Long-Term Effectiveness of Laparoscopic Antireflux Surgery as Treatment of Gastroesophageal Reflux Disease. Retrospective Analysis of a Prospective Database
Jose Walter Huaman1,2* PhD, Sebastian Videla3,4 PhD, Sorin Mocanu5 MD, Angel García San Pedro5 MD and Esteban Saperas1,2 PhD
1Department of Gastroenterology, Hospital Universitari General de Catalunya, Grupo QuirónSalud, Barcelona, Spain
2School of Medicine, Universitat Internacional de Catalunya, Barcelona, Spain
3Clinical Research Support Unit, IDIBELL, Clinical Pharmacology Department, Bellvitge University Hospital, L’Hospitalet de Llobregat, Barcelona, Spain
4Pharmacology Unit, Departament of Pathology and Experimental Therapeutics, Faculty of Medicine and Health Sciences, IDIBELL, University of Barcelona, L’Hospitalet de Llobregat, Barcelona, Spain
5Department of Surgery, Hospital Universitari General de Catalunya, Grupo QuirónSalud, Barcelona, Spain
Submission:September 09, 2020;Published:September 18, 2020
*Corresponding author:Jose Walter Huaman. Department of Gastroenterology, Hospital Universitari General de Catalunya, Grupo QuirónSalud. C/ Pedro i Pons, 1. 08190, Barcelona, Spain
How to cite this article:Jose W H, Sebastian V, Sorin M, Angel G S P, Esteban S. Long-Term Effectiveness of Laparoscopic Antireflux Surgery as Treatment of Gastroesophageal Reflux Disease. Retrospective Analysis of a Prospective Database. Adv Res Gastroentero Hepatol, 2020;15(5): 555925. DOI: 10.19080/ARGH.2020.15.555925.
Abstract
Introduction:Laparoscopic antireflux surgery (LARS) is an effective alternative to medical therapy for patients whose symptoms of gastroesophageal reflux disease (GERD) are not fully relieved. However, a subset of patients will experience postoperative relapse of GERD that might require corrective antireflux surgery. The aim of this study was to estimate long-term success of LARS.
Patients and Methods:Single-center, retrospective analysis of a prospective cohort of consecutive patients who underwent LARS from January 2005 onward. Success of LARS was defined as no necessity of revisional surgery, performed when relapse of GERD was confirmed by either endoscopic evidence of esophagitis or a 24-h pH study indicative of pathologic acid reflux. Success of LARS and predictive factors associated were assessed.
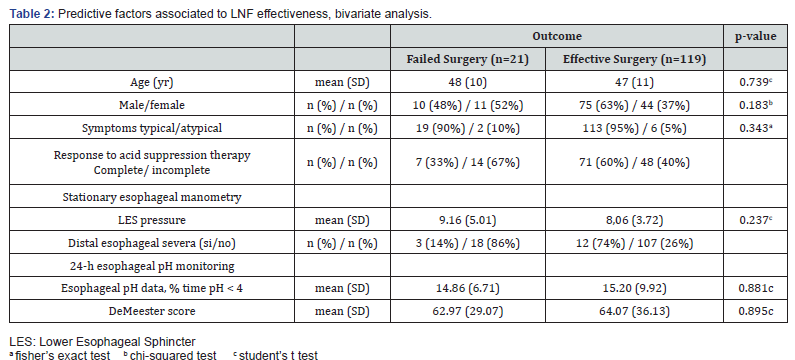
Results: A total of 140 patients [85 (61%) male, mean (SD) age: 47 [11) years] with GERD considered proper candidates for surgery underwent LARS. After a median (range) of follow-up period was 8 [5-10] years, Success of LARS was in 119 out of 140 patients (85%, 95CI:78-90%). 21 patients were considered suitable candidates for revisional antireflux surgery, but 5 declined it. The cumulated incidence of need of corrective antireflux surgery for postoperative relapse of GERD, per ‘full analysis set’ or ‘per protocol’ was 15% (21/140, 95CI:10-22%) or 12% (16/135, 95CI:7-18%), respectively. The mean (SD) time to revisional surgery was of 5.8 (2.9) years. No predictive factors for success of LARS were found in the multivariate analysis.
