Tumor Budding in Esophageal Squamous Cell Carcinomas: Correlation with Prognosis and Grade of Tumor
Zainab Waseem1, Sajid Mushtaq1, Mudassar Hussain2, Usman Hassan1 and Maryam Hameed1
1Department of Pathology, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Pakistan
2Consultant Histopathology, Shaukat khanum Memorial Cancer Hospital and Research Center, Pakistan
Submission:June 18, 2021; Published:July 09, 2021
*Corresponding author: Zainab Waseem, Department of Pathology, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Pakistan
How to cite this article: Zainab W, Sajid M, Usman H, Maryam H. Tumor Budding in Esophageal Squamous Cell Carcinomas: Correlation with Prognosis and Grade of Tumor. Adv Res Gastroentero Hepatol, 2021; 17(3): 555962. DOI: 10.19080/ARGH.2021.17.555962.
Abstract
Introduction: Tumor Budding has emerged as a promising indicator of prognosis in various carcinomas of the body. Since it is a H&E feature, its evaluation can and should be included in pathology reports wherever feasible.
Aims: The aim of this study is to analyze the correlation of tumor budding with various prognostic factors of esophageal squamous cell carcinomas.
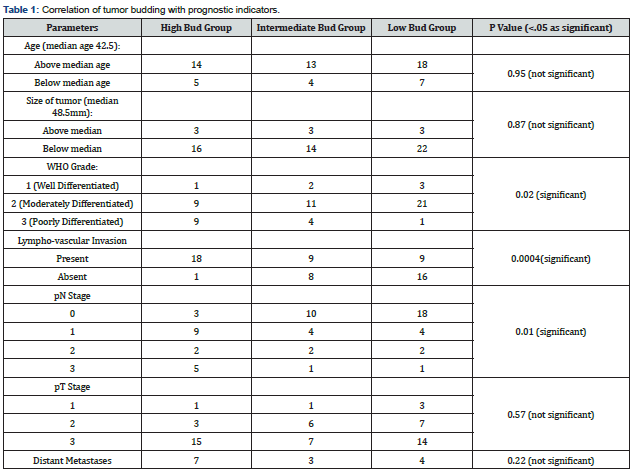
Method and Results: Sixty-one cases of esophageal resections reported as squamous cell carcinoma at Shaukat Khanum Memorial Cancer Hospital and Research Centre between 2018 and 2019 were reviewed for tumor budding. Budding was classified into high (10 or more buds), intermediate (5 to 9 buds) and low (4 and less) bud groups. Each group was then correlated with lympho-vascular invasion, pN, pT stages, grade of the tumor and 1-year survival. P values were obtained from chi square tests (significant if less than 0.05) and showed statistical significance of tumor budding with tumor grade (p= 0.02), lympho-vascular invasion (p= 0.0004) and pN stage (p= 0.03). Survival graphs were plotted using Kaplan Meier plot in SPSS software.
Conclusion: A higher bud group is a promising predictor of poor prognosis in esophageal squamous cell carcinomas and can assist in the planning of treatment in the patients.
Keywords: Keywords: Esophageal; Squamous Cell Carcinoma; Tumor Budding; Prognosis
Introduction
Introduction
Squamous cell carcinoma constitutes 90% of all the tumors in esophagus. It is the sixth most common cancer diagnosed and treated at Shaukat Khanum Hospital, Lahore, Pakistan. The current practice of estimating prognosis in these patients utilizes the grade and stage of tumor as determined by the AJCC TNM system, however, the search for better and more reliable prognostic markers continue. Tumor budding is one of the emerging and promising prognostic markers which is currently being studied in several carcinomas. A higher bud group has been reported to confer an adverse prognosis. In this study, we aim to correlate tumor budding with pT, pN, lympho-vascular invasion, grade of tumor and prognosis in patients diagnosed with esophageal squamous cell carcinoma at Shaukat Khanum Hospital, Lahore.
Materials and Methods
Ethical issues
The Institutional Review Board Approval was taken (IRB# EX-28-05-20-02).
Case selection
Sixty-one esophageal resections grossed at Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore, Pakistan from January 2018 to December 2019 were included in the study. These excluded cases of review originally grossed outside the hospital, and cases with suboptimal fixation. The blocks were fixed in 10% buffered formalin and embedded in paraffin. The tissues were then sectioned at 0.5um thickness and stained with hematoxylin and eosin. The slides were retrieved from hospital archives and reviewed by two pathologists.
