Drug Development Challenges in Hepatitis Clinical Trials
Mohamed El Mouelhi*
Independent Consultant for Health Care Industry, USA
Submission: July 29, 2019; Published: August 13, 2019
*Corresponding author:Mohamed El Mouelhi, MD, PhD, FACP, Independent Consultant for Health Care Industry, 86 Van Cleef Drive, White House Station, NJ08889, New Jersey, USA
How to cite this article: Mohamed El Mouelhi. Drug Development Challenges in Hepatitis Clinical Trials. Adv Res Gastroentero Hepatol. 2019;13(4): 555869. DOI: 10.19080/ARGH.2019.13.555869.
Abstract
Drug development goes through 3 phases and encounters many challenges. Only 10% of compounds entering human testing makes it to the market after an average of 14 years with a cost of approximately $3 billion. Interestingly, prescription drugs constituted only 10 % of each health care dollar spent in USA. Challenges encountered in liver clinical trials include lack of validated patient-reported outcome tools, blinding issues, and the use of placebo in addition to lack of health authority guidance for many diseases including orphan diseases. In about 80% of clinical trials, enrollment of the right patient is the leading cause of missed clinical trial deadlines, resulting in delayed availability of potentially life-saving therapies. Other additional challenges specific to hepatitis trials include the need to evaluate various drug regimens, identification of specific treatment for each genotype of the hepatitis C virus, associated co-morbidities, and any previous anti-viral usage. Another recent challenge introduced by current technology is the use of social media and risk of bias. Sharing personal experiences while in the study could easily introduce bias among patients that would interfere with accurate interpretation of collected data. Cure of hepatitis C infection went a long way over the years due to drug development effort, and consideration of all these challenges will allow timely response to the remaining unmet medical needs for many hepatitis patients.
Keywords: Drug development; Clinical trials; Hepatitis; Hepatitis C virus; Liver; Clinical testing; Patients; Drug regimens; Liver diseases; New drug; Animal toxicity; Chemical stability; Disease control; Absorption; Elimination
Abbrevations: IND: Investigational New Drug Application; CTA: Clinical Trial Application; EMA in EU: European Medicines Agency in Europe; NDA: New Drug Application; BLA: Biologic License Application; HAS: Health Authorities; HCV: Hepatitis C Virus; PR: Pegylated Interferon Alfa and Ribavirin; AASLD: American Association for Study of Liver Diseases; IDSA: Infectious Diseases Society of America; EASL: European Association for the Study of the Liver; GT: Genotype; RASs: Resistance-Associated Substitutions; AEs: Adverse Events; PRO: Patient Reported Outcome; CLDQ: Chronic Liver Disease Questionnaire; HRQoL: Health-Related Quality Of Life; SWE: Shear Wave Elastography; DAA: Direct Acting Antiviral; IFN: Interferon
Introduction
Drug development challenges for liver diseases were described in a previous review [1]. Here challenges encountered in developing drugs for hepatitis are discussed. In a recent survey conducted by Kaiser Family Foundation Health Tracking Poll [2], almost three quarters of the Americans feel that drug costs are unreasonable (72 %). On the other hand, participants in this poll largely value the role prescription drug companies play, with most (62 %) saying that prescription drugs developed in the past two decades have made the lives of people in the USA better. This impression about prescription drug costs was highlighted in a more recent poll conducted in 2019 by Kaiser Family Foundation [3].
Nearly seven in ten Americans (68 percent) say lowering prescription drug costs for as many Americans as possible is a “top priority” for Congress. For the majority of people, they have no information about the process of drug development. It is for them like a “black box”, including patients, who are the main stakeholder for this process. To develop a drug, it goes through lengthy and costly scientific, medical, and regulatory processes. The main objective of developing drugs is to respond to unmet medical needs. The ideal objective is to cure the disease. However, another scenario—cessation or slowing down the disease progression—is more common. A last resort is to provide some symptomatic relief. Of course, disease aggravation should be avoided at all cost. Achieving an earlier diagnosis will provide a good chance for the treatment to be more effective with a higher rate of disease control.
Drug Discovery and Development
A common question for many people when picking up their prescriptions from the pharmacy is why just a month supply of tablets or capsules cost that much? In fact, in order to reach the consumer, to develop any drug it takes about 14 years and the average cost is around $ 3 billion. The average cost to bring a new drug to the market has become more expensive over the years. Recently, costs more than doubled compared to the last estimate 13 years ago. Tufts Center for the Study of Drug Development in a report published in 2016, found the cost to have averaged $2.87 billion (in 2013 dollars) [4].