Results: LARS is a lasting effective treatment for the management of GERD in most well selected patients.
Keywords: Funduplication; Anti-reflux surgery; Gastroesophageal reflux disease; Manometry; pH monitoring and Nissen
Introduction
Laparoscopic antireflux surgery (LARS) is an effective alternative to medical therapy for patients whose symptoms of gastroesophageal reflux disease (GERD) are not fully relieved, as well as for those who reject lifelong medication [1]. LARS has been shown to lead to effective symptom reduction, overall patient satisfaction and an improved quality of life in both short- and medium-term in numerous studies [2–5]. Current evidence suggests that laparoscopic fundoplication is more effective than medical therapy, providing good short-term outcome in most patients [6,7]. However, a subset of patients will eventually experience recurrence of reflux symptoms after surgery. Among them, some will need to reinitiate acid-reducing medications while others will need to undergo another operation to improve symptom control [8,9]. Moreover, recent data of increasing recurrence of reflux symptoms [10] or use of PPIs [11] with longer follow-up suggests that symptom control after surgery may be waning over time [12]. Actually, a recent populationbased register study found that more than half of the patients regain antisecretory therapy within 10-15 years after surgery [13]. This figure is much higher than that reported by clinical trials and challenges the favorable long-term outcome of LARS, though the significance of the use of acid-reducing medication is questionable without objective measurements and may not be a reliable marker of surgical failure [14]. However, reoperation for recurrence of symptoms of reflux is still necessary in an average of 8% of patients and appears to be more frequent in series with longer follow-up [1].
Improvement, and ideally, relief of GERD symptoms are the main goals of any treatment for GERD. New data on the long-term benefits of surgery allows knowing its real impact in patients who are complete responders to PPI therapy [15]. Thus, the aim of this study was provide new data on the success of laparoscopic antireflux surgery (no necessity of revisional surgery) and the predictive factors associated.
Patients and Methods
Study design
This study was a single-centre, retrospective analysis from a prospectively maintained database of a cohort of consecutive patients who underwent LARS between January 2005 and December 2017. Database was closed when the last patient included had at least a year of follow-up. This database gathers our clinical practice. The project received approval from the Research Ethical Committee of the Hospital Universitario General de Catalunya.
Study population
The eligibility criteria for inclusion in the study were: age ≥18 years and previous LARS with at least 1 year of follow-up. According to our clinical practice protocol, criteria for surgery included abnormal 24-hour pH esophageal monitoring results or endoscopic evidence of erosive esophagitis.
Clinical examinations: preoperative and follow-up
Based on our clinical practice protocol, preoperative evaluation included clinical characteristics, upper gastrointestinal endoscopy, stationary esophageal manometry and ambulatory 24- hour pH esophageal monitoring. Optionally, follow-up evaluation could include stationary esophageal manometry and ambulatory 24-hour pH esophageal monitoring at 6 months after surgery.
Upper gastrointestinal endoscopy. A standard upper gastrointestinal (GI) endoscopy was performed before surgery in all patients. Hiatal hernia size and the Los Angeles classification esophagitis grade were determined endoscopically [16].
Stationary esophageal manometry. A water-perfused system with a multiple lumen catheter with an incorporated sleeve sensor (Dentsleeve, Adelaide, Australia) was used. To determine the distal and proximal border of the lower esophageal sphincter (LES), the catheter was introduced transnasally and subsequently retracted. Thereafter, the manometric response to 10 swallows of 5-ml ambient temperature water spaced by > 20 s were taken [17].
