Hepatocellular Carcinoma: Epidemiologic, Clinical Features And Treatment In Senegal
Salamata Diallo1*, Marie Louise Bassène1, Mamadou Ngoné Gueye2, Hornella Bonning Noumen Petga1,Mariéme Polele Fall1, Mame Aissé Thioubou3, Cheikh Ahmadou Bamba Cissé2, Alioune Badara Fall1, Atteib Fall1, Alsine Yauck1 and Fatou Fall4
1Cheikh Anta Diop university of Dakar, Hospital Aristide Le Dantec, Senegal
2Cheikh Anta Diop university of Dakar, Hospital Idrissa Pouye, Senegal
3Assane Seck university of Ziguinchor, Hospital de la paix, Senegal
4Hospital Principal of Dakar, Senegal
Submission:January 01, 2022; Published:January 24, 2022
*Corresponding author:Salamata Diallo, Cheikh Anta Diop university of Dakar, Department of gastroenterology and hepatology of Hospital Aristide Le Dantec Dakar Senegal
How to cite this article: GSalamata D, Marie L B, Mamadou N G, Hornella B N P, Mariéme Polele F, et al. Hepatocellular Carcinoma: Epidemiologic, Clinical Features And Treatment In Senegal. Adv Res Gastroentero Hepatol, 2022; 18(3): 555988. DOI: 10.19080/ARGH.2022.18.555988.
Abstract
Background:Liver cancer is the third leading cause of cancer deaths worldwide. Hepatocellular carcinoma (HCC) is the most common primary liver tumour. The aim of our study was to evaluate the epidemiological, diagnostic, therapeutic and outcome aspects of HCC.
Material and Method :We conducted a bicentric, retrospective, descriptive and analytical study over 19 months from January 1, 2018 to July 31, 2019 in the department of gastroenterology and hepatology of hospital Aristide le Dantec (Dakar, Senegal) and at the Marie Louise BREVIE clinic at the hospital Principal of Dakar (Dakar, Senegal). We included all patients with a diagnosis of HCC.
Results : We include 104 patients, 89 were men (85.6%). The mean age was 46.7 years [18-78 years]. The most frequent clinical signs were abdominal pain (95.2%). AFP levels were elevated in 77.88 % of cases. The CT scan revealed a wash-in-wash-out appearance in 100 cases (96.1%). Histology revealed HCC in 5 cases (4.8%). HCC developed in a cirrhotic liver in 99 cases (95.1%). This cirrhosis was viral B in 69 cases (69.7 %). Patients were BCLC C in 70.2 %. One patient underwent a left hepatectomy. Chemoembolisation was performed in three patients. Eighty-three patients (79.8%) were lost to follow-up and 21 (20.2%) died. The mean survival of deceased patients was 5 months [ 2-13 months].
Conclusion : In our country HCC, frequently affects young adult males with viral B cirrhosis. Its prognosis is poor because most patients are diagnosed at advanced stage of the disease.
Keywords: Liver cancer; Hepatocellular carcinoma; Cirrhosis; Hepatitis B virus
Abbreviations: HCC: Hepatocellular Carcinoma; NASH: Non-Alcoholic Steatohepatitis; TACE: Trans-Arterial Chemoembolization; ALT: Alanine Aminotransferase; AST: Aspartate Aminotransferase; CP: Child-Pugh; BCLC: Barcelona Liver Cancer Clinic
Introduction
Hepatocellular carcinoma (HCC) is the most common liver cancer. HCC represents more than 90% of primary liver cancers and is a major global health problem. According to GLOBOCAN 2020, liver cancer ranked sixth in the world in terms of incidence. Africa is the 3rd most affected continent with 70542 new cases after Asia (656992 new cases), Europe (87630 new cases). In Senegal, the incidence was 1084 new cases. In the world, liver cancer is the third in cancer-related deaths after lung cancer and colorectal cancer. It accounts for 13.8% of cancer-related deaths for all ages and sexes combined [1].
