Stage II/III Rectal Cancer Post-Treatment Surveillance Patterns of Care: A SEER- Medicare Study
Catherine Chioreso1, Mary C Schroeder2, Irena Gribovskaja Rupp3, Eric Ammann1, Knute D Carter4, Charles F Lynch1,5, Elizabeth A Chrischilles1 and Mary E Charlton1,5*
1Department of Epidemiology, University of Iowa College of Public Health, USA
2Department of Pharmacy Practice and Science, University of Iowa College of Pharmacy, USA
3Department of Surgery, University of Iowa Carver College of Medicine, USA
4Department of Biostatistics, University of Iowa College of Public Health, USA
5Iowa Cancer Registry, University of Iowa College of Public Health, USA
Submission:September 13, 2021; Published:September 30, 2021
*Corresponding author:Mary E Charlton, Iowa Cancer Registry, University of Iowa College of Public Health, Iowa City, IA, USA
How to cite this article:Catherine C, Mary C S, Irena Gribovskaja R, Eric A, Knute D C, et al. Stage II/III Rectal Cancer Post-Treatment Surveillance Patterns of Care: A SEER- Medicare Study.Adv Res Gastroentero Hepatol, 2021; 17(5): 555972. DOI: 10.19080/ARGH.2021.17.555972.
Abstract
Introduction:Despite high rectal cancer recurrence rates, knowledge on post-treatment surveillance utilization is limited. Hence, this study aims to estimate patterns of post-treatment surveillance and determine associated factors.
Patients and Methods:Retrospective study of 1,024 SEER-Medicare patients >65 years old diagnosed with stage II/III rectal cancer between 2007-2013. Logistic regression was used to determine factors associated with ≥1 colonoscopy, ≥2 physician visits, ≥2 carcinoembryonic antigen (CEA) tests and ≥2 computed tomographic colonography (CT) within 14 months after primary treatment.
Results:Fifty-five percent had ≥1 colonoscopy, 54% had ≥2 physician visits, 47% had ≥2 CEA tests and 20% had ≥2 CTs. In multivariable logistic models, younger age and receipt of chemoradiation therapy (vs none) were significant across all surveillance procedures while clinical factors such as comorbidity were not. Being married (OR=1.69; 95% CI: 1.26-2.26) and proximity to a high-volume hospital (≤15 vs >30 minutes, OR=1.56; 95% CI: 1.00-2.43) were associated with ≥1 colonoscopy. Female gender (OR=1.56; 95% CI: 1.17-2.09), being married (OR=1.56; 95% CI: 1.17-2.08), white race (OR=1.79; 95% CI: 1.23- 2.62) and surgery from high-volume surgeon (OR=1.47; 95% CI: 1.06-2.04) were associated with ≥2 physician visits. Female gender (OR=1.45; 95% CI: 1.08-1.95), being married (OR=1.46; 95% CI: 1.08-1.96) and surgery from high-volume surgeon (OR=1.55; 95% CI: 1.10-2.17) had higher ≥2 CEA tests.
Conclusion: Post-treatment surveillance remains low but is more common among younger patients and recipients of chemoradiation. Distinct profiles of patient characteristics and provider volume were associated with individual surveillance procedures suggesting the need for multicomponent strategies to increase surveillance.
Keywords: Rectal cancer; Follow-up; Surveillance; Post-treatment patterns of care
Abbreviations: CEA: Carcinoembryonic Antigen Tests; Chemo: Chemotherapy; CI: Confidence Interval; CMS: Centers for Medicare and Medicaid Services; CPT: Current Procedural Terminology; CRT: Chemoradiation Therapy; CT: Computed Tomographic Colonography; DME: Durable Medical Equipment; FRI: Function-Related Indicator; HHA: Home Health Agency; HVH: High-Volume Hospital; HVS: High-Volume Surgeon; ICD_9: International Classification of Diseases, Ninth Revision Clinical Modification; MEDPAR: Medicare Provider Analysis and Review; NCCN: National Comprehensive Cancer Network; NCH: National Claims History; NCI: National Cancer Institute; OR: Odds Ratio; PEDSF: Patient Entitlement and Diagnosis Summary File; RUCA: Rural-Urban Commuting Area Classification; SEER: Surveillance Epidemiology and End Results; TME: Total Mesorectal Excision; US: United States of America
Introduction
Approximately 45,000 rectal cancer cases in the US are expected in 2021 [1]. Advances in rectal cancer management, such as total mesorectal excision (TME) and neoadjuvant therapy, have been associated with reduced recurrences and better survival outcomes [2,3]. Despite these rectal cancer management advances, 5-year survival rates average 64% mostly due to high stage II/III rectal cancer recurrence rates (≈ 40%) [4,5]. Previous research has suggested that early asymptomatic recurrence detection via post-treatment surveillance doubles the odds of receiving curative surgery [6] and can ultimately improve survival outcomes [4,7-9].
Between 2007 and 2020, the National Comprehensive Cancer Network (NCCN) recommended post-treatment colonoscopy one year after primary rectal cancer therapy while physician visits, carcinoembryonic antigen (CEA) tests and pelvic computed tomographic colonography (CT) were recommended every 3-6 months in the first two years after primary rectal cancer therapy [10]. Previous studies have reported inconsistent colorectal cancer post-treatment surveillance uptake [11-15]. and there is limited knowledge on factors associated with receipt of stage II/ III rectal cancer surveillance. Therefore, the objective of this study is to determine utilization rates and factors associated with posttreatment colonoscopy, physician visits, CEA tests and CTs for stage II/III rectal cancer patients.
