Efficacy and Safety of Tenofovir Disoproxil Fumarate in Senegalese Patients with Chronic Hepatitis B
Salamata Diallo1, Marie Louise Bassène1, Mamadou Ngoné Gueye2, Mariéme Polele Fall1, Mame Aissé Thioubou3, Cheikh Ahmadou Bamba Cissé2, Alioune Badara Fall1, Atteib Fall1, Alsine Yauck1, Daouda Dia1, Mouhamadou Mbengue1 and Mamadou Lamine Diouf1
1Cheikh Anta Diop university of Dakar, Hospital Aristide Le Dantec, Senegal
2Cheikh Anta Diop university of Dakar, Hospital Idrissa Pouye, Senegal
3Assane Seck university of Ziguinchor, Hospital de la paix Ziguinchor, Senegal
Submission:October 28, 2021; Published:November 02, 2021
*Corresponding author: Salamata Diallo, Cheikh Anta Diop university of Dakar, Department of gastroenterology and hepatology of Hospital Aristide Le Dantec Dakar Senegal
How to cite this article: Salamata D, Marie Louise B, Mamadou N G, Mariéme P F, Mame A T, et al. Efficacy and Safety of Tenofovir Disoproxil Fumarate in Senegalese Patients with Chronic Hepatitis B. Adv Res Gastroentero Hepatol, 2021; 18(1): 555976. DOI: 10.19080/ARGH.2021.18.555976.
Abstract
Background: Tenofovir disoproxil fumarate (TDF) demonstrated a high antiviral efficacy and a good safety profile in patients with chronic hepatitis B (CHB). The objectives of this study were to evaluate the efficacy and safety of TDF in CHB in Senegal.
Materials and methods: We retrospectively analyzed CHB who were treated with TDF therapy at the Department of Gastroenterology and hepatology of Aristide Le Dantec hospital of Dakar (Senegal), from January 2015 to December 2019. Demographic, clinical, biochemical, virological, compliance and safety data were collected. The data were entered and analysed with Microsoft office Excel and R2.2.9 software.
Results: In total, 162 patients were examined in this study. The mean age was 40.38 ± 11, 41 years old and 64.2 % of the patients were males. Eighty-eight patients (54.3 %) had cirrhosis and 12 patients (7.4 %) were positive for HBeAg. After 48 weeks, 83.7 % of patients in non-liver cirrhosis group achieved virologic response (HBV DNA< 26 IU/mL) and 93.1 % in cirrhosis group. At 48 weeks, 90.7% patients had normalization of ALT. TDF was well tolerated in the study. During the study, median in serum creatinine remained relatively stable over 48 weeks of treatment and twenty-seven patients have hypophosphatemia. Three patients developed HCC during treatment.
Conclusion: TDF was effective in achieving complete viral suppression in Senegalese patients with CHB in both cirrhotic and non-liver cirrhosis patients. The TDF was safe and well tolerated in the study.
Keywords: Hepatitis B; Cirrhosis; Tenofovir; Senegal
Abbreviations: HBV: Hepatitis B Virus; CHB: Chronic Hepatitis B; HCC: Hepatocellular Carcinoma; LC: Liver Cirrhosis; ETV: Entecavir; TDF: Tenofovir Disoproxil Fumarate; TAF: Tenofovir Alafenamide; Non-LC: Non-Liver Cirrhosis; EASL: European Association for the Study of the Liver; ALT: Alanine Aminotransferase; HBeAg: Hepatitis B e Antigen; CVR: Complete Virological Response
Introduction
Hepatitis B infection is a serious global public health problem and a leading cause of death. Worldwide, some 257 million people have chronic hepatitis B virus (HBV) [1]. The prevalence was highest in the African (6.1%) and Western Pacific regions (6.2%) [2]. The spectrum of liver disease in persons with chronic hepatitis B (CHB) ranges from minimal fibrosis to cirrhosis and hepatocellular carcinoma (HCC). Without intervention, 15- 40% of patients with CHB are at high risk of developing serious complications, such as liver cirrhosis (LC), liver failure, and hepatocellular carcinoma (HCC) [2]. Worldwide, it is estimated that around 650 000 people die each year from the complications of CHB [3]. Sub-Saharan Africa is an area of high endemicity of VHB with 60 million of patients with chronic hepatitis B (CHB) [2]. It is estimated that 87890 HBV carriers die each year from cirrhosis and hepatocellular carcinoma (HCC) in this area [2]. In Senegal, the prevalence of CHB is estimated at 11% [1].
