Chlorogenic Acid: A Promising Natural Agent for Non-Alcoholic Fatty Liver Disease Management
Stella M Honoré1,2* and Sara S Sánchez1
1Instituto Superior de Investigaciones Biológicas (INSIBIO, CONICET-UNT),
2Facultad de Bioquímica, Química y Farmacia, Universidad Nacional de Tucumán. Chacabuco 461, T4000ILI – San Miguel de Tucumán, Argentina
Submission:November 29, 2021; Published:December 03, 2021
*Corresponding author: Stella M Honoré, Instituto de Biología “Dr. Francisco D. Barbieri”, Facultad de Bioquímica, Química y Farmacia, Universidad Nacional de Tucumán. Chacabuco 461, T4000ILI – San Miguel de Tucumán, Tucumán, Argentina
How to cite this article: Honoré SM, Sánchez SS. Chlorogenic Acid: A Promising Natural Agent for Non-Alcoholic Fatty Liver Disease Management. Adv Res Gastroentero Hepatol, 2021; 18(1): 555980. DOI: 10.19080/ARGH.2021.18.555980.
Abstract
Emerging and relatively consistent evidence shows that chlorogenic acid (CGA), a phenolic acid, positively modulates a variety of contributors to the non-alcoholic fatty liver (NAFLD) phenotype, through diverse and complementary mechanisms of action. Therefore, we believe that CGA is a good candidate for the management of NAFLD that deserves a review to aid future research.
Keywords: Non-alcoholic fatty liver disease; Liver steatosis; Polyphenol; Chlorogenic acid; Antioxidant; Gut microbiota
Abbreviations: NAFLD: Non-Alcoholic Fatty Liver Disease; NASH: Non-Alcoholic Steatohepatitis; T2DM: Type 2 Diabetes; IR: Insulin Resistance; CGA: Chlorogenic Acid; LPS: Lipopolysaccharides
Introduction
As a consequence of the growing epidemics of obesity and Type 2 Diabetes Mellitus (T2DM), Nonalcoholic fatty liver disease (NAFLD) has become the most common form of chronic liver disease in the world [1-3]. NAFLD initially presents as a relatively benign, non-progressive hepatic steatosis, but can, in certain individuals, progress to a potentially serious condition [4]. At the early stages, patients can restore liver function by changing their lifestyle (diet and exercise). However, as the disease progresses, pharmacological intervention or even a liver transplant is required [5]. Despite NAFLD is a major public health problem, it remains difficult to treat, as there is not yet a large, validated treatment [2]. Recent trials suggest that different approaches may be beneficial in subgroups of patients with this condition [4].
Flavonoids and phenolic acids have recently gained substantial attention due to their various biological and pharmacological effects [6]. Accumulating evidence suggests that these bioactive compounds may represent a complementary and integrative natural therapy for the management of obesity and its associated disorders such as T2DM and NAFLD [7-9].
In this review, we explore the medicinal promises of chlorogenic acid (CGA) on NAFLD management based on in vitro and in vivo reported studies. This phenolic acid is able to positively modulate a variety of steps from NAFLD pathogenesis, through diverse and complementary mechanisms of action.
Nonalcoholic fatty liver disease
NAFLD is defined by the presence of steatosis in more than 5% of hepatocytes with little or no alcohol consumption [10,11]. The spectrum of the hepatic abnormalities ranging from a simple isolated steatosis to nonalcoholic steatohepatitis (NASH) characterized by steatosis along with hepatocellular injury, inflammation, and varying degrees of fibrosis [12] which sometimes can progress to cirrhosis and hepatocellular carcinoma [13].
The worldwide prevalence of NAFLD increased substantially along with increased rates of obesity and other components of the metabolic syndrome [3,5]. However, a proportion of cases have revealed a normal body mass index (BMI), a phenomenon known as “non-obese NAFLD” [11]. In any case, the presence of T2DM in patients, displays a very high risk of developing NASH and fatty liver associated complications, evidencing an additive detrimental liver outcome [14].
It is now accepted that the metabolic and molecular changes that lead to NAFLD results from “multiple impacts” on the liver as a consequence of complex interactions between genetic susceptibility, environmental factors, insulin resistance, and changes in the gut microbiota [12,15]. In this sense, several of the triggers of fatty liver can be traced back to events that occur outside the liver in distant organs such as the intestine, adipose tissue, and muscles, among others [12,16]. In this sequence of multiple events, metabolic alterations in lipid homeostasis (free fatty acids, non-HDL-cholesterol) have been reported to precede fatty liver onset [17]. This initial impact results in the development of macrovesicular steatosis with an accumulation of liver fat [18].
