Occurrence of Extended Spectrum β-Lactamase and Carbapenemase Producing Enterobacteria Infection and Antimicrobial Susceptibility Patterns among HIV-Positive and HIV-Negative Individuals with Clinical Signs of Various Tract Infection in Western-Cameroon
Kouitcheu Mabeku Laure Brigitte1*, Efon Ekangouo Arnauld2 and Kouam Mewa Jeannette Euranie3
1Department of Microbiology, Medical Microbiology Laboratory, University of Yaoundé, Cameroon
2Department of Biochemistry, Faculty of Science, Microbiology and Pharmacology Laboratory, University of Dschang, Cameroon
3Regional Hospital Bafoussam, Cameroon
Submission:January 03 2022; Published:January 21, 2022
*Corresponding author:Kouitcheu Mabeku Laure Brigitte, Department of Microbiology, Medical Microbiology Laboratory, University of Yaoundé, Cameroon
How to cite this article:Kouitcheu M L B, Efon E A, Kouam ewa J E. Occurrence of Extended Spectrum β-Lactamase and Carbapenemase Producing Enterobacteria Infection and Antimicrobial Susceptibility Patterns among HIV-Positive and HIV-Negative Individuals with Clinical Signs of Various Tract Infection in Western-Cameroon. Adv Res Gastroentero Hepatol, 2022; 18(3): 555987. DOI: 10.19080/ARGH.2022.18.555987.
Abstract
Introduction :Resistance linked to the production of extended spectrum β-lactamase (ESBL) is a particular problem in the treatment of Enterobacteria infections. The situation can become alarming for the immune-compromised patients as HIV-positive individuals, who are at risk of repeated infections. The objective of this study was to determinate the prevalence of beta-lactamase producing Enterobacteria infection among HIV-positive individuals in Cameroon.
Material and Method :Based on the symptoms, urine, stool, blood, vaginal exudates and wound swab sample were collected from 204 HIV-negative and 104 HIV-positive individuals attending the Regional Hospital of Bafoussam-Cameroon from September 2016 to June 2017. Specimen were culture using dilution streak technique and Enterobacteria were isolated based on their morphological features, Gram staining and biochemical characteristics using API 20E galleries (Biomerieux, France). The antimicrobial susceptibility was determined by Kirby-Bauer disc diffusion test, double disk synergy and combination disk tests for beta-lactamase detection.
Results :The prevalence of Enterobacteria infection among HIV-positive individuals was 46.2% versus 27.5% among HIV-negative ones (P=0.0014). Resistance rate of 35.5% versus 27.7% was recorded among isolates from HIV-positive versus HIV-negative participants (P=0.0059). The prevalence of Enterobacteria harbouring β lactamase enzyme was 32.7% in HIV-positive subjects and 9.3% in HIV-negative ones (P <0.0001), with significantly higher prevalence of ESBL and carbapenemase producing Enterobacteria infection among HIV-positive participants (P=0.0215 and 0.0186 respectively).
Conclusion :Our findings showed that HIV positive patients are more likely to be infected by highly resistant organisms and had the highest carriage rate of ESBL, AmpC and carbapenemase producing Enterobacteriaceae in Western-Cameroon.
Keywords:Extended-spectrum beta-lactamase producing Enterobacteriaceae; Carbapenemase-producing Enterobacteriaceae; HIV infection; Cameroon
Abbreviations:Pen TEM: Penicillinase TEM; Pen RI: Penicillinase Resistant To Inhibitors ; ESBL: Extended Spectrum Beta Lactamases ; AmpC: AmpC Beta Lactamases ; Carbap A: Carbapenemase Class A ; MBL: Metallo Beta Lactamases ; E-ESBLs: Extended-Spectrum Beta-Lactamase-Producing Enterobacteriaceae ; CPE: Carbapenemase-Producing Enterobacteriaceae
Introduction
Human immunodeficiency virus (HIV) particularly attacks and kills CD4 T cells, resulting in the dysfunction and deregulation of the immune system and ultimately AIDS.1 Because of the weakened immune system, HIV-positive individuals without ART can no longer ward off opportunistic infections [1-3]. Several studies showed that Enterobacteriaceae infections are widely increased in HIV positive individuals [4-8]. Enterobacteriaceae infection in immune compromised individual are associated with considerable morbidity, mortality and associated health care costs [9]. In such patients, the spectrum of Enterobacteriaceae infection is diverse, ranging from asymptomatic colonization to gastroenteritis,meningitis, wound infections, urinary tracts infections, septicaemia and urinary tract infections. Enterobacteriaceae are now recognized as one of the most important threat for public health, due to the ambiguous resistance of these strains even to last classes of antibiotics. For example, some Enterobacteriaceae produce extended-spectrum beta-lactamases (ESBLs) which break down antibiotics such as penicillins and cephalosporins and alter their activity [10], thus making infections caused by ESBLproducing Enterobacteriaceae more difficult to manage.
Studies on the prevalence of Enterobacteriaceae infection among HIV-positive individuals and their susceptibility patterns to antimicrobial agents have been conducted in some African countries [11,12]. Concerning resistance mechanism, few studies on the prevalence of extended spectrum β-lactamase (ESBL) and carbapenemase producing members of Enterobacteriaceae (CPE) have been documented [13-15], despite the fact that Sub-Saharan Africa is the epicenter of the HIV epidemic [16].
Growing concerns exists over the prevalence of HIV infection in Cameroon. The prevalence of HIV infection in Cameroon has progressively risen from 0.4% in 1987 to 1.2% in 1990 and from 4% in 1992 [17] to about 7% in 1997 and 11% in 2000 [18]. In 2004, with the gratuitously of the antiretroviral treatment, the national prevalence based on a Demographic and Health Survey decreases to 5.5% [19]. In 2018-2020, prevalence rate of HIV infection was estimated at 4.3% with women and children the most vulnerable subjects [20]. In fact, the mother-to-child transmission rate of 14% is still too high, while the antiretroviral treatment rate among children and adolescents remains extremely low (13%) [20]. In addition, the mortality rate linked to HIV/AIDS continues to rise among adolescents [20]. Despite this great negative impact of HIV infection in Cameroon, there is a lack of data concerning the resistance pattern to antimicrobial agents among HIV-positive individuals. Also data on the prevalence of ESBL-E and CPE infection among these immuno-compromised subjects is scarce.
To address these knowledge gaps, the present study is intended to estimate the prevalence of Enterobacteriaceae infection among HIV-positive individuals attending the Regional Hospital of Bafoussam, Western Cameroon, to elucidate the antimicrobial susceptibility patterns of Enterobacteriaceae isolated from these individuals to β-lactam antibiotics and also to detect among these isolates those harbouring β-lactamase enzyme.
Material and Methods
Study design and setting
This cross sectional study was conducted at the Bafoussam Regional Hospital, in the western region of Cameroon. The study population consisted of HIV-positive individuals and HIV-negative individuals attending the selected health facility from September 2016 to June 2017 and presenting signs and symptoms of various infections. The detected type of infections were bacteremia, wound infections and those from the urinary, genital and digestive tract. We employed a consecutive sampling for data collection, requesting consent from all volunteer patients (or from children’s parents or guardian if participant was a child or an adolescent) who fulfilled the eligibility criteria of the study. Exclusion criteria were
a) Patients under antibacterial therapy within the last two weeks
b) Patients with mixed types of infection
c) Non-cooperative patients who refused to give their consent or to participate to the study
d) HIV-positive and HIV-negative individuals who were severely ill and unable to provide sample. Pregnant and breastfeeding women were also excluded from the study.