Approval of a drug requires proving that it is safe, effective, and the dose proposed is the optimal one for the intended use. The process starts with pre-clinical phase including research where animal and laboratory studies are conducted to identify the proper target, characterize it then test the proper molecule. Usually around 10,000 compounds are identified in the beginning, out of which 1000 compounds are chosen for in vitro testing. Superior compounds are selected based on many factors including animal toxicity, chemical stability, and in vivo handling (such as absorption and elimination). This pre-clinical and research testing step takes about 5 years.
At the end of this pre-clinical testing phase, an Investigational New Drug Application known as IND in the USA and Clinical Trial Application (CTA) in EU is submitted to the Health Authority (FDA in US; and EMA in EU) prior to any clinical testing in human. As a next step, 1 to 10 compounds are selected for clinical testing in man. During the pre-clinical and early exploratory clinical phases of drug development (phases 1 and 2), a large number of programs up to 90% do not reach the registration phase [5].
The clinical testing is composed of three phases, namely phases I, II, and III (Figure 1). Phase I generally tests the investigational compound in a small group of individuals (around 50) with the goal of identifying how the drug behaves in the body and a first assessment of its tolerability. It could be conducted in healthy volunteers or patients with the intended disease indication. Based on phase I findings, this investigational compound may proceed to phase II, where a larger number of patients with the intended disease indication (usually several hundreds) are enrolled to evaluate the proper dosage and preliminary efficacy. The last clinical phase (phase III) is “confirmatory” in nature requiring a large number of patients, usually between 1000 and 3000. Phase III studies generally use either a proper comparator or placebo and to provide a large data base for safety of the investigational drug. These three clinical phases take approximately 9 years to complete.
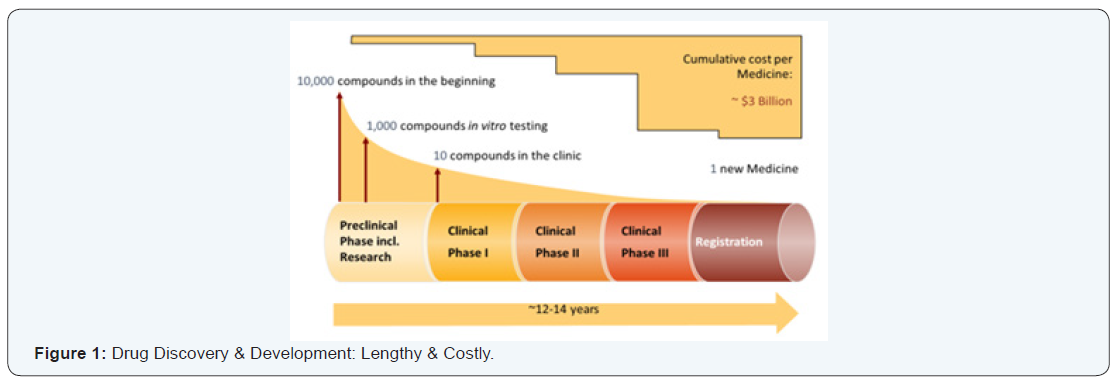
At the end of the clinical phases, preparation and submission of data (depending on drug nature either New Drug Application: NDA; or Biologic License Application: BLA) to the Health Authorities for drug registration approval. The registration process could take up to 1.5 years. On average, it takes testing 10 compounds in humans to yield one marketed drug. There are several reasons that a compound fails during the development process. These reasons include animal toxicity, lack of chemical stability, human pharmacokinetic properties, intolerance, lack of efficacy, poor differentiation from available treatments, or lack of long-term safety. One final reason for a drug not reaching the market is non-approval by the health authority. Interestingly, some drugs are approved in USA and not in Europe and vice versa. In certain cases where thousands of patients are included in pre-registration studies, rare adverse events may not be detected until after approval. The Health Authorities (HAs) in these cases request a post-approval testing that is known as “Post approval Commitment.”