Ambulatory 24-h pH esophageal monitoring. Ambulatory 24-h pH testing was performed with the patient at least 7 days free of medication that could affect results. pH catheters with an antimony electrode (Medtronic A/S, Kobenhavn, Denmark) were passed nasally and positioned such that the distal electrode was 5 cm above the manometrically determined lower esophageal sphincter. The 24-h pH recordings were stored in a digital data logger (Digitrapper MK III, Medtronic), and patients registered reflux symptoms and body position in a diary. The data were downloaded and analyzed using PolyNet 4.0 computer software (Medtronic Gastroenterology and Urology, Shoreview, Minnesota, USA) [18].
Classification into pathological or physiological esophageal acid exposure was based on the percentage of time with pH below 4. Pathological acid exposure was defined as an acid exposure time of 4.2 percent or more of total time with pH below 4 [18].
Surgical procedure
Laparoscopic Nissen fundoplication (LNF) was performed under general anesthesia in all patients operated and included in this study. A five-port laparoscopic approach was used in all cases: 11-mm epigastric port for the optical device, 5-mm port in the anterior right axillary line for liver retractor and three working ports (one 5-mm and two 11-mm ports) placed in the right upper and left quadrants and in the anterior left axillary line respectively. Various types of advanced surgical energy devices were used to minimize the bleeding risk during the dissection (Thunderbeat®, Ultracision® or Ligasure®). Endo StitchÔ suturing device was used to perform the cruroplasty and gastric fundoplication.
The intervention starts with the anatomization of the hiatal region and dissection of the distal esophagus in the lower mediastinum until an abdominal esophageal segment of at least 5 cm is obtained. During the dissection, both vagus nerves are identified and preserved, the hernia sac is partially or completely resected and the short gastric vessels in the fundic region are divided using the energy device. Diaphragmatic cruroplasty is performed over a 36 Ch Lavacuator® tube using 2 or 3 nonabsorbable braided polyester stitches, followed by a 360º “floppy” Nissen fundoplication with 2 or 3 stitches of the same material. Occasionally, the wrap is tethered to the esophagus with one of the fundoplication stitches or to the diaphragm with an additional stitch. At the end of the procedure, the Lavacuator tube is removed. Nasogastric tube and intraabdominal drains are not routinely used. Gastric antisecretory drugs are discontinued in patients operated for gastroesophageal reflux [1].
The most prominent symptom present at the time of surgical referral was recorded as the primary symptoms driving the need for operative therapy. Heartburn, regurgitation, and dysphagia were considered typical symptoms, and hoarseness, cough, wheezing, and chest pain were considered atypical symptoms.
Study definitions
Treatment success (success of LARS or effective surgery) was defined as the no necessity of revisional surgery. Revisional surgery was offered routinely to patients with recurrent reflux symptoms not controlled by medical therapy, or when medical therapy was not tolerated. Acid reflux was confirmed by endoscopic evidence of esophagitis or a positive 24-h pH study (pH less than 4 for more than 4.2 per cent of the study). Hence, failed surgery was defined as LARS followed by necessity of revisional surgery for recurrent reflux.
Response to acid suppression therapy was defined (recorded) as complete if the patient reported 100% relief of symptoms and incomplete in all other situations.
LES resting pressure was defined as the mean of five recordings.
Statistical analysis
No formal sample size was calculated. The sample size was defined as all patients operated by LARS during the inclusion period.
Baseline characteristics were summarized using standard descriptive statistics, and a descriptive analysis was carried out. Continuous variables were described as mean (standard deviation) or median (range) and categorical data were summarized as absolute frequency and percentages. The treatment success (effectiveness of LARS) was estimated and its 95% confidence interval (95%CI) was calculated. The mean (range) time to reoperate was analyzed using the Kaplan–Meier method. Bivariate and multivariate logistic analyses and Cox proportional hazard regression models were performed, when appropriate, to determine potential factors associated with treatment success. The association between potential explanatory variables and treatment success was tested in 2 steps. First, a bivariate regression model was performed to obtain an indication of the relevance of the explanatory variables. Second, factors showing a P value less than 0.3 were used to perform a multiple regression model using a stepwise selection of variables. Hazard ratios (HRs) were estimated as well as their 95%CI. A p-value of 0.05 or less was considered statistically significant. Data were analyzed using SPSS version 15.0 statistical software (SPSS, Inc., Chicago, Illinois, USA) and StatXact-8 (Cytel Inc., Cambridge, Massachusetts, USA).