Variables
The socio-demographic data (age, gender) as well as others factors (previous history of antibiotics treatments, diagnosis for opportunistic infections) were requested from the volunteer subjects in a structured questionnaire. HIV test was conducted for every volunteer eligible participant and they were then divided according to HIV status as HIV-positive individuals and HIVnegative individuals.
Research ethics and patient consent
The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Institutional Review Board of the Regional Hospital of Bafoussam (Ref. 1853/L/ MINSANTE/SG/DRSPO/HRB/D) and the Centre Regional of Ethics Committee for Human Health Research (CRERSHC) (Ref. 00618/ CRERSHC). Each patient provided written informed consent to participate in the study. Children were enrolled after their parents or legal guardians provided a written informed consent.
Sample collection
Due to the fact that, Enterobacteriaceae affects several systems of the body, various clinical specimens were collected based on symptoms or clinical signs presented by participants. Patients with mixed types of infection were excluded from the study, so one sample type was collected per participant at the location that correlated with symptoms. They were midstream urine sample for participants with clinical signs of urinary tract infection, stool sample for those with gastrointestinal disorders, blood for those with clinical signs of bacteraemia,vaginal exudates for those with clinical signs of genital tract infections and wound swab for those with wound infections.
Participants with urinary or digestive tract infection were instructed to collect stool and urine samples aseptically by the health personnel and were provided with sterile containers labelled with unique identification numbers for the collection of specimens. The collection of vaginal exudates, wound swab and blood samples was done under aseptic conditions by well trained personnel of the hospital. For patients with clinical signs of bacteraemia, 2ml of blood was collected into a clean test tube. All the collected samples were processed for culture and the result were analyzed according to HIV status of participants.
HIV infection detection
For each participant, two milliliters (2ml) of venous blood was drawn for HIV serology detection. The blood was transferred into a clean test tube and allowed to clot naturally by standing the sample at room temperature for 10mn. Clear serum sample was obtained by spinning the tubes at 3000rpm for 5mn and used for HIV serology detection using OnSite HIV-1/2 Ab plus Comb Rapid Test-Cassette kit (CTK BIOTECH, San Diego USA).
Culture and identification of Enterobacteriaceae
The Clinical specimens were plated on solid cultures media; urine samples, vaginal swab and wound swab on Mac Conkey agar and blood agar, stools samples on Salmonella/Shigella (SS) agar and Mac Conkey agar, and blood samples on blood agar. Fecal samples were dissolved in sterile physiological water to obtain a 10% diluted solution and the mixture was homogenized using a Vortex mixer before culture.
Each specimen was culture using dilution streak technique. The first quadrant of the plate was streaked using a spot of the collected sample and each successive quadrant was streaked using a new bacteriologic loop in order to dilute the number of bacteria in each quadrant. Inoculated plates were incubated at 37oC for 24 hours. Quantification was expressed as 0, 1+, 2+, 3+, or 4+ based on the number of quadrants with bacterial growth: 0 for no bacterial growth, bacterial growth limited on quadrant 1 was taken as 1+, bacterial growth on quadrants 1 and 2 as 2+, bacterial growth on quadrants 1, 2, and 3 as 3+, and bacterial growth on all the 4 quadrants as 4+. Enterorobacteria in a sample was considered as a colonizer or a pathogen according to the extent of the bacteria growth to quadrant. Enterobacteria growth even only on quadrant 1 in blood sample was taken as pathogen. For others clinical samples, Enterobacteria growth of categories 3+ and 4+ was taken as pathogen and Enterobacteria growth less than the above limits were taken as a colonizer. Participant which sample demonstrated Enterorobacteria growth considered as pathogen was taken as infected and non-infected those with Enterorobacteria growth considered as a colonizer [21].
Isolated colonies from culture sample of participants considered as infected were processed for sub-culturing and identified as Enterobacteriaceae species based on their morphological features, Gram staining and using API 20E galleries (Biomerieux, France).
Antibacterial susceptibility testing
All the isolates were subjected to antibiotic sensitivity test. Susceptibility of isolates to antibiotics was evaluated on Mueller Hinton Agar (MHA) using Kirby-Bauer disc diffusion method according to the Clinical and Laboratory Standards Institute guidelines 2016 [22]. The antibiotic tested (Bioanalyse) were: Amoxicillin (25μg); amoxicillin + clavulanic acid (25/10μg); Piperacillin (100μg); Piperacillin + tazobactam (100/10μg); Cefuroxim (30μg); Cefuroxim + sulbactam; Cefotaxim (30μg), Ceftazidim (30μg); cefoxitin (30μg); Aztreonam (30μg), Imipenem (10μg). The tested Enterobacteriaceae species were classified as sensible, intermediate and resistant according to the cut-off point value of the inhibition zone recommended by the Clinical and Laboratory Standard Institute 2017 [23].
Screening for potential ESBL-producing isolates
The isolates that showed an inhibition zone size of ≤ 22 mm with ceftazidim (30μg) and/or ≤ 27 mm with cefotaxim (30μg) were considered as potential ESBL-producer and were selected for confirmation of ESBLs production. E. coli (ATCC 25922) and Klebsiella pneumoniae (ATCC 700603) were used as ESBLs negative and positive reference strains respectively.
Confirmation of extended spectrum β-lactamase (ESBL) and AmpC production
Isolates resistant to cephalosporin of the 3rd generation (cefotaxim, ceftazidim) or to monobactam (aztreonam) in antibiotic sensitivity test were tested for ESBL and AmpC production by the Double Disc Synergy Test [24].
In a lawn culture of the strain into Mueller Hinton agar plate (MHA), amoxicillin/clavulanic acid (amoxyclav) disc (20μg+10μg) was placed in the centre of petri dish and cefotaxim (30μg), ceftazidim (30μg) and aztreonam (30μg) were placed on either side of amoxicillin/clavulanic acid disc at a distance of 18 to 20mm. A disc of cephalosporin of the 4th generation; cefoxitin (30μg) was also placed at a distance of 15mm from cefotaxim and ceftazidim disc. Plates were incubated at 37°C for 16 to 18 hours. Organism which showed extension inhibition zone of cefotaxim or ceftazidim or aztreonam towards amoxyclav disc was taken as ESBL screen positive. Blunting of inhibition zone of ceftazidim towards cefoxitin was taken as AmpC screen positive. Blunting of zone of inhibition of ceftazidim towards amoxyclav was taken as inducible AmpC positive.
Screening and confirmation for carbapenemase producing isolates
Isolates selected for this test were those known as ESBL producers from the above screening and those which were not susceptible to imipenem in antibiotic sensitivity test. Confirmation of the carbapenemase class A production was evaluated using Double Disc Synergy Test [24]. In a lawn culture of the selected isolates, amoxicillin/clavulanic acid (20μg+10μg) and imipenem (10μg) discs were placed in petri dish at a distance of 20mm from each other. Plates were incubated at 37°C for 16 to 18 hours. Organism which showed extension of inhibition zone of imipenem towards amoxyclav disc was taken as carbapenemase class A screen positive.
