The Cankerous Breadbasket-Gastric Adenocarcinoma
Anubha Bajaj*
Histopathologist in A.B. Diagnostics, India
Submission: May 23, 2022; Published: June 01, 2022
*Corresponding author: Anubha Bajaji, Histopathologist in A.B. Diagnostics, New Delhi, India
How to cite this article: Anubha Bajaj. The Cankerous Breadbasket-Gastric Adenocarcinoma. Adv Res Gastroentero Hepatol, 2022; 18(5): 555996. DOI: 10.19080/ARGH.2022.18.555996.
Keywords: Gastric carcinoma; Gastric mucosal epithelium; Smoking; Obesity; Weight loss; Jaundice; Vomiting; Dysphagia; Diarrhoea; Constipation
Abbreviations: EB: Epstein-Barr; GERD: Gastro-Oesophageal Reflux Disease; HIV: Human Immunodeficiency Virus; CEA: Carcinoembryonic Antigen; PET-CT: Positron Emission Computerized Tomography; HER2: Human Epidermal Growth Factor Receptor 2
Introduction
Gastric carcinoma engendered from gastric mucosal epithelium exhibits specific subtypes as preponderant gastric adenocarcinoma (~90%), intestinal subtype or diffuse subtype and signet ring adenocarcinoma. Contributing factors such as infection with Helicobacter pylori, Epstein-Barr (EB) virus or human immunodeficiency virus (HIV), smoking, obesity with associated gastro-oesophageal reflux disease (GERD), iodine deficiency or consumption of alcohol, smoked or salt-rich foods, pickled vegetables, brackens and red or processed meat are encountered [1,2]. Additionally, diabetes mellitus, pernicious anaemia, chronic atrophic gastritis, Menetrier’s disease or hyperplastic, hyper-secretory gastropathy, intestinal metaplasia or Foxp3 polymorphism may contribute to disease emergence [1,2]. Consumption of fresh or citrusy fruits, vegetables, antioxidants, Mediterranean diet, administration of aspirin, cessation of smoking and treating Helicobacter pylori infection may decimate emergence of gastric carcinoma [1,2]. Cutaneous conditions associated with gastric carcinoma are acanthosis nigricans with pigmented hyperplasia of axilla and groin, "tripe palms" with pigmented hyperplasia of palms and Leser-Trelat sign engendered due to rapidly progressive cutaneous lesions as seborrheic keratosis. Gastric carcinoma can be asymptomatic or exemplify non-specific symptoms. Clinical symptoms generally appear with locally advanced or metastatic neoplasms [1,2]. Antecedent clinical symptoms occur as indigestion, heartburn, abdominal discomfort, upper abdominal pain, nausea, anorexia, unexplained weight loss, jaundice, vomiting, dysphagia, diarrhoea, constipation or haematochezia [1,2]. Enlarged, infiltrative gastric carcinoma can engender weakness, fatigue, bloating, upper abdominal pain, nausea and occasional vomiting. Additional tumour expansion is associated with weight loss, hematemesis, melena and consequent anaemia [1,2]. Clinical symptoms of gastric viral infection, gastric ulcer or tropical sprue may accompany manifestations of gastric carcinoma [1,2].
a) stage 0 exhibits tumour confined to mucosa, can be suitably treated with endoscopic mucosal resection or gastrectomy with lymphadenectomy.
b) stage I demonstrates tumour infiltration to submucosa or muscularis propria, designated as stage IA which can be managed with surgical extermination and omentectomy. Submucosa with regional lymph node involvement, expounded as stage IB is treated with 5-fluorouracil and radiotherapy.
c) stage II displays tumefaction infiltrating to submucosa and distant lymph nodes or muscularis propria and regional lymph nodes or may extend to serosa in the absence of lymph node involvement. Surgical extermination and omentectomy along with radiotherapy and neoadjuvant chemotherapy may be employed.
d) stage III delineates tumour extension into muscularis propria and distant lymph nodes or into serosa along with adjacent viscera or regional and distant lymph nodes. Therapeutic option akin to stage II may be adopted.
e) stage IV exemplifies neoplastic extension to adjacent viscera and distant lymph nodes. Distant metastasis can occur. Therapeutic strategies such as surgical intervention, laser, stenting the digestive tract and chemotherapy with 5-fluouracil, cisplatin, epirubicin, etoposide, docetaxel, oxaliplatin, capecitabine or irinotecan are utilized [1,2].
Gastric adenocarcinoma originates from malignant mucosal glandular epithelium. Aggressive gastric adenocarcinomas infiltrate gastric wall comprised of muscularis mucosae, submucosa and muscularis propria. Intestinal subtype of gastric adenocarcinoma is composed of irregular, tubular structures with several lumens, stratification and pseudo-stratification of neoplastic cells with decimated intervening stroma and "back-to-back" accumulation of neoplastic glands. Adjacent mucosa exhibits intestinal metaplasia [1,2].
Contingent to glandular architecture, cellular pleomorphism and mucus secretion, gastric adenocarcinoma is segregated into well-differentiated, moderately- differentiated and poorly differentiated categories [1,2].