The main goal of therapy for patients with chronic HBV infection is to improve survival and quality of life by preventing disease progression, and consequently HCC development. The goal of antiviral therapy for CHB is to reduce (or reverse) necroinflammatory change and hepatic fibrosis leading to progressive liver disease, cirrhosis, decompensated cirrhosis and liver failure, HCC and death [3]. According to several international guidelines entecavir (ETV), tenofovir disoproxil fumarate (TDF), Tenofovir alafenamide (TAF) are recommended as first line therapy in the treatment of naïve CHB because of their higher antiviral potency and higher genetic barriers [3,4]. TDF is a selective inhibitor of HBV DNA polymerase reverse transcriptase. Several studies have reported high efficacy and a favorable safety profile with TDF treatment. TDF has been available in Senegal since 2012. To our knowledge, few studies have been conducted on the efficacy and safety of TDF in CHB in Sub-Saharan Africa. The objectives of this study were to evaluate the efficacy and safety of TDF in CHB in Senegal.
Materials And Methods
Study design and data collection
We retrospectively analyzed CHB who were treated with TDF therapy at the Department of Gastroenterology and hepatology of Aristide Le Dantec hospital of Dakar (Senegal), from January 2015 to December 2019. The study enrolled treatment- naïve patients ≥ 18 years of age with CHB. All patients did not take any other antiviral agent except TDF during follow up period.
A total of 162 CHB cases treated with TDF for at least 12 months were screened for the study and evaluated. Non-Liver Cirrhosis (NonLC) was defined as chronic necroinflammatory liver disease caused by HBV infection, without cirrhosis. Cirrhosis was defined based on the ultrasound or histopathological test or non-invasive markers of fibrosis. Cirrhosis at ultrasonography was defined as heterogeneous liver echotexture, irregular surface, splenomegaly, and the features of portal hypertension. Decompensated cirrhosis was established if there was ascites, variceal bleeding, hepatic encephalopathy.
Treatment eligibility
The treatment eligibility criteria were based on the European Association for the Study of the Liver (EASL) guidelines which recommend treatment in the following situations [3]:
a) HBeAg positive or negative chronic CHB, defined by HBV DNA > 2,000 IU/ml, ALT >ULN and/or at least moderate liver necroinflammation or fibrosis
b) Compensed or decompensated cirrhosis
c) HBV DNA >20,000 IU/ml and ALT > 2xULN
d) HBeAg-positive chronic HBV infection, defined by
persistently normal ALT and high HBV DNA levels, when they are older than 30 years
e) Family history of HCC or cirrhosis and extrahepatic manifestations
f) Candidates for chemotherapy and immunosuppressive therapy
Inclusion criteria were
CHB patient who are eligible to treatment, who continued antiviral treatment for > 12 months and available basal and visit clinical and laboratory data.
Exclusion criteria were
Patients with immunodeficiency diseases, autoimmune diseases, co-infection with other hepatitis viruses, alcoholic liver disease or cancer. Demographic, clinical, biochemical, and serologic test parameters including age, sex, prior history of variceal bleeding, encephalopathy, ascites, and hospitalization as a result of CHB, hemogram, alanine aminotransferase (ALT), aspartate aminotransferase, total bilirubin, albumin, prothrombin, creatinine, phosphorus, hepatitis B e antigen (HBeAg) status, HBV DNA, Child-Pugh score were recorded were collected at baseline 12th, 24th, 48th weeks.
Biochemical response was defined as a normalized ALT (≤ 1 times ULN). Complete virological response (CVR) was defined as a decline in HBV‐DNA levels to < 26 UI/mL. Serum HBV DNA was measured using the Roche COBAS TaqMan assay (lower limit of quantification of 26 UI/mL). The virological breakthrough was confirmed ≥ 1 log10 increase in HBV DNA level from nadir. Serological response was defined as the disappearance of HBe Ag positivity (HBeAg loss) and then HBeAc positivity (HBeAg seroconversion).
Statistical analysis
The data were entered and analysed with Microsoft office Excel and R2.2.9 software.