Phenotypic detection for Metallo beta Lactamases (MBL) or carbapenemase class B production was done by combination disc test using discs of Ceftazidim (30μg) and imipenem (10μg) alone and those combined with EDTA [25]. The stock solution of EDTA (Sigma-Aldrich, Germany) was prepared by dissolving anhydrous EDTA in distilled water at a concentration of 0.1M. From this solution, 10μl was dispensed onto Ceftazidim (30μg) and imipenem (10μg) discs. The discs were dried and used within 60 minutes. The test was performed by inoculating the test organism on Mueller Hinton Agar and placing one disc of Ceftazidim (30μg) and imipenem (10μg) without any inhibitor and two discs of Ceftazidim (30μg) and imipenem (10μg), each containing EDTA. The agar plates were incubated at 37°C overnight. The diameter of the growth inhibitory zone seen around the Ceftazidim (30μg) and imipenem (10μg) disc with EDTA was compared with that seen around the plain Ceftazidim (30μg) and imipenem (10μg) disc [25]. Regardless of the diameter zone, a 5mm increase in zone diameter of Ceftazidim (30μg) and imipenem (10μg) tested in combination with EDTA versus Ceftazidim (30μg) and imipenem (10μg) zone size when tested alone was taken as carbapenemase class B or Metallo beta lactamases production [25].
Statistical analysis
Data were analysed using IBM SPSS Statistics 20. The association between variable and the outcome was done taken HIV-negative participants as reference group. We tested for association in categorical variables such as prevalence of enteric bacterial infection, rate of Enterobacteria positivity per sample type, frequency of enteric bacterial species, resistance rate, prevalence of β lactamase producing enteric bacteria among HIV-positive and HIV-negative individuals using the chi-square test or fisher’s exact test, reporting corresponding p-values. The level of statistical significance for the study was set at p < 0.05. The outcome measure was the detection of the presence of Enterobacteriacea among clinical samples from HIV-positive and HIV-negative participants with clinical signs of various tract infection, the susceptibility of the isolated Enterobacteriaceae to β-lactam antimicrobial agents and the detection of beta-lactamase and carbapenemase enzyme production.
Results
Prevalence of Enterobacteriaceae infection in the study population
The study included three hundred and eight (308) participants with clinical sign of infection, their age ranged from 1 to 85 years with mean 36.5±16.6 years. From these participants, 49.4, 34.1 and 9.5% were with clinical signs of urinary, genital and gastrointestinal tract infection respectively, while 4.6, and 2.6% of them were with those related to wound and blood infection. Participants were made up of 104 HIV-positive individuals (64 females and 40 males, mean age 35.64 ± 15.87 years) and 204 HIVnegative individuals (116 females and 88 males, mean age 34.18 ± 16.48 years) (Table 1).
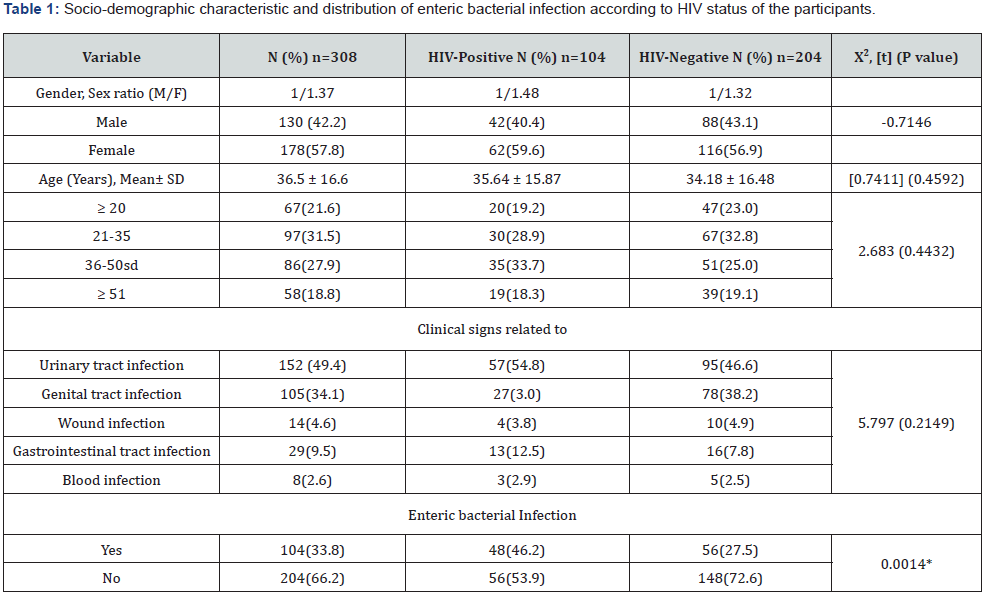
From these participants, 308 non-repetitive clinical specimen were collected based on the symptoms. Urine samples, which constituted 49.4% of the overall collected specimens were the most present, following by vaginal swab (34.1%) (Table 2). Out of the 308 participants enrolled, 104 were with sample positive or sample that demonstrated enteric bacterial growth quantification to be consider them as Enterobacteria infected subjects, given an infection prevalence of 33.8% in our sample population. Out of the 104 HIV-positive individuals, 48 (46.2%) were with positive sample versus 56 out of the 204 (27.5%) among HIV-negative individuals. This difference was significant (P=0.0014).
The proportion of positive sample per sample type was as follow: 62.1, 50.0, 50.0, 30.9 and 26.7% respectively for stool, wound swab, blood, urine and vaginal swab samples. Regarding the proportion of positive sample among sample type according to group of participants; excepted stool (46.2 vs 75.0), urine (40.4 vs 25.3%), vaginal swab (55.6 vs 16.7%), blood (66.7 vs 40.0) from HIV-positive participants were with a higher risk of Enterobacteria positivity compared to sample from HIV-negative individuals. But the different was statically significant only with vaginal swab sample (p= 0.0002) (Table 2).

Frequency of Enterobacteriaceae isolates
A total of 104 consecutive, non-repetitive clinical isolates of Enterobacteriaceae were recovered from the samples. Fourteen Enterobacteria species were identified among the overall isolates in variable proportion; Escherichia coli (36.5%), Klebsiella pneumoniae (8.7%), Klebsiella rhinoscleromatis (1.9%), Klebsiella oxytoca (7.5%), Serratia odorifera (5.8%), Serratia mascecens (4.8%), Enterobacter aerogenes (4.8%), Enterobacter cloacae (4.8%), Enterobacter agglomerans (3.8%), Citrobacter freundii (7.7%), Proteus mirabillis (2.9%), Proteus vulgaris (7.7%), Salmonella spp (2.9%), and Providencia rettgeri (1.0%) (Table 3). Among the overall identified species, 48 were from the 104 HIVpositive participants (46.2%) and 56 from the 204 HIV-negative ones (27.5%). Escherichia coli was the most predominant species among both HIV-positive (16.4%) and HIV-negative individuals (10.3%). This species was followed by Klebsiella oxytoca (4.8%), Proteus vulgaris (4.8%), Enterobacter aerogenes (3.9%) and Citrobacter freundii (3.9%) among HIV positive individuals and by Klebsiella pneumonia (2.9%), Serratia odorifera (2%), Enterobacter agglomerans (2%) and Citrobacter freundii (2%) among HIV negative individuals.