Health Care Dollar and Cost of Prescription Drugs
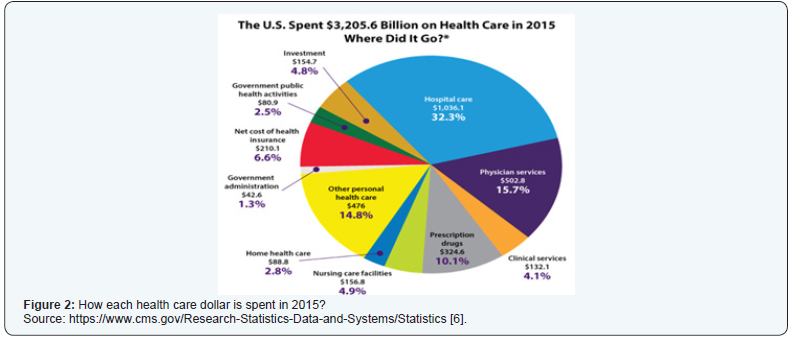
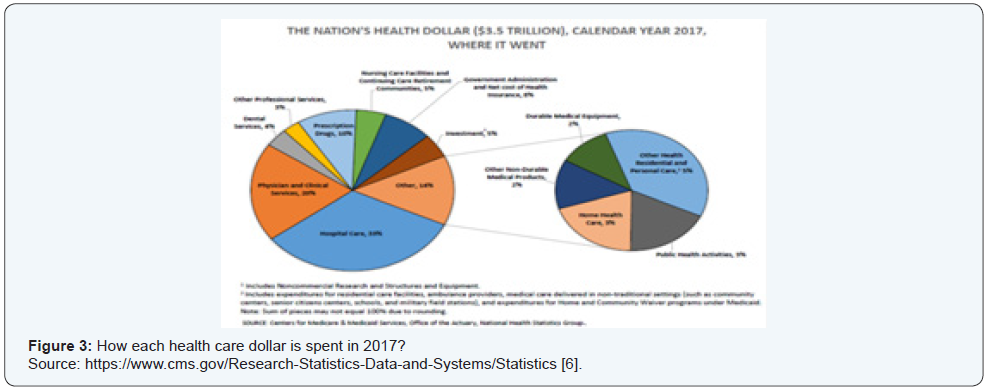
The Centers for Medicare & Medicaid Services assessed the proportion of each health care dollar being spent for prescription drug in the USA in 2017 [6]. In comparison to that in 2015, the total expenditure in 2017 increased to 3.5 Trillion dollars from 3.2 Trillion dollars in 2015, however the distribution remained almost unchanged. Hospital care constituted the highest portion, reaching 33 % of health care spending, followed by physicians and clinical services with 20 %. Interestingly, prescription drugs constituted only 10 % of health care spending (Figures 2 & 3). Nursing care facilities and home health care contributed the lowest percentage of spent health care dollars (around 8 %).
Challenges with Clinical Trials
Clinical studies are crucial for any new drug approval where its safety and efficacy need to be established. Current regulation by the health authorities usually requires two phase III studies for any marketing approval. Over 75% of clinical studies fail to meet their timelines due to lack of patient enrollment in these studies resulting in a delayed availability of potentially life-saving treatments to the overall patient population.
Challenges for Hepatitis C Clinical Trials
Clinical trials in liver diseases and specially hepatitis C have some peculiar challenges. Till recently viral hepatitis and parasitic liver diseases including schistosomiasis were a major health concern in some geographies. However, these diseases became less of a health concern after discovery of new antiviral therapies, adoption of effective control measures and the use of oral therapy for schistosomiasis [7,8].
Hepatitis C virus (HCV) infection predisposes to liver fibrosis, cirrhosis, and liver cancer. It infects approximately 3% of the world population (over 71 million individuals) representing a public health challenge. However, in high endemic countries, the HCV prevalence is much higher and could reach 20%. The highest virus prevalence was registered in Egypt, where 22% of population is infected as a result of mass treatment campaign with parenteral anti-schistosomal treatment prior to prior to availability of oral therapy [9,10]. Understanding of host and viral factors could speed up the availability of an efficient vaccine [11].
In advanced countries such as the US, other factors drove the rise in HCV infection. The role of the opioid epidemic and, in particular, the 2010 introduction of an abuse-deterrent version of OxyContin was evaluated. The OxyContin reformulation led some users of the drug to switch to heroin, which could have exposed them to the hepatitis C virus. States with above-median OxyContin misuse before the reformulation experienced a 222% increase in hepatitis C infection rates in the post-reformulation period, while states with below-median misuse experienced only a 75% increase. These results suggest that interventions to deter opioid misuse can have unintended long-term public health consequences [12].
HCV has seven genotypes (gt 1-7) and numerous subtypes that complicated the process of developing effective therapy. Recently, multiple therapies with a pan-genotypic activity became available [11].