Case review
The definition of tumor budding as per the UICC (Union for International Cancer Control) was employed. As per the UICC protocol, tumor buds are defined as groups of 4 cells or less dissociating at the tumor-host junction (Figure 1). Tumor Budding was counted and grouped according to the ITBCC guidelines. One hotspot field with the highest number of buds was selected after scanning all sections of tumor and they were counted on H&E sections. Eyepiece with a diameter of 20mm and 20X objective lens were used. Buds were then grouped as follows: (0-4) low bud group, (5-9) intermediate bud group and (10 and more) high bud group. Factors tabulated and correlated with budding group included age, gender, size of tumor, grade of tumor, pT and pN stages, lympho-vascular invasion and prognosis. P values were obtained using chi-square contingency tables and statistical significance was considered if p value was less than 0.05. Survival graph was plotted using Kaplan-Meier plot in SPSS software.
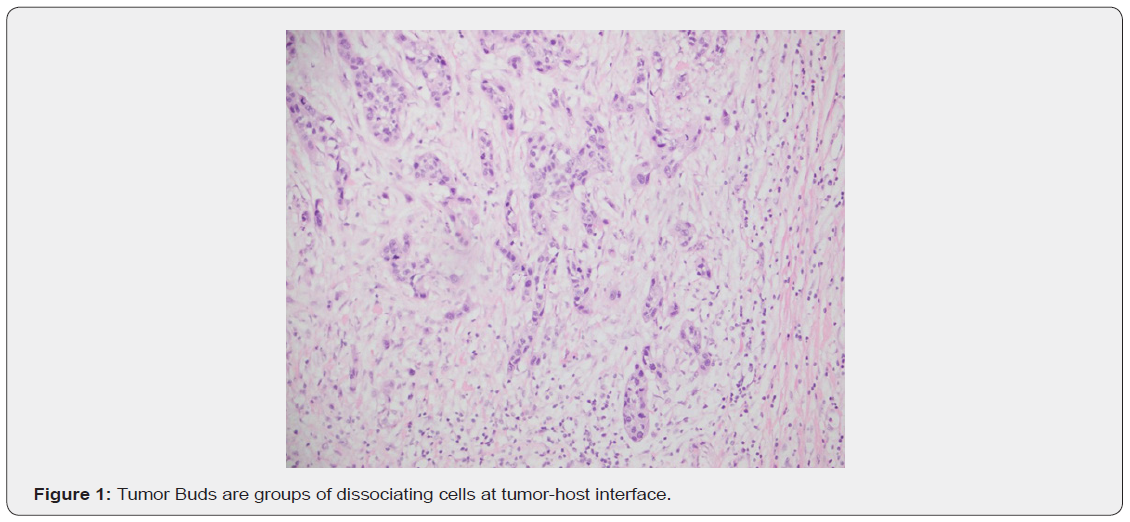
Results
A total of 19 out of 61 cases (31.15%) showed high budding, 17 of 61 (27.8%) showed intermediate budding and 25 of 61(40.9%) showed low budding.
Among the high bud group, 9 of 19(47.3%) were poorly differentiated tumors, 9 of 19 (47.3%) were moderately differentiated and 1of 19 (5.3%) showed well differentiation. 18 of 19 (94.7%) high bud group cases showed lymph-lympho-vascular invasion; 1 of 19 (5.3%) did not show lympho-vascular invasion; 16 out of 19 (84.2%) had nodal metastases while 3 of 19 (15.8%) did not. Of these 15 cases, 9 (56.3%) were staged pN1, 2 (12.5%) were pN2 and 5 (31.3%) were pN3. 7 of the 19 (50%) cases showed distant metastases. In the intermediate bud group, 4 of 17 (23.5%) cases showed poor differentiation, 11 of 17 (64.7%) were moderately differentiated and 2 out of 17 (11.8%) were well differentiated. 9 of 17 cases had lympho-vascular invasion while 8 of 17 did not show any lympho-vascular invasion. Of the 17 cases, 10 (58.8%) were pN0, 4 (23.5%) were pN1, 2 (11.8%) were pN2, and 1(5.9%) was staged as pN3. 3 of 17 cases showed distant metastases (21.4%). Out of 17 cases, one case showed pT4a stage, and one was not graded due to a size of 1mm. Out of the remaining 15 cases, 1 (6.7%) was pT1, 6/15 (33.3%) were pT2 and 7/15(46.7%) were pT3.
In the low budding group, 1 of 25 (4%) case had poor differentiation, 21 of 25 (84%) cases were moderately differentiated and 3 of 25 (12%) were well differentiated. 9 out of 25 cases had lympho-vascular invasion while 16 out of 25 did not. 18 of 25 (72%) cases were staged pN0, 4 out of 25 (16%) were pN1, 2 out of 25 (8%) were pN2 and 1 (4%) was staged pN3. 4 of 25 (28.6%) cases showed distant metastases. One case in the low bud group was not graded due to a size of less than 1 mm of the tumor. The remaining 24 cases were staged as follows: 3/24 (12.5%) were pT1, 7/24 (29.2%) were pT2 and 14/24 (58.3%) were pT3. P values were obtained from chi square 5 × 5 contingency tables and a cut-off of .05 was considered significant. Statistical significance of tumor budding with tumor grade (p= 0.02), lympho-vascular invasion (p= 0.0004) and pN stage (p= 0.01) was observed. The presence of nodal metastases also showed a statistical significance (0.0008). The statistical correlation with pT stage was not significant (p=0.4). Survival graph was plotted using the Kaplan Meier plot (Figure 1 & 2). The results are summarized in the Table 1.

