo Jazr1, Emad Al-Hout3, Mahmoud Al ShaikhAli2, Hassan Rosros1, Salah Al Shami2, Rami Salute2, Mohammed Hejazi1 and Ayman M H AbuHelal2*
1Department of Gastroenterology and Endosocpy, European Gaza Hospital, Palestine
2Department of Internal Medicine, European Gaza Hospital, Palestine
3Department of General Surgery, European Gaza Hospital, Palestine
Submission:January 01, 2023; Published:January 26, 2023
*Corresponding author:Khaled Matar, Department of Gastroenterology and Endosocpy, European Gaza Hospital, Gaza Strip, Palestine
How to cite this article:Khaled M, Nour I, Iyad Abo J, Emad Al-H, Salah Al S, et al. Characteristics and Outcomes in Patients with Gastrointestinal Symptoms: A Cross-sectional Study at a Tertiary Care Hospital. Adv Res Gastroentero Hepatol, 2023; 19(2): 556010. DOI: 10.19080/ARGH.2023.19.556010.
Abstract
Introduction: Upper gastrointestinal (UGI) symptoms are among the commonest complaints for which patients seek medical attention mainly the gastroenterology services. Association of patient demographics, clinical features with endoscopic outcomes are of paramount importance for better understanding and practice of endoscopy and to establish a local guideline guiding the practice of endoscopy including indications to perform upper endoscopy in patients with upper GI symptoms.
Aim: This study was to document the demographic characteristics, indications and endoscopic findings of patients undergoing UGIE at the endoscopic unit in European Gaza Hospital.
Materials and Methods: This study is a retrospective analytical study design. The study is descriptive and correlational study. Since the aim of the study is to identify the causes of UGIB, presenting complaint of patients and the findings of the gastroscopic procedure among patients undergoing gastroscopy at endoscopy unit in European Gaza Hospital, this is the most appropriate design for the study objectives. The study was conducted at the endoscopy unit in European Gaza Hospital. Data were collected retrospectively for the first three month of the year 2022 during the period from April 1st to April 15th, 2022. The study included all patients undergoing gastroscopy during the first three month of the year 2022.
Results: The study included 95 patients who were undergone gastroscopy and were admitted from either outpatient department or through the emergency department. The mean age of study participants was 45.03 + 17.51 years with median age of 47 years. The patients’ age ranged from 13 to 86 years. The study included 49 males and 46 females The most common symptom was dyspepsia (n= 28, 29.5%) followed by abdominal pain mainly epigastric (n= 25, 26.3%). Abdominal pain (mainly epigastric) was the most frequent indication (n= 35, 36.8%) followed by dyspepsia (n= 23, 24.2%). Active upper gastrointestinal bleeding was the indication among three cases (3%). The most common findings were gastritis, duodenitis and hiatus hernia.
Conclusion: The main indication for UGIE in the studied patients was dyspepsia and most of these patients had gastritis on endoscopy. Only few patients had normal findings.
Keywords: Upper gastrointestinal bleeding; Esophageal varices; Treitz ligament; Endoscopy
Abbreviations: UGI: Upper Gastrointestinal; UGIB: Upper Gastrointestinal Bleeding; PUD: Peptic Ulcer Disease; EGD: Esophagogastroduodenoscopy; EAC: Esophageal Adenocarcinoma; BE: Barrett’s Esophagus; LGIB: Lower Gastrointestinal Bleeding; GBS: Glasgow-Blatchford Score; PPI: Proton Pump Inhibitor; OBGIB: Obscure Gastrointestinal Bleeding; RS: Rockall Score; EVL: Endoscopic Variceal Ligation; MELD: Model for End-Stage Liver Disease; TIPS: Transjugular Intrahepatic Portosystemic Shunt; OTSC: Over-The-Scope Clip; ESGE: European Society of Gastrointestinal Endoscopy; ASA: American Society of Anesthesiology; ACG: American College of Gastroenterology; ASGE: American Society for Gastrointestinal Endoscopy; ESCC: Esophageal Squamous Cell Carcinoma; GERD: Gastroesophageal Reflux Disease; GEJ: Gastroesophageal Junction; AFI: Autofluorescence Imaging; CLE: Confocal Laser Endomicroscopy; IPCLs: Intrapapillary Hairlike Circles; WLE: White-Light Endoscopy; AA: Acidic Corrosive; FOBT: Fecal Occult Blood Test; TPB: Theory of Planned Behavior; PPI: Proton Pump Inhibitor
Background
Upper gastrointestinal bleeding (UGIB) is a major source of morbidity and mortality across the world, and acute UGIB is a common medical emergency addressed by gastroenterologists [1,2]. It has an annual incidence of 50-150/100,000 people and an overall death rate of 6-10%, which can reach up to 30% in Sub- Saharan Africa [3-5]. UGIB is four times more prevalent than lower GI bleed and occurs more frequently in men and the elderly [3,6,7].
Because of the disparities in therapeutic techniques and prognosis, UGIB is frequently categorized as variceal or nonvariceal [8]. Peptic ulcer disease (PUD) is the most prevalent cause of UGIB worldwide, accounting for roughly half of all cases, followed by esophageal varices [9,10]. However, research has revealed that esophageal varices are the most prevalent cause in Sub-Saharan Africa [4-11]. Gastritis, esophagitis, duodenitis, Mallory-Weiss tear, and upper GI cancers are other important causes of UGIB [12].
The identification of the etiology of UGIB is critical in patient care. Upper GI endoscopy is the recommended method of examining individuals with UGIB. It has the potential to be both diagnostic and therapeutic [13]. There have been few investigations on UGIB in Nigeria, and these studies have revealed a distinct pattern in the most prevalent cause across the Northern and Southern sections of the nation [14,15]. Prior research that revealed etiology of UGIB in Zaria occurred almost three decades ago [16]. As a result, there is a need to reassess the causes of UGIB in order to update current information for appropriate UGIB management in our environment. Upper Gastrointestinal Bleeding is described as bleeding that originates between the esophagus and the Treitz ligament [17]. It is one of the most common gastrointestinal crises, with a 10% fatality rate [18,19]. Despite breakthroughs in UGIB diagnosis and care, the death rate has remained stable over the previous 50 years [18-20].
Effective management necessitates the use of a riskstratification tool to divide patients into low- and high-risk groups, which can then be used to guide therapy and follow-up [19]. Various research has discovered a plethora of risk factors for death. Several risk ratings have also been established. The Rockall score, introduced in 1996, is the most widely used risk rating system. This score, which integrates the patient’s age, systolic blood pressure, heart rate, presence of additional comorbidities, and endoscopic results, estimates the probability of mortality following UGIB [19]. The Glasgow Blatchford Score [19] is another popular scoring system. Upper GI hemorrhage is still a major source of morbidity and mortality in Tanzania. In one retrospective trial, for example, 130 hospitalized patients with UGIB were included within two years [21]. Similarly, in another research in northern Tanzania, 18.7 percent of all patients who had a fiberoptic upper GI endoscopy were found to have UGIB [22]. In both investigations, the most prevalent causes were esophageal varices and peptic ulcer disease (PUD). According to studies conducted in Tanzania, the death rate of patients hospitalized with UGIB ranges between 10% and 17% [21-23].
Research Problem
Proper management and quick intervention are crucial in order to save life. On the other hand, majority of patients are undergoing diagnostic gastroscopy during their outpatient follow up. There is lack of research addressing the characteristics of patients undergoing gastroscopy. This study spotlight on the clinical characteristics of patients undergoing gastroscopy at endoscopy unit in European Gaza Hospital.
Objectives
a) To spotlight on the clinical characteristics of patients undergoing gastroscopy at endoscopy unit in European Gaza Hospital.
b) To determine the chief complaint of patients undergoing gastroscopy.
c) To address the findings of gastroscopy procedure among patients selected in this study.
d) To assess the relationship between patient’s clinical characteristics and the gastroscopy findings as well as their chief complaint.
Literature Review
Gastrointestinal endoscopy provides a significant clinical service, with a constant increase in demand [1]. Esophagogastroduodenoscopy (EGD) has emerged as a critical diagnostic method in the upper gastrointestinal (UGI) tract, primarily for the diagnosis and monitoring of premalignant diseases and malignancies. Endoscopy usage surged by more than 50% in the first decade of the new millennium, according to data from the United States (US), a trend that continues to this day. For example, it was projected that 6.9 million EGDs were conducted in the United States in 2009, at a cost of $12.3 billion (in an outpatient environment alone) [1]. More recent data from the United Kingdom (UK) have shown that over 1.2 million EGDs (both therapeutic and diagnostic) were performed in the UK in 2016 [2].
The utility of EGD correlates to many epidemiological challenges all over the world. EGD has not only become an established alternative to X-ray photofluorography as a firstline screening modality in high-risk locations for gastric cancer, such as Japan and South Korea, but is increasingly replacing it [3]. In the Western world, on the other hand, the rapidly rising prevalence of esophageal adenocarcinoma (EAC) has become a major epidemiological concern [4]. Because this cancer has an endoscopically visible (and perhaps treatable) precursor lesion - Barrett’s esophagus (BE) – screening for high-risk patients is suggested [5-7]. Clearly, the value of EGD is rising on a global scale, and with increased demand comes the necessity to assure the highest quality standard. In the realm of colonoscopy, the relevance of quality evaluation has previously been studied. Since its inception as a colorectal cancer screening test, a large amount of study has been devoted to creating quality measures. These are measurable parameters of individual performance that are usually related to procedural diagnostic yield (e.g., a positive correlation between colonoscopy withdrawal time and the rate of detected adenomas) and patient-oriented outcomes (e.g., an inverse relationship between adenoma detection rate and postcolonoscopy cancer incidence) [8-10].