Insulin resistance (IR) plays a key role in the development and progression of steatosis / NASH, promoting de novo hepatic lipogenesis and failing to suppress adipose tissue lipolysis, with the consequent rise of fatty acids in the liver [16]. In an attempt to compensate for these changes, both hepatic fatty acid β-oxidation and VLDL secretion are initially upregulated. However, this is insufficient to decrease the continuous flow of fatty acids to the liver that leads to tissue injury [19]. In some patients, steatosis even worsens IR by engaging in a vicious cycle once NAFLD develops. The cytokines derived from the dysfunctional adipose tissue, the free fatty acid-induced ectopic fat deposition, and lipotoxicity increase insulin resistance with the consequent changes in glucose and lipid metabolism [20]. The accumulation of fat in the liver, specifically in the form of triglycerides, impacts the production of reactive oxygen species and endoplasmic reticulum stress along with mitochondrial dysfunction [21]. Excess nutrients overwhelm the endoplasmic reticulum (ER), which activates the unfolded protein response and triggers the development of IR through various mechanisms, including activation and inflammation of c-jun kinase N-terminal (JNKs) [22]. Oxidative stress can promote lipid peroxidation in the hepatocytes and induce the secretion of pro-inflammatory cytokines and the activation of stellate cells through multiple signaling pathways, which in turn lead to fibrosis [4].
In recent years, a crosstalk between the gut microbiota and multiple organs of the host has been demonstrated with a beneficial role in physiological regulation [22]. Nevertheless, changes in the microbiota composition are recognized as key players in the pathogenesis of NAFLD [23]. The intestinal microbiota not only influences the absorption and elimination of nutrients to reach the liver. It also induces changes in the liver microenvironment by supplying gut-derived factors that stimulate hepatocytes to release free oxygen radicals and inflammatory cytokines that activate downstream signaling pathways such as nuclear factor NF-κB [24,25].
Chlorogenic acid
Chlorogenic acid (CGA) also known as 5-O-caffeoylquinic acid (5-CQA) (IUPAC numbering) or 3-CQA (pre-IUPAC numbering) (Figure 1) [26], is one of the most abundant isomers of caffeoylquinic acid in nature. It is produced by the esterification of caffeic acid and L-quinic acid in certain plant species, in response to environmental stress [27].
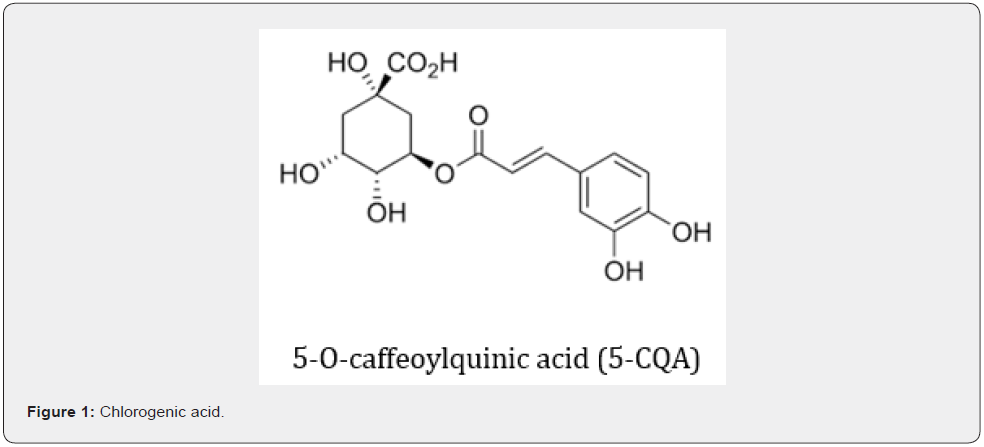
CGA can be found in varying amounts in seeds, leaves, fruits, roots, and tubers forming part of the human diet [7,28-30]. Substantial amounts of CGA have been reported to be available in green tea and coffee extracts [27] and lower in apples, blueberries, strawberries, tomatoes, and potatoes [8]. Chlorogenic acid is also present in dairy products, as part of the phenolic content of milk influenced by animal grazing [31].
As a natural plant extract from a wide range of sources, CGA exhibits many biological properties including antioxidant, antiinflammatory, antimutagenic, anticancer, immunomodulatory, antibacterial, antiviral, particularly hypoglycemic and hypolipidemic effects [32]. Recently, the functions and applications of CGA, in relation to liver metabolism, have been highlighted in both the biological and medical fields [9,33].
Chlorogenic acid in the line of NAFLD “multiple impacts”
Several lines of evidence indicate that CGA could play vital roles in regulating metabolic dysfunction closely associated with the onset and progression of NAFLD [33,34].