Discussion
Tumor budding was first described in 1954, coined as the term “sprouting”. Imai T. recognized the phenomenon to be related to a clinically aggressive tumor behavior. Gabbert H, et al, in 1985, proposed that the peritumoral buds were distinct from the tumor bulk. Thus, tumor budding came to be broadly divided into peritumoral and intratumoral budding, of which the former is predominantly studied.
The formation of tumor buds is currently explained by Epithelial-Mesenchymal transition, defining a shift of cells towards embryological cell migration method. A downregulation of e-cadherin and activation of WNT pathway has been noted. There is also an increased expression of anti-apoptotic proteins. This can explain the resistance of cancers with higher buds to chemotherapy.
The phenomenon of tumor budding is currently being studied in various carcinomas of the body, including squamous cell carcinomas of the oral cavity, carcinomas of the gynecological tract, and adenocarcinomas of the lung. However, the reporting of tumor bud group in pathological reports is still not a standardized practice, even though an adverse prognosis is consistently reciprocated in these studies in conjunction with a higher bud group.
In a study of 135 patients by Jesinghaus et al. [1] a higher bud group in esophageal squamous cell carcinomas showed a strong association with a decreased disease-specific and disease-free survival. The same study proposed a unique quantification and grading system by introducing the assessment of cell nest size (TBNS: tumor-budding-cell-nest-grading system). A similar result is seen in our study, with a higher bud group showing a decreased overall survival. Furthermore, Jesinghaus et al. [1] also stated that the current WHO grading system of tumors did not correlate with prognosis (p=0.053). However, our study showed that a higher bud group correlated with a higher grade of tumor (p=0.02), and by inference, a higher grade correlated with poor prognosis.
In our study, bud group correlated positively with the pN stage (p=0.01) and lympho-vascular invasion (p=0.0004). This is consistent with the findings of Seki M et al. who studied tumor budding in oral squamous cell carcinomas in 209 patients. The study claimed that tumor budding had a statistically significant correlation with lymphatic vessel invasion, and in the event of intermediate or high bud group in cases of cN0, a prophylactic neck dissection is warranted. This confirms that tumor budding is a universal prognostic marker with reproducible results in various tumors. pT stage remained statistically insignificant in our study (p=0.57). This is also reciprocated in a few earlier research. Koelzer H et al. [2] however, found in a separate study that a higher bud group correlated with a higher pT stage in esophageal squamous cell carcinomas, after a thorough review of previously conducted studies. Moreover, our study did not show any statistically significant correlation between tumor budding and distant metastases (p=0.22). This is in stark difference to the EMT logic explained in detail by Koelzer V et al. [3].
In our opinion, the above-mentioned differences can be attributed to the regional differences in which the studies were conducted. The genetics and different carcinogens in Pakistani population may contribute to these differences. The assessment of tumor budding as a part of prognostic markers is highly facilitated by the fact that this is an H&E feature, and therefore, readily visible to a trained pathologist’s eye. The UICC proposed a quantitative method to analyze tumor buds, one which was employed in the data collection of this paper. Several variations exist. The most used method in the esophagus was devised by Ueno et al. [4] in which buds were categorized into high (more than 10 buds) and low (less than 10) groups. The attempt to quantify the tumor buds helps in reducing inter-observer variability and form a uniform model [5-10].
During our study, a major limitation that was felt was the almost compulsive use of resection specimens rather than the small, pre-operative biopsies. In our setting, the endoscopic mucosal biopsies received for pre-therapeutic histopathology usually yield only epithelium, with little lamina propria or muscularis mucosae [11-15]. This scanty tissue, often sent without any orientation, is a hurdle to the identification of tumor buds. The general recommendations are to send 3 biopsies, taken from the edge of the cancer with 5mm vertical and 8mm horizontal size 15. Tangential cuts fragmented and autolyzed biopsies continue to pose a further challenge to the pathologist’s eye [16-20].
Conclusion
Tumor budding is a reliable prognostic marker in cases of esophageal squamous cell carcinomas and should be reported alongside the grade of tumor. It is imperative to devise a way to assess tumor buds in small biopsy specimens to assist in management of the patients.
Acknowledgement
Zainab Waseem and Sajid Mushtaq wrote the paper. Mudassar Hussain provided additions for analysis. Usman Hassan and Maryam Hameed provided valuable data for analysis.