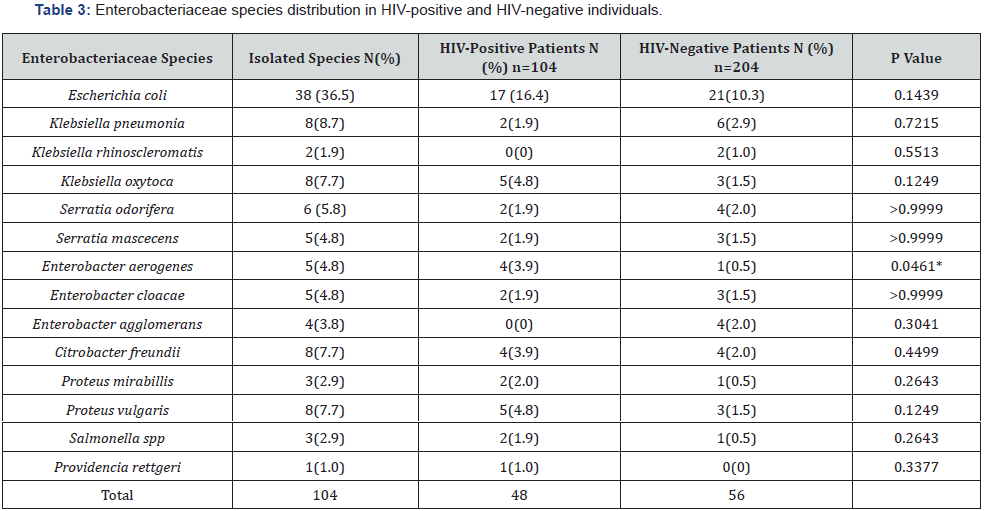
n: Number, *: Significants
The distribution of the identified species according to HIV status of the participants showed that Escherichia coli, Klebsiella oxytoca, Enterobacter aerogenes, Citrobacter freundii, Proteus spp and Salmonella spp were highly accounted among isolates from HIV-positive than HIV-negative participants, but the difference was significant only with Enterobacter aerogenes (p=0.0461) (Table 3). Some species were specific for a group of participants, Providencia rettgeri in HIV-positive participants, Klebsiella rhinoscleromatis and Enterobacter agglomerans in HIV-negative participants (Table 3).
Antibiotic resistance pattern of Enterobacteria isolates according to HIV status
Resistance rate ranged from 4.2 to 72.9% among isolates from HIV-positive participants and from 0.0 to 73.2% among those coming from HIV-negative ones. The overall resistance rate were 27.7 and 35.5% respectively among isolates from HIV-negative and HIV-positive participants. This difference was significant (P= 0.0059), indicating that HIV-positive subjects are more in risk to develop resistance to antibiotics than HIV-negative individuals.
When examining resistance to single antibiotic, we noticed that with the exception of Piperacillin + Tazobactam (4.2 vs 10.7%), higher resistance rate was detected among isolates from HIV-positive participants compared to those from HIV-negative participants. Resistance rate of 10.4 vs 0.0%, 33.33 vs 25% , 37.5 vs 10.7%, 41.7 vs 35.75, 58.3 vs 35.7% and 50 vs 35.7% were noticed against imipenem, cefotaxim, cefoxitin, piperacillin, ceftazidim and cefuroxim respectively among isolates from HIV-positive vs HIV-negative participants. This difference was significant regarded imipenem (P = 0.0186), cefoxitin (P = 0.002) and ceftazidim (P = 0.0298). Resistance rate detected against amoxicillin, amoxicillin/ clavulanic acid and aztréonam were independent of the HIV status of patients. Resistance rate against imipenem and cefuroxine/ sulfobactam were detected only among isolates from HIV-positive patients (Table 4).
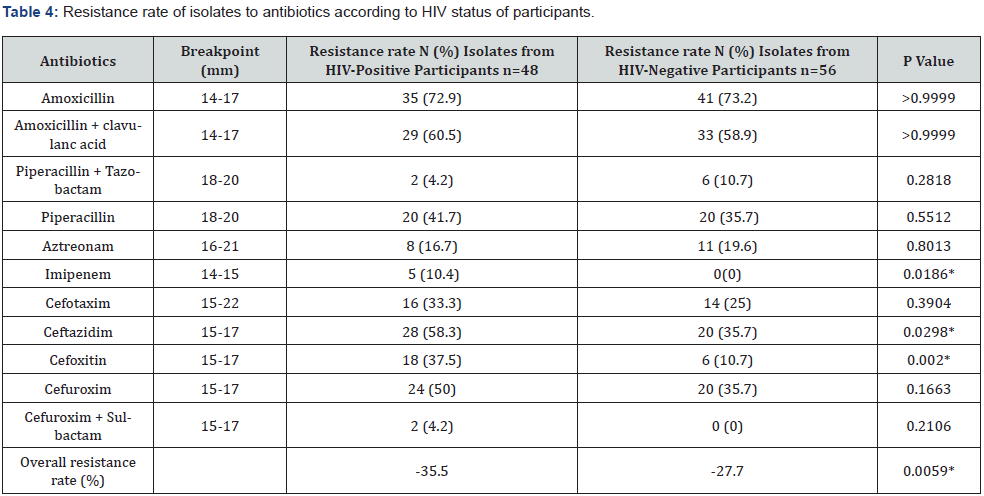
n: Number, *: Significant.
Magnitude of ESBL, AmpC, and Carbapenemase producing Enterobacteriaceae
Of all the enteric bacteria isolates, 25% (26/104) were positive for the screening test of ESBL production. For all the isolates detected as ESBL producer, E. coli accounted for 34.6% (9/26), Enterobacter spp for 15.4% (4/26), Klebsiella spp, Proteus spp and Citrobacter freundi for 11.5% (3/26) each. Only P. rettgeri was not detected as ESBL producer.
The overall prevalence of AmpC producing Enterobacteria infection among the isolates was found to be 21.2% (22/104). For all the isolates detected as AmpC producer, E. coli accounted for 40.9% (9/22) followed by Enterobacter spp 18.2% (4/22)and Klebsiella spp. 18.2% (4/22). P. rettgeri and Salmonella spp were not detected as AmpC producers. Approximately 5% of isolates (4.8%; 5/104) were detected as Carbapenemase producing Enterobacteria, 1.9% (2/104) for class A and 2.9% (3/104) for class B or MBL. E. coli and Klebsiella pneumonia were the only carbapenemase producing Enterobacteria detected, in the proportion of 80% and 20% respectively of the overall carbapenemase isolates producer detected (Table 5).