Results
Baseline patient characteristics
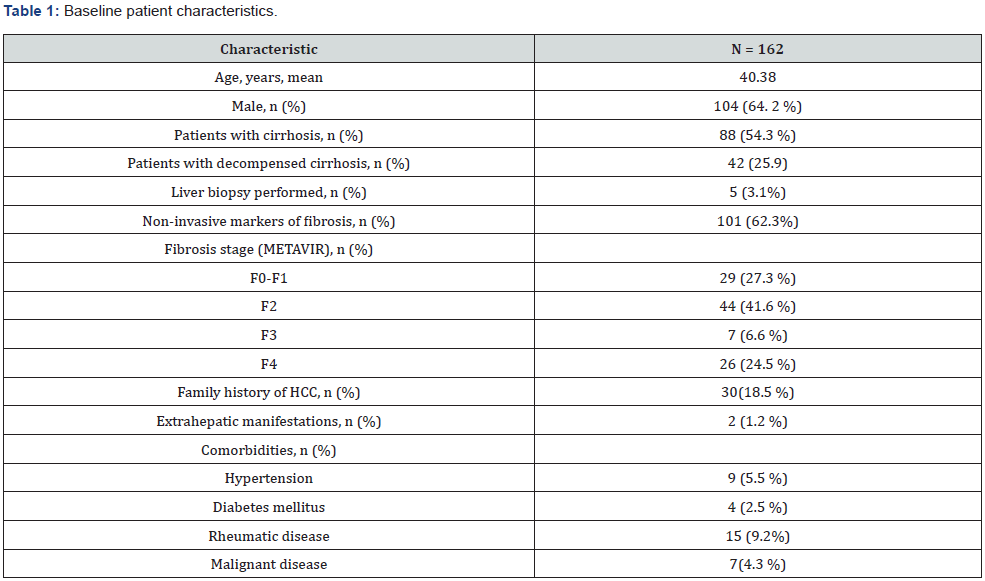
In total, 162 patients were examined in this study from January 2015 to December 2019. The baseline characteristics of the patients are summarized in Table 1 & Table 2. The mean age was 40.38 ± 11.41 years old and 64.2 % of the patients were males. Eighty-eight patients had cirrhosis and 74 patients had non liver cirrhosis. Forty-two patients had decompensed cirrhosis. Twelve patients (7.4 %) were positive for HBeAg.
Thirty patients had a family history of HCC and twentytwo patients were candidates for chemotherapy and immunosuppressive therapy. Two patients had extrahepatic manifestations.
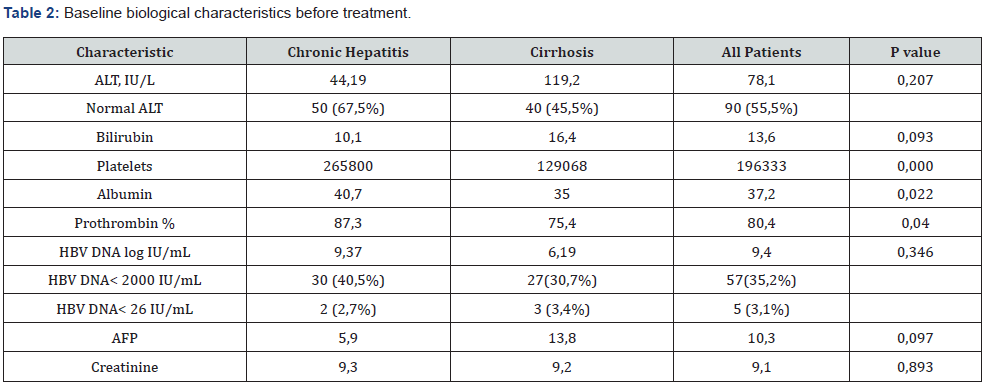
The mean HBV DNA levels and ALT levels were 9.4 log10 IU/ mL and 78.05 IU/ L, respectively. The mean HBV DNA level was 9.37 log10 IU/mL in non-LC group and 6.19 log10 IU/mL in cirrhosis group. Mean serum levels of ALT was 119.2 IU/L in the cirrhosis and 44.19 IU/L in the non-LC group. One hundred and one patients had a report of non-invasive markers of fibrosis (62.3 %) and five patients had histopathological test at baseline (3.1 %). The majority had moderate to severe fibrosis (METAVIR score 41.6 % (F2), 6.6 % (F3), 24.5 % (F4).