Results
Patient characteristics and effectiveness of LARS
Between January 2005 and December 2017, a total of 140 consecutive patients with GERD considered suitable candidates for surgery (60.7%; 85/140, men) underwent LARS.
Table 1 shows the baseline (demographic and physiologic) characteristics. The median (range) of follow-up period was 8 [5-10] years One hundred twenty-three (91.1%) patients had more than 3 years of follow-up. LARS was successful in 119 out of 140 patients; (85%, 95CI: 78%-90%) whereas 21 patients were considered suitable candidates for revisional antireflux surgery. Among them, 16 patients (96%) eventually underwent revisional surgery whereas the other 5 patients declined additional surgery. The cumulated incidence of need of revisional antireflux surgery for postoperative relapse of GERD per ‘full analysis set’ or ‘per protocol’ was 15% (21/140, 95CI:10-22%) or 12% (16/135, 95CI:7-18%), respectively.
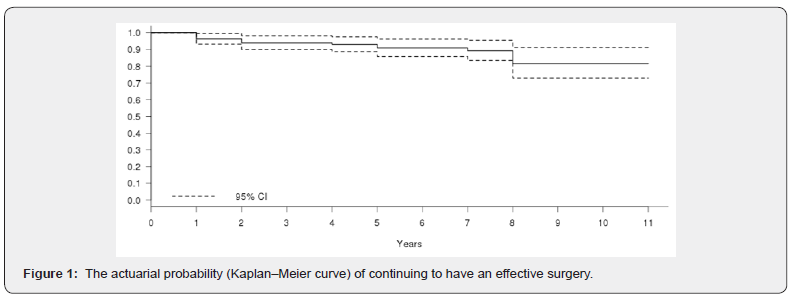
Recurrence of GERD in the 16 patients submitted to revisional surgery occurred because of fundoplication failure in 10 out of 16 patients (62%, 95%CI: 39-83%): 7 patients due to intrathoracic wrap migration and 3 patients due to wrap disruption. In the other 6 patients (38%, 95%CI: 18-61%), GERD relapsed despite of a successful intact fundoplication. The mean (SD) time to revisional surgery was of 5.8 (2.9) years. Figure 1 depicts the actuarial probability (Kaplan–Meier curve) of remaining free of revisional surgery. Only ‘response to acid suppression therapy’ previous LARS was a protective factor for revisional surgery in bivariate analysis, p-value=0.025 (Table 2). But, in multivariate analysis, only was found a trend (HR: 0.42, 95%CI: 0.15-1.22, p-value=0.110).