Despite an increasing interest in quality measures for EGD, as seen by recently published recommendations and position statements [11-14], high-quality indicators for this operation remain unavailable. The majority of the measures offered are based on a lack of evidence and are rarely connected with patientoriented outcomes [11-14]. Given the high rate of missed UGI malignancies during endoscopy, 11.3 percent, according to a recent meta-analysis [15], the problem of EGD quality control is critical. GIB, along with abdominal discomfort, is one of the most prevalent disorders seen in the emergency room. Upper GIB (UGIB) is a severe condition that has been decreasing over the last 20 years but still has a 2.1 percent mortality rate [1]. Lower GIB (LGIB) has a mortality rate of less than 5%, however it is more prevalent in older patients, those with intestinal ischemia, and those with comorbidities [2].
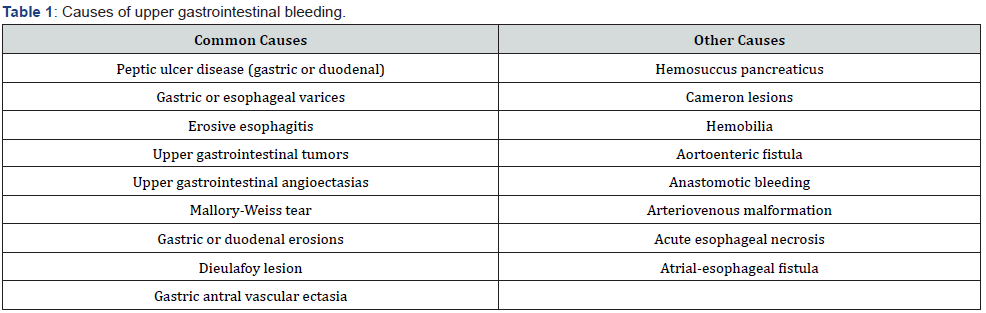
Hematemesis (vomiting of blood or coffee-ground-like substance), melena (black or tarry stools), and hematochezia are common symptoms of GIB. UGIB manifests as hematemesis in 40% to 50% of cases, and as melena or hematochezia in 90% to 98 percent of cases, particularly hematochezia in major UGIB [3]. Patients with LGIB, on the other hand, often present with hematochezia, however right-sided colonic or small intestinal bleeding may manifest as melena [4]. As a result, distinguishing between UGIB and LGIB is usually difficult based just on the patient’s early symptoms [5]. In true clinical practice, the patient with melena and hematochezia should be approached based on the major symptom rather than UGIB or LGIB. The physician in the emergency room normally performs early resuscitation of acute GIB, although highly competent endoscopists are required to establish the source and location of bleeding. Endoscopic therapy should be used in many situations to halt bleeding and avoid recurrence [6]. When endoscopy should be performed in individuals with suspected acute GIB is still debatable. It is very difficult to determine the etiology of UGIB from clinical signs alone. Table 1 summarizes the causes of UGIB by frequency.
Endoscopy should be conducted as soon as the patient is hemodynamically stable following adequate resuscitation when variceal hemorrhage is highly suspected [7]. Endoscopy should be done within 12 hours of the onset of acute variceal bleeding, according to the American Association for the Study of Liver Diseases recommendations [8]. Because gastric varix can be seen in more than half of patients with liver cirrhosis and varix hemorrhage has a 6-week mortality rate of roughly 20%, immediate endoscopy should be explored in patients with hematemesis and liver cirrhosis [7,9]. In the case of non-variceal UGIB, a previous Asia-Pacific Working Group consensus recommended that endoscopic intervention be performed within 24 hours of the onset of bleeding in patients who were at high risk of preendoscopic assessment (e.g., Glasgow-Blatchford Score (GBS) 12; The GBS is a composite score of blood urea, hemoglobin, systolic blood pressure, pulse, history, and comorbidities) [10]. This consensus is comparable to a recent cascade guideline published in 2018 by the European Society of Gastrointestinal Endoscopy, which stated that endoscopy should be conducted within 24 hours following acceptable first treatment [11]. As a result, it has been suggested that in patients who are hemodynamically unstable and have significant hematemesis, endoscopy should be delayed until the patient has been stabilized by resuscitation [12].
Several studies have looked into the role of emergency endoscopy within 12 hours in cases with non-variceal UGIB. A study of 361 patients indicated that patients who received emergency endoscopy had a 5-fold increase in the chance of adverse outcomes such as mortality, rebleeding, surgery, radiological intervention, or recurrent endoscopic treatment. Time to endoscopy was not a significant predictor of worse outcome in a subgroup analysis of that research, and it was a weaker prognostic effect in patients with a high GBS score (12 points) than in patients with a low score [13]. Because first bleeding symptoms are more severe in highrisk patients, they are more likely to get fluid treatment and fast proton pump inhibitor (PPI) medication prior to endoscopy. This emergency medical therapy may be the most important factor to prevent a poor outcome from UGIB regardless of time of endoscopy. A nationwide cohort study [14] of 12601 peptic ulcer patients found that patients with an American Society of Anesthesiology score of 3-5 points or who were hemodynamically unstable had a lower rate of hospital mortality if an endoscopic intervention was performed within 6 hours to 24 hours of admission [14]. However, the precise timing within the next 24 hours is unknown. Another national analysis in the United Kingdom, which included 4478 patients, advised an early endoscopy of less than 12 hours after admission and found no difference in mortality rate or requirement for surgery when compared to endoscopy performed within 24 hours [15]. According to Singapore cohort research, the timing of endoscopy in high-risk UGIB patients with GBS > 12 is the most important factor connected to hospital all-cause death [16]. The endoscopic cut-off time that enhanced survival was within 13 hours of the beginning of symptoms. Active hemorrhagic lesions requiring endoscopic hemostasis were common in patients who had endoscopy within 6 hours, but it did not aid with rebleeding prevention, death, transfusion rate, or hospitalization length. As a result, for any non-variceal UGIB, an emergency endoscopy is not necessary within 6 hours.
In summary, new guidelines and research imply that if variceal bleeding is evident or the patient is hemodynamically unstable, an emergency endoscopy should be performed within 12 hours [17]. Endoscopy should also be accompanied by appropriate and timely medical treatment, such as hydration therapy and intravenous PPIs [18-30]. Obscure gastrointestinal bleeding (OBGIB) is characterized as persistent or recurrent bleeding despite esophagogastroduodenoscopy or colonoscopy [30]. Overt bleeding with visible gastrointestinal hemorrhage, such as hematochezia or melena, and occult bleeding with recurring positive fecal occult blood test results or laboratory evidence of iron deficiency anemia are the two types of OBGIB [30]. This OBGIB accounts for around 5% of all gastrointestinal bleeding, with more than 80% of these bleedings occurring in the small intestine [31]. The introduction of capsule endoscopy has allowed for a comprehensive inspection of the small intestinal mucosa. Therapeutic endoscopy for these lesions is now possible because to device-assisted enteroscopy [32-34].
Treatment for individuals with suspected upper gastrointestinal bleeding, such as hematemesis or melena, may change depending on the cause of the bleeding. However, in all patients, vital signs, hemodynamic condition, and proper fluid management are critical [35]. If there is hypovolemic shock, fast pulse rate, high blood urea nitrogen level, reduced urine volume, or a history of severe bleeding at the time of presentation, more active early monitoring, fluid therapy, and blood transfusion treatment are required. However, if major bleeding is suspected, close monitoring and follow-up are required since early hemoglobin levels in acute bleeding may be normal [36]. When referred to the emergency department for upper gastrointestinal bleeding, the scoring system may be separated into two types: those that contain endoscopic findings and those that do not. In 1996, the Rockall score (RS) [37] was the most regularly used scoring system. This score method uses five parameters to predict the chance of mortality within 30 days: patient age, concomitant shock, co-morbidities such as heart, liver, and renal, bleeding causative disorders, and endoscopic bleeding stigmata. However, because the endoscopic results must be known, the pre-endoscopic RS, which may be computed using the three data except the origin of the bleeding and endoscopic findings, is utilized in practice. This is helpful in predicting rebleeding. This is useful for predicting rebleeding and mortality risk [38].
Furthermore, the Glasgow-Blatchford score (GBS), created in 1882, is extensively used to predict the requirement for transfusion, endoscopic therapy, rebleeding rate, and prognosis [39]. This scoring method, in particular, has the benefit of being able to test blood urea, hemoglobin, systolic blood pressure, pulse rate, presence of melena or syncope, liver illness, and heart failure in the emergency department swiftly and easily. AIMS65, a simplified scoring system that includes albumin, prothrombin time, mental state, systolic blood pressure, and age above 65 years, was recently presented. It is simple to remember, and it can be calculated objectively and quickly [40]. The mortality rate with AIMS65 scores ranged from 0.3 percent to 32 percent in one research [41]. The goal of the first treatment for patients with GI bleeding is to stabilize the hemodynamic circulation. To preserve blood vessel volume and hemodynamic stability, a big vein must be secured and checked to see if it is accompanied by heart, kidney, or liver problems [35]. Although the placement of a nasogastric tube is debatable, it can identify the requirement for rapid endoscopic hemostasis if blood is visible flowing from the upper gastrointestinal hemorrhage to the nasogastric tube. It should be noted, however, that a false negative owing to duodenal hemorrhage is possible [35]. One dosage of erythromycin provided 30 minutes to 120 minutes before endoscopy is not routinely suggested, although it is advised to enhance endoscopic visibility, minimize the need for transfusion and endoscopy, and shorten hospital stay [42,43].
Endoscopic procedures have improved the therapy of GIB, including peptic ulcer, variceal, diverticular, and angiodysplastic bleeding, in recent years. Furthermore, a growth in the availability of accessible and technologically sophisticated, well-trained endoscopy center-related professionals has resulted in early diagnosis by endoscopic intervention [44]. Injection (usually diluted epinephrine or a special sclerosing agent), contact and noncontact thermal devices (unipolar or bipolar electrocoagulation, heater probes, and argon plasma coagulation), and mechanical devices (endoscopic clips and band ligation) are all methods of endoscopic hemostasis for acute UGIB and LGIB [45]. Diluted epinephrine injections of 1:10000 to 1:20000 dilution aid in primary hemostasis of active bleeding; mechanical or thermal treatment to achieve certain hemostasis should be administered promptly as a secondary technique to limit the risk of rebleeding [4,46]. Endoscopic hemostatic effects in acute GIB are little studied in randomized studies. The etiology and location of the GIB, the ability to access the site, and the endoscopist’s experience all influence the manner of hemostasis used.