Materials and Methods
Data sources
The University of Iowa Institutional Review Board approved this Surveillance, Epidemiology and End Results (SEER)- Medicare retrospective cohort study. SEER data contain demographic, tumor, cancer treatment and survival information from 18 populationbased cancer registries representing approximately 28% of the US [16,17]. Medicare data contain diagnoses and procedure information for 94% of the US population aged ≥65 years.17 The National Cancer Institute (NCI) and Centers for Medicare and Medicaid Services (CMS) link SEER and Medicare data by date of birth, social security number and gender
Study population
The study population (Figure 1) met the following inclusion criteria:

a) primary stage II/III rectal (ICD-O-3 site: C209) adenocarcinoma (histology: 8140-8571) patients who aged into Medicare and were not diagnosed via autopsy or death certificate between January 2007 and August 2013 at age 66+
b) no simultaneous cancer diagnosed within 6 months of diagnosis
c) identifiable Medicare rectal cancer surgery date within 6 months of diagnosis
d) consistent date of death between SEER and Medicare files
e) continuous Parts A and B Medicare coverage and no HMO coverage during study period to enable complete healthcare utilization assessment
f) no history of inflammatory bowel disease
g) sufficient 14-month follow-up time before 31 December 2014 (last Medicare follow-up date in study dataset). Since patients at the end of life have different patterns of care, those who were admitted into hospice care or died before or during the post-treatment posttreatment surveillance period were excluded [12].
Patients who had a recurrence before the post-treatment surveillance period (n=314) were excluded since they were ineligible for surveillance. Patients who had recurrence during the surveillance period (n=603) were excluded to ensure the identification of surveillance-related procedures [11,12,18]. Recurrent cancer diagnosis and treatment after primary cancer surgery were derived using International Classification of Diseases, Ninth Revision Clinical Modification (ICD-9) diagnosis and Current Procedural Terminology, Fourth Edition (CPT) codes (Appendix Table A1) [19]. Recurrent cancer surgery was identified >90 days after surgery to factor in surgical complications. If a patient had surgery but no adjuvant therapy, chemotherapy or radiotherapy received >120 days after surgery was identified as recurrent treatment. Among patients who had surgery and adjuvant therapy, chemotherapy or radiotherapy received >90 days after last adjuvant therapy claim was considered recurrent treatment. The date of recurrence was identified as the minimum date of recurrence diagnosis or treatment.19 Patients whose adjuvant treatment was >6 months (i.e., start date to end date) were excluded since it could be indicative of recurrence.
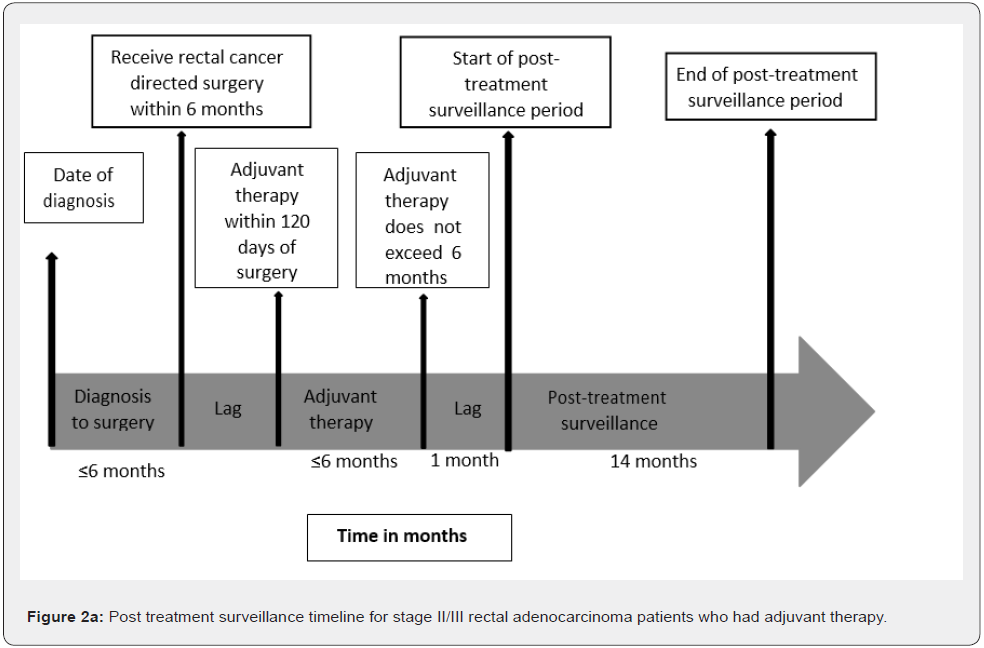
Rectal cancer treatment and post-treatment surveillance
Rectal cancer surgery was derived from CPT and ICD-9 codes (Appendix Table A1) [20]. Medicare CPT and ICD-9 codes and their respective dates were used to derive chemoradiation therapy (CRT) sequence (i.e., no CRT, neoadjuvant CRT plus adjuvant chemotherapy, neoadjuvant CRT, and adjuvant CRT); adjuvant therapy had to be within 120 days of primary surgery treatment. The last date of treatment for patients who did not receive adjuvant therapy was date of surgery (Figure 2a), but for those who received adjuvant therapy, it was the last adjuvant chemotherapy or radiotherapy claim (Figure 2b). The posttreatment surveillance period began 30 days after the last rectal cancer primary treatment date (i.e., surgery, chemotherapy or radiation). Patients were observed for 14 months after primary rectal cancer treatment to allow for scheduling delays encountered in real-world clinical care.