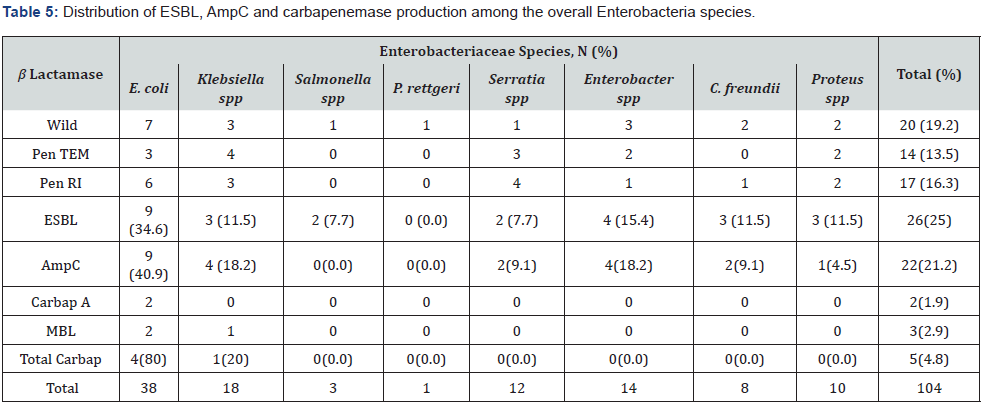
N: Number, Pen TEM: Penicillinase TEM, Pen RI: Penicillinase Resistant To Inhibitors, ESBL: Extended Spectrum Beta Lactamases, AmpC: AmpC beta Lactamases, Carbap A: Carbapenemase class A, MBL: Metallo Beta Lactamases. E. colI: Echerichia coli, P. rettgeri: Providencia rettgeri, C.fruendii: Citrobacter freundii.
The overall prevalence of β lactamase enzyme producing Enterobacteria infection was 17.2% (53/308) in our sample population, 8.4% (26/308), 7.1% (22/308) and 1.6% (5/308) respectively for ESBL, AmpC, and Carbapenemase enzyme. This prevalence of infection varied significantly (p<0.0001) according to HIV status of the participants with the pic of infection with these pathogens among HIV-positive participants. In fact, the prevalence β lactamase enzyme producing enterobacteria infection were 32.7 % (34/104) and 9.3% (19/204) respectively among HIVpositive and HIV-negative participants (p<0.0001), suggesting that Enterobacteria harbouring this enzyme were more present among HIV-positive subjects than HIV-negative ones.
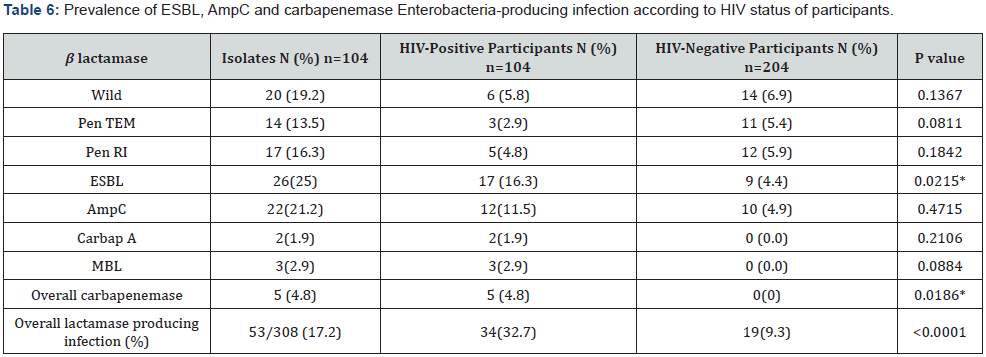
n: Number, *: Significant, Pen TEM : Penicillinase TEM, Pen RI : Penicillinase resistant to inhibitors, ESBL : Extended Spectrum Beta Lactamases, AmpC: AmpC beta Lactamases, Carbap A: Carbapenemase class A, MBL: Metallo beta Lactamase
Considering each individual β lactamase enzyme, the higher prevalence of infection with organism-producer was noticed among HIV-positive individuals compared to HIV-negative individuals. ESBL, AmpC, carbapenemase class A, carbapenemase class B (MBL) and total carbapenemase producing Enterobacteria infection prevalence of 16.3 vs 4.4%, 11.5 vs 4.9%, 1.9 vs 0.0%, 2.9 vs 0.0% and 4.8 vs 0.0% were noticed respectively among HIV-positive individuals vs HIV-negative ones. The difference was significant regarded ESBL (P = 0.0215) and total carbapenemase (P = 0.0186) producing Enterobacteria (Table 6).
Discussions
Enterobacteriaceae are a large family of bacteria that commonly cause infections both in healthcare settings and in communities. Enterobacteriaceae infections are common and more likely to cause invasive conditions in HIV-positive individuals than healthy ones [2]. The absence of these data in our population has hinder the better understanding of the harmful effect of HIV infection in our society and also slow down the planning for resources which can enable us to best fight against this disease. Thus, the present study have evaluated the prevalence of Enterobacteria infection, the antimicrobial susceptibility patterns and the prevalence of β-lactamase enzyme producing enteric bacterial infection among HIV-positive individuals compared with HIV-negative ones in a healthcare setting.
On the overall samples currently collected, urine (49.4%) and vaginal swab (34.1%) were the most frequent. Such observation indicate that affections of urinary and genital systems are the most common health complain in our milieu. Our data shown that HIVpositive individuals (46.2%, 48/104) are significantly more prone to Enterobacteria infection than HIV-negative ones (27.47%, 56/204), (P=0.0014). A compromised immunity resulting from HIV infection could help to understand the vulnerability of HIVpositive individuals to infection. Additionally, others risks factors to which HIV-positive individuals are commonly exposed such as prolonged hospitalization, intensive-care unit stay could also explained the positive relationship between HIV infection and high positivity to Enterobacteria infection. Our finding is in accordance with previous studies reporting that rates of Gramnegative bacterial enteric infections are at least 10-fold higher among HIV-positive adults than in the general population [3-6].
Approximately 34% (33.8%) of the overall sample collected were Enterobacteria positive. When examining the proportion of sample positivity according to sample type, we noticed that stool sample (62.1%) was the one with the highest positivity. This is true, since Enterobacteria are commensal enteric flora usually not pathogenic, but which in certain conditions can cause various infections such as gastroenteritis [26]. Some Enterobacteria infections of the digestive tract may lead to watery or bloody diarrhea in infected patients or necrotizing enterocolitis in infected newborn infants [27]. Urine (p= 0.069), vaginal swab (p= 0.0002) and blood samples (p>0.9999) from HIV-positive participants were with a higher risk of Enterobacteria positivity compared to sample from HIV-negative individuals, suggesting that enteric bacterial infections of the urinary, genital and blood systems are common in HIV-positive individuals compared to HIVnegative ones. Our finding is in accordance with previous reports showing that, among opportunistic infections of AIDS defined illness, urinary tract infections accounts for 60% [28]. The high positivity of enteric bacteria in blood of HIV-positive individuals compared to HIV-negative ones seem to be real, since among HIV-positive subjects the most prevalent enteric infection such as salmonellosis and shigellosis develops in more than 40 [29] and 50% [30] of cases respectively into bacteremia. Bloodstream infection (BSI) is a frequent complication found in HIV-positive individuals and is usually associated with a poor prognosis, responsible for the immediate cause of death in up to 32% of HIVpositive individuals [31].