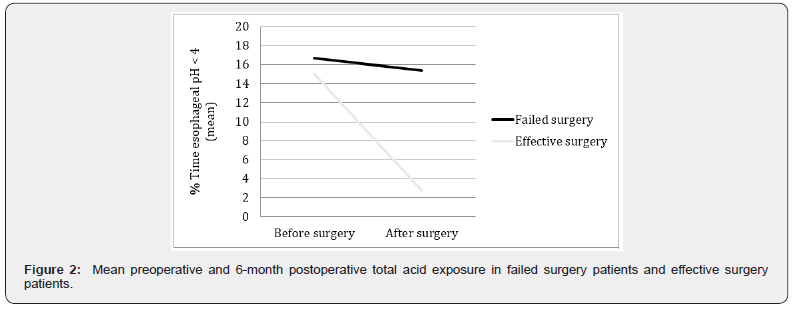
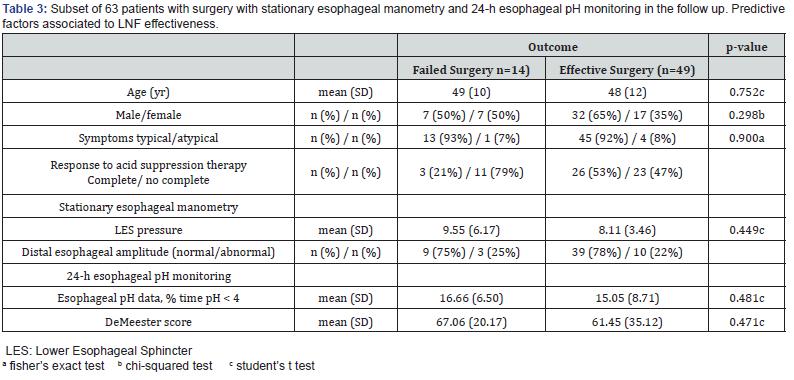
Stationary esophageal manometry and ambulatory 24-h esophageal pH monitoring were repeated 6 months after LARS in 63 out of 140 patients (45%). Among them, corrective antireflux surgery was necessary in 14 out of these 63 patients (22%). Table 3 presents the baseline (demographic and physiologic) characteristics of this subset of patients. No differences were found respect to whole population. In the analysis between previous LARS (baseline) and 6 months after, a reduction in esophageal exposure to pH<4 on 24-hour pH monitoring after fundoplication and an increase in LES pressure were both registered postoperatively, but only the former was significantly higher in patients who did not required revisional surgery for recurrence of reflux (Figure 2).
In the group effective surgery, a reduction in esophageal exposure to pH < 4 on 24-hour pH monitoring was 15.05 a 2.78, while in the group failed surgery a reduction in esophageal exposure to pH < 4 on 24-hour pH monitoring was only 16.71 a 15.43. p < 0,001.
Adverse events
No patients developed serious adverse events following LARS.
Discussion
Our results endorse that laparoscopic antireflux surgery is an effective treatment for GERD. To our knowledge, this is the first time that the long-time effectiveness of LARS as treatment of GERD is provided in our geographical area. The success of LARS varies considerably in the literature, between 79% and 98%. This variability in the rate of treatment success among studies could be explained by the definition of recurrence used, by the diagnostic workup performed and by the duration of follow-up. In our study, LARS treatment success was of 85%. This finding is comparable to that of previous studies reporting long-term control of reflux in 74%-90% of patients [12,19,20], using standardized symptom questionnaires, quality of life assessment or composite criteria including the need for additional medical or surgical treatment, patient’s perception of overall success, and long-term quality of life. Conversely, our results differ of the long-term outcome from Sweden, where the treatment failure after LARS occurred in the 25 - 29% of the patients [21]. It is noteworthy that the lack of standardization regarding the definition of treatment outcome precludes comparisons among those studies. In front of this scenario and from the clinical practice point of view, to perform a revisional surgery for reflux recurrence should be considered a strong variable for assessing the effectiveness of LARS [22,23]. We understand that this one is the most stringent, objective and less biased criteria to reliably analyze the results of a retrospective study, although is a variable which depends on follow-up period. In our study, the necessity of revisional anti-reflux surgery was slightly greater than reported in a literature review, 12% and 8%, respectively [23,24].
Various reasons may determine whether antireflux surgery will eventually improve or fully resolve the symptoms of the patient. Among them, a key issue is to know whether the symptoms are actually caused by GERD. In fact, it has been reported that almost one third of 822 consecutive patients referred for esophageal function tests because of clinical diagnosis of GERD had a normal 24-h pH esophageal monitoring study [25]. Consequently, selection of right candidates with a detailed workup and multidisciplinary evaluation is essential to avoid poor outcomes. Accordingly, the latest guidelines published in 2013 by a panel of expert gastroenterologists and surgeons recommend that an appropriate preoperative workup should include an accurate clinical history (including the evaluation of the response to PPI therapy), barium swallow, upper endoscopy, esophageal manometry, and 24-h pH esophageal monitoring [26,27]. Likewise, our findings showed that the treatment success of LARS in patients with complete response to acid-reducing medication [71/78 patients; 91%) was slightly superior to the remission rates at five years found in a randomized study comparing LARS (85%) and antisecretory medication (92%) in patients with GERD and complete response to PPI therapy [17].