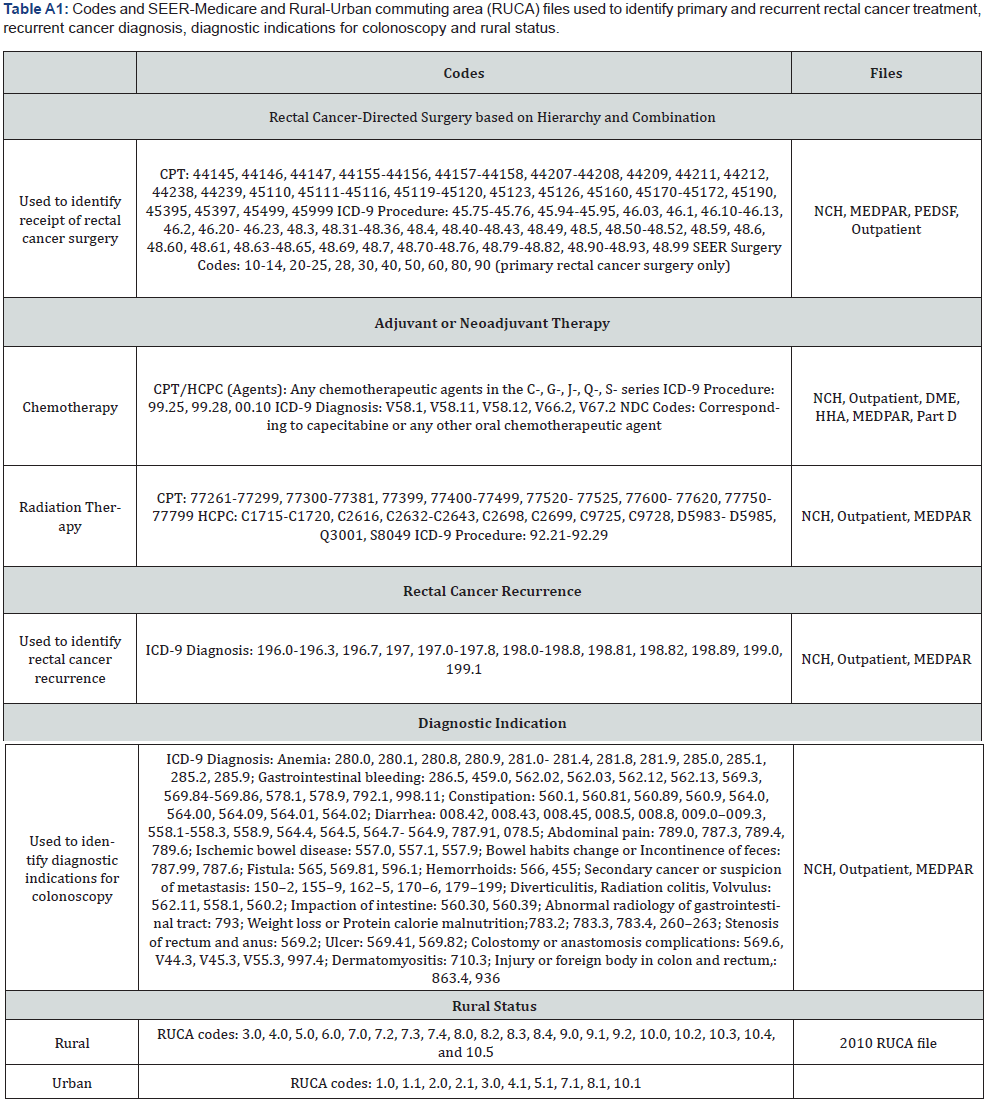
CPT and ICD-9 codes were used to identify the frequency and time to first post-treatment colonoscopy (CPT: 44388- 44389, 44392-44394, 44397, 45100, 45108, 45300, 45303, 45305, 45307-45309, 45315, 45317, 45320-45321, 45327, 45330- 45335, 45337-45342, 45345, 45355, 45378-45387, 45391-45392, 45382-45385, 45499, 45990, 45999, 74261- 74263, 74270, 74280, 82270-82272, 82274, 10021-10022, G0104-G0107, G01020, G0122, G0328, G0464; ICD-9: 45.21- 45.29, 45.41-45.43, 48.21-48.26, 48.29, 54.11, 89.34), physician visits to oncologist, primary care provider and surgeon specialties that typically perform surveillance (CPT: 99201-99245, 99381- 99397; ICD-9: V70, V70.0, V70.9, V72, V72.9), CEA test (CPT: 82378) and CT (CPT: 71250, 71260, 71270, 72191-72194, 74150, 74160, 74170, 71275, 74175-74178, 75635) [11-15]. Physician specialty was derived from National Claims History (NCH) and American Medical Association files. Subsequent procedures were counted if they occurred >90 days after prior procedure to avoid over-estimating surveillance (e.g., colonoscopy redo due to incomplete bowel preparation) [14]. Colonoscopies with ICD- 9 codes for symptoms, such as gastrointestinal bleeding and anemia (Appendix Table A1), were considered ‘indicated’ (vs. for surveillance purposes only) [21].
Patient characteristics
Patient age, gender, marital status, race and AJCC 6th edition stage were extracted from SEER Patient Entitlement and Diagnosis Summary File (PEDSF). PEDSF patient ZIP code was used to classify rural status using the 2006 Rural–Urban Commuting Area (RUCA) classification system [22]. Percent living below the federal poverty level and percent with at least a 4- year college education from the Tract census file were dichotomized by median percentage [23]. A one-year Medicare claims lookback period was used to derive Charlson comorbidity scores and Function-Related Indicators (FRIs). The Charlson score is an established predictor of one-year survival [24]. FRIs reflect diminished functional capacity based on diagnosis codes for conditions such as dementia, malnutrition and home oxygen use [25].
Surgical complications, such as surgical site infection, were defined using Hendren et al.’s algorithm [26]. Hospital and surgeon rectal cancer surgery volume was defined as the sum of 2007-2013 SEER-Medicare rectal cancer surgeries within 6 months of diagnosis [20]. this correlates well with total volume [27,28] Hospitals (≥14 surgeries) and surgeons (≥5 surgeries) in the fourth volume quartile were considered high-volume hospitals (HVH) and high-volume surgeons (HVS), respectively. As published previously, NCI designated comprehensive centers were classified as HVHs because they are an indicator of quality cancer care while colorectal cancer surgeons or surgical oncologists were considered HVSs due to their specialized training [20]. Travel time between the centroids of patient residence and the nearest HVH ZIP code were calculated [29] and used as a proxy for access to care [30].
Statistical analysis
The 4 individual measures of post-treatment surveillance (≥1 colonoscopy, ≥2 physician visits, ≥2 CEA tests, and ≥2 CTs) were the outcomes of interest. The Chi-square test was used to determine variation in surveillance uptake by patient characteristics. Multivariable logistic regression was used to ascertain factors associated with surveillance. In four separate sensitivity analyses we
a) excluded patients with ‘indicated’ colonoscopies to account for potential reason for procedure
b) restricted physician visits to oncologists and colorectal surgeon specialists to test robustness of results for visits to cancer physician specialists
c) included recurrent patients in the analysis and considered them adherent on the basis that the goal of surveillance is to detect recurrence to estimate potential maximum surveillance rates
d) excluded patients aged 80+ who tend to forgo recurrence treatment to determine extent of recurrence misclassification on surveillance rates [16,19].