More than 80% of HCCs develop in less developed regions. In Senegal, it is a public health problem. It is the leading cause of death by cancer in men and the third in women. HCC remains asymptomatic for a long time, and its diagnosis is most often delayed [2]. The presence of clinical signs most often indicates advanced disease. The diagnosis is based on non-invasive diagnostic criteria in the cirrhotic patient. Histology remains the reference in the case of atypical imaging, a healthy liver, or a nodule smaller than 2 cm [3]. HCC most often develops in a cirrhotic liver, regardless of the aetiology. The aetiologies of cirrhosis are dominated by viral hepatitis B and C, alcohol, and non-alcoholic steatohepatitis (NASH) [3]. Rapid diagnosis and early management are essential for patients with HCC.
Management of HCC has many options. Curative treatment includes liver transplantation, liver resection and percutaneous destruction (radiofrequency and percutaneous ethanol injection). Trans-arterial chemoembolization (TACE) and sorafenib are recommended as first-line treatment for unresectable HCC. HCC is a tumour with a poor prognosis with an annual mortality rate of around 96% in developing countries. In Senegal, few studies have been conducted on this pathology, showing that HCC develops most often in young adults who are chronic HBV carriers, and that the prognosis is poor with high mortality.
We conducted a retrospective, descriptive, analytical, and bicentric whose aim was to evaluate the epidemiological, diagnostic, therapeutic, prognostic, and outcome aspects of HCC to participate in a better knowledge of this cancer.
Material and Methods
We conducted a retrospective, analytical and descriptive study from January 1, 2018 to July 31, 2019 (19 months). Our study population was composed of patients followed as ambulatory patients or hospitalized in the hepato-gastroenterology department of the hospital Aristide le Dantec (Dakar, Senegal) and at the Marie Louise BREVIE clinic at the hospital Principal of Dakar (Dakar, Senegal) during the study period.
We included all patients diagnosed with HCC according of the following criteria:
a) Histological analysis of a tumour fragment obtained by ultrasound-guided liver biopsy or CT scan
b) A typical HCC appearance on imaging (CT/MRI) in cirrhotic patients (hypervascularised nodule in the arterial phase with washout in the portal phase or late phase).