HCC: Hepatocellular Carcinoma
ALT: Alanin Aminotransferase; AFP: Alfafoetoprotein
In the decompensated cirrhosis group, 25 (59.5 %) patients had ascites, 3 (7.1 %) had episodes of hepatic encephalopathy, and 17 (40.4 %) experienced variceal bleeding. Upper GI endoscopy was available for all the patients who had cirrhosis and 30 (39.4%) had esophageal varices, 17 (22.3 %) had gastric varices and 13 (17.1%) had gastropathy. On average, patients were treated for 20.7 months (12-54).
Virological and biochemical response
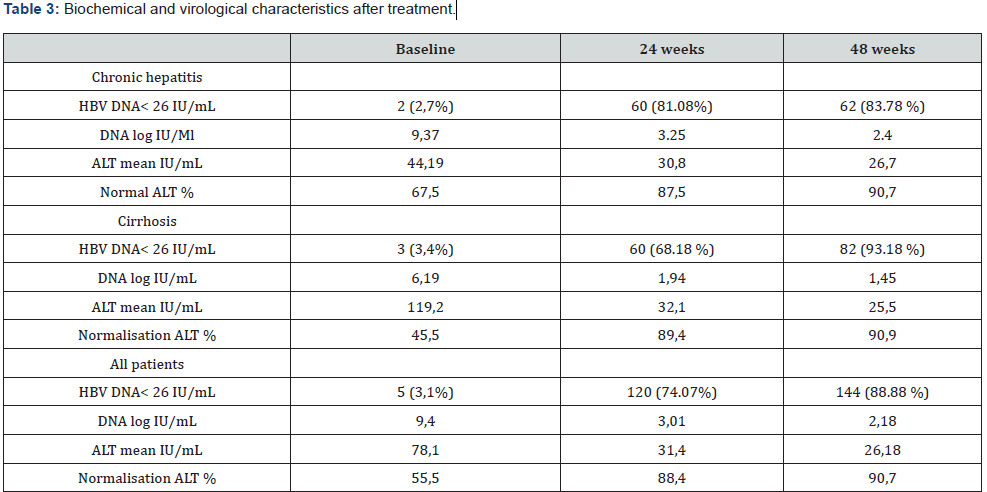
After 24 weeks the proportion of patients achieving HBV DNA levels < 26 IU/ml was 73.4 % (n=119) and rising to 88.8 % (n= 144) after 48 weeks Table 3 & Figure 1. After 24 weeks of treatment the mean change in HBV DNA levels from baseline in the non-LC group was – 6.12 log10 /IU mL and – 4.29 log10 IU /mL in cirrhosis group. After 48 weeks of treatment the mean change in HBV DNA levels from baseline in the non-LC group was – 6.96 log10 /IU mL and – 4.74 log10 IU /mL in cirrhosis group.
ALT: Alanin Aminotransferase
CVR rate at week 48 in the non-LC group was 83.7 % (n=62) and 93.1 % in cirrhosis group (n=82). Virological breakthrough developed in 2 patients. For these patients, breakthrough was transient. Mean ALT improved from 78.1 UI/ mL at baseline to 26.18 UI/ mL at week 48. ALT normalization at week 48 was achieved in 90.7%. Proportion of ALT normalization was also not significantly different compared non-LC group (91.4%) and cirrhosis group (90.9%). No case of HBeAg loss or HBsAg loss occurred in the study.
The change of mean Child-Pugh score was -0.55 after 48 weeks of treatment. Seventeen patients on cirrhosis group achieved a ≥1 point decrease in Child-Pugh score after 12 months of therapy. Two patients had aggravation of Child-Pugh score after 12 months of TDF. A problem of compliance was reported for eight patients, and they had interrupted therapy for >1 month.
