Cholangiocarcinoma surveillance in patients with Inflammatory Bowel Disease and Primary Sclerosing Cholangitis
Sujani Yadlapati* and Thomas A Judge
Division of Gastroenterology, Cooper University Hospital at Rowan University School of Medicine, USA
*Corresponding author: Sujani Yadlapati, Division of Gastroenterology, Cooper University Hospital at Rowan University School of Medicine, Camden, NJ, USA
How to cite this article: Sujani Y, Thomas A J. Cholangiocarcinoma surveillance in patients with Inflammatory Bowel Disease and Primary Sclerosing Cholangitis. Adv Res Gastroentero Hepatol, 2021; 17(1): 555954. DOI: 10.19080/ARGH.2021.17.555954.
Abstract
Inflammatory bowel disease (IBD), which comprises of Crohn’s disease and Ulcerative colitis, is characterized by chronic inflammation in the gastrointestinal tract. These patients are at increased risk of extra-intestinal malignancies. Cholangiocarcinoma, although rare in incidence, is associated with IBD and primary sclerosing cholangitis (PSC). PSC occurs in about 5% of IBD patients. Lifetime risk of cholangiocarcinoma in patients with PSC is significant. Cholangiocarcinoma occurs at a younger age in IBD patients than in general population. The aim of cancer surveillance is to detect cancer or precancerous lesions in asymptomatic, high risk individuals with the target of prevention or early cure. Unfortunately, data driven surveillance guidelines for cholangiocarcinoma in patients with IBD and PSC do not yet exist. Per most societal guidelines a combination of cross-sectional imaging such as ultrasound or magnetic resonance imaging (MRI) and serial carbohydrate antigen 19-9 (CA 19-9) every 6-12 months can be used for surveillance. These surveillance strategies will be discussed further in this review along with role for Endoscopic retrograde cholangiopancreatography (ERCP) with brush cytology sampling.
Keywords: Crohn’s disease; Ulcerative colitis; Chronic inflammation; Gastrointestinal tract; Cholangiocarcinoma; Jaundice; Abdominal pain; Pruritis
Abbreviations: ERCP: Endoscopic Retrograde Cholangiopancreatography; MRI: Magnetic Resonance Imaging; PSC: Primary Sclerosing Cholangitis; IBD: Inflammatory Bowel Disease; CD: Crohn’s Disease; UC: Ulcerative Colitis; CT: Computerized Tomography; LFTs: Liver Function Tests; AASLD: American Association for the Study of Liver Diseases; MRCP: Magnetic Resonance Cholangiopancreatography; FISH: Fluorescence in Situ Hybridization; CBD: Common Bile Duct
Introduction
Inflammatory bowel disease (IBD), which comprises of Crohn’s disease (CD) and Ulcerative colitis (UC), is characterized by chronic inflammation in the gastrointestinal tract. Chronic inflammation increases risk of gastrointestinal malignancies. Guidelines outlining screening of colorectal cancer in these patients are currently well established. Patients with IBD are also at augmented risk for developing variety of extra-intestinal cancers. Cholangiocarcinoma, although rare in incidence, is associated with IBD and primary sclerosing cholangitis (PSC). Rates for cholangiocarcinoma appear to be two to six-fold higher in IBD patients compared to general population. Crude incidence as high as 24.2 out of 100,000 cases has been reported [1]. Ten-year cumulative risk of cholangiocarcinoma in IBD is about 0.07% [2,3]. There is an increased risk of cholangiocarcinoma in patients with UC compared to those with CD by two-fold, 8.2 vs 4.3 out of 100,000 cases respectively [3]. Cholangiocarcinoma occurs at a younger age in IBD patients than in general population, fifth decade vs. seventh decade respectively. The risk of cholangiocarcinoma in IBD patients with PSC is 160-fold higher than that of general population [4]. PSC occurs in about 5% of IBD patients and is considered an intermediate step in the development of cholangiocarcinoma [3,5]. The risk of cholangiocarcinoma in patients with PSC is approximately 9% over 10 years [6].
Cholangiocarcinoma carries a grave prognosis with a 5-year survival rate of less than 10% [7] Previously many studies have aimed to identify high risk IBD and PSC patients that would benefit from aggressive surveillance strategies. Smoking, alcohol use, longer duration of IBD, presence of colorectal cancer or dysplasia in UC patients, proctocolectomy, older age at PSC diagnosis and polymorphism of the NKG2D gene are some factors that have been associated with increased risk of cholangiocarcinoma. These studies are yet to be rigorously validated [8,9].