Results
Overall study population
The median age of the eligible 1,024 patients with stage II/ III rectal cancer was 77 (IQR: 71-82). Most patients were white (86%) and lived in urban areas (78%). Half of the patients were married. Forty-seven percent had a Charlson score ≥1 and 35% had ≥1 function-related indicator (indicative of functional impairment). Fifty-two percent of patients had neoadjuvant CRT (with or without adjuvant therapy), 8% had adjuvant CRT and 25% had no CRT. More than sixty percent of patients had surgery from HVS (62%) and HVH (62%).
Colonoscopy
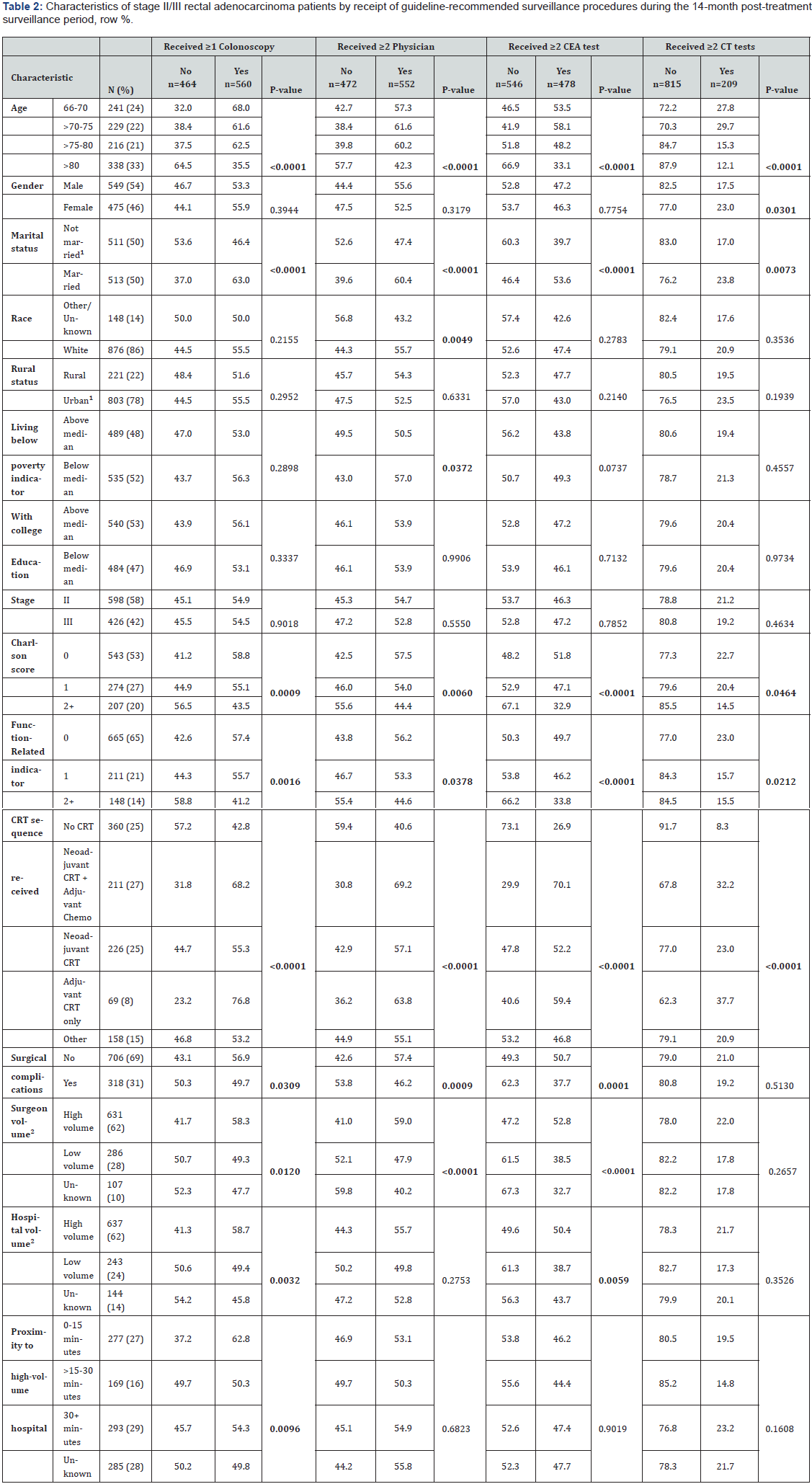
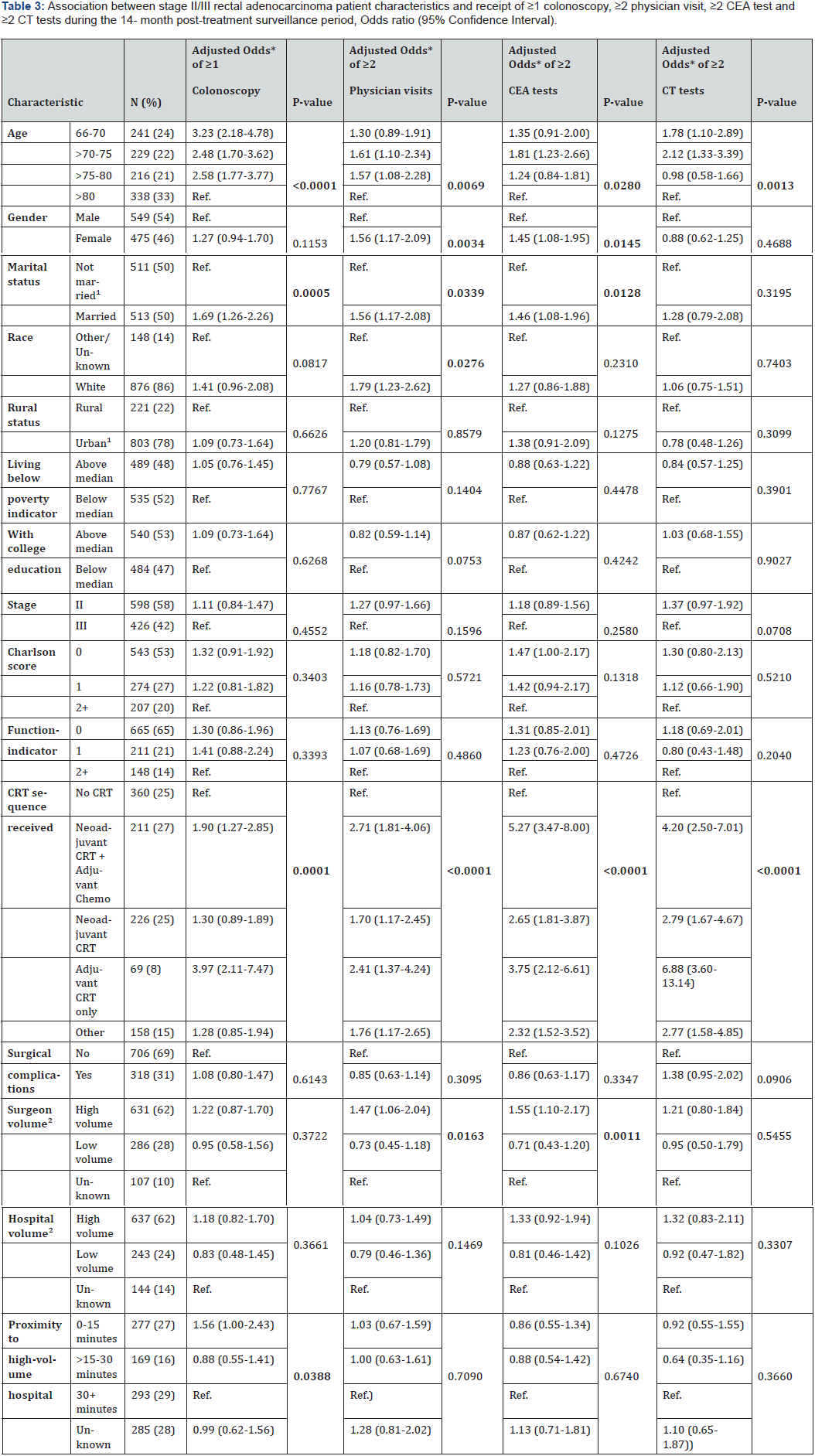
As Table 1 shows, 560 (55%) patients received ≥1 colonoscopies. The median months from the end of treatment to first colonoscopy were 5 (IQR: 3-9). The median number of colonoscopies for the entire cohort and for patients who had ≥1colonoscopies was 1 (IQR=0-1) and 1 (IQR=1-2), respectively. In bivariate analyses, younger age, being married, lower comorbidity, lower function-related indicator, any CRT sequence (vs none), not experiencing surgical complications, surgery from HVS, surgery from HVH, and proximity to HVH were associated with having ≥1 colonoscopy (Table 2).
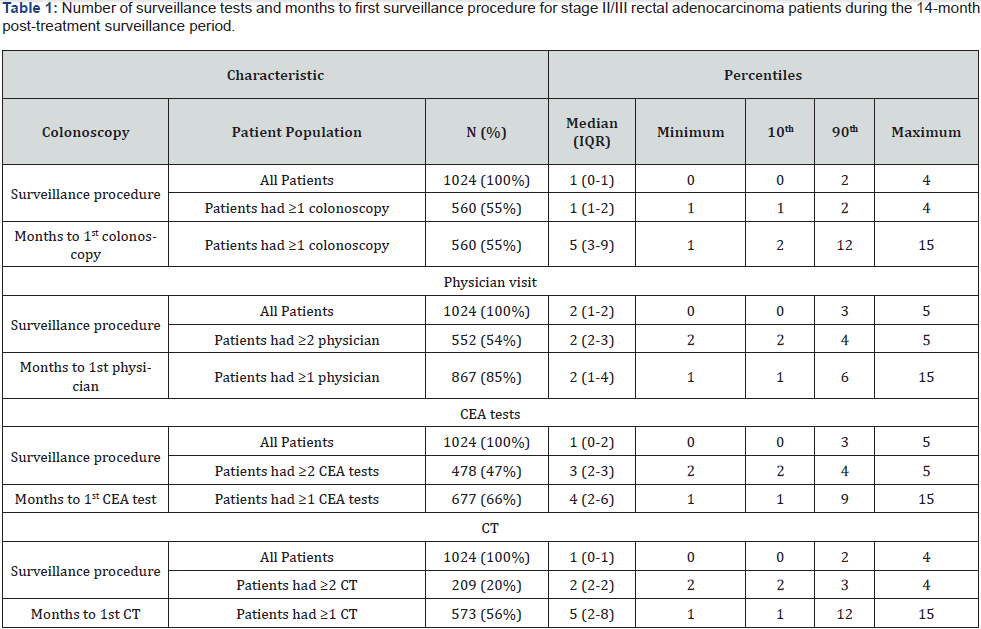
In multivariable analysis, younger age (>66-70 vs > 80 years, OR=3.23; 95% CI: 2.18-4.78; >70-75 vs > 80 years, OR=2.48; 95% CI: 1.70-3.62; >75-80 vs > 80 years, OR=2.58; 95% CI: 1.77-3.77), being married (OR=1.69; 95% CI: 1.26-2.26) and residing