LARS achieved satisfactory short-term outcomes, effectively alleviating symptoms of GERD, healing erosive esophagitis and attaining good functional improvement and patient satisfaction [28]. The success of antireflux surgery depends also on a careful preoperative evaluation and accurate identification of patients with symptomatic GERD, as well as the surgical technique employed and surgeon’s experience. In agreement with previous studies we also provide convincing objective evidence for the short-term effectiveness of surgery, suggesting that the quality of surgery has been as good in this study as elsewhere. Fundoplication significantly reduced esophageal acid exposure and increased LES pressure compared with preoperative values, with pH studies returning to normal in 49 out of 63 (78%) of patients. Like any other surgical procedure, LARS is subject to a learning curve, which may impact patient outcomes. The study was conducted over a wide time span. With improvements in operative technique it is possible that failure rates could have varied over time. However, the recurrence of reflux (failure surgery) was equally distributed over the years without any significant cluster in the early years. Recurrence of reflux may occur because of decreasing effectiveness of a successful intact fundoplication over time. However, in agreement with previous findings, in our study the need for revisional surgery occurred more often 10/16, (62%) because of fundoplication failure due to wrap slippage or disruption.
Factors associated with effective surgery have been previously shown to be strongly related to patient selection [1]. Some predictive factors have been found to lead to superior outcomes include the presence of typical GERD symptoms, abnormal 24-h pH monitoring and good response to medical therapy [9]. In our study, preoperative clinical response to acid suppression therapy showed a trend as predictive factor associated with surgery effectiveness. In contrast, the predictive value of the two former factors could not be endorsed. A possible explanation could be in that almost all patients of our study have abnormal 24-h pH monitoring, endoscopic signs of severe GERD or both. Likewise, in agreement with other authors [29], we have not found an association with age or with the presence of atypical symptoms (a very small number of patients with this indication in our study).
A subset of 63 out of 140 (45%) patients underwent objective testing with both stationary esophageal manometry and 24-h esophageal pH monitoring, 6 months after fundoplication. Among them, the reduction in 24-h esophageal acid exposure but not the increase in basal LES pressure after fundoplication was significantly higher in responders than in those eventually requiring revisional surgery. Unfortunately, a bias in this finding is likely because postoperative functional testing was performed in all symptomatic patients requiring revisional surgery but only in 41.1% of the responders.
A few limitations of this study must be acknowledged. The study design of retrospective nature (patients were not randomized) can overestimate the results. The population analyzed (all from a single geographical area and form a single center) makes it risky to extrapolate our results beyond the population and condition studied. The lack of a systematic long-term assessment by endoscopy and 24-h pH monitoring and that not all the patients were available for long-term follow-up, it makes us to be prudent to interpret the results since we cant not completely exclude the possibility that any of the patients lost to follow-up had in fact a poor outcome. However, we believe this is unlikely because our dataset was obtained from a database derived from the prospective electronic medical history. The results of the present study reflect the experience of a group with expertise in GERD diagnosis and treatment, patient’s selection and esophageal physiopathology and are likely representative of the results from the majority of laparoscopic antireflux procedures, which are currently done in general surgical practice. Because administrative data have demonstrated superior outcomes in high-volume centers for complex procedures such as esophagectomy or pancreatectomy, it can be argued that antireflux surgery carried out in expert centers might result in better outcomes than when carried out in general surgical practice, but this has not been shown for laparoscopic fundoplication.