Impact 1: Improvement of lipid metabolism:
The enhancement of de novo lipogenesis, suppression of β-oxidation, and decreased lipid export from the liver are the major reasons for the promotion of fat accumulation in the liver [18,35]. Recently, Zamani-Garmsiri [36] using a NAFLD rodent model found that CGA alone or in combination with metformin attenuates the expression of the lipogenic genes SREBP-1c and FAS. CGA also induces the expression and strengthens the activity of CPT-1, a fatty acid oxidation speed limit enzyme, and promotes the oxidation of fatty acid. A consequent decrease in plasma and triglyceride liver levels was achieved, demonstrating the antilipidemic function of CGA in HFD-fed mice. In addition, Ma et al. [37] have shown that CGA is able to reduce the transport of long-chain fatty acids into the liver by inhibiting the diet-induced expression of PPARγ and its target genes CD36 and Fabp4. Also, CGA drastically reduces the level of Mgat1 mRNA, shown to be involved in triglyceride synthesis [38]. In addition to the effect on PPARγ, other studies have shown that CGA could enhance PPARα levels in the liver and stimulate lipid utilization through adiponectin receptor-mediated signaling pathway [39-41]. It was also suggested that the effects of CGA could be linked to the modulation of cholesterol metabolism [42]. Some in vitro evidence indicates that CGA may indirectly decrease lipid accumulation in the liver by effectively inhibiting HMG-CoA by reducing cholesterol synthesis [43].
Impact 2: Improvement of oxidative status:
The appearance and persistence of oxidative stress in the liver seem to play a fundamental role in the development of inflammation and NAFLD progression to more severe stages [3]. In fact, higher production of oxygen and nitrogen radical species, a lack of endogenous antioxidant defenses, and mitochondrial structural defects within hepatocytes were observed in patients with NASH [24]. So, targeting oxidative stress and inflammation could represent one of the main pathways of innovative NAFLD therapies [44].
Several studies in vivo and in vitro have found that plant extracts containing CGA have anti-inflammatory and antioxidant activities [45-48]. It has been shown that CGA up-regulates cellular antioxidant enzymes and suppresses ROS-mediated NF-κB, AP- 1, and MAPK activation in vitro [45]. Also, CGA could promote scavenge free radical, up-regulate the expression and stimulate antioxidant enzymatic activities of SOD and GPx, CAT to attenuate NAFLD in vivo [47,49]. Additionally, Budryn et al. [50] showed a high reduced to oxidized glutathione (GSH/GSSG) ratio in the liver and a high concentration of the antioxidants in blood serum, as a result of the consumption of diets containing microencapsulated CGA.
Impact 3: Improvement of inflammation:
Abnormal lipid accumulation in hepatocytes increases oxidative stress and leads to lipotoxicity, which triggers liver inflammation [24]. The secretion of pro-inflammatory cytokines in the hepatocytes is accompanied by macrophage infiltration and a change in macrophage phenotype M2 (anti-inflammatory) to M1 (pro-inflammatory) in the infiltrated tissues, key players in the metabolic inflammation observed in NAFLD [4]. CGA reduces the transcription of TNF-α, IL-6, MCP-1, and CRR2, suppressing the NF-κB activity, reducing inflammatory responses in the liver of HFD mice [37,38]. The modulating action of CGA on the NF-kB signaling was previously demonstrated by [51] in a lipopolysaccharide (LPS)-challenge in mice. In addition, CGA also could reverse the HFD-induced activation of TLR4 signaling pathway in liver [21] and decrease macrophage marker genes (including F4/80, CD68, CD11b and CD11c) and pro-inflammatory mediator genes (MCP-1 and TNF-α) in the liver and adipose tissues [36]. Recently, it was determined that CGA is capable of suppressing inflammation by inhibiting the activation of TLR4 / sphingosine (SPK / S1P), highlighting the role of CGA in preventing progression to NASH [49].
Impact 4: Improvement of insulin sensitivity:
Insulin resistance is at the core of the pathophysiology of metabolic syndrome and T2DM. CGA is able to exert vital roles in the regulation of metabolic abnormalities closely associated with the occurrence and progression of NAFLD [52]. Accumulating evidence suggests that CGA may improve adipose tissue dysfunction and in turn reduce the development of obesity-linked IR [6,53]. According to Ma et al. [36], CGA is able to sensitize peripheral tissues for insulin response by attenuating inflammatory phenotypes in both adipose and liver tissues of obese mice.