In the following, we will discuss the most clinically relevant methods of endoscopic treatment for the four major types of GIB and describe the most effective procedure.
Peptic Ulcer Bleeding
Peptic ulcer bleeding, which accounts for 30-60% of UGIB, has received the most attention. Although peptic ulcer bleeding classifications were developed a long time ago, an endoscopic classification known as the Forrest classification is generally utilized as a benchmark for endoscopic therapy. Most Forrest classification Ia to IIa lesions have a rebleeding rate more than 50%, requiring aggressive endoscopic treatment in the majority of patients [47,48].
However, the Forrest classification is over 40 years old; lately, [49] investigated if it is beneficial in predicting rebleeding and death from peptic ulcer bleeding and conducted research to see whether it might be simplified. They have condensed the Forrest categorization into three categories: high risk (Forrest Ia), increasing risk (Forrest Ib-IIc), and low risk (Forrest Ib-IIc) (Forrest III). In a total of 397 individuals, the risk of rebleeding was greatest (59 percent) in Forrest Ia peptic ulcers, although rates in Ib and IIc were comparable. Prediction of rebleeding using the Forrest classification is more reliable for gastric ulcers than for duodenal ulcers in subgroup analysis. Simplifying this categorization can minimize interobserver variability in diagnosing lesions, although it requires further research [49].
Injection therapy, heat therapy, and mechanical therapy have traditionally been employed as endoscopic therapeutic options for peptic ulcer bleeding. Several studies and meta-analyses have been conducted to determine if monotherapy or combination modality therapy is more successful. The Cochrane review of 19 RCTs involving 2033 patients published in 2014 found that the second bleeding control approach significantly decreased the risk of rebleeding and emergency surgery when compared to epinephrine injection treatment alone [50]. Mortality was also lowered, albeit the difference was not statistically significant. Other meta-analyses [51,52] found similar results. In research including 2888 patients published in 2016 [52], hemoclips alone or injection treatment coupled with heat therapy were more successful than injection therapy alone. As a result, it was determined that epinephrine injectable therapy should be used in combination with a secondary medication rather than as a stand-alone treatment. Adverse outcomes such as perforation and therapy-induced bleeding can occur following endoscopic treatment. They may be more prevalent in endoscopic therapy than in medical therapy alone, although a meta-analysis found no statistically significant difference (0.8% vs. 0.1%) [53]. High dosage PPI is considered to be an essential medicine to avoid rebleeding after endoscopic therapy for spurting bleeding or exposed vascular lesion, both of which are known to be highly rebleeding [54]. However, recent research found that the risk of rebleeding associated with Forrest Ib is much lower than that of Forrest IIa and IIb, and that high dosage IV PPI may not be required following successful endotherapy [55].
Variceal Bleeding
hemorrhage is a common and significant side effect of portal hypertension. Previous research has found that variceal hemorrhage in individuals with liver cirrhosis can result in a 50% mortality rate [56]. Although the use of vasoactive medications, endoscopic treatment, and preventive antibiotics has reduced mortality, esophageal varix hemorrhage is linked with a 20% death rate within 6 weeks [9]. Prior to endoscopic therapy for variceal bleeding, it is critical to stabilize patients and maintain an intravenous line for hemodynamic stability and a hemoglobin level of at least 7-8 g/dL by blood volume resuscitation [57]. Prophylactic antibiotics, such as intravenous quinolones or ceftriaxone, are also required and may minimize systemic bacterial infection and death [58]. Vasoactive drugs such as octreotide, somatostatin, and terlipressin are recommended to be administered as soon as possible [56].
The therapy of choice for esophageal variceal bleeding and secondary prevention is endoscopic variceal ligation (EVL). The appearance of bleeding varices, stigmata of recent bleeding including an adherent clot over varix or platelet plug called by white nipple marks, or the presence of varices without a definite active bleeding focus are used to make the diagnosis of variceal bleeding in the setting of active bleeding [59]. According to a recent meta-analysis of 1236 patients from 14 studies published by [60], EVL is superior to endoscopic injection sclerotherapy in terms of key outcome, including rebleeding, variceal eradication, and complication rate, but not in terms of mortality. As a result, EVL is the most effective first-line treatment for esophageal varix hemorrhage. Repeated endoscopy with EVL until varix elimination is advised after acute esophageal variceal bleeding, which generally requires 2 to 4 sessions of treatment [61]. The ideal interval of each EVL for secondary prevention has not been identified, however in trials examining repeated EVLs for secondary prevention, it typically varies from 2 wk to 8 wk. As an EVL consequence, post-EVL band-induced ulcer bleeding may develop. Another research [62] found that while the incidence was just 2.8 percent, it was substantially related to death. A high MELD score (MELD stands for Model for End-Stage Liver Disease and is computed using blood bilirubin, prothrombin time, and serum creatinine) was linked to an increased risk of band-induced ulcer bleeding [62]. Band-induced ulcer bleeding can be treated with a transjugular intrahepatic portosystemic shunt (TIPS) or sclerotherapy, and pantoprazole for 10 days can reduce ulcer size [63]. Furthermore, rebleeding from band ulcers can be treated with hemostatic power or a spray similar to that used to treat peptic ulcer bleeding [64,65]. Recently, a study by [66] showed that immediate application of hemostatic powder is effective for early clinical course and endoscopic hemostasis in patients with acute initial variceal bleeding.
In addition, despite the most effective EVL therapy, we might explore alternative therapeutic option, such as esophageal balloon tamponade, in patients with recurrent or refractory variceal bleeding. The esophageal stent, which was previously utilized primarily for luminal GI stenosis, has been employed in place of the balloon in refractory variceal bleeding, with a statistically significant rate of therapeutic success and bleeding control [67]. TIPS therapy, as previously stated, is utilized for recurrent and refractory variceal bleeding in patients who meet high-risk criteria (Child-Pugh B plus active bleeding at endoscopy or Child-Pugh C). However, early TIPS for recurrent variceal bleeding in a Child- Pugh B patient may hasten the development of acute-on-chronic liver failure and/or mortality. The careful decision of patients for TIPS is essential and other parameters should be considered, such as systemic inflammation, non-selective beta blocker-nonresponse and portal vein thrombosis [68]. Various endoscopic therapies have been attempted in cases of failure of hemostasis due to general endoscopic treatment [69-78]. The OTSC (Overthe- scope clip, Ovesco AG, Tübingen, Germany) system, which is inserted at the upper end of the endoscope, has been widely used in fistulas and perforations [79-84]. However, it can be used in cases of continuous hemorrhage due to local injection or clipping [85]. In addition, the hemostasis of the bleeding site through the nano powder (Hemospray, Cook Medical, Winston-Salem, NC, USA) or starch (EndoClot Plus Inc., Santa Clara, CA, USA) can be used when other hemostasis is not treated. Previously, it was difficult to distribute the powder or starch materials to the hemorrhagic lesion, but in recent years, disposable powder roots have been developed and can be used more easily [86,87]. Endoscopic findings of GIB are observed, and successful hemostasis can improve the prognosis and increase the survival rate. However, endoscopic hemostasis is not always successful. If we know the factors that are likely to fail in endoscopic hemostasis, decisions can be made to change the modality [88-95].
Endoscopic therapy of peptic ulcer bleeding was found to be 94% successful in a prior publication [95], followed by permanent hemostasis without rebleeding (82.5 percent). Endoscopic treatment failure or rebleeding occurred in 17.5% of endoscopic procedures. Patients with active bleeding at the time of diagnosis, shock at admission, or low hemoglobin levels had considerably higher rates. The medication history, age, and location of the stomach ulcer, on the other hand, had no influence on the failure rate of endoscopic bleeding management. Another major trial including injection and heat treatment of 3386 patients with peptic ulcer bleeding found that 98.6 percent had good initial hemostasis, although 8.2 percent rebled. As a result, the overall failure rate of bleeding control was 9.6 percent. Endoscopic bleeding control failed substantially more frequently when blood pressure was low, hemoglobin was less than 10g/dL, new blood was visible in the stomach, and big or active ulcers were present [96]. In one research, 427 patients were endoscopically treated, with a failure rate of 20.1%. Endoscopic observations of spurting hemorrhage and a duodenal ulcer on the posterior side or anastomosis site indicated that therapy had failed. In summary, indicators of serious bleeding, such as shock, a low hemoglobin level, new blood at the moment of presentation, or an ulcer with a big surface area and spurting blood, might result in the endoscopic treatment failing.
Portal hypertension causes variceal hemorrhage. The most critical factor is the hepatic venous pressure gradient, which reflects portal pressure. Furthermore, the severity of liver illness as measured by the Child-Pugh class or MELD, encephalopathy, platelet count, history of drunkenness, and presence of portal vein thrombosis were revealed to be independent predictors of endoscopic variceal bleeding control failure [97-99]. As a result, in high-risk patients with a high likelihood of bleeding control failure, a preemptive TIPS should be prepared, and tamponade ballooning should be administered momentarily following endoscopic hemostasis retrials [100]. In contrast to other types of bleeding, variceal hemorrhage should be thought to be more impacted by the degree of liver cirrhosis than by endoscopic findings [100]. The pattern adopted to produce initial hemostasis is the predictor of LGIB recurrence. Primary hemostatic modality [100]; usage of antiplatelet, anticoagulant, and NSAIDs; presence of chronic renal disease or liver cirrhosis; and cause of first bleeding [4] are risk factors for recurrent LGIB. It is unclear how these individual factors’ proportionate effect may affect the incidence of recurrent bleeding. In a retrospective examination of 171 severe LGIB patients [100], the three most prevalent causes of bleeding were diverticular hemorrhage, anorectal disorders, and angiodysplasia. In particular, 15% of the individuals had previously been treated with antiplatelet medicines and 9% with anticoagulants. Approximately one-third of the individuals developed recurrent LGIB throughout the average follow-up period of 11 years. As noted in previous studies, LGIB is more likely to rebleed and affects older patients taking several medications. One of the most important points in diverticular bleeding and angiodysplasias bleeding, which comprise a large part of LGIB, is that they can stop spontaneously and rebleed at the same site or in other lesions.