closer to a HVH (≤15 vs >30 minutes, OR=1.56; 95% CI: 1.00-2.43) were associated with receiving ≥1 colonoscopy (Table 3). Compared to no CRT, receiving adjuvant CRT (OR=3.97; 95% CI: 2.11-7.47) and neoadjuvant CRT plus adjuvant chemotherapy (OR=1.90; 95% CI: 1.27-2.85) were associated with having ≥1 colonoscopy.
Physician visit
As Table 1 shows, 552 (54%) patients received ≥2 physician visits (85% had ≥1). The median months to first physician visit was 2 (1-4). The median number of physician visits for all patients was 2 (IQR=1-2) and 2 (IQR=2-3) for those who had ≥2 physician visits. In bivariate analyses, having ≥2 physician visits was associated with younger age, being married, white race, living below the federal poverty indicator level, lower comorbidity, lower function-related indicator, any CRT sequence (vs none), not experiencing surgical complications, and surgery from HVS (Table 2)
As shown in Table 3, younger age (>70-75 vs > 80 years, OR=1.61; 95% CI: 1.10-2.34; >75-80 vs > 80 years, OR=1.57; 95% CI: 1.08-2.28), female gender (OR=1.56; 95% CI: 1.17-2.09), being married (OR=1.56; 95% CI: 1.17-2.08), white race (OR=1.79; 95% CI: 1.23-2.62) and surgery from HVS (OR=1.47; 95% CI: 1.06-2.04) were associated with higher odds of receiving ≥2 physician visits. Compared to no CRT, receiving neoadjuvant CRT plus adjuvant chemotherapy (OR=2.71; 95% CI: 1.81-4.06), neoadjuvant CRT (OR=1.70; 95% CI: 1.17-2.45) and adjuvant CRT (OR=2.41; 95% CI: 1.37-4.24) were associated with ≥2 physician visits.