In conclusion, our study suggests that laparoscopic antireflux surgery is an effective therapy for the long-term management of GERD in well-selected patients. However, revisional antireflux surgery for postoperative relapse of GERD will be necessary in few patients during long-term follow-up, despite appropriate selection of patients. Further clinical prospective long-term research is needed to know the real impact of LARS on the management of GERD and to learn whether improvements in technical aspect of the operation such as the use of prosthetic meshes for reinforced hiatoplasty may have an impact on longterm reducing the number of revisional surgeries.
Acknowledgement
The authors thank Gemma Domenech and Carmen Ferrer for technical support.
References
- Stefanidis D, Hope WW, Kohn GP (2010) Guidelines for surgical treatment of gastroesophageal reflux disease. Surg Endosc 24: 2647-2669.
- Cookson R, Flood C, Koo B (2005) Short‐term cost effectiveness and long‐term cost analysis comparing laparoscopic Nissen fundoplication with proton‐pump inhibitor maintenance for gastro‐oesophageal reflux disease. Brit J Surg 92(6): 700-706.
- Anvari M, Allen C, Marshall J, David Armstrong, Ron Goeree, et al. (2006) A Randomized Controlled Trial of Laparoscopic Nissen Fundoplication Versus Proton Pump Inhibitors for Treatment of Patients with Chronic Gastroesophageal Reflux Disease: One-Year Follow-Up. Surg Innov 13(4): 238-249.
- Mehta S, Bennett J, Mahon D, Rhodes M (2006) Prospective Trial of Laparoscopic Nissen Fundoplication Versus Proton Pump Inhibitor Therapy for Gastroesophageal Reflux Disease: Seven-Year Follow-up. J Gastrointest Surg 10: 1312-1317.
- Lundell L, Attwood S, Ell C, R Fiocca, J-P Galmiche, et al. (2008) Comparing laparoscopic antireflux surgery with esomeprazole in the management of patients with chronic gastro-oesophageal reflux disease: a 3-year interim analysis of the LOTUS trial. Gut 57(9): 1207-1213.
- Hatlebakk JG, Zerbib F, Varannes SB des, Stephen E Attwood, Christian Ell, et al. (2016) Gastroesophageal Acid Reflux Control 5 Years After Antireflux Surgery, Compared with Long-term Esomeprazole Therapy. Clin Gastroenterol H 14(5): 678-685.e3.
- Frazzoni M, Piccoli M, Conigliaro R, Leonardo Frazzoni, Gianluigi Melotti, et al. (2014) Laparoscopic fundoplication for gastroesophageal reflux disease. World J Gastroentero 20(39): 14272-14279.
- Bloomston M, Nields W, Rosemurgy AS (2003) Symptoms and antireflux medication use following laparoscopic Nissen fundoplication: outcome at 1 and 4 years. Jsls J Soc Laparoendosc Surg Soc Laparoendosc Surg 7: 211-218.
- Patti MG, Allaix ME, Fisichella PM (2015) Analysis of the Causes of Failed Antireflux Surgery and the Principles of Treatment: A Review. Jama Surg 150: 585-590.
- Byrne JP, Smithers BM, Nathanson LK, I Martin, H S Ong, et al. (2005) Symptomatic and functional outcome after laparoscopic reoperation for failed antireflux surgery. Brit J Surg 92(8): 996-1001.
- Wijnhoven BPL, Lally CJ, Kelly JJ, Jennifer C Myers, David I Watson, et al. (2008) Use of Antireflux Medication After Antireflux Surgery. J Gastrointest Surg 12: 510-517.
- Ross SB, Gal S, Teta AF, Kenneth Luberice, Alexander S Rosemurgy, et al. (2013) Late results after laparoscopic fundoplication denote durable symptomatic relief of gastroesophageal reflux disease. Am J Surg 206: 47-51.