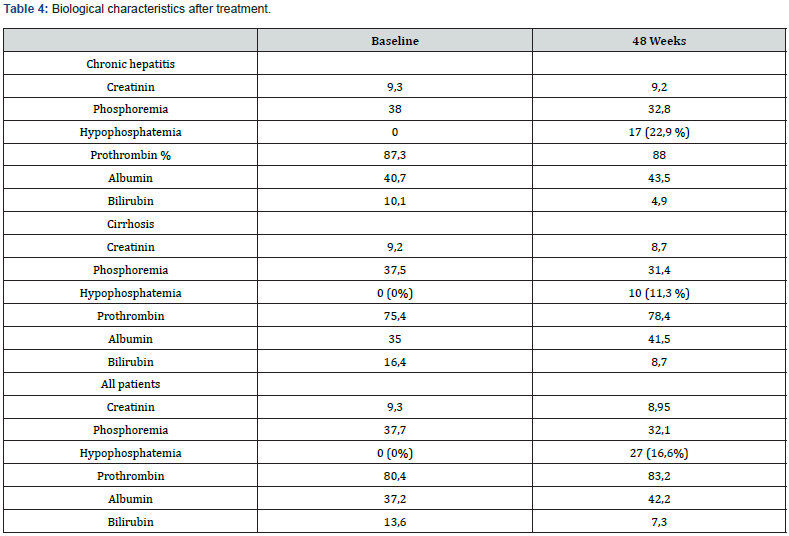
Safety
TDF was well tolerated in the study. Three patients reported 3 side effects: asthenia, dizziness, musculoskeletal pain. Serious adverse effects such as lactic acidosis was reported in any of the patients. During the study, median in serum creatinine remained relatively stable over 48 weeks of treatment. Five patients had increase serum creatinine levels. Twenty-seven patients have hypophosphatemia during the TDF treatment (Table 4). Three patients developed HCC during treatment. All patients were reported to have cirrhosis at baseline. There were 3 deaths during the study [HCC n = 1; hepatic encephalopathy n = 1; breast cancer n = 1]. Neither death was related to TDF.
Discussion
The major complication in patients with CHB is the risk of development of complications such as cirrhosis and HCC. The main goal of therapy for patients with chronic HBV infection is to improve survival and quality of life by preventing disease progression and consequently HCC development. This study evaluated the therapeutic efficacy of TDF monotherapy both in non-liver cirrhosis and cirrhosis patients. More than half of the patients were over 40 years old, many had advanced liver disease at enrolment. Majority had significant liver fibrosis, of which 46.9 % had cirrhosis.
Majority of patients in our cohort were HBeAg negative. In the past few decades, HBeAg negative CHB has become the commonest type of HBV infection in many countries of the world. The studies indicating that in African CHB patients only 10 % remain HBeAgpositive in their twenties. The precore variant emerges during HBV infection particularly in patients infected at birth or in the early years of life from infected family members [5]. Patients with HBeAg negative CHB have exacerbations with high viral load, elevated ALT, and histologic activity, and have been shown to develop cirrhosis faster than patients with HBeAg positive CHB. The higher rates of cirrhosis development in HBeAg negative CHB reflect the advanced stage of liver disease at presentation and diagnosis [5].

The study revealed that TDF monotherapy was highly effective in both groups: non liver cirrhosis and cirrhosis. The risk of cirrhosis and/or HCC in CHB increases proportionally as serum HBV DNA levels increase [6]. This finding suggests that sustained suppression of HBV replication with anti-HBV therapy may successfully prevent the progression of chronic liver diseases. Thus, the suppression of HBV replication should be considered the most important therapeutic goal for CHB patients in clinical settings. The primary target of antiviral therapy is the complete suppression of viral replication, thus delaying the progression of the liver disease. HBV replication can be sustainably suppressed by anti-HBV therapies to prevent and/or retard cirrhosis and subsequently HCC. In the present study, viral suppression occurred in majority of both non-LC group and Cirrhosis group at 48 weeks (83,7% vs 93,1%, p=0,147). There was no significant difference observed in rate of viral suppression between non-LC patients and cirrhosis patients. The mean reduction in serum HBV DNA levels from baseline was -6,96 log10 UI/mL in the non-LC group and – 4,29 in the cirrhosis group (P = 0.159)
Our result was in accordance with the evidence from the literature. Monotherapy with TDF at daily dose of 300mg has shown high rates of viral suppression in randomized trials and real-life cohorts [7-11]. Viral suppression of CHB naïve subjects under TDF reached 100% in hepatitis B e antigen (HBeAg) positive and 98% in HBeAg-negative subjects after 10-year extension phase of randomized clinical trials [12]. Viral suppression rate in field-practice studies yielded slightly lower suppression rate, generally above 90% in treatment-naïve patients for TDF regimens, regardless of HBeAg status [9,13].