CEA tests
Forty-seven percent (n=478) of the cohort had ≥2 CEA tests; 66% had ≥1 CEA test (Table 1). The median months to first CEA test was 4 (IQR=2-6). The median number of CEA tests for the entire cohort and patients who had ≥2 CEA tests was 1 (IQR=0-2) and 3 (IQR=2-3), respectively. In bivariate analyses, receiving ≥2 CEA tests was associated with younger age, being married, lower comorbidity, lower function-related indicator, any CRT sequence (vs none), not experiencing surgical complications, surgery from HVS and surgery from HVH (Table 2).
Younger age (OR=1.81; 95% CI: 1.23-2.66), female gender (OR=1.45; 95% CI: 1.08-1.95), being married (OR=1.46; 95% CI: 1.08-1.96) and surgery from HVS (OR=1.55; 95% CI: 1.10-2.17) had higher odds of having ≥2 CEA tests (Table 3). Compared to no CRT, receiving neoadjuvant CRT plus adjuvant chemotherapy (OR=5.27; 95% CI: 3.47-8.00), neoadjuvant CRT (OR=2.65; 95% CI: 1.81-3.87) and adjuvant CRT (OR=3.75; 95% CI: 2.12-6.61) were associated with ≥2 CEA tests.
CT tests
As shown in Table 1, 209 (20%) patients had ≥2 CTs (56% had ≥1). The median months to first image was 5 (IQR=2- 8). The median number of CTs for the entire cohort and those who had ≥2 CT tests was 1 (IQR=0-1) and 2 (IQR=2-2), respectively. In bivariate analyses, younger age, female gender, being married, lower comorbidity, lower function-related indicator and any CRT sequence (vs none) were associated with ≥2 CTs (Table 2).
Younger age (66-70 vs > 80 years, OR=1.78; 95% CI: 1.10- 2.89; >70-75 vs > 80 years, OR=2.12; 95% CI: 1.33-3.39) was associated with associated with ≥2 CTs (Table 3). Compared to no CRT, receiving adjuvant CRT (OR=6.88; 95% CI: 3.60-13.14), neoadjuvant CRT plus adjuvant chemotherapy (OR=4.20; 95% CI: 2.50-7.01) and neoadjuvant CRT (OR=2.79; 95% CI: 1.67-4.67) had higher odds of ≥2 CTs.
Sensitivity analyses
The results of the various sensitivity analysis are not shown. In a sensitivity analysis excluding 204 patients with ‘indicated’ colonoscopies, 43% (n=356) of patients received ≥1 colonoscopy and surgery from HVS (OR=1.64; 95% CI: 1.11- 2.43) was significantly associated with ≥1 colonoscopy (not significant in main analysis). Compared to the main analysis, a sensitivity analysis restricting physician visits to oncologists and colorectal surgeon specialists had similar post-treatment surveillance utilization rate (42%) and identical significant predictors of ≥2 physician visits.
Given that the goal of surveillance is to detect recurrence, we did a sensitivity analysis in which recurrent patients were included in the study and considered adherent; median months to recurrence was 5 (IQR=3-10) and the recalculated surveillance rates increased to 71% for ≥1 colonoscopy, 71% for ≥2 physician visits, 66% for ≥2 CEA tests and 50% for ≥2 CTs. In another sensitivity analysis excluding patients aged 80+, surveillance rates increased to 64%, 60%, 53% and 24% for colonoscopies, physician visits, CEA tests and ≥2 CTs, respectively
Discussion
Among the 1,024 patients with stage II/III rectal cancer, receipt of guideline-recommended surveillance colonoscopy (55%), physician visits (54%), CEA tests (47%) and CTs (20%) was low at 14 months post-treatment. However, since a higher percentage of these patients received at least one physician visit (85%), CEA test (66%) and CT (56%) within the recommended timeline, this suggests that most patients initiate but do not complete post-treatment surveillance. The reasons for this phenomenon are unclear but previous studies have suggested that lack of patient self-management tools, patient preferences and failure to adequately communicate with patient play a role in receipt of guideline-recommended surveillance [31,32].
A key finding consistent with previous research is that younger age was significantly associated with receipt of all four guidelinerecommended post-treatment surveillance measures [33]. The low surveillance among older patients is indicative of the clinical dilemma physicians face given the heterogeneity in physiological fitness among older patients that makes it challenging to achieve consensus on the risk-benefit of surveillance [33]. Higher incidence of side-effects from primary rectal cancer treatment among patients aged 80+ years may account for the low surveillance rates in older patients.33 It is possible that patient preferences or perceived risk-benefit by either the patient or physician may account for lower surveillance rates among older patients [33,34]. While clinical factors, such as comorbidities and functional impairment, could explain lower surveillance among older patients, after adjusting for multiple factors, those clinical factors were not significant predictors of receipt of any surveillance procedures in this study. Higher risk pathology or healthy-adherer bias may be driving the association between receipt of CRT and post-treatment surveillance.
Patient demographic characteristics and surgeon volume were not consistently significant across all surveillance procedures; this suggests that the weight of these factors in the decision to receive surveillance varies by surveillance procedure. Being married was associated with having guideline-recommended colonoscopies, physician visits and CEA tests likely because it offers psychosocial support, greater economic and logistical access [35,36]. Females had higher receipt of physician visits and CEA tests; the reason for this remains speculative but variation in gender preferences to cancer care [37] may explain this finding. The significant association between urban status and CEA test adherence may suggest variation in practice patterns by rurality as reported previously [38]. The significance of drive time to nearest HVH (colonoscopy model) and race (physician visits model) suggest the significance of access to quality care in surveillance receipt [20,39]. The higher odds of physician visits and CEA tests among HVSs suggest that surgeons play a major role in post-treatment surveillance referrals [40] this is more apparent in the absence of an imperative to seek care on the patient’s part by the significance of HVS in the sensitivity analysis of colonoscopies without indications (i.e., HVS not significant in main model including ‘indicated’ colonoscopies).