After considering the colony morphology and biochemical characteristics, our finding revealed that Escherichia coli, Klebsiella oxytoca, Enterobacter aerogenes, Citrobacter freundii, Proteus spp and Salmonella spp were highly accounted among isolates from HIV-positive than HIV-negative participants, with a significant difference regarded Enterobacter aerogenes (p = 0.0461). Our findings concerning the commonest of Salmonella and Escherichia coli species among enteric bacteria isolates from HIV-positive individuals agree with previous finding. In fact, it is reported that Salmonella, Shigella, Campylobacter and Escherichia coli are the most common enteric bacterial pathogens in HIV-positive individuals [42] and etiologic agents with potential cause of severe illness among them [32]. Additionally, 66.7% of the overall Salmonella spp isolates were recorded from HIVpositive individuals. Our findings correlated with previous reports that HIV-positive individuals have a higher risk of salmonellosis than healthy individuals [29]. The present rate of Salmonella infection among HIV-positive individuals (1.9%) is similar to that reported in Peru (1%) [36] and Southern Ethiopia (2.8%) [33], but rather lower than a result from Southern Ethiopia (5.1%) [34] and Uganda (8.1%) [37]. In the present study, blood sample were not cultured on specific medium for Samonella spp. This may be the reason of the discrepancy between the current rate of Salmonella isolate and those of the previous studies, since in immune compromised individuals, salmonellosis mostly develops into bacteremia [30].
Resistant of bacteria to common antibiotics reduce the efficacy of these drugs to treat these infections and increase the frequency of therapeutic failure. The situation can become alarming for the immune-compromised patients as HIV-positive individuals, who are at risk of repeated infections. In the present study, resistance rate of 27.7 and 35.5% were detected respectively among isolates from HIV-negative and HIV-positive participants (P = 0.0059), indicating that HIV-positive subjects are more in risk to develop resistance to antibiotics than HIV-negative participants. Similarly, previous studies revealing higher resistance rate to antimicrobials among enteric bacterial coming from HIV-positive individuals has been documented [43,44].
Regarding resistance to single antibiotic, significant higher resistance rate was detected against imipenem (P = 0.0186), cefoxitin (P = 0.002) and ceftazidim (P = 0.0298) among isolates from HIV-positive participants compared to those from HIVnegative participants. Moreover, resistance rate against imipenem and cefuroxine/sulfobactam were detected only among isolates from HIV-positive participants. Recurrent used of antimicrobials and long term exposure to antimicrobials in order to cure or to prevent opportunistic infections in HIV-positive subjects may lead to the emergence of multidrug-resistant bacteria [45]. In fact, co-trimoxazole is recommended to HIV-positive adults and children born to HIV-positive women as prophylaxis measures against opportunistic infection [46]. The continuous exposure to this drug has result to the emergence of co-trimoxazoleresistant bacteria in HIV-positive population and the therapeutic failure against bacterial infections. This highlights the need for susceptibility testing of Enterobacteria isolates prior to therapy initiation, since antibiotics given empirically without proper antibiotic susceptibility testing, a clinical common practice in our milieu is one of the major cause in the clonal selection of resistantorganisms.
β-lactam antibiotics such as penicillins, cephalosporins and carbapenems are the most commonly used antibacterial drugs. The predominant drug resistance mechanism against β-lactam antibiotics among Gram-negative bacteria is the production of β-lactamase enzymes. Most important β-lactamase enzymes are extended spectrum β-lactamases (ESBLs), AmpC β- lactamase (AmpC) and Metallo β-lactamase (MBL). In this study, bacterial isolates from HIV -positive participants were highly resistant to cephalosporins (cefotaxim, ceftazidim, cefoxitin, cefuroxim) and lowly to cefuroxine/sulfobactam and piperacillin/tazobactam, characteristics which are specific to extended spectrum β-lactam antibiotics producing microorganisms. Few of them were also resistant to imipenem. Thus, the magnitude of extended-spectrum beta-lactamases (ESBLs) and carbapenemase production were evaluated among these isolates according to HIV status of the participants.
Our data showed that 25% (26/104), 21.1% (22/104) and 4.8% (5/104) of the isolated Enterobacteriaceae were found to be ESBL, AmpC and carbapenemase producing bacterial infection independently of the HIV status of the participants. The highest frequency of ESBL and AmpC production was observed among E. coli, followed by Enterobacter spp whereas carbapenemase producing Enterobacteriaceae were predominantly E. coli and Klebsiella pneumonia (Table 5).
The overall prevalence of infection with β lactamase enzyme producing member was 17.2% (53/308) in our sample population, 8.4% (26/308), 7.1% (22/308) and 1.6% (5/308) respectively for ESBL, AmpC, and Carbapenemase producing Enterobacteriaceae. Enterobacteria harboring β lactamase enzyme were more present among isolates from HIV-positive subjects (32.7%) than HIVnegative ones (9.3%), (P <0.0001). Taking individual lactamase enzyme, significantly higher prevalence of infection with ESBL (16.3 vs 4.4%, P= 0.0215) and carbapenemase (4.8 vs 0.0%, P= 0.0186) producing Enterobacteria were noticed among HIVpositive vs HIV-negative participants (Table 6).
Among the Enterobacteria isolates from HIV–positive participants, 35.4, 25 and 10.4% respectively were ESBL, AmpC and carbapenemase producing enteric bacterial infection. The current findings on the prevalence of extended spectrum β-lactamases producing Enterobacteria among HIV infected patients (35.4%) is comparable with studies from Ethiopia like Jimma (38.4%) [47], and Harar (33.3%) [48]. However this prevalence was higher compared with that documented in studies from others parts of Ethiopia (25%) [49], (21.4%) [50] and Saudi Arabia (22%) [51] and far higher than reports in the United States (8.6%) [52] and the United Kingdom (1%) [53]. On the other hand, it was lower than reports from different African countries such as Ghana (49.3%) [54], Addis Ababa (52%) [55] and Uganda (62%) [56]. This variation might be due to the difference in sampling population, the policy of antibiotics prescription, as well as sociocultural and economic factors.
In Cameroon, few studies on the extent of E-ESBL circulating strains among the general population have been investigated but none among HIV-positive individuals. A ESBL carriage of 12% and 16% was reported among enteric bacteria isolates respectively in HIV-negative patients in Yaoundé and in the community of Ngaoundéré [57,58]. Approximately 45% (45.3%) enteric bacteria produced ESBL was isolated from women with urinary tract infections in Yaoundé-Cameroon prior to antibiotics use [59]. The discrepancy in the prevalence of extended spectrum β-lactamases producing Enterobacteria of this study compared with the aforementioned reports may be due to the difference in sampling population. In fact, HIV-positive individuals are in high risk for ESBL carriage because they are commonly subjected to infections and hospitalization, and more likely to consume antimicrobial agents than HIV- negative persons. This may also justify the present high prevalence of AmpC (25%) and carbapenemase (10.4%) producing Enterobacteria infection among HIV-positive participants compared to HIV-negative ones. Carbapenems are antibiotics usually used to treat extendedspectrum and AmpC β-lactamases producing pathogens [60]. However, the spread of enzymes that break down these antibiotics has been reported [61]. This situation may further compromise therapeutic alternatives using every beta-lactam antibiotics but also those using non-beta-lactam, since carbapenemase mediated resistance are usually transposon- and/or integron-encoded determinants which can easily disseminated [62]. The use of appropriate infection prevention practices via minimizing risks of β lactamase producing organisms transmission such as intensivecare unit stay, sharing a room with a known carrier of β lactamase strain, prolonged hospitalization may reduce the spread of these pathogens in our healthcare setting.