On the other hand, hepatic IR is known to be associated with dysregulated glucose metabolism, as a consequence of an increase in gluconeogenesis and a reduction in glycogen synthesis. Recent findings showed that AMP-activated protein kinase (AMPK) plays a major role in the control of hepatic metabolism [54]. AMPK activation in the liver has metabolic consequences on lipids and glucose due to its ability to integrate nutritional and hormonal signals [55]. CGA has been shown to regulate glucose overproduction by inhibiting glucose-6-phosphatase (G-6-Pass) activity through AMPK stimulation [56]. Meanwhile, by specific binding to AKT, CGA is able to promote glucose uptake in liver cells by stimulating glycogen synthesis through phosphorylation of molecules downstream of GSK3β / FOXO1 signaling [57]. In addition, the increased expression and translocation of glucose transporter type-4 (GLUT-4) in skeletal muscle mediated by AMPK activation, could also facilitate glucose clearance in peripheral tissues, maintaining fasting glucose levels, glucose tolerance, and insulin sensitivity [35,58]. In this way, CGA could be considered a novel insulin sensitizer capable of maintaining glucose homeostasis similar to metformin [59].
The improvement in systemic glucose control can also be attributed to CGA inhibitory action on glucose-6-phosphate translocase that delays absorption in the small intestine [60]. It was reported that CGA also inhibited the activities of 𝛼-amylase and 𝛼-glucosidase contributing to reducing the glycemic impact of food and chronically lowering blood glucose levels in patients with T2DM at high risk of developing NAFLD [61,62].
Impact 5: Improvement of the gut–liver axis:
The gut microbiota and intestinal permeability have been demonstrated to be the key players in the gut-liver cross-talk in NAFLD [63]. In NAFLD, overgrowth of the gut microbiota contributes to the disease progression, through the leaky gut barrier [64]. The increased permeability allows translocation of intestinal luminal antigens, including LPS to the liver, where they bind to their specific CD14 and TLR4 receptors on Kupffer cells aggravating inflammation and oxidative stress damage in the liver [65]. So, targeting the gut-liver axis and modulation of gut microbiota metabolites using specific prebiotics could represent an additional therapeutic approach for the treatment of NAFLD [66]. In line with this, several studies showed that CGA is able to reverse intestinal dysbiosis, increasing the metabolic activity and/ or numbers of the beneficial Bifidobacterium spp. in both humans and mice [39,66-68]. Simultaneously, it was found that CGA in combination with Genistoside could enhance the intestinal barrier function, preventing leakage of LPS derived from the intestine and reducing plasma D-lactate in NAFLD [69]. The reduction of gut permeability by CGA is closely related to the restoration of the expression of the tight junction proteins occludin, claudin-1, and zonula occludens-1 (ZO-1) in the intestinal mucosa, together with the inhibition of tight junction disassembly promoted by downregulation of RhoA/ ROCK signaling [25].
Furthermore, the interactions between the CGA and the gut microbiota can impact intestinal L-cell metabolism, increasing the GLP-1 levels in the portal vein [25]. An abnormal incretin system has been found in nondiabetic NAFLD and NASH patients [70]. By binding to GLP-1R, GLP-1 could act on β-cells, through cAMP-dependent mechanisms, helping to maintain the response capacity of these cells to increased plasma glucose [71]. GLP- 1 could also improve NAFLD, increasing liver lipid oxidation, improving insulin sensitivity and inhibiting liver fat synthesis through AMPK-activation [72].
Impact 6: Improvement of progression to liver fibrosis:
As seen above, oxidative and ER stresses play an important role in the development of liver complications [73]. NASH is characterized by fatty liver, liver inflammation and substantial hepatocyte cell death. The activation of hepatic stellate cells (HSCs) by apoptotic bodies, ROS, or by TGFβ from activated-kupffer cells, produces liver collagen accumulation leading to fibrosis. In the line with this, Shi et al. [47] showed that CGA could significantly improve liver matrix remodeling reducing hydroxyproline content and collagen Ⅰ, collagen Ⅲ, and TIMP-1 expression in CCl4-injected rats. Moreover CGA, also prevents HSCs activation, by suppressing PDGF/ROS generation in these cells, suggesting that the antifibrogenic mechanisms might be related to CGA-antioxidant and anti-apoptotic effects.
At last, it is well established that apoptotic hepatocytes define the progression of the severity of liver disease [20]. Thus, limiting liver injury could be a therapeutic way to prevent the progression of hepatic complications. Recent evidence indicates that CGA is able to reduce oxidative stress-mediated cell death via Nrf2 activation in HepG2 cells [74]. Consistent with this, the consumption of products with a high CGA content, improves oxidative stress and reduce liver cell death constituting promising agents for NAFLD management [27,30,46,48,75].