Clinical , epidemiological data and liver function test were collected including gender, age at diagnosis, prior history of HCC, family history of HCC, presence of cirrhosis, liver disease aetiology , the Child-Pugh (CP) score , hemogram, alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin, albumin, prothrombin .The data about the HCC were collected , treatments carried out and evolution after treatment including characteristics of the tumours (number, site, diameter of masses and presence of vascular invasion), the alpha-fetoprotein level and the type of therapy used. Data entry was done with EXCEL 2016, statistical analysis was done with STATA 15.1 and graphs were done with Excel 2016. The means were calculated using the chi2 test.
Results
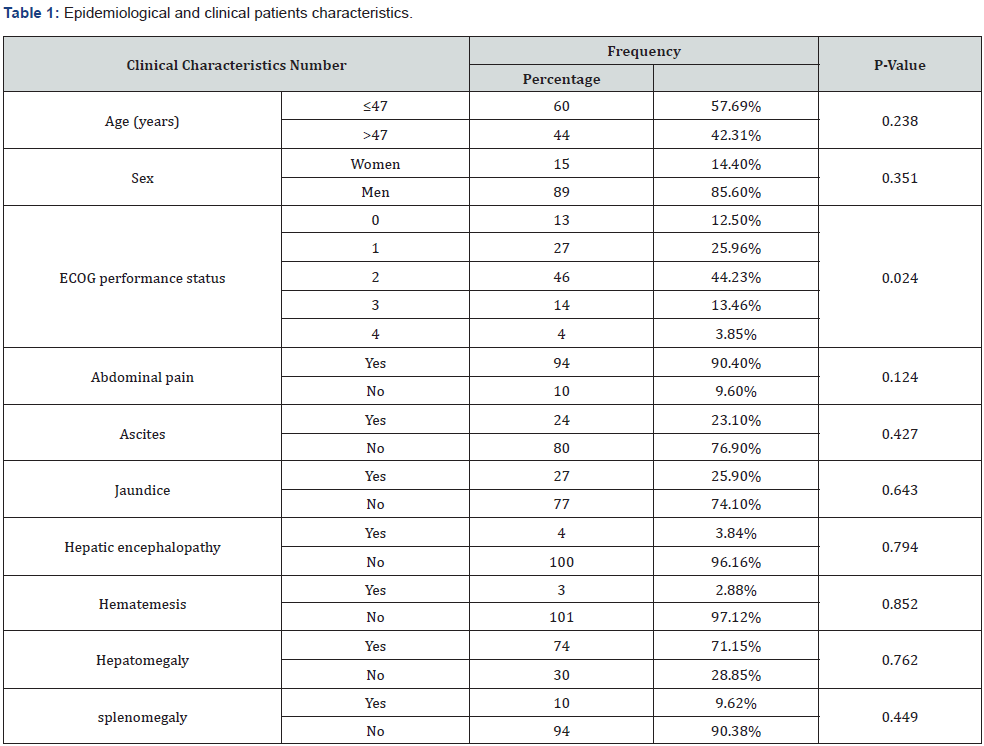
ECOG: Eastern Cooperative Oncology Group.
This study included 104 HCC patients. During the study period, 1393 consultations were recorded representing a prevalence of 7.46 %. There were 89 men (85.6%) and 15 women (14.4%). The gender ratio man for every woman was 5.9. The mean age was 46.7 years ranging from 18-78 years (Table 1).
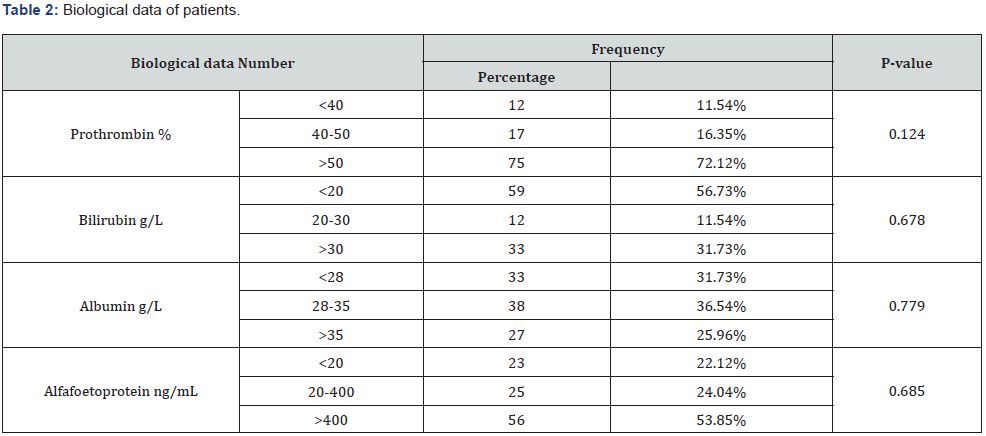
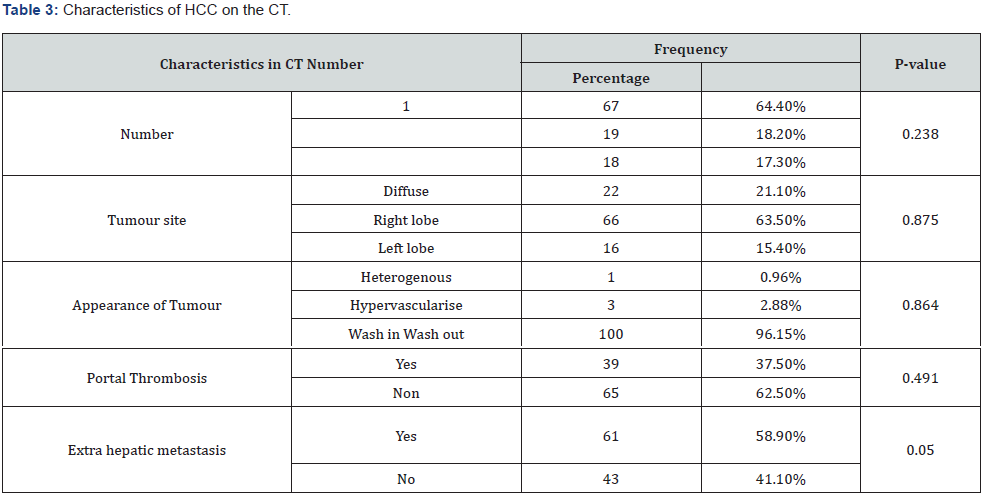
A family history of first-degree HCC was present in 11 patients or 10.6%. Alcohol consumption was present in 7 patients (6.7 %). The clinical signs were dominated by abdominal pain (90,4%), followed by jaundice (25.9%) %, ascites (23.1 %), hepatic encephalopathy (3.84%) %, hematemesis (2,88 %). Table 1 AFP levels were elevated in 81 patients (77.88 %) with a mean level of 12,217.6 ng/ml [13-298,586 ng/ml]. Twenty-Three patients (22.12 %) had normal AFP levels. Table 2 The diagnosis of HCC was made by CT in 99 patients (95.2%) and in five patients (4.8%) a biopsy was performed. The patients had one tumour in 64.4 % and several tumours in 35.6 %. The tumour was found in 63.5% of cases in the right lobe, in 15.4% of cases in the left lobe and in 21.1% of cases it was diffuse. Seventy-five (75%) patients had details on the diameter of the largest mass. Table 3 In patients with single tumour the mean nodule size was 13cm [ 2.3-20.8cm]. The mean size of the largest nodule was 10.8cm [2.4-22cm] in patients with multinodular tumours. Portal thrombosis was present in 39 patients (37.5 %%) and 61 patients (58,9 %) patients were metastatic. The pulmonary metastasis was present in 50.8 %, bone metastasis in 14.7% and pleural metastasis in 13.1 %. HCC occurred in a cirrhotic liver in 99 cases (95.2%). The Child Pugh score was A in 38.9%, B in 50% and C in 11.1%. In 69 cases (66.3 %) the cirrhosis was viral B cirrhosis, one case was viral C cirrhosis (0.96 %) and unspecified in 34 patients (32.74%). According to the Barcelona Liver Cancer Clinic (BCLC) classification, the patients were stage A in 1,9 %, B in 2.88 %, C in 70.2 % and D in 25.02 % . Left hepatectomy was performed in one patient classified BCLC A. Three patients classified BCLC C underwent unspecified liver surgery. Three patients classified BCLC B underwent TACE. The number of sessions and the interval were not specified. Sorafenib was prescribed in 2 patients classified BCLC C. Symptomatic treatment was administered in 98 patients (94.2%). The average follow-up time was 2.2 months [extremes: 1-13 months]. We noted 83 patients lost to followup and 21 patients died. The circumstances of the deaths were hepatic encephalopathy in 6 patients and upper GI bleeding in one patient. They were not specified in 14 patients.
Discussions
Hepatocellular carcinoma (HCC) is the most common liver cancer and represents more than 90% of primary liver cancers. HCC is a major global health problem, and he is responsible for high mortality. There are few studies on this disease in Senegal. This study analysed the data 104 HCC. In this study 85.6% of patients were male and 14.4% were female. Worldwide, HCC is predominant in male, especially in countries with a high prevalence. The predominance of men is reported in the publishing data with a ratio man for every woman that varied from 1.6 to 6.9 [4-6].
Several hypotheses have been proposed to explain the high male prevalence of HCC. Men are more exposed to carcinogens such as tobacco or alcohol. During B viral infection, sex hormones and their corresponding receptors (AR and ER-𝛼) play a role in the development of HCC [7]. Oestrogen appears to have a protective effect in women, whereas in men the androgenic signal may significantly increase the risk of developing HCC [7]. The age of onset of HCC varies across the world. In this study, most of patients were young adults with a mean age of 46.7 years [18-78 years]. In Senegal, a previous study found a mean age of 47 years [6]. In the literature, in sub-Saharan Africa, the average age varied between 46 and 49 years [5,8-10].