Conclusion
Our findings showed that HIV-positive individuals are significantly more affected by Enterobacteriaceae infection than HIV-negative ones. The present study also indicates that HIV positive individuals are more likely to be infected by highly resistant organisms and that they are more prone to be infected by ESBL, AmpC and carbapenemase producing Enterobacteriaceae than HIV-negative individuals in our milieu. Ongoing studies on the prevalence of ESBL, AmpC and carbapenemase infection among HIV-positive individuals in other region of the country are performing in order to fully elucidate this issue and to establish a well-defined national surveillance program that monitor the evolution of lactamase producing Enterobacteria in Cameroon. Future efforts should focus on determining the presence of transferable resistance plasmids through genomic analysis in order to track the spread of these resistance genes within the population.
Acknowledgment
We acknowledge the support of the staffs of the Regional Hospital of Bafoussam-Cameroon who facilitated recruitment of patients for this research. We thank the participating patients for their cooperation.
References
- Woerther PL, Burdet C, Chachaty E, Andremont A (2013) Trends in human fecal carriage of extended-spectrum β-lactamases in the community: toward the globalization of CTX-M. Clin Microbiol Rev 26(4): 744-758.
- (1992) National AIDS/STD control program. Handbook on HIV Infection and AIDS for Health Workers. Federal Ministry of Health, Abuja, Nigeria pp. 12-14.
- Oyeyipo OO, Azuounwu O, Owhoeli O (2010) Prevalence of HIV antibodies in patients with clinical tuberculosis in Port Harcourt. International J Biosci 5(30): 65-68.
- Downs JH (2010) The gastrointestinal tract and HIV pathogenesis. S Afr J Clin Nutr 23: 565-568.
- Angulo FJ, Swerdlow DL (1995) Bacterial enteric infections in persons infected with human immunodeficiency virus. Clin Infect Dis 21(Suppl 1): 84-93.
- Sanchez TH, Brooks JT, Sullivan PS, Juhasz M, Mintz E, et al. (2005) Bacterial diarrhea in persons with HIV infection, United States, 1992-2002. Clin Infect Dis 41(11): 1621-1627.
- Wilcox CM, Saag MS (2008) Gastrointestinal complications of HIV infection: changing priorities in the HAART era. Gut 57(6): 861-870.
- Hung CC, Hung MN, Hsueh PR, Chang SY, Chen MY, et al. (2007) Risk of recurrent nontyphoid Salmonella bacteremia in HIV-infected patients in the era of highly active antiretroviral therapy and an increasing trend of fluoroquinolone resistance. Clin Infect Dis 45(5): e60-67.
- Zimmerman FS, Assous MV, Bdolah AT, Lachish T, Yinnon AM, et al. (2013) Duration of carriage of carbapenem-resistant Enterobacteriaceae following hospital discharge. Am J Infect Control 41(3): 190-194.
- Rampha lR, Ambrose PG (2006) Extended-spectrum beta-lactamases and clinical outcomes: current data. Clin Infect Dis 42 Suppl 4: S164-172.
- Cotton MF, Wasserman E, Smit J, Whitelaw A, Zar HJ (2008) High incidence of antimicrobial resistant organisms including extended spectrum beta-lactamase producing Enterobacteriaceae and methicillin-resistant Staphylococcus aureus in nasopharyngeal and blood isolates of HIV-infected children from Cape Town, South Africa. BMC Infect Dis 8: 40.
- Mwansa J, Mutela K, Zulu I, Amadi B, Kelly P (2002) Antimicrobial sensitivity in enterobacteria from AIDS patients, Zambia. Emerg Infect Dis 8(1): 92-93.
- Tansarli GS, Poulikakos P, Kapaskelis A, Falagas ME (2014) Proportion of extended-spectrum β-lactamase (ESBL)-producing isolates among Enterobacteriaceae in Africa: evaluation of the evidence-systematic review. J Antimicrob Chemother 69(5): 1177-1184.
- Storberg V (2014) ESBL-producing Enterobacteriaceae in Africa–a non-systematic literature review of research published 2008-2012. Infect Ecol Epidemiol 4: 20342.
- Bourjilat F, Bouchrif B, Dersi N, Claude JD, Amarouch H, et al. (2011) Emergence of extended-spectrum beta-lactamases-producing Escherichia coli in community-acquired urinary infections in Casablanca, Morocco. J Infect Dev Ctries 5(12): 850-855.
- unaids.org/sites/default/files/media_asset/20150901_FactSheet_2015_en.pdf.
- Garcia CJM, Abbenyi S (1993) Review of HIV prevalence studies in Cameroon. AIDS Inform. Bull 1: 5-6.
- (2000) Sentinel Surveillance of the Ministry of Public Health, Cameroon.
- (2004) National Institute of Statistics Demographic and Health Survey Cameroon.
- UNICEF Cameroon Country Program 2018-2020. Strategy Note HIV/AIDS Program 2018-2020 DRAFT.
- Soshi H, Nobuaki S (2013) Evaluation of semi-quantitative scoring of Gram staining or semi-quantitative culture for the diagnosis of ventilator-associated pneumonia: a retrospective comparison with quantitative culture. Journal of Intensive Care 1(1): 2.
- Clinical Laboratory Standards Institute (2016) Performance standards for antimicrobial susceptibility testing. (26th), CLSI supplement M100S. Wayne, PA, USA.
- (2017) Antibiogram Committee of the French Society of Microbiology Recommendation. CASFM 117.
- Clinical Laboratory Standards Institute (2011) Performance standards for antimicrobial susceptibility testing: twenty-first Informational Supplement M100-S21. Wayne, PA, USA.
- Tsakris A, Poulou A, Pournaras S, Voulgari E, Vrioni G, et al. (2010) Simple phenotypic method for the differentiation of metallo-lactamases and class A KPC carbapenemases in Enterobacteriaceae clinical isolates. J Antimicrob Chemother 65(8): 1664-1671.
- Paton AW, Paton JC (1996) Enterobacter cloacae producing a shiga-like toxin. H-related cytotoxin associated with a case of haemolytic uremic syndrome. J Clin Microbiol 34(2): 463-465.
- Raphael MM, Peter HG (2011) Frequency of isolation of Enterobacter species from a variety of clinical specimens in a teaching hospital in Nigeria. Tropical Journal of Pharmaceutical Research 10(6): 793-800.
- Hidron AI, Kempker R, Moanna A, Rimland D (2010) Methicillin-resistant Staphylococcus aureus in HIV-infected patients. Infection and Drug Resistance 3: 73-86.
- Cummings PL, Sorvillo F, Kuo T (2010) Salmonellosis-related mortality in the United States, 1990-2006. Foodborne Pathog Dis 7(11): 1393-1399.
- Framm Sr, Soave R (1997) Agents of diarrhea. Med Clin North Am 81(2): 427-447.
- Tumbarello M, Tacconelli E, Caponera S, Cauda R, Ortona L (1995) The impact of bacteremia on HIV infection. Nine years experience in a large Italian university hospital. The Journal of Infection 31(2): 123-131.
- Datta D, Gazzard B, Stebbing J (2003) The diagnostic yield of stool analysis in 525 HIV-1-infected individuals. AIDS 17(11): 1711-1713.
- Ayele AA, Tadesse D, Manilal A, Yohanes T, Seid M, et al. (2020) Prevalence of enteric bacteria and enteroparasites in human immunodeficiency virus-infected individuals with diarrhoea attending antiretroviral treatment clinic, Arba Minch General Hospital, southern Ethiopia. New Microbes and New Infections 38: 100789.