Conclusion
Based on the results obtained from various studies, we believe that CGA, a prebiotic phenolic compound with multifunctional properties, protects against steatosis, oxidative stress, and liver inflammation. The combination of these health benefits makes CGA an excellent candidate for the prevention and treatment of NAFLD. Future research should focus both on stimulating clinical studies on NAFLD and on analyzing the inclusion of CGA in different matrices to ensure its bioavailability.
Acknowledgments
This research was supported by PICT 2017 No. 3941 (ANPCyT, Argentina) and PIUNT D619 grants to SMH; and PIP No. 183 (CONICET, Argentina) grants to SSS and SMH.
References
- Younossi ZM (2019) Non-alcoholic fatty liver disease - A global public health perspective. J Hepatol 70(3): 531-544.
- Huang TD, Behary J, Zekry A (2020) Non-alcoholic fatty liver disease: a review of epidemiology, risk factors, diagnosis and management. Intern Med J 50(9): 1038-1047.
- Ye Q, Zou B, Yeo YH, Li J, Huang DQ, et al. (2020) Global prevalence, incidence, and outcomes of non-obese or lean non-alcoholic fatty liver disease: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol 5(8): 739-752.
- Peng C, Stewart AG, Woodman OL, Ritchie RH, Qin CX (2020) Non-Alcoholic Steatohepatitis: A Review of Its Mechanism, Models and Medical Treatments. Frontiers in pharmacology 11: 603926.
- Patel SS, Siddiqui MS (2019) Current and Emerging Therapies for Non-alcoholic Fatty Liver Disease. Drugs 79(1): 75-84.
- Dinda B, Dinda M, Roy A, Dinda S (2020) Dietary plant flavonoids in prevention of obesity and diabetes. Adv Protein Chem Struct Biol 120: 159-235.
- Naveed M, Hejazi V, Abbas M, Kamboh AA, Khan GJ, et al. (2018) Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomedicine & pharmacotherapy 97: 67-74.
- Kumar R, Sharma A, Iqbal MS, Srivastava JK (2020) Therapeutic Promises of Chlorogenic Acid with Special Emphasis on its Anti-Obesity Property. Curr Mol Pharmacol 13(1): 7-16.
- Abenavoli L, Larussa T, Corea A, Procopio AC, Boccuto L, (2021) Dietary Polyphenols and Non-Alcoholic Fatty Liver Disease. Nutrients 13(2): 494.
- Cobbina E, Akhlaghi F (2017) Non-alcoholic fatty liver disease (NAFLD) - pathogenesis, classification, and effect on drug metabolizing enzymes and transporters. Drug Metab Rev 49(2): 197-211.
- Ahadi M, Molooghi K, Masoudifar N, Namdar AB, Vossoughinia H, Farzanehfar M (2021) A review of non-alcoholic fatty liver disease in non-obese and lean individuals. J Gastroenterol Hepatol 36(6): 1497-1507.
- Chen YY, Yeh MM (2021) Non-alcoholic fatty liver disease: A review with clinical and pathological correlation. J Formos Med Assoc 120(1 Pt 1): 68-77.
- Yan Y, Liu N, Hou N, Dong L, Li J (2017) Chlorogenic acid inhibits hepatocellular carcinoma in vitro and in vivo. J Nutr Biochem 46: 68-73.
- Hazlehurst JM, Woods C, Marjot T, Cobbold JF, Tomlinson JW (2016) Non-alcoholic fatty liver disease and diabetes. Metabolism: clinical and experimental 65(8): 1096-1108.
- Fierbinteanu-Braticevici C, Sinescu C, Moldoveanu A, Petrisor A, Diaconu S, et al. (2017) Nonalcoholic fatty liver disease: one entity, multiple impacts on liver health. Cell Biol Toxicol 33(1): 5-14.
- Byrne CD, Olufadi R, Bruce KD, Cagampang FR, Ahmed MH (2009) Metabolic disturbances in non-alcoholic fatty liver disease. Clin Sci (Lond) 116(7): 539-564.
- Than NN, Newsome PN (2015) A concise review of non-alcoholic fatty liver disease. Atherosclerosis 239(1): 192-202.
- Ipsen DH, Lykkesfeldt J, Tveden-Nyborg P (2018) Molecular mechanisms of hepatic lipid accumulation in non-alcoholic fatty liver disease. Cell Mol Life Sci 75(18): 3313-3327.
- Fujita K, Nozaki Y, Wada K, Yoneda M, Fujimoto Y, et al. (2009) Dysfunctional very-low-density lipoprotein synthesis and release is a key factor in nonalcoholic steatohepatitis pathogenesis. Hepatology 50(3): 772-780.
- Lavallard VJ, Gual P (2014) Autophagy and non-alcoholic fatty liver disease. Biomed Res Int 2014: 120179.