- Lødrup A, Pottegård A, Hallas J, Bytzer P (2014) Use of proton pump inhibitors after antireflux surgery: a nationwide register-based follow-up study. Gut 63(10): 1544.
- Lord RVN, Kaminski A, Öberg S, David J Bowrey, Jeffrey A Hagen, et al. (2002) Absence of gastroesophageal reflux disease in a majority of patients taking acid suppression medications after Nissen fundoplication. J Gastrointest Surg 6(1): 3-10.
- Khan F, Maradey‐Romero C, Ganocy S, R Frazier, R Fass, et al. (2016) Utilization of surgical fundoplication for patients with gastro‐oesophageal reflux disease in the USA has declined rapidly between 2009 and 2013. Aliment Pharm Therap 43: 1124-1131.
- Lundell LR, Dent J, Bennett JR, A L Blum, D Armstrong, et al (1999) Endoscopic assessment of oesophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut 45(2): 172.
- Broeders JA, Draaisma WA, Bredenoord AJ, A J Smout, I A Broeders, et al. (2010) Long‐term outcome of Nissen fundoplication in non‐erosive and erosive gastro‐oesophageal reflux disease. Brit J Surg 97: 845-852.
- Charbel S, Khandwala F, Vaezi MF (2005) The Role of Esophageal pH Monitoring in Symptomatic Patients on PPI Therapy. Am J Gastroenterology 100(2): 200549.
- Dallemagne B, Perretta S (2011) Twenty Years of Laparoscopic Fundoplication for GERD. World J Surg 35: 1428-1435.
- Furnée EJB, Draaisma WA, Broeders IAMJ, A J P M Smout, A L M Vlek, et al. (2008) Predictors of symptomatic and objective outcomes after surgical reintervention for failed antireflux surgery. Brit J Surg 95(11): 1369-1374.
- Sandbu R, Sundbom M (2009) Nationwide survey of long-term results of laparoscopic antireflux surgery in Sweden. Scand J Gastroentero 45: 15-20.
- Broeders JA, Draaisma WA, Vries DR de, Albert J Bredenoord, André J Smout, et al. (2009) The Preoperative Reflux Pattern as Prognostic Indicator for Long-Term Outcome After Nissen Fundoplication. Am J Gastroenterology 104(8): 1922.
- Valiati W, Fuchs KH, Valiati L (2000) Laparoscopic fundoplication - short- and long-term outcome. Langenbeck’s Archives Surg 385: 324-328.
- Lamb PJ, Myers JC, Jamieson GG, S K Thompson, P G Devitt, et al. (2009) Long‐term outcomes of revisional surgery following laparoscopic fundoplication. Brit J Surg 96: 391-397.
- Patti MG, Diener U, Tamburini A, D Molena, L W Way, et al (2001) Role of Esophageal Function Tests in Diagnosis of Gastroesophageal Reflux Disease. Digest Dis Sci 46: 597-602.
- Katz PO, Gerson LB, Vela MF (2013) Guidelines for the Diagnosis and Management of Gastroesophageal Reflux Disease. Am J Gastroenterology 108: 308.
- Jobe BA, Richter JE, Hoppo T, Jeffrey H Peters, Reginald Bell, et al. (2013) Preoperative Diagnostic Workup before Antireflux Surgery: An Evidence and Experience-Based Consensus of the Esophageal Diagnostic Advisory Panel. J Am Coll Surgeons 217(4): 586-597.
- Maret-Ouda J, Wahlin K, El-Serag HB, Lagergren J (2017) Association Between Laparoscopic Antireflux Surgery and Recurrence of Gastroesophageal Reflux. Jama 318: 939-946.
- Tolone S, Gualtieri G, Savarino E, Marzio Frazzoni, Nicola de Bortoli, et al. (2016) Pre-operative clinical and instrumental factors as antireflux surgery outcome predictors. World J Gastrointest Surg 8(11): 719.






