- Ayele K, Solomon A, Techalew S (2017) The common enteric bacterial pathogens and their antimicrobial susceptibility pattern among HIV-infected individuals attending the antiretroviral therapy clinic of Hawassa university hospital, southern Ethiopia. Antimicrobial Resistance and Infection Control 6: 128.
- Chhin S, Harwell JI, Bell JD, Rozycki G, Ellman T, et al. (2006) Etiology of chronic diarrhea in antiretroviral-naïve patients with HIV infection admitted to Norodom Sihanouk hospital, Phnom Penh, Cambodia. Clin Infect Dis 43(7): 925-932.
- Cárcamo C, Hooton T, Wener MH, Weiss NS, Gilman R, et al. (2005) Etiologies and manifestations of persistent diarrhea in adults with HIV-1 infection: a case-control study in lima, Peru. JID 191(1): 11-19.
- Brink AK, Mahe C, Watera C, Lugada E, Gilks C, et al. (2002) Diarrhea, CD4 counts and enteric infections in a community-based cohort of HIV-infected adults in Uganda. J Inf Secur 45(2): 99-106.
- Uppal B, Kashyap B, Bhalla P (2009) Enteric pathogens in HIV/AIDS from a tertiary care hospital. Indian J Community Med 34(3): 237-242.
- Awole M, Gebre SS, Kassa T, Kibru G (2002) Isolation of potential bacterial pathogens from the stool of HIV-infected and HIV-non-infected patients and their antimicrobial susceptibility patterns in Jimma hospital, south west Ethiopia. Ethiop Med J 40(4): 353-364.
- Obi CL, Bessong PO (2002) Diarrhoeagenic bacterial pathogens in HIV-positive patient with diarrhea in rural communities of Limpopo Province, South Africa. JHPN 20(3): 230-234.
- Huang DB, Zhou J (2007) Effect of intensive hand washing in the prevention of diarrheal illness among patients with AIDS: a randomized controlled study. J Med Microbiol 56(5): 659-663.
- Shah S, Kongre V, Kumar V, Bharadwaj R (2016) A Study of parasitic and bacterial pathogens associated with diarrhea in HIV-positive patients. Cureus 8(9).
- Kownhar H, Shankar EM, Rajan R, Vengatesan A, Rao UA (2007) Prevalence of campylobacter jejuni and enteric bacterial pathogens among hospitalized HIV- infected versus non-HIV infected patients with diarrhoea in southern India. Scand J Infect Dis 39(10): 862-866.
- Mwansa J, Mutela K, Zulu I, Amadi B, Kelly P (2002) Antimicrobial sensitivity in Enterobacteria from AIDS patients, Zambia. Emerg Infect Dis 8(1): 92-93.
- Rossit ARB, Gonçalves ACM, Franco C, Machado RLD, et al (2009) Etiological agents of diarrhea in patients infected by the HIV-1. a review. Rev Inst Med trop S Paulo 51(2): 59-65.
- (2000) World Health Organization. Provisional WHO/ UNAIDS Secretariat Recommendations on the Use of Cotrimoxazole Prophylaxis in Adults and Children Living with HIV/AIDS in Africa. WHO/UNAIDS, Geneva, Switzerland.
- Siraj SM, Ali S, Wondafrash B (2015) Extended- spectrum β-lactamase production in Klebsiella pneumoniae and Escherichia coli at Jimma University Specialized Hospital, South-West, Ethiopia. Mol Microbiol Res 5(1): 1-9.
- Seid J, Asrat D (2005) Occurrence of extended spectrum beta-lactamase enzymes in clinical isolates of Klebsiella species from Harar region, Eastern Ethiopia. Acta Trop 95(2): 143-148.
- Mulisa G, Selassie LG, Jarso G, Shiferew T, Zewdu A, et al. (2016) Prevalence of extended spectrum beta-lactamase producing Enterobacteriaceae: a cross sectional study at Adama Hospital, Adama. J Emerg Infect Dis 1(1): 1-6.
- Mulualem Y, Kasa T, Mekonnen Z, Suleman S (2012) Occurrence of extended spectrum beta-lactamases in multidrug resistant Escherichia coli isolated from a clinical setting in Jimma University specialized hospital, Jimma, Southwest Ethiopia. East Afr J Public Heal 9(2): 58-61.
- Kandeel A (2014) Prevalence and risk factors of extended-spectrum β-lactamases producing Enterobacteriaceae in a general hospital in Saudi Arabia. J Microbiol Infect Dis 4(2): 50-54.
- Reuland EA, Al Naiemi N, Kaiser AM, Heck M, Kluytmans JA, et al. (2016) Prevalence and risk factors for carriage of ESBL-producing Enterobacteriaceae in Amsterdam. J Antimicrob Chemother 71(4): 1076-1082.
- Enoch DA, Brown F, Sismey AW, Mlangeni DA, Curran MD, et al. (2012) Epidemiology of extended-spectrum beta-lactamase-producing Enterobacteriaceae in a UK district hospital; an observational study. J Hosp Infect 81(4): 270-277.
- Obengnkrumah N, Twumdanso K, Krogfelt KA, Newman MJ (2013) High levels of extended-spectrum beta-lactamases in a major teaching hospital in Ghana: the need for regular monitoring and evaluation of antibiotic resistance. Am J Trop Med Hyg 89(5): 960-964.
- Desta K, Woldeamanuel Y, Azazh A, Mohammod H, Desalegn D, et al. (2016) High gastrointestinal colonization rate with extended-spectrum β-Lactamase-Producing Enterobacteriaceae in hospitalized patients: emergence of carbapenemase-producing K. pneumoniae in Ethiopia. PLoS One Med 11(8): 1-14.
- Kateregga JN, Kantume R, Atuhaire C, Lubowa MN, Ndukui JG (2015) Phenotypic expression and prevalence of ESBL-producing Enterobacteriaceae in samples collected from patients in various wards of Mulago Hospital, Uganda. BMC Pharmacol Toxicol pp. 14-19.
- Gangoue PJ, Bedenic B, Koulla SS, Randegger C, Adiogo D, et al. (2005) Enterobacteriaceae producing extended-spectrum beta-lactamases in Yaoundé, Cameroon. J Clin Microbiol 43(7): 3273-3277.
- Carine ML, Cécile M, Joseph GP, Raphaël B, Marie CO, et al. (2012) Proportion of extended-spectrum ß-lactamase-producing Enterobacteriaceae in community setting in Ngaoundere, Cameroon. BMC Infect Dis 9: 53.
- Djuikoue IC, Njajou O, Kamga HG, Fokunang C, Bongoe A, et al. (2017) Prevalence of CTX-M β-lactamases in Escherichia coli from community-acquired urinary tract infections and associated risk factors among women in Cameroon. J Epidemiol Res 3(1): 51.
- Ikonomidis A, Tokatlidou D, Kristo I, Sofianou D, Tsakris A, et al. (2005) Outbreaks in distinct regions due to a single Klebsiella pneumoniae clone carrying a blaVIM-1 metallo-b-lactamase gene. J Clin Microbiol 43(10): 5344-5347.
- Queenan AM, Bush K (2007) Carbapenemases The versatile β-lactamases. Clin Microbiol Rev 20(3): 440-458.
- Thomson KS (2010) Extended-Spectrum-Lactamase, ampC and carbapenemase issues. J Clin Microbiol 48(4): 1019-1025.






