Surveillance Strategy
The aim of cancer surveillance is to detect cancer or precancerous lesions in asymptomatic, high risk individuals with the target of prevention or early cure. Up to the present time no proven screening tool for early detection of cholangiocarcinoma exists. Diagnosis of cholangiocarcinoma particularly in setting of PSC can often be technically challenging because the abnormalities of the biliary tree due to PSC obscure early cholangiocarcinoma recognition. PSC may display two different patterns. PSC may involve distal bile ducts in the absence of visible abnormalities on computerized tomography (CT) or magnetic resonance imaging (MRI). Diagnosis in these cases is often supported by presence of altered liver function tests (LFTs) and possibly a biopsy. These patients carry lower risk of cholangiocarcinoma compared to PSC involving large bile ducts. PSC involving intrahepatic ducts and biliary confluence is at high risk of malignant transformation.
Unfortunately, data driven surveillance guidelines for cholangiocarcinoma in patients with IBD and PSC do not yet exist. The position of American Association for the Study of Liver Diseases (AASLD) and American College of Gastroenterology (ACG) are similar. Per these societal guidelines a combination of cross-sectional imaging such as ultrasound or MRI and serial carbohydrate antigen 19-9 (CA 19-9) every 6-12 months can be used for surveillance [10,11]. On the other hand, European Association for Study of Liver Disease (EASL) states that no specific biochemical marker or imaging is recommended for early detection of cholangiocarcinoma [12]. Other surveillance strategies that will be discussed in this review include Endoscopic retrograde cholangiopancreatography (ERCP) with brush cytology sampling.
Imaging
A reasonable approach to screening is an interval radiologic assessment of biliary tree which is more commonly done with MRI with magnetic resonance cholangiopancreatography (MRCP) or ultrasound. CT is associated with radiation and contrast exposure, therefore is a less suitable alternative. Features of cholangiocarcinoma on imaging include distinctive signal intensity and enhancement of a mass. Small mass lesions seen on MRI are often not visualized by ultrasound exam. MRCP unaided is associated with a sensitivity of 78% and specificity of 76%. Combination of MRCP with MRI improves sensitivity to 89% without a change in test accuracy. Ultrasound on the other hand has a sensitivity and specificity of 57% and 94% respectively. Accuracy of ultrasound exceeds that of MRI/MRCP although sensitivity remains low [13]. A summary of sensitivity and specificity of diagnostic modalities for cholangiocarcinoma screening can be found in Table 1. This data was previously highlighted by charatcharoenwithhya et al. [13].
Table 1: Summary of diagnostic performance of the various surveillance modalities for cholangiocarcinoma detection in patients with PSC [13].
|
Testing Modality [13] |
Sensitivity |
Specificity |
|
Ultrasound |
57% |
94% |
|
Ultrasound plus CA 19-9* |
91% |
62% |
|
MRI |
63% |
79% |
|
MRCP |
78% |
76% |
|
MRI/MRCP |
89% |
75% |
|
MRI/MRCP plus CA 19-9* |
100% |
38% |
|
CT scan |
75% |
80% |
|
CT scan plus CA 19-9* |
100% |
38% |
|
ERCP |
91% |
66% |
|
ERCP plus CA 19-9* |
100% |
43% |
*CA 19-9 more than or equal to 20 U/mL and either test being positive
PSC: Primary Sclerosing Cholangitis; MRI: Magnetic Resonance Imaging; MRCP: Magnetic Resonance Cholangiopancreatography; CT: Comput- erized Tomography; CA 19-9- carbohydrate antigen 19-9; ERCP- endoscopic retrograde cholangiopancreatography.
CA 19-9
Only current available serum biomarker for cholangiocarcinoma is CA 19-9 assay. The testing of CA 19-9 with a cutoff value more than or equal to 20 U/mL enhances MRI/MRCP sensitivity to 100% but lowers specificity (38%) and accuracy (47%) [13]. A cut off value of 129 U/mL or more improves the specificity but decreases the sensitivity [13]. This needs to be interpreted carefully since one third of the patients with this cut off do not have cholangiocarcinoma. About 7% of the general population has negative Lewis antigen and therefore would not benefit from CA 19-9 testing [14].