A real-life multicenter prospective study of 440 CHB patients from France after 12 months, 92 % of the overall cohort achieved virologic response [7]. A study in China, viral suppression after 96 weeks of treatment with TDF was achieved in >95% of patients [14]. Studies from Burkina Faso and Mali suppressed viremia after 6 months treatment with TDF was achieved in respectively72.7% and 84.3% [10,11]. Alanine aminotransferase (ALT) normalization under antiviral treatment has been associated with a decrease in viral replication, tissue damage and necroinflammation. In our study after 48 weeks 90,7 % of the patients had a normal ALT. Conventional ALT cut offs in most laboratories are established at 40IU/ ml, although the American Association for the Study of Liver Diseases (AASLD) settled ALT cut-offs at 30IU/L for men and 19IU/L for women. A real-world cohort study of 21,182 patients receiving TDF or ETV show that an ALT decline below AASLD thresholds during the first year of treatment was associated with fewer hepatic events after 6-year follow-up (3.51% versus 5.70%, p<0.001), including HCC [15]. The patients with CHB would have a lower risk of hepatic events if they achieve normal on-treatment ALT in the first 12 months of NA treatment. The higher the ALT levels during treatment, the higher the risk of hepatic events [15].
HBsAg loss is the ideal endpoint of therapy because it indicates suppression of both HBV replication and viral protein expression and is, therefore, associated with better clinical outcomes. A large multicenter nationwide study performed in Hong Kong including 20,263 treated patients showed that HBsAg clearance confers additional benefits over viral suppression on reducing HCC risk (0.6% versus 5.6% at 8years, p < 0.001) but not on liver decompensation, liver transplantation and liver related mortality [16].
Unfortunately, HBsAg loss is achieved infrequently with the currently available antiHBV agents, including TDF. In our study no patient had loss HBsAg. The benefit of an effective antiviral treatment is to improve patient survival by reducing liver decompensation, liver transplantation and mortality. Benefits in CHB patients are illustrated by several studies and seem to be more remarkable in patients with cirrhosis [7,17]. A multicenter study on 1088 cirrhotic patients also showed benefits of TDFtreated patients compared with a historical cohort of untreated individuals; TDF treatment was independently associated with reduced risks of HCC (aHR 0.46, p <0.01), liver decompensations (aHR 0.28, p=0.01) and death or liver transplant (aHR 0.06, p<0.01) [18]. TDF therapy in decompensated cirrhosis patients was effective for decreasing HBV DNA levels and improving hepatic function with relatively lower CVR than in compensated cirrhosis [19].
HCC development is still a subject of concern; since oncogenic risk seems to decrease but not disappear in non-cirrhotic CHB patients that achieve treatment response. Persistence of cccDNA with damage in cellular repair and oxidative stress have been proposed as underlying mechanisms that could explain carcinogenesis in patients without significant fibrosis. HCC is currently considered the main threat for CHB patients’ survival [8].
During the study 03 patients developed HCC. All patients the 03 patients were cirrhotic and had achieved virologic response. This finding confirmed that cirrhotic patients should be carefully undergone surveillance for HCC regardless of achieving virological response. During the study, median in serum creatinine remained relatively stable over 48 weeks of treatment. This finding is report in previous controlled studies in which few patients who received TDF present significant renal adverse [7,8,20]. Our study has some limitations. The study was retrospective, and the number of patients was small. Our results need to be validated in future by long term multicentric, a prospective randomized clinical trial with larger numbers of patients.
Conclusion
TDF was effective in achieving complete viral suppression in Senegalese patients with CHB in both cirrhotic and nonliver cirrhosis patients. Cirrhotic patients should be carefully undergone surveillance for HCC regardless of achieving virologic response. The TDF was safe and well tolerated in the study.
References
- Schweitzer A, Horn J, Mikolajczyk RT, Krause G, Ott JJ (2015) Estimations of worldwide prevalence of chronic hepatitis B virus infection: a systematic review of data published between 1965 and 2013. The Lancet Oct 386(10003): 1546‑15
- WHO | Global hepatitis report (2017) WHO World Health Organization.
- (2017) EASL 2017 Clinical Practice Guidelines on the management of hepatitis B virus infection. J Hepatol 67(2): 370-398.
- Sarin SK, Kumar M, Lau GK, Abbas Z, Chan HLY, et al. (2016) Asian-Pacific clinical practice guidelines on the management of hepatitis B: a 2015 update. Hepatol Int. Janv 10(1): 1‑
- Shimakawa Y, Lemoine M, Njai HF, Bottomley C, Ndow G, et al. (2016) Natural history of chronic HBV infection in West Africa: a longitudinal population-based study from The Gambia. Gut 65(12): 2007‑20
- Chen CJ, Hwai IY, Jun S, Chin LJ, San LY, et al. (2006) Risk of Hepatocellular Carcinoma Across a Biological Gradient of Serum Hepatitis B Virus DNA Level. JAMA 295(1): 65.