- Liu X, Green RM (2019) Endoplasmic reticulum stress and liver diseases. Liver research 3(1): 55-64.
- He LH, Yao DH, Wang LY, Zhang L, Bai XL (2021) Gut Microbiome-Mediated Alteration of Immunity, Inflammation, and Metabolism Involved in the Regulation of Non-alcoholic Fatty Liver Disease. Front Microbiol 12: 761836.
- Da Silva HE, Teterina A, Comelli EM, Taibi A, Arendt BM, et al. (2018) Nonalcoholic fatty liver disease is associated with dysbiosis independent of body mass index and insulin resistance. Sci Rep 8(1): 1466.
- Delli Bovi AP, Marciano F, Mandato C, Siano MA, Savoia M, et al. (2021) Oxidative Stress in Non-alcoholic Fatty Liver Disease. An Updated Mini Review. Frontiers in medicine 8: 595371.
- Shi A, Li T, Zheng Y, Song Y, Wang H, et al. (2021) Chlorogenic Acid Improves NAFLD by Regulating gut Microbiota and GLP-1. Front Pharmacol 12: 693048.
- Pimpley V, Patil S, Srinivasan K, Desai N, Murthy PS (2020) The chemistry of chlorogenic acid from green coffee and its role in attenuation of obesity and diabetes. Prep Biochem Biotechnol 50(10): 969-978.
- Tajik N, Tajik M, Mack I, Enck P (2017) The potential effects of chlorogenic acid, the main phenolic components in coffee, on health: a comprehensive review of the literature. Eur J Nutr 56(7): 2215-2244.
- Santana-Gálvez J, Cisneros-Zevallos L, Jacobo-Velázquez DA (2017) Chlorogenic Acid: Recent Advances on Its Dual Role as a Food Additive and a Nutraceutical against Metabolic Syndrome. Molecules 22(3): 358.
- Honoré SM, Genta SB, Sánchez SS (2015) Smallanthus sonchifolius (Yacon) leaves: an emerging source of compounds for diabetes management. Journal of Research in Biology 5.
- Honoré SM, Grande MV, Gomez Rojas JR, Sánchez SS (2018) Smallanthus sonchifolius (Yacon) Flour Improves Visceral Adiposity and Metabolic Parameters in High-Fat-Diet-Fed Rats. Journal of obesity 2018: 5341384.
- Hilario MC, Puga CD, Ocana AN, Romo FPG (2010) Antioxidant activity, bioactive polyphenols in Mexican goats' milk cheeses on summer grazing. The Journal of dairy research 77(1): 20-26.
- Miao M, Xiang L (2020) Pharmacological action and potential targets of chlorogenic acid. Adv Pharmacol 87: 71-88.
- Meng S, Cao J, Feng Q, Peng J, Hu Y (2013) Roles of chlorogenic Acid on regulating glucose and lipids metabolism: a review. Evidence-based complementary and alternative medicine: eCAM 2013: 801457.
- Rodriguez-Ramiro I, Vauzour D, Minihane A (2016) Polyphenols and non-alcoholic fatty liver disease: Impact and mechanisms. Proc Nutr Soc 75(1): 47-60.
- Ong KW, Hsu A, Tan BK (2013) Anti-diabetic and anti-lipidemic effects of chlorogenic acid are mediated by ampk activation. Biochemical pharmacology 85(9): 1341-1351.
- Zamani-Garmsiri F, Ghasempour G, Aliabadi M, Hashemnia S, Emamgholipour S, et al. (2021) Combination of metformin and chlorogenic acid attenuates hepatic steatosis and inflammation in high-fat diet fed mice. IUBMB life 73(1): 252-263.
- Ma Y, Gao M, Liu D (2015) Chlorogenic acid improves high fat diet-induced hepatic steatosis and insulin resistance in mice. Pharm Res 32(4): 1200-1209.
- Yu JH, Song SJ, Kim A, Choi Y, Seok JW, et al. (2016) Suppression of PPARγ-mediated monoacylglycerol O-acyltransferase 1 expression ameliorates alcoholic hepatic steatosis. Sci Rep 6: 29352.
- Jin S, Chang C, Zhang L, Liu Y, Huang X, et al. (2015) Chlorogenic acid improves late diabetes through adiponectin receptor signaling pathways in db/db mice. PloS one 10(4): e0120842.
- Wang Z, Lam KL, Hu J, Ge S, Zhou A, et al. (2019) Chlorogenic acid alleviates obesity and modulates gut microbiota in high-fat-fed mice. Food Sci Nutr 7(2): 579-588.