Individuals having endoscopy should be well educated about the technique, its indications, the dangers it entails, and how to handle any consequences. According to the European Society of Gastrointestinal Endoscopy (ESGE), correct fasting instructions are part of patient preparation [12]. Based on existing evidence, ESGE defines proper fasting period as at least 2 hours for liquids and 6 hours for solids [12]. Patients with known delayed stomach emptying (gastroparesis) may benefit from a longer time of fasting (e.g. overnight fasting) on a case-by-case basis, notwithstanding the recommendations [16]. Before EGD, patients should be given an easily comprehensible take-home information sheet on the technique, and, unless the surgery is an emergency, informed signed consent should be obtained. An evaluation of the patient’s health state is also part of the procedure’s preparation. Diabetes control, anticoagulant medication, the necessity for prophylactic antibiotics, and sedation needs are the most prevalent clinical concerns requiring consideration before to endoscopy. The American Society of Anaesthesiology (ASA) score is the most widely used grading system for categorizing patients based on their physiological condition in order to anticipate surgical risk [17].
Before doing any endoscopic examination, it is critical to review the endoscopy referral form as well as the reasons for the treatment. Endoscopists should be conversant with the standard EGD indication list and should personally assess any deviations from it. The comprehensive list of indications is beyond the scope of this paper, but it may be found in prior documents, such as the quality guidelines of the American College of Gastroenterology (ACG) and the American Society for Gastrointestinal Endoscopy (ASGE) [13,22]. According to ACG/ASGE, indications for EGD must be documented and correspond to the published standard list in at least 80% of instances [23].
Furthermore, assessing the indication and relevant clinical data allows for the identification of individuals at higher risk of UGI malignancies. In such cases, a lower threshold for biopsying suspected lesions should be explored, and suitable imageenhancing modalities should be planned ahead of time. The Asian agreement highlights the importance of risk assessment for patients prior to any diagnostic EGD [14]. Patients of the male gender, for example, who have a history of past head and neck cancer as well as a known history of heavy alcohol intake or cigarette smoking, are at an elevated risk of esophageal squamous cell carcinoma (ESCC). These individuals should have a thorough examination of the whole length of their esophagus, ideally using sophisticated imaging methods. On the other hand, obese men of Caucasian origin with a long-standing history of gastroesophageal reflux disease (GERD) are at increased risk of BE-related EAC and require a meticulous inspection of the gastroesophageal junction (GEJ) and BE segment, if present.
Although EGD is a less technically difficult operation than colonoscopy, the range of mucosal alterations seen during EGD is substantially broader since it encompasses the assessment of three separate organs and epithelia in a single examination. In recent years, advanced imaging methods have emerged with the goal of improving mucosal visibility and fine structural and microvascular architecture [48].
The ongoing range of laid out picture improving (IE) methods incorporate ordinary chromoendoscopy, restricted range endoscopy, for example, tight band imaging (NBI; Olympus Medical Systems, Tokyo, Japan) and blue laser imaging (BLI; Fujifilm, Tokyo, Japan), yet in addition a few different frameworks, for example, connected variety imaging (LCI; Fujifilm, Tokyo, Japan), I-Scan advanced contrast (I-SCAN; Pentax, Tokyo, Japan), autofluorescence imaging (AFI), and confocal laser endomicroscopy (CLE). These advances are at an alternate degree of clinical application and won’t be entirely examined inside this article; be that as it may, not many of them play a laid-out part in routine practice and portray an excellent method [48]. In the throat, both Lugol staining and restricted range endoscopy have been displayed to further develop location and portrayal of squamous neoplasia [49-51]. Lugol responds with glycogen, which is plentiful in typical squamous epithelium, however altogether diminished or missing in dysplastic tissue. Consequently, ordinary regions are strongly stained by the Lugol color to an earthy colored tone, though dysplastic regions have diminished or totally missing staining. In NBI imaging, then again, squamous neoplasia shows up as dull earthy colored patches on the esophageal mucosa. NBI, combined with amplification, can be very useful in deciding the possible profundity of attack by evaluating the morphology of intrapapillary hairlike circles (IPCLs) [52]. Both BSG and ESGE suggest the utilization of Lugol’s chromoendoscopy if squamous neoplasia is thought, [11,12] which alludes for the most part to patients recently treated with remedial plan for an essential head and neck malignant growth.
IE strategies are utilized broadly in the reconnaissance of BE, albeit (up until this point) not a solitary one of them have been shown convincingly to be better than standard superior quality white-light endoscopy (WLE) [5,7-53]. Acidic corrosive (AA) is a normally utilized color that initiates the “acetowhitening” impact on BE mucosa, it its surface to feature. One review study showed a better neoplasia recognition rate with AA-directed biopsies when contrasted and ordinary convention directed planning biopsies [54]. A randomized get over preliminary looking at AA-directed biopsies versus non-designated biopsies according to Seattle convention is in progress (the ABBA study), and the outcomes might give more conclusive proof to advance this procedure overflow standard of care [55,56].
For NBI, a basic approved framework to distinguish dysplasia and EAC inside BE sections has been fostered [Barrett’s global NBI bunch (BING) criteria] [57]. This grouping depends on a straightforward assessment of the BE surface and vascular examples (ordinary versus unpredictable). The BING rules had a general precision of 85% in recognizing patients with dysplasia, which expanded to 92% with high certainty expectations [57]. In the stomach, virtual chromoendoscopy has been applied broadly in the observation of premalignant conditions. NBI, specifically, can be useful in featuring AG and discrete areas of GIM. The refreshed ESGE rules on the administration of epithelial precancerous circumstances and injuries in the stomach (MAPS II) suggest that virtual chromoendoscopy (±magnification) ought to be utilized for the conclusion of the premalignant stomach by directing biopsy for arranging atrophic and metaplastic changes and by aiding objective neoplastic sores [58]. LCI, then again, is an arising imaging methodology with developing utility in the UGI plot. LCI separates the red variety range, and builds the differentiation among aroused and typical regions; LCI is useful in exact identification and outline of neoplastic sores, especially inside gastric mucosa. In a new report, LCI was shown more viable for acknowledgment of early gastric disease contrasted and WLE [59]. Additionally, past investigations have shown that LCI can be useful in the ongoing finding of dynamic Helicobacter pylori disease [60].
The side effects of draining in the GI parcel that are experienced in genuinely clinical practice are basically melena, hematemesis, and hematochezia. Whenever a patient with these side effects presents to the trauma center, endoscopic finding and treatment are viewed as along with fitting beginning revival. For better forecast in instances of thought variceal dying, it is central that endoscopy is performed following the patient is balanced out, and it would be adequately compelling for endoscopy to be embraced inside 24h from side effect improvement for non-variceal UGIB. In instances of thought LGIB, sigmoidoscopy might be at first performed assuming there is a solid doubt of anorectal dying.
Notwithstanding, overall, full colonoscopy after gut planning is compelling for recognizing the reason and area of draining and treating with hemostasis. Taking everything into account, the job of endoscopy in GIB is vital, and numerous rules have been created about endoscopic treatment for explicit draining infections. In any case, there are still parts that poor person been laid out. Particularly, further examinations on prophylactic intubated endoscopy, schedule second-look endoscopy and crisis case endoscopy issues are required.
Methodology
Study design
This study is a retrospective analytical study design. The study is descriptive and correlational study. Since the aim of the study is to identify the causes of UGIB, presenting complaint of patients and the findings of the gastroscopic procedure among patients undergoing gastroscopy at endoscopy unit in European Gaza Hospital, this is the most appropriate design for the study objectives.
Study setting
The study was conducted at the endoscopy unit in European Gaza Hospital. Data were collected retrospectively for the first three month of the year 2022 during the period from April 1st to April 15th, 2022.
Participants and sampling
The study included all patients undergoing gastroscopy during the first three month of the year 2022. The study data were collected from medical records. However, there are certain exclusion criteria:
a) Patients undergoing double scope (gastroscopy and coroscopy).
b) Patients undergoing colonoscopy alone.
c) Patients undergoing gastroscopy during previous years.
The flow chart in figure 1 is showing the process of selecting patients for this study.
Data collection
Data were collected from medical records retrospectively for the first three months of the year 2022 using a self-designed questionnaire based on most recent published literature.
Instruments
The study tool is a self-designed questionnaire based on most recent published literature. The questionnaire included basic information about the patients, gastroscopy findings and laboratory work done for the patients.
Statistical analysis
Data obtained from questionnaire were entered and analyzed using SPSS program version 23 computer software. Sociodemographic data were presented using descriptive statistics as means, median, percentages and standard deviation. Independent T test and one-way Anova are used to show statistical significance among patients’ characteristics and tool scores. Chi square test is used to show relationship between categorical variables.
Permission and ethical considerations
An approved permission was gained from Helsinki Committee at the MoH in Gaza Strip to collect data from patients’ medical records.