- Marcellin P, Zoulim F, Hézode C, Causse X, Roche B, et al. (2016) Effectiveness and Safety of Tenofovir Disoproxil Fumarate in Chronic Hepatitis B: A 3-Year, Prospective, Real-World Study in France. Dig Dis Sci 61(10): 3072‑30
- Liang X, Gao Z, Xie Q, Zhang J, Sheng J, et al. (2019) Long-term efficacy and safety of tenofovir disoproxil fumarate in Chinese patients with chronic hepatitis B: 5-year results. Hepatol Int. Mai 13(3): 260‑26
- Riveiro BM, Tabernero D, Calleja JL, Lens S, Manzano ML, et al. (2017) Effectiveness and Safety of Entecavir or Tenofovir in a Spanish Cohort of Chronic Hepatitis B Patients: Validation of the Page-B Score to Predict Hepatocellular Carcinoma. Dig Dis Sci. mars 62(3): 784‑7
- Samaké DKW, Coulibaly SHW, Dicko MY, Diallo AM, Sidibe SSDW, et al. (2019) Effectiveness and Tolerance of Tenofovir Disoproxil Fumarate in Sub-Saharan Viral B Cirrhotic Patients. Open J Gastroenterol 9(2): 43‑4
- Bobilwindé SSMO, Fahosiata N, Léonce ZS, Damien OZ, Mali K, et al. (2021) Viral Hepatitis B under Tenofovir in Ouagadougou (Burkina Faso). Open J Gastroenterol 11(8): 141‑
- Chen M, Wang H, Zheng Q, Zheng X, Fan J, et al. (2019) Comparative efficacy of tenofovir and entecavir in nucleos(t)ide analogue-naive chronic hepatitis B: A systematic review and meta-analysis. 14(11): e0224773.
- Lampertico P, Soffredini R, Yurdaydin C, Idilman R, Papatheodoridis GV, et al. (2014) Four years of tenofovir monotherapy for NUC naïve field practice European patients suppresses HBV replication in most patients with a favorable renal safety profile but does not prevent HCC in patients with or without cirrhosis. Dig Liver Dis 46: e14.
- Zheng S, Liu L, Lu J, Zhang X, Shen H, et al. (2019) Efficacy and safety of tenofovir disoproxil fumarate in Chinese patients with chronic hepatitis B virus infection: A 2-year prospective study. Medicine (Baltimore). 98(42): e17590.
- Wong GLH, Chan HLY, Tse YK, Yip TCF, Lam KLY, et al. (2018) Normal on-treatment ALT during antiviral treatment is associated with a lower risk of hepatic events in patients with chronic hepatitis B. J Hepatol 69(4): 793‑
- Yip TCF, Wong GLH, Chan HLY, Tse YK, Lam KLY, et al. (2019) HBsAg seroclearance further reduces hepatocellular carcinoma risk after complete viral suppression with nucleos(t)ide analogues. J Hepatol mars 70(3): 361‑
- Buti M, Fung S, Gane E, Afdhal NH, Flisiak R, et al. (2015) Long-term clinical outcomes in cirrhotic chronic hepatitis B patients treated with tenofovir disoproxil fumarate for up to 5 years. Hepatol Int. avr 9(2): 243‑2
- Liu H, Shi Y, Hayden JC, Ryan PM, Rahmani J, et al. (2020) Tenofovir Treatment Has Lower Risk of Hepatocellular Carcinoma than Entecavir Treatment in Patients with Chronic Hepatitis B: A Systematic Review and Meta-Analysis. Liver Cancer 9(4): 468‑4
- Lee SK, Song MJ, Kim SH, Lee BS, Lee TH, et al. (2017) Safety and efficacy of tenofovir in chronic hepatitis B-related decompensated cirrhosis. World J Gastroenterol 23(13): 2396-2403.
- Gish RG, Clark MD, Kane SD, Shaw RE, Mangahas MF, et al. (2012) Similar risk of renal events among patients treated with tenofovir or entecavir for chronic hepatitis B. Clin Gastroenterol Hepatol 10(8): 941-946.






