- Huang K, Liang XC, Zhong YL, He WY, Wang, Z (2015) 5-Caffeoylquinic acid decreases diet-induced obesity in rats by modulating PPARα and LXRα transcription. J Sci Food Agric 95(9): 1903-1910.
- Chen Q, Wang T, Li J, Wang S, Qiu F, et al. (2017) Effects of Natural Products on Fructose-Induced Nonalcoholic Fatty Liver Disease (NAFLD). Nutrients 9(2): 96.
- Hao S, Xiao Y, Lin Y, Mo Z, Chen Y, et al. (2016) Chlorogenic acid-enriched extract from Eucommia ulmoides leaves inhibits hepatic lipid accumulation through regulation of cholesterol metabolism in HepG2 cells. Pharm Biol 54(2): 251-259.
- Salomone F, Godos J, Zelber-Sagi S (2016) Natural antioxidants for non-alcoholic fatty liver disease: molecular targets and clinical perspectives. Liver Int 36(1): 5-20.
- Feng R, Lu Y, Bowman LL, Qian Y, Castranova V, et al. (2005) Inhibition of activator protein-1, NF-kappaB, and MAPKs and induction of phase 2 detoxifying enzyme activity by chlorogenic acid. J Biol Chem 280(30): 27888-27895.
- Habib NC, Serra-Barcellona C, Honoré SM, Genta SB, Sánchez SS (2015) Yacon roots (Smallanthus sonchifolius) improve oxidative stress in diabetic rats. Pharmaceutical biology 53(8): 1183-1193.
- Shi H, Shi A, Dong L, Lu X, Wang Y, et al. (2016) Chlorogenic acid protects against liver fibrosis in vivo and in vitro through inhibition of oxidative stress. Clin Nutr 35(6): 1366-1373.
- Aleman MN, Sánchez SS, Honoré SM (2019) Smallanthus sonchifolius roots ameliorate non-alcoholic fatty liver disease by reducing redox imbalance and hepatocyte damage in rats fed with a high fructose diet. Asian Pacific Journal of Tropical Biomedicine 2019: 365-372.
- Alqarni I, Bassiouni YA, Badr AM, Ali RA (2019) Telmisartan and/or chlorogenic acid attenuates fructose-induced non-alcoholic fatty liver disease in rats: Implications of cross-talk between angiotensin, the sphingosine kinase/sphingoine-1-phosphate pathway, and TLR4 receptors. Biochem Pharmacol 164: 252-262.
- Budryn G, Zaczyńska D, Żyżelewicz D, Grzelczyk J, Zduńczyk Z, et al. (2017) Influence of the Form of Administration of Chlorogenic Acids on Oxidative Stress Induced by High fat Diet in Rats. Plant Foods Hum Nutr 72(2): 184-191.
- Xu Y, Chen J, Yu X, Weiwei Tao, Fengrong Jiang, et al. (2010) Protective effects of chlorogenic acid on acute hepatotoxicity induced by lipopolysaccharide in mice. Inflamm Res 59(10): 871-877.
- Bhandarkar NS, Brown L, Panchal SK (2019) Chlorogenic acid attenuates high-carbohydrate, high-fat diet-induced cardiovascular, liver, and metabolic changes in rats. Nutr Res 62: 78-88.
- Zhang LT, Chang CQ, Liu Y, Chen ZM (2011) Effect of chlorogenic acid on disordered glucose and lipid metabolism in db/db mice and its mechanism. Zhongguo yi xue ke xue yuan xue bao. Acta Academiae Medicinae Sinicae 33(3): 281-286.
- Zhao P, Saltiel AR (2020) From overnutrition to liver injury: AMP-activated protein kinase in nonalcoholic fatty liver diseases. J Biol Chem 295(34): 12279-12289.
- Viollet B, Foretz M, Guigas B, Horman S, Dentin R, et al. (2006) Activation of AMP-activated protein kinase in the liver: a new strategy for the management of metabolic hepatic disorders. J Physiol 574(Pt 1): 41-53.
- Li SY, Chang CQ, Ma FY, Yu CL (2009) Modulating effects of chlorogenic acid on lipids and glucose metabolism and expression of hepatic peroxisome proliferator-activated receptor-alpha in golden hamsters fed on high fat diet. Biomed Environ Sci 22(2): 122-129.
- Gao J, He X, Ma Y, Zhao X, Hou X, et al. (2018) Chlorogenic Acid Targeting of the AKT PH Domain Activates AKT/GSK3β/FOXO1 Signaling and Improves Glucose Metabolism. Nutrients 10(10): 1366.
- Ong KW, Hsu A, Tan BK (2012) Chlorogenic acid stimulates glucose transport in skeletal muscle via AMPK activation: a contributor to the beneficial effects of coffee on diabetes. PloS one 7(3): e32718.