This study has several limitations such as lack of information on physician recommendations, patient preferences and reasons for surveillance. While lack of information on reasons for surveillance procedures could mean that physician visits could be for noncancer surveillance reasons, the similarity in results between the main analysis and sensitivity analysis restricting physician visits to oncologists and colorectal surgeon specialists suggests the extent of this is minimal. As published previously [19] the identification of recurrence based partly on receipt of treatment for the recurrence may not capture patients who decided to forgo treatment resulting in under-estimated surveillance rates for patients who opted out of recurrent cancer treatment, or an overestimation of surveillance if procedures were performed due to suspected recurrence. To minimize the impact of this, we excluded patients whose adjuvant treatment was >6 months or those who sought hospice treatment before or during the surveillance period. Similar to the algorithm by Deshpande et al. [19] which identified 18.4% of recurrent cancer, our study identified 16.7% rectal cancer recurrences. This published algorithm was reported to have 81% sensitivity and 99% specificity,19 supporting that our algorithm correctly excluded cases it detected as recurrent but under-identified them. While this could explain lower surveillance rates among patients aged 80+, exclusion of this population showed marginally higher but still sub-optimal surveillance rates. Even though an argument can be made that exclusion of recurrent patients may underestimate surveillance, the sensitivity analyses including recurrent cancer patients showed slightly higher but sub- optimal surveillance rates. Furthermore, since the median of 5 months to recurrence diagnosis suggests tests for recurrent patients were for diagnostic versus surveillance purposes, this supports the argument to exclude recurrent patients. Despite these limitations, the post-treatment surveillance estimates in this study are similar to recent studies [41,42] and add relevant knowledge to the ongoing problem of sub-optimal surveillance by describing factors associated with surveillance.
Conclusion
In conclusion, our study indicates sub-optimal post-treatment surveillance. This is a significant public health challenge given the high rectal cancer recurrence rates and sub-optimal survival outcomes [4,43]. Although associations with age and CRT treatment were relatively consistent across guidelinerecommended surveillance procedures, associations with patient characteristics and surgeon volume were specific to particular surveillance procedures suggesting that multicomponent interventions may be necessary to increase post-treatment surveillance uptake. Future studies should determine the types of multicomponent interventions that can bridge the gap between guideline-recommended care and real-world challenges to receipt of post-treatment surveillance.
Grant Support
This work was supported by NIH/NCI K07 Cancer Prevention, Control, Behavioral Sciences and Population Sciences Career Development Award: 1K07CA197067 and in part by NIH/NCI contract number HHSN261201300020I.
Author Contributions
All authors have made:
a) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data.
b) drafting the article or revising it critically for important intellectual content.
c) final approval of the version to be published.
References
- Siegel RL, Miller KD, Fuchs HE, Jemal A (2021) Cancer Statistics, 2021. CA Cancer J Clin 71(1): 7-33.
- Heald R, Ryall R (1986) Recurrence and survival after total mesorectal excision for rectal cancer. Lancet 327: 1479-1482.
- Kapiteijn E, Putter H, Van de Velde CJ, Cooperative investigators of the Dutch ColoRectal Cancer G (2002) Impact of the introduction and training of total mesorectal excision on recurrence and survival in rectal cancer in The Netherlands. Br J Surg 89(9): 1142-1149.
- Moy B, Jacobson BC, Tanabe KK, Goldberg RM, Laurence S, et al. (2009) Surveillance after colorectal cancer resection. UpToDateR, c2016.
- Siegel RL, Miller KD, Goding SA, Fedewa SA, Butterly LF, et al. (2020) Colorectal cancer statistics, 2020. CA Cancer J Clin 70(3): 145-164.
- Pita FS, Alhayek AM, Gonzalez MC, Lopez CB, Seoane PT, et al. (2015) Intensive follow-up strategies improve outcomes in nonmetastatic colorectal cancer patients after curative surgery: a systematic review and meta-analysis. Ann Oncol 26(4): 644-656.
- Steele SR, Chang GJ, Hendren S, Weiser M, Irani J, et al. (2015) Practice Guideline for the Surveillance of Patients After Curative Treatment of Colon and Rectal Cancer. Dis Colon Rectum 58(8): 713-725.
- Benson AB, Venook AP, Al Hawary MM, Cederquist L, Chen YJ, et al. (2018) NCCN Guidelines (R) Insights Colon Cancer, Version 2.2018. Featured Updates to the NCCN Guidelines. J Natl Compr Canc Netw 16(4): 359-369.
- Kahi CJ, Boland CR, Dominitz JA, Giardiello FM, Johnson DA, et al. (2016) Colonoscopy Surveillance after Colorectal Cancer Resection: Recommendations of the US Multi-Society Task Force on Colorectal Cancer. Am J Gastroenterol 111(3): 337-346.
- Benson AB, Venook AP, Al Hawary MM, Arain MA, Chen YJ, et al. (2020) NCCN Guidelines Insights: Rectal Cancer, Version 6.2020. J Natl Compr Canc Netw 18(7): 806-815.
- Vargas GM, Sheffield KM, Parmar AD, Han Y, Brown KM, et al. (2013) Physician follow-up and observation of guidelines in the post treatment surveillance of colorectal cancer. Surgery 154(2): 244-255.
- Paulson EC, Veenstra CM, Vachani A, Ciunci CA, Epstein AJ (2015) Trends in surveillance for resected colorectal cancer, 2001-2009. Cancer 121(19): 3525-3533.
- Cooper GS, Kou TD, Reynolds HL (2008) Receipt of guideline-recommended follow-up in older colorectal cancer survivors: a population-based analysis. Cancer 113(8): 2029-2037.
- Mollica MA, Enewold LR, Lines LM, Halpern MT, Schumacher JR, et al. (2017) Examining colorectal cancer survivors' surveillance patterns and experiences of care: a SEER-CAHPS study. Cancer Causes Control 28(10): 1133-1141.
- Snyder RA, Hu CY, Cuddy A, Francescatti AB, Schumacher JR, et al. (2018) Association Between Intensity of Posttreatment Surveillance Testing and Detection of Recurrence in Patients with Colorectal Cancer. JAMA 319(20): 2104-2115.
