Tacrolimus Induced Acute Renal Failure Caused by Ledipasvir (Harvoni) In Hepatitis C Infected Renal Transplant Patient
Mohammad Raza Khan1*, Aemen Shams Khakwani2, Areebah Qadir1, Nabiha Mustafa1, Sarah Siddiqui3,Subhan Shah4 and Usman Ahmad Khan5
1Bakhtawar Amin Medical and Dental College, Pakistan
2Resident Internal Medicine, Suburban Community Hospital Norristown, USA
3BR Ambedkar University,India
4Aston University, United Kingdom
5Department of Internal Medicine, University of Oklahoma and VA Medical Center, USA
Submission:April 07, 2022; Published:April 18, 2022
*Corresponding author:Mohammad Raza Khan, Bakhtawar Amin Medical and Dental College, Pakistan
How to cite this article: Mohammad Raza K, Aemen Shams K, Areebah Q, Nabiha M, Sarah S, et al. Tacrolimus Induced Acute Renal Failure Caused by Ledipasvir (Harvoni) In Hepatitis C Infected Renal Transplant Patient. Adv Res Gastroentero Hepatol, 2022; 18(4): 555993. DOI: 10.19080/ARGH.2022.18.555993.
Abstract
Hepatitis C viral infection is associated with loss of liver function due to liver cirrhosis and increased risk for the development of liver carcinoma. HCV is quite prevalent and has various extra-hepatic manifestations including loss of renal function. Earlier, HCV patients were treated with pegylated interferon and ribavirin, which showed a decline in the risk of development of end-stage renal disease in HCV patients, but it was discouraged as a primary mode of treatment because of adverse effects and intolerance [1]. After the emergence of Direct-acting antivirals (DAAs) HCV treatment has revolutionized. The goal of treatment was to attain sustained virologic response (SVR) and with DAA regimens, it exceeded to over 90%. Numerous DAAs such as NS5A and NS5B inhibitors with protease inhibitors are used to target different aspects of the HCV genome and prevent replication of the virus. Ledipasvir (Harvoni) is a commonly used inhibitor of HCV NS5A protein, which is required for the viral replication process and hence is widely used for HCV treatment worldwide. However, these regimens were only allowed for HCV patients with mild to moderate renal loss because of the increased risk of accumulation of toxic metabolites in patients with eGFR < 30 ml/min/1.73 m^2 [2,3].
We present a case of treatment of hepatitis C viral infection with Direct-acting anti-virals that has been linked to nephrotoxicity in a renal transplant patient due to drug interactions between Tacrolimus and ledipasvir. Tacrolimus is a widely used immunosuppressant for renal transplant patients. Tacrolimus and Harvoni share the same P-gp transporter and if administered together, they may cause nephrotoxicity. We will discuss how direct-acting antivirals interact in patients who are also taking certain immunosuppressants and why this can lead to induced acute renal failure in a hepatitis C infected patient with a renal transplant.
Keywords:Acute kidney injury; Hepatitis C virus; Direct-acting antiviral agents; Sustained virological response; Urea and creatinine
Abbreviations:DAAs: Direct-Acting Antivirals; SVR: Sustained Virologic Response; AKI: Acute Kidney Injury; HCV: Hepatitis C Virus; DAAs: Direct-Acting Antiviral Agents
Introduction
Acute kidney injury (AKI) is characterized by the rapid loss of the kidney’s function and is typically diagnosed by the accumulation of end products of nitrogen metabolism (urea and creatinine) or decreased urine output, or both [4]. There are many possible aetiologias for AKI, and the following case report will investigate an apparent association of Tacrolimus and DAAs resulting in AKI in a renal transplant patient who also suffers from unrelated hepatitis C. Hepatitis C virus (HCV) is a single-stranded enveloped RNA virus that causes hepatitis along with cirrhosis (for which hepatitis C viral infection is the second most common cause of in the USA) and hepatocellular carcinoma, an estimated 2.4 million people are suffering from chronic hepatitis C infection in the USA. Historically, chronic hepatitis C were treated with pegylated interferon and ribavirin; however adverse effects from the interferons were the major limiting factor for this regimen of treatment [5]. Recent treatments for hepatitis C utilize direct-acting antiviral agents (DAAs), and the patient in this case report was on the HARVONI treatment combination of the DAAs ledipasvir and sofosbuvir for his type 1 genotype HCV infection. These DAA combination treatments, including the glecavaprvir/pibrentasvir combination for type 2 and 3 genotypes, have shown remarkable efficacy with a sustained virological response (SVR) of over 90% across genotypes, an immensely significant improvement on the 50% SVR of the pegylated interferon/ribavirin treatment. [6,7] However, this treatment regime is associated with several toxicities in renal transplant patients on Tacrolimus. We report a case of acute kidney injury associated with increased Tacrolimus plasma levels, secondary to the unique drug interactions of ledipasvir (HARVONI) and Tacrolimus [8].