One of the Non-structural proteins NS5A is present inside HCV infected cell and plays a role in HCV replication, virus pathogenesis, modulation of cell signaling pathways, virus propagation and the interferon response. It can be used to predict the resistance and the sensitivity of HCV to the IFN treatment [13,14]. In the course of the last two decades, treatment of the HCV infection has significantly improved. For nearly 15 years, the standard of care was the combination of pegylated interferon alfa and ribavirin (PR) with a moderate sustained virologic response (SVR) ranging from 45 to 85% depending on the viral genotype [15]. Development of the direct-acting antiviral drugs (DAAs) targeting viral proteins was achieved as a result of better understanding of the HCV life cycle. As a result of continuous drug development effort in the hepatitis area, the standard of care in 2015 became a combination of DAAs (Sofosbuvir, daclatasvir and the sofosbuvir /ledipasvir combination) with excellent cure rates above 95% [16-18]. Addition of ribavirin to the combination of Sofosbuvir-Ledipasvir provided broader range of efficacy against multiple genotypes (1,4, 5, and 6) with enhanced efficacy (SVR12 >95%) [19].
The American Association for the Study of Liver Diseases (AASLD) and the Infectious Diseases Society of America (IDSA) in 2018 developed a web-based process for the expeditious formulation and dissemination of evidence-based recommendations for the treatment of HCV patients. Their recommendations were based on patient factors (treatment naive vs experienced, cirrhosis status, comorbidities) and viral characteristics (genotype [GT], subtype, resistance-associated substitutions [RASs]) [20].
As per the recommendations of the European Association for the Study of the Liver (EASL) on treatment of hepatitis C in 2018, the goal of therapy is to cure HCV infection in order to: (i) prevent the complications of HCV-related liver and extra-hepatic diseases, including hepatic necroinflammation, fibrosis, cirrhosis, decompensation of cirrhosis, HCC, severe extra-hepatic manifestations and death; (ii) improve quality of life and remove stigma; (iii) prevent onward transmission of HCV. They clarified in their recommendation the endpoint of therapy being an undetectable HCV RNA in serum or plasma 12 weeks (SVR12) or 24 weeks (SVR24) after the end of therapy [21]. This expectation represented another challenge for developing HCV successful treatment.
The use of placebo has been one of the main concerns for patients. Understandably, they prefer to be on the active arm. However, because many measured endpoints such as fatigue, physical activities, work productivity, emotional impact and quality of life are subjective, the placebo arm is crucial for these studies to minimize bias by the patients and investigators. In addition, placebo arm inclusion allows comparing adverse events (AEs) in the active arm versus placebo arm, as some of the reported AEs could be related to the studied disease and not related to the investigational drug.
Development of a validated Patient Reported Outcome (PRO) instrument for liver diseases to complement clinical outcomes of liver disease patients is needed. These various instruments must be validated using a larger sample and a wider range of patients. Once validated, the outcomes which are captured by PRO will add to clinical endpoints of upcoming clinical trials and assure that the comprehensive assessment of treatment options includes accurately measured patients’ experience [22].
Recent efforts were directed to identify relevant PROs in liver diseases. Among others, fatigue and psychologic disturbances occur frequently in chronic liver diseases. The fatigue experienced by patients with HCV infection is more severe and responds poorly to relieving factors [22,23]. Moreover, patients with HCV infection are more depressed and harbor greater feelings of anger and hostility compared with those with non-liver chronic diseases. These observations are important because proper management of the psychologic symptoms may have a favorable impact on the quality of life of patients with HCV infection [23]. In addition, the degree of fatigue varies among patients and often does not correlate with disease severity. Fatigue differs from and should be distinguished from other common conditions including depression, sleepiness, drowsiness, and weakness [22]. Numerous patient-reported outcomes (PROs) instruments such as the Chronic Liver Disease Questionnaire (CLDQ) have been used to measure health-related quality of life (HRQoL) in liver disease trials [24,25]. These great efforts are just the beginning to establish tools in the liver disease arena that would need further validation and discussion with HAs to become acceptable.
Blinding of the investigational drug is another important feature, especially when a comparator is being used. Blinding increases cost and complexity. For example, some of these comparators are available in a dosage form such as a capsule or pre-filled syringe while the investigational drug is only available as pills or in vials. Furthermore, most liver studies are being conducted in the outpatient setting, often with a self-administered study drug that makes the blinding matter more difficult but even more important.
The duration of clinical study represents another challenge. Usually shorter studies are preferable by patients, investigators, and sponsor. However, the speed with which a disease is expected to respond to treatment drives the duration of most studies. Most of the liver diseases are slowly progressing and accordingly, liver disease trials usually require longer treatment duration to show efficacy, in addition to the need to collect long-term safety data as well as maintained efficacy on chronic term usage of the investigational drug. One of the challenges specific for hepatitis clinical trials is the identification of shorter regimens that achieve a sustained cure after completion of treatment (SVR). The duration of these studies had to be extended for a period of 8 to 24 weeks after end of treatment.