ERCP with Cytology
Positive ERCP with brush cytology in combination with CA 19-9 above 20 U/mL is 100% sensitive with an accuracy of 49% for detection of cholangiocarcinoma. Positive ERCP findings include biliary stenosis with polypoid duct lesion, dominant duct stricture, or marked proximal bile duct dilation [13]. One of the drawbacks of ERCP with conventional cytology is the lack of sensitivity of this cytology for the diagnosis of cholangiocarcinoma. Bile duct brushing can be obtained safely and are specific (84-89%) but lack sensitivity (8-100%) which limits its utility as a screening tool [13]. Flurosence in situ hybridization (FISH) analysis may enhance the value of standard cytology. FISH from bile duct brushings is used to recognize chromosomal irregularities such as aneuploidy and polysomy. FISH can increase the sensitivity of typical cytology with sensitivity ranging from 64% to 68% and specificity from 70% to 94 [15,16]. This does increase diagnostic yield over conventional cytology. However, overall, there is lack of concrete evidence to advocate for ERCP based surveillance based on performance of test combinations. ERCP is a technically challenging procedure with potential risk for complications such as pancreatitis and cholangitis. This makes it less desirable as a standard surveillance strategy.
Summary
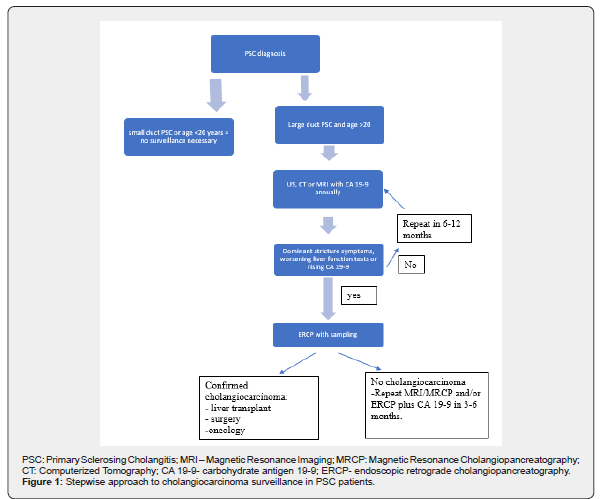
Lifetime probability of cholangiocarcinoma in PSC patients is elevated. Prospective studies evaluating cholangiocarcinoma in PSC patients are lacking. However prior studies reveal that regular surveillance was associated with a higher 5-year survival rate compared with patients who did not receive regular surveillance (68% vs 20%; P<.0061) [17]. Early detection of cholangiocarcinoma can lead to curative liver transplantation. As discussed above imaging used in surveillance includes US liver, MRI/MRCP, CT or ERCP. Serologic screening is limited to use of CA 19-9. As mentioned above detection of hepatobiliary malignancies in PSC patients is variable across modalities. The combination of serum tumor markers and imaging is associated with an improvement in sensitivity. A rational approach for screening is interval radiologic assessment using US or MRI/MRCP in conjunction with CA19-9 every 6 to 12 months. Because of higher sensitivity of MRI compared with US to detect cholangiocarcinoma, MRI is the imaging mode preferred by many experts for cholangiocarcinoma surveillance. ERCP should not be used for routine surveillance. Stepwise approach to cholangiocarcinoma screening summarized in Figure 1.
Patients with painless jaundice, abdominal pain or pruritis no longer fit within the model of cholangiocarcinoma surveillance. These patients should undergo evaluation with ERCP for diagnosis of cholangiocarcinoma. Abnormalities including dominant strictures, mass lesions or increasing CA 19-9 levels identified during surveillance, particularly in the first year of diagnosis, should be followed up with ERCP with brush cytology. Biopsy specimens and fluorescence in situ hybridization (FISH) should be obtained for further evaluation of cholangiocarcinoma. Dominant strictures are defined as strictures of the common bile duct (CBD) and right and left confluence of the hepatic ducts. On the basis of stenosis these ducts measure 1.5 mm or less in the common bile duct and/or 1.0 mm or less in a hepatic duct within 2 cm of main hepatic confluence by ERCP [18]. About 6 to 25% of PSC patients with a dominant stricture will be diagnosed with cholangiocarcinoma over 6 to 9 year follow up period [19]. Direct sampling to rule out underlying cholangiocarcinoma should be considered in such cases.
Fine needle aspiration of biliary strictures should be used with caution in PSC patients being considered for liver transplant candidacy due to concern for tumor seeding. Surveillance for cholangiocarcinoma should not be performed in PSC patients with small duct PSCs or those younger than 20 years of age due to low incidence of cholangiocarcinoma. Overall, the low prevalence of cholangiocarcinoma presents barriers to better understanding surveillance strategies and its long-term impact. Management of this patient population is further complicated by competing risks of liver disease progression and other malignancies associated with PSC. The current data on best practices for biliary malignancy surveillance in PSC patients is constructed on limited observational data. Development of large patient cohorts, advances in genetic testing and development of effective treatments for PSC may further alter our current practice in the future.