Results
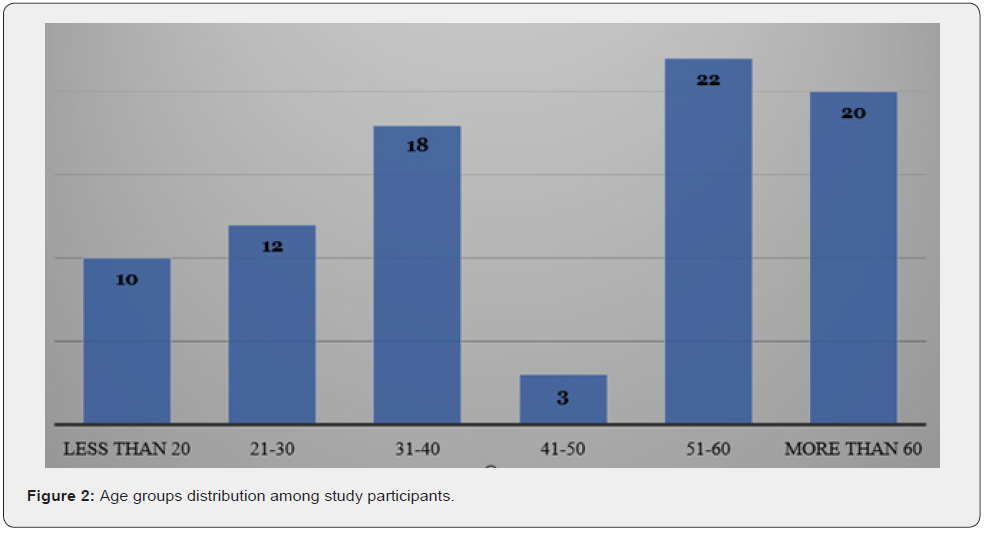
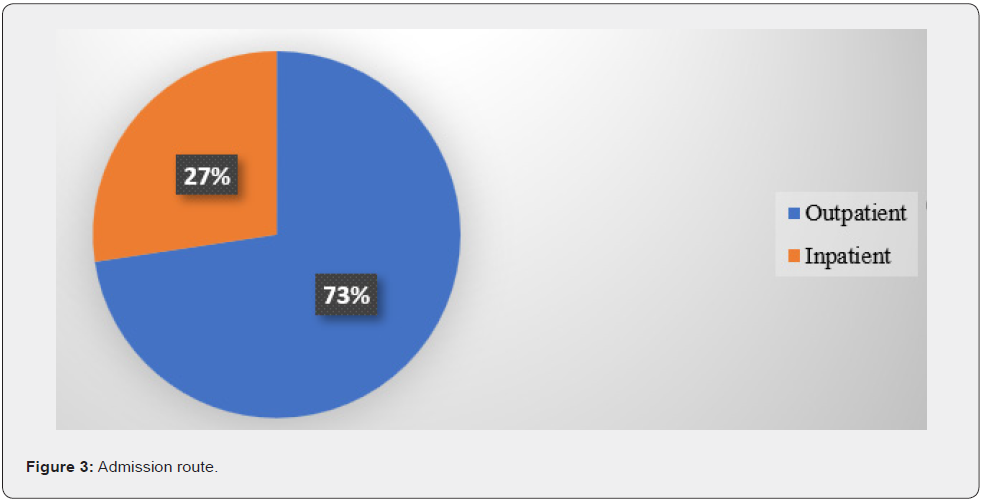
The study included 95 patients who were undergone gastroscopy and were admitted from either outpatient department or through the emergency department. The mean age of study participants was 45.03 + 17.51 years with median age of 47 years. The patients’ age ranged from 13 to 86 years. Age groups distribution is presented in figure 2. As shown, the peak age group is at older population. The male to female ratio was close; the study included 49 males and 46 females. The route of admission is presented in figure 3 (outpatient, n- 69, 73%).
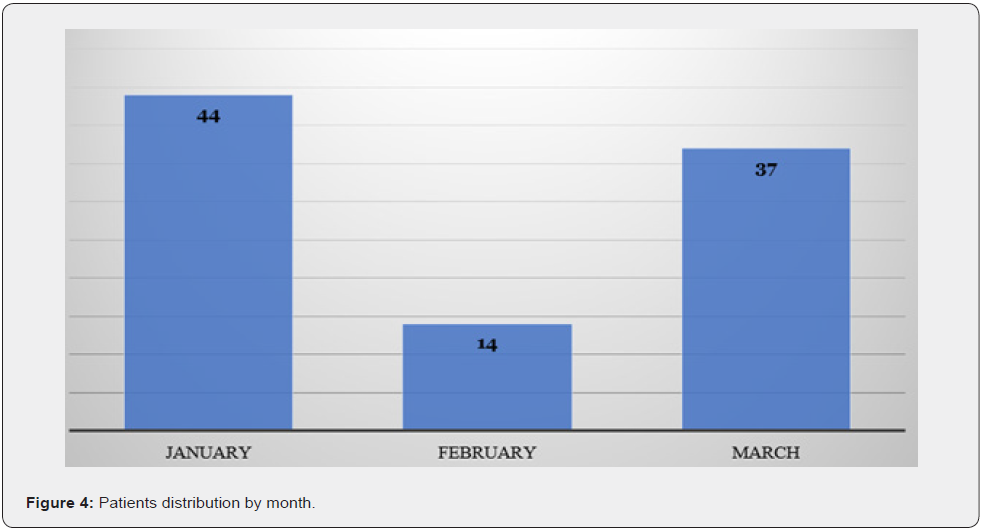
Since this study was conducted on patients during the period of the first three months of the year 2022, the month with most procedures was January (n= 44, 46.3%) (Figure 4). Patients suffered from various comorbidities. Some patients suffered from more than one comorbidity. Some of these comorbidities were related to the gastrointestinal system and others were not. Hypertension was prevalent among 17 patients while diabetes mellitus among 8 patients. Gastrointestinal related comorbidities were hepatitis B among 4 patients, liver cirrhosis among 4 patients, peptic ulcer disease among 2 patients and colon cancer among 1 patient. Other comorbidities included ischemic heart disease and rheumatic diseases. On the other hand, there was very poor documentation regarding the medications patients take for their chronic conditions.
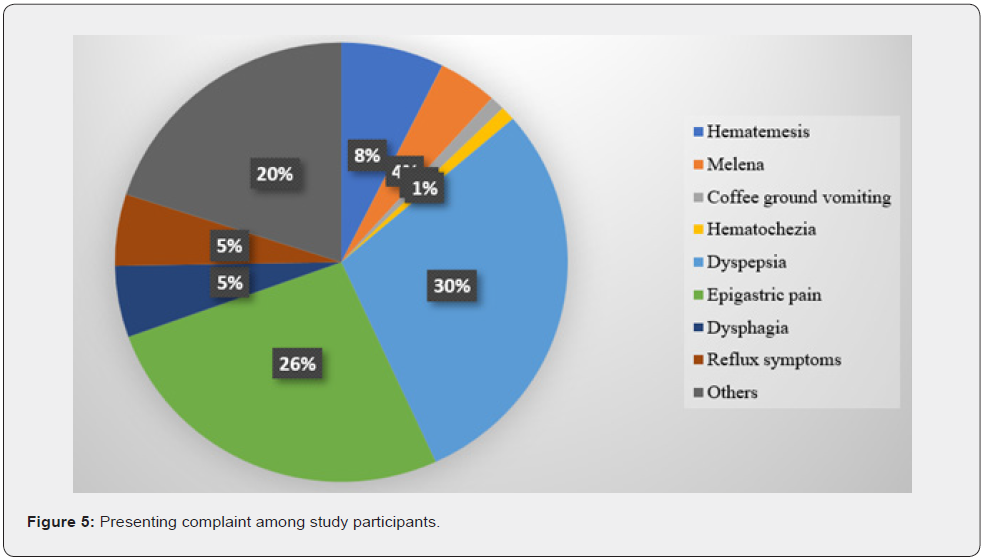
The presenting symptom that led to the decision for gastroscopy varied among study participants. The most common symptom was dyspepsia (n= 28, 29.5%) followed by abdominal pain mainly epigastric (n= 25, 26.3%). Figure 5 shows the frequency of presenting complaint among study participants. Other symptoms included postprandial disorder, heart burn, stoke for percutaneous endoscopic gastrostomy insertion (PEG) and chest pain. The indication for gastroscopy was almost the same as the presenting symptom. Abdominal pain (mainly epigastric) was the most frequent indication (n= 35, 36.8%) followed by dyspepsia (n= 23, 24.2%). Active upper gastrointestinal bleeding was the indication among three cases (3%). The anesthetic agent used for the procedure was mainly midazolam (87, 91.6%).
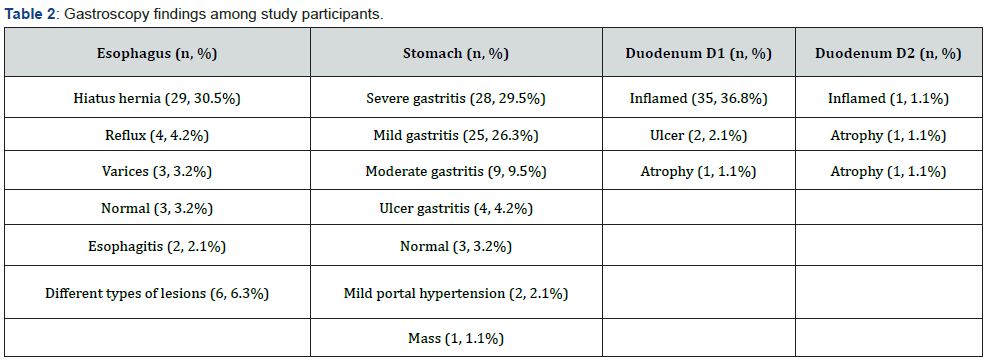
Other agents used were propofol and Ezra Cain. Findings of gastroscopy procedure was divided according to the examined part. Table 2 shows the findings of gastroscopy procedure among study participants. Biopsy was taken during 12 procedures only (12.6%). The site of biopsy varied among study participants. The most common site was the second portion of duodenum (n= 7) followed by gastric biopsy (n= 4) and one biopsy from first portion of duodenum. Gastroscopy procedure was diagnostic among most of cases (n= 88, 92.6%). However, there were two unstable cases in which band ligation was done for them. Other interventions performed during the gastroscopy were PEG insertion among two cases and PEG tube exchange among three cases.