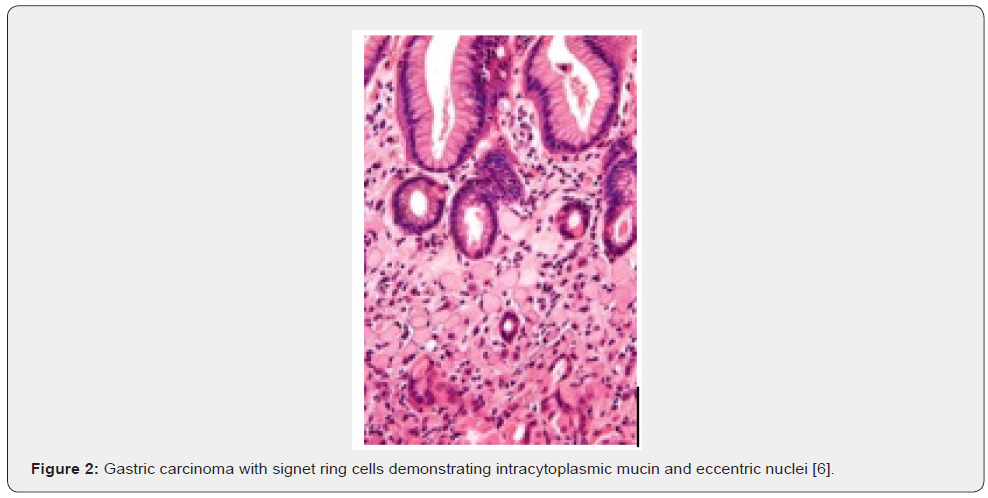
Diffuse(infiltrating)- subtype of gastric adenocarcinoma is poorly- differentiated and can denominate as mucinous (colloid) carcinoma and linitis plastic or leather-bottle stomach. Mucus-secreting neoplastic cells are dis-cohesive and depict enlarged interstitial spaces pervaded with mucus or colloid [1,2].
Signet ring cell carcinoma is configured of tumour cells with intracytoplasmic mucin and an eccentric nucleus, thus articulating characteristic signet ring cells [1,2] (Figure 1,2 & Table 1).


Comprehensive clinical history, physical examination and laboratory investigations as complete blood count and faecal occult blood are required to appropriately determine gastric carcinoma [3,4]. Tumour markers as carcinoembryonic antigen (CEA) appear to correlate with possible distant metastasis [3,4].
Gastroscopic examination of the neoplasm is an optimal, recommended technique for evaluating gastric adenocarcinoma [3,4]. Barium studies of upper gastrointestinal tract or barium roentgenogram is beneficial. Imaging techniques can determine distant tumour metastasis [3,4]. Computerized tomography (CT) of the abdomen is advantageous in discerning tumour invasion into adjacent tissues or regional lymph nodes. Features such as focal, eccentric and enhancing thickening of gastric wall exceeding > 1 centimetre appear indicative of malignant metamorphosis [3,4].
Additionally, positron emission computerized tomography (PET-CT) or endoscopic ultrasonography can be adopted [3,4]. Ultra-high magnification endo-cytoscopy can appropriately discern cellular structures and focal dysplasia. Optimal coherence tomography can be employed to determine gastric carcinoma [3,4]. Tumefaction can be detected with tissue samples from anomalous areas visualized by gastroscopy and obtained with endoscopic manoeuvers [3,4]. Distant metastasis to hepatic or pulmonary parenchyma, pancreas, bones, adjacent viscera, abdominal wall and lymph nodes may ensue. Cogent treatment options are surgical intervention, chemotherapy, radiotherapy, targeted therapy, cancer immunotherapy or gene therapy [3,4]. Curative surgical procedures are comprised of techniques such as endoscopic mucosal resection, endoscopic submucosal dissection and subtotal or total gastrectomy [3,4].
Following total gastrectomy emerging complications such as dumping syndrome and reflux esophagitis can treated with construction of a "pouch" or "gastric substitute"[3,4]. Specific chemotherapeutic regimen for treating the insensitive gastric carcinoma is debatable. However, chemotherapy is utilized to decimate tumour magnitude, relieve clinical symptoms and enhance proportionate survival [3,4].
Agents such as carmustine, semustine, doxorubicin, mitomycin C, cisplatin or taxotere appear advantageous [3,4]. Human epidermal growth factor receptor 2 (HER2) inhibitor as trastuzumab may enhance overall survival in inoperable or metastatic carcinomas [3,4]. Palliative care is optimal for advanced cancers with delayed detection. Advanced carcinoma depicts proportionate 5-year survival at <10%. Prognostic outcomes are inferior and distant metastasis are observed upon initial tumour discernment. Unfavorable outcome is associated with expression of ITGAV, DUSP1 or P2RX7 genes [3,4].
References
- Jaffer A Ajani, Thomas A D'Amico, David J Bentrem, Joseph Chao, David Cooke, et al. (2022) Gastric Cancer, Version 2.2022, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 20(2): 167-192.
- Recio-Boiles A, Babiker HM (2022) Gastric Cancer. Stat Pearls International. Treasure Isand, Florida, USA.
- Mocan L (2021) Surgical Management of Gastric Cancer: A Systematic Review. J Clin Med 10(12): 2557.
- Ilson DH (2018) Advances in the Treatment of Gastric Cancer. Curr Opin Gastroenterol 34(6): 465-468.
- Image 1 Courtesy: BMJ.com.
- Image 2 Courtesy: Wikipedia.com.






