HCC develops earlier in sub-Saharan Africa than in other regions. An intercontinental longitudinal cohort study called BRIDGE of 18031 HCC patients found a mean age of 69, 65 and 62 years in Japan, Europe, and North America respectively. The age was younger in South Korea (57 years) and China (52 years) [11]. This difference in the age of onset of HCC is related to the heterogeneous distribution of risk factors. In sub-Saharan Africa and Asia (China and South Korea), B virus infection is the leading cause of HCC [6,9-12]. It is generally acquired during the perinatal period and during childhood with a frequent progression to chronicity and complications including HCC. In northern countries, alcohol and HCV are the main aetiologies [3]. A family history of HCC in the first degree was present in 10.6% of cases in our study. Studies in the USA and Italy showed that the risk of developing HCC in cases of a family history was multiplied by 4.1 [13] and by 2.38 [14] respectively. In the case of B viral infection, the presence of a family history of HCC in the first degree increases the risk of HCC. The cumulative risk of developing HCC with a family history of HCC and HbeAg positivity is as high as 40% [15]. A systematic search for a family history of HCC in patients with chronic viral hepatitis B helps to identify patients at high risk of HCC and to establish appropriate management.
In this study, abdominal pain was the most frequent sign (90.4 %). Our results were like those found by Diallo et al in Senegal (93.2%), Umoh et al in Gambia (94%) and Bekondi et al in Central African Republic (100%) [5,6,9]. The Algerian and the Mongolian studies reported lower rates of 64.6% and 15.3% respectively [16,17]. HCC remains asymptomatic for a long time. The presence of a clinical manifestation is most often indicative of advanced disease. In sub-Saharan Africa, diagnosis is often made at a late stage. This could be explained by several factors such as limited financial resources and the use of traditional medicine [5,18]. AFP levels were elevated in 81 patients (77.88 %) with a mean level of 12217.6ng/ml [13-298586 ng/ml]. Twenty-Three patients (22.12 %) had normal AFP levels. The studies carried in Africa often found a significant increase in AFP with a mean level that varied from 47 to 5675.8ng/ml. In northern countries, the authors reported a normal or low level. This difference can be explained by the diagnosis in Northern countries, is most often early, whereas in Africa it is usually late. In Africa at diagnosis, HCC is often large and AFP level increases with tumour size. But tumour can be large and not secrete a high level of AFP [19] and elevated serum AFP is not specific to HCC. Thus, in northern countries, AFP is excluded from the diagnostic strategy for HCC [19]. Nevertheless, the significant elevation of AFP remains an important diagnostic argument in our countries where the diagnosis is usually made at an advanced stage. In this study, 99 patients (95.2%) had cirrhosis. This result is in accordance with published studies who show that HCC develops in 85-95% of cases in cirrhotic patients [20]. Lower rates were found by Diallo et al in Dakar (57%) and Yang et al (66%) in sub-Saharan Africa [6,10]. Studies from Tunisia, Egypt and Italy found cirrhosis in 73.6%, 100% and 91.5% of cases respectively [4,10,21]. Cirrhosis is the main risk factor for HCC. The incidence rate of HCC in cirrhotic patients varies from 2 to 4% per year [20]. In the present study 69 patients (66.3 %) were HbsAg positive. Studies in sub-Saharan Africa reported proportions ranging from 36% to 75.5% [8-10]. In North Africa, North America and Europe, lower rates have been reported [10,11]. Viral hepatitis B and C are major factors in liver cancer. Africa is an area of high prevalence of viral B infection. Chronic hepatitis B progresses to cirrhosis in 8-20% of cases and then to HCC in 2-5% of cases [22]. However, HCC can develop in a non-cirrhotic liver in 20% of cases [23]. In this study, locoregional and distant extension of the disease was noted in 61 patients or 58.9%. Pulmonary metastases