Discussion
Gastrointestinal disorders, specifically dyspepsia, are normal issues inside essential consideration around the world, [1-3] as well as in Greece [4,5]. Exploratory proof on dyspepsia the board is scant and rules depend on data drawn from preliminaries and clinical investigations led in scholar or expert settings. It affects general practices might be prominently basic to refute the execution interaction [6]. Current rules propose that all patients with dyspepsia north of 45 or 55 years old or those with side effects ought to go through brief esophagogastroduodenoscopy (EGD) [7]. In any case, an effective execution of proposals that remember obtrusive evaluating for regular essential consideration practice is by all accounts connected with elements, for example, the specialist patient relationship and the patient’s consistence to the specialist’s suggestions [8,9].
For instance, factors like family ancestry, saw risk, self-viability, information on the sickness, or the utilization of instructive recorded material, were not demonstrated to impact patients’ choice about colorectal disease (CRC) screening with a Fecal Occult Blood Test (FOBT) [8]. Then again, inside essential consideration, consistence to colonoscopy and FOBT has been exhibited to increment by the straightforward utilization of customized empowering pamphlets [9,10]. In this manner, making sense of and changing patients’ perspectives to acquire higher consistence rates requires additionally an exhaustive information on the elements that might impact their dynamic cycle. Accentuation is given presently on quiet focused correspondence and shared dynamic that appear to prompt a critical expansion in tolerant information, work on personal satisfaction and patient’s fulfillment towards clinical consideration, and furthermore to lessen the nervousness and decisional struggle [10,11]. The human dynamic interaction has been investigated completely during the previous many years and models have been fostered that could make sense of the consistence of the patients towards the specialists’ suggestions [11-13]. Different hypotheses towards understanding and altering human way of behaving have been applied and among them the Theory of Planned Behavior (TPB) has been built [14]. A PhD concentrate on that focused on evaluating for upper gastrointestinal side effects in an essential consideration populace was planned and executed into two Greek areas. Patients who visited chosen country rehearses were evaluated; the individuals who were positive for upper gastrointestinal side effects were alluded for upper endoscopy.
This study expected to report the segment qualities, signs and endoscopic discoveries of patients going through UGIE at the endoscopy unit in European Gaza Hospital. This study addresses the very first report on UGIE signs. Dyspepsia was the commonest sign for upper GI endoscopy in by far most of our members. This is like investigations directed in Ghana and other West African and East African nations [12,14-17]. Different explanations behind UGIE among our patients were side effects of UGI dying, evaluating for esophageal varices in cirrhotic patients and repetitive heaving. Just 1.1% of our patients went through upper GI endoscopy for dysphagia which is like 1.0% detailed by concentrate on done in Kumasi, Ghana [12]. This contrasts from a review done in Malawi [18] which announced dysphagia as the most widely recognized sign for UGIE. 37% (37%) of their patients had dysphagia as a sign for UGIE. The high commonness of esophageal malignant growth in Malawi might represent this distinction [19]. Additionally, endoscopy administrations are confined in Malawi and as such just patients with caution side effects are alluded for upper GI endoscopy [18].
Gastritis was the most successive endoscopic finding in our patients, trailed by duodenitis. This is equivalent to past Ghanaian examinations [3,10], which detailed gastritis and duodenitis as normal endoscopic discoveries among their patients. Gastric ulcer was analyzed more much of the time than duodenal ulcers among our patient populace [12] in Kumasi. This is as opposed to the discoveries of one review from Accra that detailed more duodenal ulcers than gastric ulcers [3]. Gastric holes were more normal than duodenal holes among the Kumasi populace as indicated [20]. They additionally noticed that patients giving gastric holes were more successive clients and victimizers of NSAIDS and natural medications or inventions [20]. The level of esophageal varices recognized in this study was more than past investigations distributed in this nation [12]. This is on the grounds that as a feature of the signs for endoscopy, patients with liver cirrhosis without draining were alluded for endoscopy in this review. Typical discoveries in this study were far lower than before concentrates on that have been distributed in this nation [3,12]. The distinction may likely be because of worked on endoscopic procedures in distinguishing UGI pathology or further develop techniques in clinical determination over the course of the ten years or might be because of scant accessibility of endoscopy benefits so individuals are alluded fittingly for endoscopy. The utilization of Proton Pump Inhibitor (PPI) and NSAID could likewise alter the discoveries of endoscopy and data about this was not accessible in every one of the examinations.
H. pylori colonization of the stomach is quite possibly the most widely recognized disease worldwide. A few scientists portrayed it as the most widely recognized ongoing human bacterial contamination [21,22]. It is the fundamental driver of constant gastritis and the primary etiological specialist of gastric malignant growth and peptic ulcer illness. In numerous nations, the rate of H. pylori has been diminishing in relationship with worked on way of life and further developed omen anti-microbial. The predominance of H. pylori in this study was 44.9%. This is practically identical to 45.2% announced [13], however rather than 74.8% [23], in the country. Other past investigations in Ghana [24], Nigeria [25] and other agricultural nations [26] have additionally announced high pervasiveness of H. pylori. Potential explanations behind this distinction might be the rising compelling annihilation treatment of the disease with anti-microbial mix and proton siphon inhibitors (PPI) and furthermore the far and wide and unpredictable utilization of anti-microbial and PPI. This study did not reject patients who were at that point on anti-infection agents and PPI or have consumed these medications preceding the review. It might likewise be related with further developed sterilization among the occupants [27].
Despite the lessening in predominance of H. pylori among patients in this review, the ongoing predominance of 45.2% is still high contrasted with rates in created nations [28]. The commonness of H. Pylori disease is related with lower financial status, sterilization, fundamental cleanliness, terrible eating routine; stuffing, nationality, orientation and mature, low degrees of training and geographic area likewise assume a significant part in the dispersion of the contamination [29,30]. This might make sense of the greater predominance of H. Pylori in agricultural nations.
Conclusion
The commonest indication for UGIE in the studied patients was dyspepsia and most of these patients had gastritis on endoscopy. Only few patients had normal findings. Gastric ulcers were commoner than duodenal ulcers in this patient population. The outcomes of this study have implications for policy and planning. There is a need to identify the common causes of dyspepsia/ gastritis in the community. This will help formulate and put in place community-based interventions including education to avoid these precipitating factors. Patients with dyspepsia with no warning symptoms should undergo endoscopy, particularly males, patients with body mass index > 25, epigastric pain. Gastroscopy has significant implications in dyspeptic patients, especially for those with independent risk factors. Therefore, gastroscopy should not be performed based on warning symptoms exclusively.
References
- Abougergi MS, Travis AC, Saltzman JR (2015) The in-hospital mortality rate for upper GI hemorrhage has decreased over 2 decades in the United States: a nationwide analysis. Gastrointest Endosc 81(4): 882-888.
- Strate LL, Ayanian JZ, Kotler G, Syngal S (2008) Risk factors for mortality in lower intestinal bleeding. Clin Gastroenterol Hepatol 6(9): 1004-1010.
- Fallah MA, Prakash C, Edmundowicz S (2000) Acute gastrointestinal bleeding. Med Clin North Am 84(5): 1183-1208.
- Strate LL, Gralnek IM (2016) ACG Clinical Guideline: Management of Patients with Acute Lower Gastrointestinal Bleeding. Am J Gastroenterol 111(4): 459-474.
- Wilcox CM, Alexander LN, Cotsonis G (2007) A prospective characterization of upper gastrointestinal hemorrhage presenting with hematochezia. Am J Gastroenterol 92(2): 231-235.
- Kumar NL, Travis AC, Saltzman JR (2016) Initial management and timing of endoscopy in nonvariceal upper GI bleeding. Gastrointest Endosc 84(1): 10-17.
- Kapoor A, Dharel N, Sanyal AJ (2015) Endoscopic Diagnosis and Therapy in Gastroesophageal Variceal Bleeding. Gastrointest Endosc Clin N Am 25(3): 491-507.
- Garcia-Tsao G, Abraldes JG, Berzigotti A, Bosch J (2017) Portal hypertensive bleeding in cirrhosis: Risk stratification, diagnosis, and management: 2016 practice guidance by the American Association for the study of liver diseases. Hepatology 65(1): 310-335.
- R de Franchis, Primignani M (2001) Natural history of portal hypertension in patients with cirrhosis. Clin Liver Dis 5(3): 645-663.
- Sung JJ, Chan FK, Chen M, Ching JY, Ho KY, et al. (2011) Asia-Pacific Working Group. Asia-Pacific Working Group consensus on non-variceal upper gastrointestinal bleeding. Gut 67(10): 1170-1177.
- Karstensen JG, Ebigbo A, Aabakken L, Dinis-Ribeiro M, Gralnek I, et al. (2018) Nonvariceal upper gastrointestinal hemorrhage: European Society of Gastrointestinal Endoscopy (ESGE) Cascade Guideline. Endosc Int Open 6(10): E1256-E1263.
- Cipolletta L, Cipolletta F, Granata A, Ligresti D, Barresi L, et al. (2018) What is the Best Endoscopic Strategy in Acute Non-variceal Gastrointestinal Bleeding? Curr Treat Options Gastroenterol 16(4): 363-375.
- Kumar NL, Cohen AJ, Nayor J, Claggett BL, Saltzman JR (2017) Timing of upper endoscopy influences outcomes in patients with acute nonvariceal upper GI bleeding. Gastrointest Endosc 85(5): 945-952.
- Laursen SB, Leontiadis GI, Stanley AJ, Møller MH, Hansen JM, et al. (2017) Relationship between timing of endoscopy and mortality in patients with peptic ulcer bleeding: a nationwide cohort study. Gastrointest Endosc 85(5): 936-944.