References
- Scharl S, Barthel C, Rossel JB, Biedermann L, Misselwitz B, et al. (2019) Malignancies in Inflammatory Bowel Disease: Frequency, Incidence and Risk Factors—Results from the Swiss IBD Cohort Study. Am J Gastroenterol 114(1): 116-126.
- Beaugerie L, Itzkowitz SH (2015) Cancers Complicating Inflammatory Bowel Disease. N Engl J Med 372(15): 1441-1452.
- Erichsen R, Jepsen P, Vilstrup H, Ekbom A, Sørensen HT (2009) Incidence and prognosis of cholangiocarcinoma in Danish patients with and without inflammatory bowel disease: a national cohort study, 1978–2003. Eur J Epidemiol 24(9): 513-520.
- Singal AK, Stanca CM, Clark V, Dixon L, Levy C, et al. (2011) Natural history of small duct primary sclerosing cholangitis: a case series with review of the literature. Hepatol Int 5(3): 808-813.
- Singh S, Talwalkar JA (2013) Primary Sclerosing Cholangitis: Diagnosis, Prognosis, and Management. Clinical Gastroenterology and Hepatology 11(8): 898-907.
- Claessen MM, Vleggaar FP, Tytgat KM, Siersema PD, van Buuren HR (2009) High lifetime risk of cancer in primary sclerosing cholangitis. J Hepatol 50(1): 158-164.
- Rosen CB, Nagorney DM (1991) Cholangiocarcinoma complicating primary sclerosing cholangitis. Semin Liver Dis 11(1): 26-30.
- Bergquist A, Ekbom A, Olsson R, Kornfeldt D, Lööf L, et al. (2002) Hepatic and extrahepatic malignancies in primary sclerosing cholangitis. J Hepatol 36(3): 321-327.
- Burak K, Angulo P, Pasha TM, Egan K, Petz J, et al. (2004) Incidence and risk factors for cholangiocarcinoma in primary sclerosing cholangitis. Am J Gastroenterol 99(3): 523-526.
- Chapman R, Fevery J, Kalloo A, Nagorney DM, Boberg KM, et al. (2010) Diagnosis and management of primary sclerosing cholangitis. Hepatology 51(2): 660-678.
- Lindor KD, Kowdley KV, Harrison ME (2015) ACG Clinical Guideline: Primary Sclerosing Cholangitis. Am J Gastroenterol 110(5): 646-659.
- (2009) EASL Clinical Practice Guidelines: management of cholestatic liver diseases. J Hepatol 51(2): 237-267.
- Charatcharoenwitthaya P, Enders FB, Halling KC, Lindor KD (2008) Utility of serum tumor markers, imaging, and biliary cytology for detecting cholangiocarcinoma in primary sclerosing cholangitis. Hepatology 48(4): 1106-1117.
- Nehls O, Gregor M, Klump B (2004) Serum and bile markers for cholangiocarcinoma. Semin Liver Dis 24(2): 139-154.
- Navaneethan U, Njei B, Venkatesh PG, Vargo JJ, Parsi MA (2014) Fluorescence in situ hybridization for diagnosis of cholangiocarcinoma in primary sclerosing cholangitis: a systematic review and meta-analysis. Gastrointest Endosc 79(6): 943-950.
- Salomao M, Gonda TA, Margolskee E, Eguia V, Remotti H, et al. (2015) Strategies for improving diagnostic accuracy of biliary strictures. Cancer Cytopathol 123(4): 244-252.
- Ali AH, Tabibian JH, Nasser-Ghodsi N, Lennon RJ, DeLeon T, et al. (2018) Surveillance for hepatobiliary cancers in patients with primary sclerosing cholangitis. Hepatology 67(6): 2338-2351.
- Stiehl A, Rudolph G, Klöters-Plachky P, Sauer P, Walker S (2002) Development of dominant bile duct stenoses in patients with primary sclerosing cholangitis treated with ursodeoxycholic acid: outcome after endoscopic treatment. J Hepatol 36(2): 151-156.
- Chapman RW, Williamson KD (2017) Are Dominant Strictures in Primary Sclerosing Cholangitis a Risk Factor for Cholangiocarcinoma? Current Hepatology Reports 16(2): 124-129.






