- McCarty MF (2005) A chlorogenic acid-induced increase in GLP-1 production may mediate the impact of heavy coffee consumption on diabetes risk. Med Hypotheses 64(4): 848-853.
- Yukawa GS, Mune M, Otani H, Tone Y, Liang XM, et al. (2004) Effects of coffee consumption on oxidative susceptibility of low-density lipoproteins and serum lipid levels in humans. Biochemistry (Mosc) 69(1): 70-74.
- Bassoli BK, Cassolla P, Borba-Murad GR, Constantin J, Salgueiro-Pagadigorria CL, et al. (2008) Chlorogenic acid reduces the plasma glucose peak in the oral glucose tolerance test: effects on hepatic glucose release and glycaemia. Cell Biochem Funct 26(3): 320-328.
- Oboh G, Agunloye OM, Adefegha SA, Akinyemi AJ, Ademiluyi AO (2015) Caffeic and chlorogenic acids inhibit key enzymes linked to type 2 diabetes (in vitro): a comparative study. J Basic Clin Physiol Pharmacol 26(2): 165-170.
- Gut inflammation exacerbates hepatic injury in the high-fat diet induced NAFLD mouse: Attention to the gut-vascular barrier dysfunction. Life sciences 209: 157-166
- Wigg AJ, Roberts-Thomson IC, Dymock RB, McCarthy PJ, Grose RH, et al. (2001) The role of small intestinal bacterial overgrowth, intestinal permeability, endotoxaemia, and tumour necrosis factor alpha in the pathogenesis of non-alcoholic steatohepatitis. Gut 48(2): 206-211.
- Hussain M, Umair Ijaz M, Ahmad MI, Khan IA, Bukhary SUF, et al. (2020) Gut inflammation exacerbates hepatic injury in C57BL/6J mice via gut-vascular barrier dysfunction with high-fat-incorporated meat protein diets. Food Funct 11(10): 9168-9176.
- Dai X, Hou H, Zhang W, Liu T, Li Y, et al. (2020) Microbial Metabolites: Critical Regulators in NAFLD. Front Microbiol 11: 567654.
- Mansour A, Mohajeri-Tehrani MR, Karimi S, Sanginabadi M, Poustchi H, Enayati S (2020) Short term effects of coffee components consumption on gut microbiota in patients with non-alcoholic fatty liver and diabetes: A pilot randomized placebo-controlled, clinical trial. EXCLI J 19: 241-250.
- Rodríguez-Daza MC, Pulido-Mateos EC, Lupien-Meilleur J, Guyonnet D, Desjardins Y, et al. (2021) Polyphenol-Mediated Gut Microbiota Modulation: Toward Prebiotics and Further. Front Nutrition 8: 689456.
- Peng JH, Leng J, Tian HJ, Yang T, Fang Y, et al. (2018) Geniposide and Chlorogenic Acid Combination Ameliorates Non-alcoholic Steatohepatitis Involving the Protection on the Gut Barrier Function in Mouse Induced by High-Fat Diet. Front Pharmacol 9: 1399.
- Bernsmeier C, Meyer-Gerspach AC, Blaser LS, Jeker L, Steinert RE, et al. (2014) Glucose-induced glucagon-like Peptide 1 secretion is deficient in patients with non-alcoholic fatty liver disease. PLoS One 9(1): e87488.
- Teshome G, Ambachew S, Fasil A, Abebe M (2020) Efficacy of Glucagon-Like Peptide-1 Analogs in Nonalcoholic Fatty Liver Disease: A Systematic Review. Hepat Med 12: 139-151.
- Lutz TA, Osto E (2016) Glucagon-like peptide-1, glucagon-like peptide-2, and lipid metabolism. Curr Opin Lipidol 27(3): 257-263.
- Sánchez-Valle V, Chávez-Tapia NC, Uribe M, Méndez-Sánchez N (2012) Role of oxidative stress and molecular changes in liver fibrosis: a review. Curr Med Chem 19(28): 4850-4860.
- Chen X, Yang JH, Cho SS, Kim JH, Xu J, et al. (2020) 5-Caffeoylquinic acid ameliorates oxidative stress-mediated cell death via Nrf2 activation in hepatocytes. Pharm Biol 58(1): 999-1005.
- Lu H, Tian Z, Cui Y, Liu Z, Ma X (2020) Chlorogenic acid: A comprehensive review of the dietary sources, processing effects, bioavailability, beneficial properties, mechanisms of action, and future directions. Compr Rev Food Sci Food Saf 19(6): 3130-3158.






