were more frequent according to the publishing data [24]. Yang et al found extra-hepatic metastases in 18% of cases in sub-Saharan Africa [10]. The BRIDGE study found 8% of metastases in North America, 4% in Europe and 2-10% in Asia [11]. The extension determines the therapeutic strategy for HCC. The high frequency of metastases in our study corroborates the late diagnosis observed in our countries. According to the BCLC classification patients were stage A in 1.9 %, B in 2.88 %, C in 70.2 %) and D in 25.02%. In studies from sub-Saharan Africa, the results were like ours, with majority of patients being in stages C and D. In North Africa, notably Algeria and Morocco, patients were most often in stages A and B. In Egypt and Tunisia, however, the results were close to ours, with most patients being in stages C and D [10,16]. Also, in Europe and North America most patients in the BRIDGE study were in stages C and D [11]. In Asia, notably Japan and Taiwan, the highest rates of patients diagnosed with early (BCLC 0 or BCLC A) and intermediate (BCLC B) stages of the disease were found, with percentages of 87 and 82% respectively [11]. In countries where diagnosis is made at an early stage, such as Taiwan and Japan, there are effective screening programmes for patients at risk [11]. In this study, curative treatment was carried out in only one patient. This was a hepatectomy. In studies conducted in sub- Saharan Africa, curative treatment was most often not indicated [5,10]. In North Africa, curative treatment was prescribed in 35% of cases in Egypt and in 10.6% of cases in Tunisia [4,10]. The BRIDGE study reported a higher prescription of curative treatment in North America, Europe, and Asia, respectively 36%, 44% and 42.5% [11]. The treatment strategy for HCC depends on the stage of the disease, which is most often assessed by the BCLC classification. In Africa, particularly south of the Sahara, late diagnosis is the main limitation to access to curative treatment. The average follow-up time for patients was 2.2 months [1-13 months]. The survival of the deceased patients was 5 months on average [2-13 months]. This confirms the poor prognosis of HCC [10]. Similarities were observed with the survival results reported by Yang et al in sub-Saharan Africa [10]. In North America, Europe and Asia, the BRIDGE study reported better survival rates with a 5-year survival rate ranging from 0.4% to 6.9%.
Our study has some limitations. The study was retrospective, and the number of patients was small. Our results need to be validated in future by long term multicentric clinical trial with larger numbers of patients.
Conclusion
HCC is a common disease in our country. It most frequently affects young adult males with viral B cirrhosis. Its prognosis is poor because most patients are diagnosed at an advanced stage of the disease and can usually only benefit from palliative treatment.
References
- https://gco.iarc.fr/today/home.
- Chan AWH, Zhong J, Berhane S, Toyoda H, Cucchetti A, et al. (2018) Development of pre and post-operative models to predict early recurrence of hepatocellular carcinoma after surgical resection. J Hepatol 69(6): 1284-1293.
- (2018) EASL Clinical Practice Guide: Management of hepatocellular Carcinoma. J Hepatol 69(1): 182-236.
- Bibani N, Trad D, Sabbah M, Ouakaa A, Elloumi H, et al. (2018) Facteurs prédictifs de survie au cours du carcinome hépatocellulaire. Tunis Med 96(6): 379-384.
- Umoh NJ, Lesi OA, Mendy M, Bah E, Akano A, et al. (2011) Aetiological differences in demographical, clinical and pathological characterictics of hepatocarcinoma in the Gambia. Liver Int 31(2): 215-222.
- Ibrahima Diallo, Bineta Ndiaye, Mouhamed Touré, Abdoul Sow, Ababacar Mbengue, et al. (2021) Hepatocellular carcinoma in Senegal: epidemiological, clinical and etiological aspects about 229 cases at Hospital Principal de Dakar. Pan African Medical Journal 38(99).
- Montella M, D'Arena G, Crispo A, Capunzo M, Nocerino F, et al. (2015) Role of sex hormones in the development and progression of hepatitis B virus-associated hepatocellular carcinoma. Int J Endocrinol. 2015: 854530.