- Jairath V, Kahan BC, Logan RF, Hearnshaw SA, Doré CJ, et al. (2012) Outcomes following acute nonvariceal upper gastrointestinal bleeding in relation to time to endoscopy: results from a nationwide study. Endoscopy 44(8):723-730.
- Lim LG, Ho KY, Chan YH, Teoh PL, Khor CJ, aet al. (2011) Urgent endoscopy is associated with lower mortality in high-risk but not low-risk nonvariceal upper gastrointestinal bleeding. Endoscopy 43(4): 300-306.
- Sung JJ, Chiu PW, Chan FKL, Lau JY, Goh KL, et al. (2018) Asia-Pacific working group consensus on non-variceal upper gastrointestinal bleeding: an update 2018. Gut 67(10): 1757-1768.
- Korkis AM, McDougall CJ (2005) Rectal bleeding in patients less than 50 years of age. Dig Dis Sci 40(7): 1520-1523.
- Angtuaco TL, Reddy SK, Drapkin S, Harrell LE, Howden CW (2001) The utility of urgent colonoscopy in the evaluation of acute lower gastrointestinal tract bleeding: a 2-year experience from a single center. Am J Gastroenterol 96(6): 1782-1785.
- Lhewa DY, Strate LL (2012) Pros and cons of colonoscopy in management of acute lower gastrointestinal bleeding. World J Gastroenterol 18(11): 1185-1190.
- Seth A, Khan MA, Nollan R, Gupta D, Kamal S, et al. (2017) Does Urgent Colonoscopy Improve Outcomes in the Management of Lower Gastrointestinal Bleeding? Am J Med Sci 353(3): 298-306.
- Kouanda AM, Somsouk M, Sewell JL, Day LW (2017) Urgent colonoscopy in patients with lower GI bleeding: a systematic review and meta-analysis. Gastrointest Endosc 86(1): 107-117.
- Sengupta N, Tapper EB, Feuerstein JD (2017) Early Versus Delayed Colonoscopy in Hospitalized Patients With Lower Gastrointestinal Bleeding: A Meta-Analysis. J Clin Gastroenterol 51(4): 352-359.
- Jensen DM, Machicado GA, Jutabha R, Kovacs TO (2000) Urgent colonoscopy for the diagnosis and treatment of severe diverticular hemorrhage. N Engl J Med 342(2): 78-82.
- Wong Kee Song LM, Baron TH (2008) Endoscopic management of acute lower gastrointestinal bleeding. Am J Gastroenterol 103(8): 1881-1887.
- Laine L, Shah A (2010) Randomized trial of urgent vs. elective colonoscopy in patients hospitalized with lower GI bleeding. Am J Gastroenterol 105(12): 2636-2641.
- Green BT, Rockey DC, Portwood G, Tarnasky PR, Guarisco S, et al. (2005) Urgent colonoscopy for evaluation and management of acute lower gastrointestinal hemorrhage: a randomized controlled trial. Am J Gastroenterol 100(100): 2395-2402.
- Repaka A, Atkinson MR, Faulx AL, Isenberg GA, Cooper GS, et al. (2012) Immediate unprepared hydroflush colonoscopy for severe lower GI bleeding: a feasibility study. Gastrointest Endosc 76(2): 367-373.
- Niikura R, Nagata N, Shimbo T, Sakurai T, Aoki T, et al. (2015) Adverse Events during Bowel Preparation and Colonoscopy in Patients with Acute Lower Gastrointestinal Bleeding Compared with Elective Non-Gastrointestinal Bleeding. PLoS One 10(9): e0138000.
- Pennazio M, Spada C, Eliakim R, Keuchel M, May A, et al. (2015) Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy 47(4): 352-376.
- Lin S, Rockey DC (2005) Obscure gastrointestinal bleeding. Gastroenterol Clin North Am 34(4): 679-698.
- Kim JH, Moon W (2016) Optimal Diagnostic Approaches for Patients with Suspected Small Bowel Disease. Clin Endosc 49(4): 364-369.
- Gralnek IM, Ching JY, Maza I, Wu JC, Rainer TH, et al. (2013) Capsule endoscopy in acute upper gastrointestinal hemorrhage: a prospective cohort study. Endoscopy 45(1): 12-19.
- Sung JJ, Tang RS, Ching JY, Rainer TH, Lau JY (2016) Use of capsule endoscopy in the emergency department as a triage of patients with GI bleeding. Gastrointest Endosc 84(6): 907-913.
- Cai JX, Saltzman JR (2018) Initial Assessment, Risk Stratification, and Early Management of Acute Nonvariceal Upper Gastrointestinal Hemorrhage. Gastrointest Endosc Clin N Am 28(3): 261-275.
- Kim JS, Kim BW (2016) Risk Strategy in Non-Variceal Upper Gastrointestinal Bleeding. Korean J Helicobacter Up Gastrointest Res 16(4): 173-177.
- Rockall TA, Logan RF, Devlin HB, Northfield TC (2006) Risk assessment after acute upper gastrointestinal haemorrhage. Gut 38:(3): 316-321.
- Wang CY, Qin J, Wang J, Sun CY, Cao T, et al. (2013) Rockall score in predicting outcomes of elderly patients with acute upper gastrointestinal bleeding. World J Gastroenterol 19(22): 3466-3472.
- Blatchford O, Davidson LA, Murray WR, Blatchford M, Pell J (2007) Acute upper gastrointestinal haemorrhage in west of Scotland: case ascertainment study. BMJ 315(7107): 510-514.
- Yaka E, Yılmaz S, Doğan NÖ, Pekdemir M (2015) Comparison of the Glasgow-Blatchford and AIMS65 scoring systems for risk stratification in upper gastrointestinal bleeding in the emergency department. Acad Emerg Med 22(1): 22-30.
- Saltzman JR, Tabak YP, Hyett BH, Sun X, Travis AC, et al. (2011) A simple risk score accurately predicts in-hospital mortality, length of stay, and cost in acute upper GI bleeding. Gastrointest Endosc 74(6): 215-1224.
- Na HK, Jung HY, Seo DW, Lim H, Ahn JY, et al. (2017) Erythromycin infusion prior to endoscopy for acute nonvariceal upper gastrointestinal bleeding: a pilot randomized controlled trial. Korean J Intern Med 32(60): 1002-1009.
- Carbonell N, Pauwels A, Serfaty L, Boelle PY, Becquemont L, et al. (2006) Erythromycin infusion prior to endoscopy for acute upper gastrointestinal bleeding: a randomized, controlled, double-blind trial. Am J Gastroenterol 101(6): 1211-1215.
- Troland D, Stanley A (2018) Endotherapy of Peptic Ulcer Bleeding. Gastrointest Endosc Clin N Am 28(3): 277-289.
- Conway JD, Adler DG, Diehl DL, Farraye FA (2009) Endoscopic hemostatic devices. Gastrointest Endosc 69(6): 987-996.
- Pasha SF, Shergill A, Acosta RD, Chandrasekhara V, Chathadi KV, Early D, et al. (2014) The role of endoscopy in the patient with lower GI bleeding. Gastrointest Endosc 79(6): 875-885.
- Forrest JA, Finlayson ND, Shearman DJ (2004) Endoscopy in gastrointestinal bleeding. Lancet 304(7877): 394-397.
- Laine L. Clinical Practice (2016) Upper Gastrointestinal Bleeding Due to a Peptic Ulcer. N Engl J Med 374(24): 2367-2376.
- de Groot NL, van Oijen MG, Kessels K, Hemmink M, Weusten BL, et al. (2014) Reassessment of the predictive value of the Forrest classification for peptic ulcer rebleeding and mortality: can classification be simplified? Endoscopy 46(1): 46-52.
- Vergara M, Bennett C, Calvet X, Gisbert JP (2014) Epinephrine injection versus epinephrine injection and a second endoscopic method in high-risk bleeding ulcers. Cochrane Database Syst Rev.
- Barkun AN, Martel M, Toubouti Y, Rahme E, Bardou M (2009) Endoscopic hemostasis in peptic ulcer bleeding for patients with high-risk lesions: a series of meta-analyses. Gastrointest Endosc 69(4): 786-799.
- Baracat F, Moura E, Bernardo W, Pu LZ, Mendonça E, et al. (2016) Endoscopic hemostasis for peptic ulcer bleeding: systematic review and meta-analyses of randomized controlled trials. Surg Endosc 30(6): 2155-2168.
- Laine L, McQuaid KR (2009) Endoscopic therapy for bleeding ulcers: an evidence-based approach based on meta-analyses of randomized controlled trials. Clin Gastroenterol Hepatol 7(1): 33-47.
- Gralnek IM, Dumonceau JM, Kuipers EJ, Lanas A, Sanders DS, et al. (2015) Diagnosis and management of nonvariceal upper gastrointestinal hemorrhage: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 47(10): 1-46.
- Jensen DM, Eklund S, Persson T, Ahlbom H, Stuart R, et al. (2017) Reassessment of Rebleeding Risk of Forrest IB (Oozing) Peptic Ulcer Bleeding in a Large International Randomized Trial. Am J Gastroenterol 112(3): 441-446.
- Hwang JH, Shergill AK, Acosta RD, Chandrasekhara V, Chathadi KV, et al. (2014) Cash BD American Society for Gastrointestinal Endoscopy. The role of endoscopy in the management of variceal hemorrhage. Gastrointest Endosc 80(2): 221-227.
- De Franchis R, Baveno V (2010) Revising consensus in portal hypertension: report of the Baveno V consensus workshop on methodology of diagnosis and therapy in portal hypertension. J Hepatol 53(4): 762-768.
- Soares WK, Brezis M, Tur KR, Leibovici L (2002) Antibiotic prophylaxis for cirrhotic patients with gastrointestinal bleeding. Cochrane Database Syst Rev CD002907.