References
- Jesinghaus M, Boxberg M, Konukiewitz B, Slotta HJ, Schlitter AM, et al. (2017) A Novel Grading System Based on Tumor Budding and Cell Nest Size is a strong predictor of patient outcome in Esophageal Squamous Cell Carcinoma. Am J Surg Pathol 41(8): 1112-1120.
- Koelzer V, Assarzadegan N, Dawson H, Mitrovic B, Grin A, et al. (2017) Cytokeratin‐based assessment of tumour budding in colorectal cancer: analysis in stage II patients and prospective diagnostic experience. J Pathol Clin Res 3(3): 171-178.
- Koelzer V, Langer R, Zlobec I, Lugli A (2014) Tumor budding in Upper Gastrointestinal Carcinomas. Front Oncol 4: 216.
- Ueno H, Murphy J, Jass J, Mochizuki H, Talbot IC (2002) Tumour ‘budding’ as an index to estimate the potential of aggressiveness in rectal cancer. Histopathology 40(2): 127-132.
- Abnet C, Arnold M, Wei W (2018) Epidemiology of Esophageal Squamous Cell Carcinoma. Gastroenterology 154(2): 360-373.
- Khan M, Urooj N, Syed A, Khattak S, Kazmi A, et al. (2020) Prognostic Factors for recurrence in Esophageal Cancer Patients Treated with Neoadjuvant Therapy and Surgery: A Single-Institution Analysis. Cureus 12(5): e1808.
- Amin M, Edge S, Greene F (2017) AJCC Cancer Staging Manual (8th), Springer International Publishing New York, USA, pp. 185-202.
- Roh M, Lee J, Choi P (2004) Tumor Budding as a useful prognostic marker in Esophageal Squamous Cell Carcinoma. Dis Esophagus 17(4): 333-337.
- Lugli, A, Kirsch R, Ajioka Y, Bosman F, Cathomas G, et al. (2017) Recommendations for reporting tumor budding in colorectal cancer based on the International Tumor Budding Consensus Conference (ITBCC) 2016. Mod Pathol 30(9): 1299-1311.
- Imai T (1954) The Growth of Human Carcinoma: A Morphological Analysis. Fukuoka Igaku Zasshi 45: 13-43.
- Gabbert H, Wagner R, Moll R, Gerharz C (1985) Tumor Dedifferentiation: An Important Step in Tumor Invasion. Clin Exp Metastases 3(4): 257-279.
- Berg K, Schaeffer D (2018) Tumor budding as a standardized parameter in gastrointestinal carcinomas: more than just the colon. Mod Pathol 31(6): 862-872.
- Dawson H, Koelzer V, Karamitopoulou E, Economou M, Hammer C, et al. (2014) The Apoptotic and Proliferation rate of Tumor budding cells in Colorectal Cancer outlines a heterogeneous population of cells with various impacts on clinical outcome. Histopathology 64(4): 577-584.
- Rogers A, Gibbons D, Hanly A, Hyland J, Connell OP, et al. (2014) Prognostic significance of tumor budding in rectal cancer biopsies before neoadjuvant therapy. Mod Pathol 27(1): 156-162.
- Wang C, Huang H, Huang Z, Wang A, Chen X, et al. (2011) Tumor budding correlates with poor prognosis and epithelial-mesenchymal transition in tongue squamous cell carcinoma. J Oral Pathol Med 40(7): 545-551.
- Zare S, Aisagbonhi O, Hasteh F, Fadare O (2020) Independent Validation of Tumor Budding Activity and Cell Nest Size as Determinants of Patient Outcome in Squamous Cell Carcinoma of the Uterine Cervix. Am J Surg Pathol 44(9): 1151-1160.
- Yamaguchi Y, Ishii G, Kojima M, Yoh K, Otsuka H, et al. (2010) Histopathologic Features of the Tumor Budding in Adenocarcinoma of the Lung: Tumor Budding as an Index to Predict the Potential Aggressiveness. J Thorac Oncol 5(9): 1361-1368.
- Seki M, Sano T, Yokoo S, Oyama T (2017) Tumour budding evaluated in biopsy specimens is a useful predictor of prognosis in patients with cNo early stage oral squamous cell carcinoma. Histopathology 70(6): 869-879.
- Almangash A, Youssef O, Pirinen M, Sundström J, Leivo I, et al. (2019) Does Evaluation of Tumor Budding in Diagnostic Biopsies have a Clinical Relevance? A Systematic Review. Histopathology 74(4): 536-544.
- Barresi V, Bonetti LR, Ieni A, Branca G, Baron L, et al. (2014) Histologic grading based on counting poorly differentiated clusters in preoperative biopsy predicts nodal involvement and pTNM stage in colorectal cancer patients. Hum Pathol 45(2): 268-275.






