Case Presentation
A 65-year-old patient with a past medical history of Type 2 diabetes mellitus, essential hypertension, end-stage renal disease status post recent cadaveric renal transplant, and chronic hepatitis C infection, initiated on Harvoni (ledipasvir). He had recently undergone an unrelated cadaveric renal transplant 1 month ago and was hospitalized 2 weeks for it. The patient was being followed outpatient by the transplant team, gastroenterology team for the hepatitis C treatment initiation, and the vascular surgery clinic for his left arteriovenous fistula site swelling, which had aneurysmal changes that were successfully stented and repaired by Interventional Radiology. But patient reported back to the vascular surgery clinic with worsening left arm pain and during his visit had atypical left-sided chest pain. The patient was then transferred to the emergency department over concerns of acute coronary syndrome and underwent a coronary angiogram, which was found to be normal During his hospitalization, the patient was found to have acute creatinine elevation from his baseline of 1.5mg/dl to 2.15mg/dl on admission before heart catheterization. No significant changes were reported by the patient regarding the amount of urination or character of urine and denied any fever, dysuria, urgency, or frequency. The had remained consistent with taking his immunosuppressive medications and the gastroenterology team had started him on Harvoni (ledipasvir) 2 weeks ago for his chronic hepatitis C infection. The urinalysis performed showed no hematuria, proteinuria, red blood cells, or any casts. The fractional excretion of sodium and urea were less than 1% and 35% respectively, with the rest of the lab being within normal limits. Ultrasound was negative for hydronephrosis, and the resistive indices were within normal limits. During preceding visits, tacrolimus and creatinine levels were obtained, showing creatinine of 1.6 when tacrolimus was [9] and a creatinine of 2.15 when tacrolimus was 14. Tacrolimus toxicity was the most likely cause of elevated creatinine and renal failure due to the absence of any known nephrotoxins or other causes. Research showed that tacrolimus and Harvoni (ledipasvir) share the same transporter, P-gp Protein for absorption and the elevation of tacrolimus levels and renal were secondary to tacrolimus interacting with Harvoni (ledipasvir) that initiated for patient’s hepatitis C. Our hypothesis was tested by holding the next dose of tacrolimus and cutting back the 5mg twice daily to 4mg in the AM and 3mg in the PM. The level of tacrolimus fell to [10] and the creatinine level started to drop, i.e., 2.02, strengthening our hypothesis. Daily blood draws were done to check the levels and after 5 days the tacrolimus level fell to 6.3 and the creatinine level was back to a baseline of 1.5 (Figure 1).
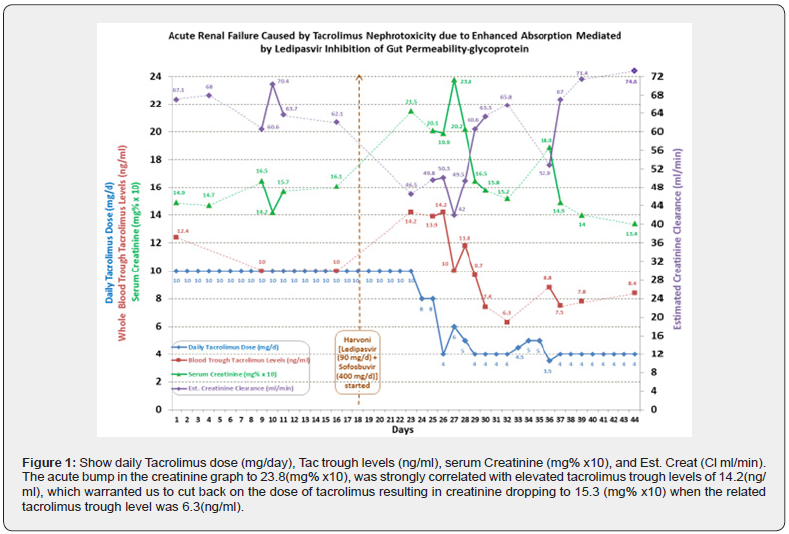
Case Discussion
The results show increased Tacrolimus trough level and increased serum creatinine following the initiation of Harvoni (Ledipasvir) for Hepatitis C infection in our patient who recently underwent a cadaveric renal transplant 1 month ago. The literature search was done, and it was instituted that Tacrolimus and Ledipasvir share the same transporter (P-gp Protein) for absorption, and the elevated levels of tacrolimus and renal failure were secondary to the interaction between Tacrolimus and Ledipasvir.