- Warren JL, Klabunde CN, Schrag D, Bach PB, Riley GF (2002) Overview of the SEER-Medicare data: content, research applications, and generalizability to the United States elderly population. Med Care 40(8): 3-18.
- Thomas KS, Boyd E, Mariotto AB, Penn DC, Barrett MJ, et al. (2018) New Opportunities for Cancer Health Services Research: Linking the SEER-Medicare Data to the Nursing Home Minimum Data Set. Med Care 56(12): e90- e96.
- Salz T, Weinberger M, Ayanian JZ, Brewer NT, Earle CC, et al. (2010) Variation in use of surveillance colonoscopy among colorectal cancer survivors in the United States. BMC Health Serv Res 10: 256.
- Deshpande AD, Schootman M, Mayer A (2015) Development of a claims-based algorithm to identify colorectal cancer recurrence. Ann Epidemiol 25(4): 297-300.
- Chioreso C, Gao X, Gribovskaja RI, Lin C, Ward MM, et al. (2019) Hospital and Surgeon Selection for Medicare Beneficiaries with Stage II/III Rectal Cancer: The Role of Rurality, Distance to Care, and Colonoscopy Provider. Ann Surg.
- Singh A, Kuo YF, Goodwin JS (2013) Many patients who undergo surgery for colorectal cancer receive surveillance colonoscopies earlier than recommended by guidelines. Clin Gastroenterol Hepatol 11(1): 65-72.
- Hart LG, Larson EH, Lishner DM (2005) Rural definitions for health policy and research. Am J Public Health 95(7): 1149-1155.
- Zeigler JCM, Tierney A, Rebbeck TR, Rundle A (2011) Prostate cancer severity associations with neighborhood deprivation. Prostate Cancer 846263.
- Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5): 373-383.
- Chrischilles EA, Schneider KM, Schroeder MC, Letuchy E, Wallace RB, et al. (2016) Association Between Preadmission Functional Status and Use and Effectiveness of Secondary Prevention Medications in Elderly Survivors of Acute Myocardial Infarction. J Am Geriatr Soc 64(3): 526-535.
- Hendren S, Birkmeyer JD, Yin H, Banerjee M, Sonnenday C, et al. (2010) Surgical complications are associated with omission of chemotherapy for stage III colorectal cancer. Dis Colon Rectum 53(12): 1587-1593.
- Bach PB, Cramer LD, Schrag D, Downey RJ, Gelfand SE, et al. (2001) The influence of hospital volume on survival after resection for lung cancer. N Engl J Med 345(3): 181-188.
- Begg CB, Cramer LD, Hoskins WJ, Brennan MF (1998) Impact of hospital volume on operative mortality for major cancer surgery. JAMA 280(20): 1747-1751.
- Schroeder MC, Chapman CG, Nattinger MC, Halfdanarson TR, Abu Hejleh T, et al. (2016) Variation in geographic access to chemotherapy by definitions of providers and service locations: a population-based observational study. BMC Health Serv Res 16(1): 274.
- Buhn S, Holstiege J, Pieper D (2020) Are patients willing to accept longer travel times to decrease their risk associated with surgical procedures? A systematic review. BMC Public Health 20.
- O Malley DM, Davis SN, Devine KA, Sullivan B, Bator A, et al. (2020) Development and usability testing of the e- EXCELS tool to guide cancer survivorship follow-up care. Psychooncology 29(1): 123-131.
- Goldenberg BA, Carpenter KT, Gingerich JR, Nugent Z, Sisler JJ (2020) Moving forward after cancer: successful implementation of a colorectal cancer patient-centered transitions program. J Cancer Surviv 14(1): 4-8.
- Li Destri G, Cavallaro M, Trovato MA, Ferlito F, Castaing M, et al. (2012) Colorectal cancer treatment and follow-up in the elderly: an inexplicably different approach. Int Surg 97(3): 219-223.
- Millan M, Merino S, Caro A, Feliu F, Escuder J, et al. (2015) Treatment of colorectal cancer in the elderly. World J Gastrointest Oncol 7(10): 204-220.
- Syse A, Lyngstad TH (2017) In sickness and in health: The role of marital partners in cancer survival. SSM Popul Health 3: 99-110.
- Martinez ME, Unkart JT, Tao L, Kroenke CH, Schwab R, et al. (2017) Prognostic significance of marital status in breast cancer survival: A population-based study. PLoS One 12(5): e0175515.
- Wessels H, De Graeff A, Wynia K, De Heus M, Kruitwagen CL, et al. (2010) Gender-related needs and preferences in cancer care indicate the need for an individualized approach to cancer patients. Oncologist 15(6): 648-655.
- Charlton M, Schlichting J, Chioreso C, Ward M, Vikas P (2015) Challenges of Rural Cancer Care in the United States. Oncology (Williston Park) 29(9): 633-640.
- Doubeni CA, Laiyemo AO, Major JM, Schootman M, Lian M, et al. (2013) Socioeconomic status and the risk of colorectal cancer: An analysis of more than a half million adults in the National Institutes of Health-AARP Diet and Health Study 118(114): 3636- 3644.
- Alfano CM, Jefford M, Maher J, Birken SA, Mayer DK (2019) Building Personalized Cancer Follow-up Care Pathways in the United States: Lessons Learned from Implementation in England. Northern Ireland, and Australia. Am Soc Clin Oncol Educ Book 39: 625-639.
- Boehmer U, Potter J, Clark MA, Winter M, Berklein F, et al. (2021) Follow-up surveillance among colorectal cancer survivors of different sexual orientations. J Cancer Surviv 1-10.
- Ramphal W, Boeding JRE, Schreinemakers JMJ, Gobardhan PD, Rutten HJT, et al. (2020) Colonoscopy Surveillance After Colorectal Cancer: the Optimal Interval for Follow-Up. J Gastrointest Cancer 51(2): 469-477.
- Siegel RL, Miller KD, Fedewa SA, Ahnen DJ, Meester RGS, et al. (2017) Colorectal cancer statistics, 2017. CA Cancer J Clin, 67(3): 177-193.






