- Nikièma Z, Sawadogo A, Kyelem CG, Cissé R (2010) Carcinomes hépatocellulaires en milieu africain burkinabé: contribution de l’échographie à propos de 58 cas. Pan Afr Med J 7: 10.
- Bekondi C, Mobima T, Ouavènè JO, Koffi B, Konamna X, Béré A et al. (2010) Etiopathologie du carcinome hépatocellulaire à Bangui, République centrafricaine: caractéristiques cliniques, biologiques et aspects virologiques des patients. Pathol Biol (Paris) 58(2): 152-155.
- Yang JD, Mohamed EA, Aziz AO, Shousha HI, Hashem MB, et al. (2017) Characteristics, management, and outcomes of patients with hepatocellular carcinoma in Africa: A multicounty observational study from the Africa Liver Cancer Consortium. Lancet Gastroenterol Hepatol 2(2): 103-111.
- Park JW, Chen M, Colombo M, Roberts LR, Schwartz M, et al. (2015) Global patterns of hepatocellular carcinoma management from diagnosis to death: the BRIDGE Study. Liver Int 35(9): 2155-2166.
- Kew MC (2010) Epidemiology of chronic hepatitis B virus infection, hepatocellular carcinoma, and hepatitis B virus-incluced hepatocellular carcinoma. Path Biol (Paris) 58(4): 273-277.
- Hassan MM, Spitz MR, Thomas MB, Curley SA, Patt YZ, et al. (2009) The association of family history of liver cancer with hepatocellular carcinoma: A case-control study in the United States. J Hepatol 50(2): 334-341.
- Turati F, Edefonti V, Talamini R, Ferraroni M, Malvezzi M, et al. (2012) Family history of liver cancer and hepatocellular carcinoma. Hepatology 55(5): 1416-1425.
- Harris PS, Hansen RM, Gray ME, Massoud OI, McGuire BM, et al. (2019) Hepatocellular carcinoma surveillance: An evidence-based approach. World J Gastroenterol 25(13): 1550-1559.
- Chikhi Y, Cheraitia S, Ould Gougam R, Lounes F, Zemmouchi C, et al. (2019) Wide Sexual Dimorphism of Hepatocellular Carcinoma Presentation in Algeria. Gastrointest Tumors 6(3-4): 122-136.
- Baatarkhuu O, Kim DY, Nymadawa P, Kim SU, Han KH, et al. (2012) Clinical features and prognosis of hepatocellular carcinoma in Mongolia: A multicentre study. Hepatol Int 6(4): 763-769.
- Ladep NG, Lesi OA, Mark P, Lemoine M, Onyekwere C, et al. (2014) Problem of hepatocellular carcinoma in West Africa. World J Hepatol 6(11): 783-792.
- Ding H, Tu H, Qu C, Cao G, Zhuang H, et al. (2021) Guideline for stratified screening and surveillance in patients with high risk of primary liver cancer (2020). Hepatoma Res 7: 17.
- Marrero JA, Kulik LM, Sirlin CB, Zhu AX, Finn RS, et al. (2018) Diagnosis, Staging, and Management of Hepatocellular Carcinoma: Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology 68(2): 723-750.
- Fenoglio L, Serraino C, Castagna E, Cardellicchio A, Pomero F, et al. (2013) Epidemiology, clinical-treatment patterns, and outcome in 256 hepatocellular carcinoma cases. World J Gastroenterol 19(21): 3207-3216.
- Bruix J, Sherman M (2011) American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology 53(3): 1020-1022.
- Desai A, Sandhu S, Lai JP, Sandhu DS (2019) Hepatocellular carcinoma in non-cirrhotic liver: A comprehensive review. World J Hepatol 11(1): 1-18.
- Zimmermann A (2017) Invasion Patterns and Metastatic Patterns of Hepatocellular Carcinoma. Springer International Publishing Switzerland 4: 91-107.






