- Sarin SK, Kumar A, Angus PW, Baijal SS, Baik SK, et al. (2011) Asian Pacific Association for the Study of the Liver (APASL) Working Party on Portal Hypertension. Diagnosis and management of acute variceal bleeding: Asian Pacific Association for Study of the Liver recommendations. Hepatol Int 5: 607-624.
- Dai C, Liu WX, Jiang M, Sun MJ (2015) Endoscopic variceal ligation compared with endoscopic injection sclerotherapy for treatment of esophageal variceal hemorrhage: a meta-analysis. World J Gastroenterol 21(8): 2534-2541.
- Garcia TG, Sanyal AJ, Grace ND, Carey W (2007) Prevention and management of gastroesophageal varices and variceal hemorrhage in cirrhosis. Practice Guidelines Committee of the American Association for the Study of Liver Diseases; Practice Parameters Committee of the American College of Gastroenterology Hepatology 46(6): 922-938.
- Sinclair M, Vaughan R, Angus PW, Gow PJ, Parker F, et al. (2015) Risk factors for band-induced ulcer bleeding after prophylactic and therapeutic endoscopic variceal band ligation. Eur J Gastroenterol Hepatol 27(8): 928-932.
- Shaheen NJ, Stuart E, Schmitz SM, Mitchell KL, Michael WF, et al. (2005) Pantoprazole reduces the size of postbanding ulcers after variceal band ligation: a randomized, controlled trial. Hepatology 41(3): 588-594.
- Sanglodkar UA, Jothimani D, Rela M (2018) Hemospray for recurrent esophageal band ulcer bleeding. Clin Exp Hepatol 4(1): 46-48.
- Ibrahim M, El Mikkawy A, Abdalla H, Mostafa I, Devière J (2015) Management of acute variceal bleeding using hemostatic powder. United European Gastroenterol J 3: 277-283.
- Ibrahim M, El Mikkawy A, Abdel Hamid M, Abdalla H, Lemmers A, et al. (2018) Early application of haemostatic powder added to standard management for oesophagogastric variceal bleeding: a randomised trial. Gut 68(5): 844-853.
- Escorsell À, Pavel O, Cárdenas A, Morillas R, Llop E, et al. (2016) Variceal Bleeding Study Group. Esophageal balloon tamponade versus esophageal stent in controlling acute refractory variceal bleeding: A multicenter randomized, controlled trial. Hepatology 63(6): 1957-1967.
- Trebicka J (2017) Emergency TIPS in a Child-Pugh B patient: When does the window of opportunity open and close? J Hepatol 66(2): 442-450.
- Feuerstein JD, Falchuk KR (2016) Diverticulosis and Diverticulitis. Mayo Clin Proc 91(8): 1094-1104.
- Wensaas KA, Hungin AP (2016) Diverticular Disease in the Primary Care Setting. J Clin Gastroenterol 50(Suppl 1): S86-S88.
- Lee KK, Shah SM, Moser MA (2011) Risk factors predictive of severe diverticular hemorrhage. Int J Surg 9(1): 83-85.
- McGuire HH (2004) Bleeding colonic diverticula. A reappraisal of natural history and management. Ann Surg 220(5): 653-656.
- Jensen DM, Ohning GV, Kovacs TO, Jutabha R, Ghassemi K, et al. (2016) Natural history of definitive diverticular hemorrhage based on stigmata of recent hemorrhage and colonoscopic Doppler blood flow monitoring for risk stratification and definitive hemostasis. Gastrointest Endosc 83(2): 416-423.
- Ishii N, Omata F, Nagata N, Kaise M (2018) Effectiveness of endoscopic treatments for colonic diverticular bleeding. Gastrointest Endosc 87(1): 58-66.
- Soetikno R, Ishii N, Kolb JM, Hammad H, Kaltenbach T (2018) The Role of Endoscopic Hemostasis Therapy in Acute Lower Gastrointestinal Hemorrhage. Gastrointest Endosc Clin N Am 28(3): 391-408.
- Ishii N, Setoyama T, Deshpande GA, Omata F, Matsuda M, et al. (2012) Endoscopic band ligation for colonic diverticular hemorrhage. Gastrointest Endosc 75(2): 382-387.
- Nagata N, Ishii N, Kaise M, Shimbo T, Sakurai T, et al. (2018) Long-term recurrent bleeding risk after endoscopic therapy for definitive colonic diverticular bleeding: band ligation versus clipping. Gastrointest Endosc 88(5): 841-853.
- Foutch PG, Rex DK, Lieberman DA (2005) Prevalence and natural history of colonic angiodysplasia among healthy asymptomatic people. Am J Gastroenterol 90(4): 564-567.
- Boley SJ, Sammartano R, Adams A, DiBiase A, Kleinhaus S et al. (2007) On the nature and etiology of vascular ectasias of the colon. Degenerative lesions of aging. Gastroenterology 72(4 Pt 1): 650-660.
- Sekino Y, Endo H, Yamada E, Sakai E, Ohkubo H, et al. (2012) Clinical associations and risk factors for bleeding from colonic angiectasia: a case-controlled study. Colorectal Dis 14(10): e740-e746.
- Strate LL, Liu YL, Huang ES, Giovannucci EL, Chan AT (2011) Use of aspirin or nonsteroidal anti-inflammatory drugs increases risk for diverticulitis and diverticular bleeding. Gastroenterology 140(5): 1427-1433.
- Strate LL (2005) Lower GI bleeding: epidemiology and diagnosis. Gastroenterol Clin North Am 34(4): 643-664.
- Brandt LJ, Spinnell MK (2009) Ability of naloxone to enhance the colonoscopic appearance of normal colon vasculature and colon vascular ectasias. Gastrointest Endosc 49(1): 79-83.
- Kwan V, Bourke MJ, Williams SJ, Gillespie PE, Murray MA, et al. (2006) Argon plasma coagulation in the management of symptomatic gastrointestinal vascular lesions: experience in 100 consecutive patients with long-term follow-up. Am J Gastroenterol 101(1): 58-63.
- Kirschniak A, Kratt T, Stüker D, Braun A, Schurr MO, et al. (2007) A new endoscopic over-the-scope clip system for treatment of lesions and bleeding in the GI tract: first clinical experiences. Gastrointest Endosc 66(1): 162-167.
- Sinha R, Lockman KA, Church NI, Plevris JN, Hayes PC (2016) The use of hemostatic spray as an adjunct to conventional hemostatic measures in high-risk nonvariceal upper GI bleeding (with video) Gastrointest Endosc 84(6): 900-906. e3.
- Beg S, Al-Bakir I, Bhuva M, Patel J, Fullard M, et al. (2015) Early clinical experience of the safety and efficacy of EndoClot in the management of non-variceal upper gastrointestinal bleeding. Endosc Int Open 3(6): E605–E609.
- Kim JH, Kim JH, Chun J, Lee C, Im JP, et al. (2018) Early versus late bedside endoscopy for gastrointestinal bleeding in critically ill patients. Korean J Intern Med 33: 304-312.
- Jean-Baptiste S, Messika J, Hajage D, Gaudry S, Barbieri J, et al. (2018) Clinical impact of upper gastrointestinal endoscopy in critically ill patients with suspected bleeding. Ann Intensive Care 8(75).
- Arrowsmith JB, Gerstman BB, Fleischer DE, Benjamin SB (2001) Results from the American Society for Gastrointestinal Endoscopy/U.S. Food and Drug Administration collaborative study on complication rates and drug use during gastrointestinal endoscopy. Gastrointest Endosc 37(4): 421-427.
- Sharma VK, Nguyen CC, Crowell MD, Lieberman DA, de Garmo P, et al. (2007) A national study of cardiopulmonary unplanned events after GI endoscopy. Gastrointest Endosc 66(1): 27-34.
- Rudolph SJ, Landsverk BK, Freeman ML (2003) Endotracheal intubation for airway protection during endoscopy for severe upper GI hemorrhage. Gastrointest Endosc 57(1): 58-61.
- Rehman A, Iscimen R, Yilmaz M, Khan H, Belsher J, et al. (2009) Prophylactic endotracheal intubation in critically ill patients undergoing endoscopy for upper GI hemorrhage. Gastrointest Endosc 69(7): e55-e59.
- Hayat U, Lee PJ, Ullah H, Sarvepalli S, Lopez R, et al. (2017) Association of prophylactic endotracheal intubation in critically ill patients with upper GI bleeding and cardiopulmonary unplanned events. Gastrointest Endosc 86(3): 500-509.e1.
- Choudari CP, Rajgopal C, Elton RA, Palmer KR (2004) Failures of endoscopic therapy for bleeding peptic ulcer: an analysis of risk factors. Am J Gastroenterol 89(1): 1968-1972.
- Wong SK, Yu LM, Lau JY, Lam YH, Chan AC, et al. (2002) Prediction of therapeutic failure after adrenaline injection plus heater probe treatment in patients with bleeding peptic ulcer Gut 50(3): 322-325.
- Thomopoulos KC, Mitropoulos JA, Katsakoulis EC, Vagianos CE, Mimidis KP, et al. (2001) Factors associated with failure of endoscopic injection haemostasis in bleeding peptic ulcers. Scand J Gastroenterol 36(6): 664-668.
- La Mura V, Nicolini A, Tosetti G, Primignani M (2015) Cirrhosis and portal hypertension: The importance of risk stratification, the role of hepatic venous pressure gradient measurement. World J Hepatol 7(4): 688-695.
- Moitinho E, Escorsell A, Bandi JC, Salmerón JM, García-Pagán JC, et al. (2009) Prognostic value of early measurements of portal pressure in acute variceal bleeding. Gastroenterology 117(3): 626-631.
- Hernández-Gea V, Berbel C, Baiges A, García-Pagán JC (2018) Acute variceal bleeding: risk stratification and management (including TIPS) Hepatol Int 12(Suppl 1): 81-90.






