P-glycoprotein (MDR1; ABCB1), the permeability glycoprotein or plasma glycoprotein is an active, 170kDa efflux membranebound transport protein pump discovered in 1976. P-gp is a member of the ATP binding cassette (ABC) superfamily [9]. P-glycoprotein is expressed in tissues such as intestinal enterocytes, hepatocytes, renal proximal tubular cells, the blood-brain, and blood-testis barrier, and cancer cells [10]. The P-glycoprotein can be inhibited, activated or its expression can be induced by some drugs, food components, and herbs, affecting its activity. P-glycoprotein inhibitors or substrates such as Ledipasvir, increase the permeability, and as a result serums levels of P-glycoprotein substrates such as Tacrolimus, resulting in its toxicity, which was presented in our patient as acute renal injury (AKI). Also, studies have shown that the use of Tacrolimus posttransplant has seen to be associated with downregulating P-gp expression on CD4 lymphocytes, influencing its own concentration [11] and concomitant use of Ledipasvir can exaggerate that effect. In line with our hypothesis, prescribing DAAs with tacrolimus should be given careful consideration with close follow-up of their tacrolimus levels while on therapy with DAAs for Hepatitis C infection 13. Our hypothesis could’ve been tested further by monitoring Tacrolimus trough level on a varied dosage of Harvoni (Ledipasvir) [12].
Conclusion
Prescribing direct-acting antiviral agents (DAA’s) for Hepatitis C infections in renal transplant or renal disease patients needs contemplation because there is a chance of interaction with tacrolimus and P-glycoprotein. Newer DAAs that have been recently developed have a lower risk of adverse effects because of their less frequent drug interactions. This will increase their use in the treatment of Hepatitis C treatment in patients who have received renal transplants. The dose titration of immunosuppressive medications such as tacrolimus should be strictly monitored in renal transplant HCV infected patients who are being treated with DAA’s. This should be done to prevent nephrotoxicity of these immunosuppressive drugs at higher plasma levels, and to avoid the risk of renal transplant rejection at their lower plasma levels.
References
- Kao CC, Lin YS, Chu HC, Fang TC, Wu MS, et al. (2018) Association of Renal Function and Direct-Acting Antiviral Agents for HCV: A Network Meta-Analysis. Journal of Clinical Medicine 7(10): 314.
- Sharma R, Brown RS (2017) Treatment of hepatitis C virus in advanced renal disease. Clinical Liver Disease 10(3): 79-81.
- Sise ME, Chute DF, Oppong Y, Davis MI, Long JD, et al. (2020) Direct-acting antiviral therapy slows kidney function decline in patients with Hepatitis C virus infection and chronic kidney disease. Kidney Int 97(1): 193-201.
- Bellomo R, Kellum JA, Ronco C (2012) ACUTE Kidney Injury. Lancet 380(9843): 756-766.
- Edlin BR, Eckhardt BJ, Shu MA, Holmberg SD, Swan T (2015) Toward a more accurate estimate of the prevalence of hepatitis C in the United States. Hepatology 62(5): 1353-1363.
- Sulkowski MS, Cooper C, Hunyady B, Jia J, Ogurtsov P, et al. (2011) Management of adverse effects of Peg-IFN and ribavirin therapy for hepatitis C. Nat Rev Gastroenterol Hepatol 8(4): 212-223.
- Reig M, Mariño Z, Perelló C, Iñarrairaegui M, Ribeiro A, et al. (2016) Unexpected high rate of early tumor recurrence in patients with HCV-related HCC undergoing interferon-free therapy. J Hepatol 65(4): 719-726.
- Hill L (2015) Hepatitis C Virus Direct-Acting Antiviral Drug Interactions and Use in Renal and Hepatic Impairment. Top Antivir Med 23(2): 92-96.
- Ambudkar SV, Kimchi SC, Sauna ZE, Gottesman MM (2003) P-glycoprotein: from genomics to mechanism. Oncogene. 22(47): 7468-7485.
- Fromm MF (2002) The influence of MDR1 polymorphisms on P-glycoprotein expression and function in humans. Adv Drug Deliv Rev 54(10): 1295-1310.
- Parasrampuria DA, Lantz MV, Birnbaum JL, Vincenti FG, Benet LZ (2002) Effect of Calcineurin Inhibitor Therapy on P-gp Expression and Function in Lymphocytes of Renal Transplant Patients: A Preliminary Evaluation. J Clin Pharmacos 42(3): 304-311.
- Muzica CM, Stanciu C, Huiban L, Singeap MA, Sfarti C, et al. (2020) Hepatocellular carcinoma after direct-acting antiviral hepatitis C virus therapy: A debate near the end. World J Gastroenterol 26(43): 6770-6781.






