Another challenge specific to hepatitis studies is the identification of the efficacious and safe combination of drugs. There are several combinations of regimens some are fixed dose regimens that were under consideration and assessed for their benefit. In addition, the presence of 7 genotypes of the hepatitis C virus with various subtypes added another specific challenge to the development of efficacious regimen for this specific genotype and subtype. The associated morbidity resulting from HCV infection such as presence of liver cirrhosis or status post-liver transplant added another challenge to clinical trials in hepatitis area. Also, the status and the exposure to previous antiviral therapy is an additional peculiar challenge. The need to develop therapy for children and adolescent patients has to be put into consideration, and safety and efficacy in this special population have to be established. All these factors contributed to the large number of studies listed on ClinicalTrials.gov website; there are more than 2,000 clinical trials reported on this official website.
Identification of relevant biomarkers or acceptable surrogate endpoints would be crucial in guiding the drug development process and providing supporting evidence of efficacy. A surrogate endpoint is a marker, such as a laboratory measurement, radiographic image, physical sign, or other measure that is thought to predict clinical benefit but is not itself a measure of clinical benefit. These acceptable biomarkers or surrogate endpoints will facilitate the “go no-go” decision and will dedicate long-term studies for confirmatory purposes. Of course, these biomarkers and surrogate endpoints will need validation as well as ongoing discussion with HAs for acceptance. To investigate shear wave (SW) propagation velocity in patients with untreated hepatitis C and patients with sustained virologic response (SVR). Liver stiffness measurements with SWE were reported to be useful in diagnosing fibrosis in hepatitis C [26]. The authors found that in hepatitis C patients, liver stiffness is higher in treatment-naïve patients than in those showing SVR and concluded SWE may be a predictor of hepatic complications in HCV infected patients [27].
For early identification and surveillance of HCV patients with liver disease progression, the availability of suitable diagnostic and prognostic biomarkers is still an unmet clinical need. Alfa-fetoprotein together with imaging techniques is commonly used, however its specificity and sensitivity are not satisfactory. The risk of disease progression in HCV infected patients and new biomarkers have been proposed, including post-transcriptionally modified molecules and genetic biomarkers [28].
Increased levels of chemokine interferon-gamma (IFN-γ)-inducible protein-10 (CXCL10), soluble CD163 (sCD163) and soluble CD14 (sCD14) have been reported in HCV infection. It was found that IFN-free regimens including newer DAA induce an early and marked decrease in these circulating inflammatory biomarkers. However, the full normalization of biomarkers was not obtained, especially in patients with advanced fibrosis, thus underlying the need for a treatment in the early stages of HCV infection before the progression to fibrosis [29].
Fujita et al. [30] in 2018 compared the diagnostic abilities of markers of fibrosis to histopathological analysis of liver biopsy samples from patients with positive Hepatitis C Virus (HCV) infection. All four biomarkers of liver fibrosis were influenced by histopathological activity grading. The authors concluded liver biopsy should be the gold standard to evaluate liver fibrosis staging even though several noninvasive biomarkers have been investigated.
The oral route of administration is the easiest one for most disease conditions. This is followed by the subcutaneous route (s.c.), which can be self-administered with some training. The intravenous route (i.v.) is the hardest, generally cannot be self-administered, and usually requires to be administered by a health care professional. However, based on the nature of the investigational compound, the i.v. route may be the only feasible route of administration. This would require either a clinical site visit or a specialized visiting nurse. Of course, depending on the degree of efficacy and benefit from the investigational drug, patients are willing to go with chronic intravenous treatments.
We live in a new era when social media and technology play an important communication role. This novel technology had been embraced by many patient advocacy groups and had been a valuable communication tool among patients, caregivers, and support groups. However, the risk to clinical trial integrity is increased by the use of this tool to share personal experience during the actual participation of patients in an ongoing study. Here is one scenario: In an ongoing study, one patient is randomly assigned to placebo and the other to active treatment. The first patient does not observe any benefit (+/− worsening of his condition). The second patient experiences some adverse events. These two patients communicate through social media and share their experiences. At this stage, you can imagine the impact and consequences of this interaction. Would you be confident in the collected data or would you expect some bias, either falsely reported AEs or low/ lack of efficacy? Because of the serious consequences to the integrity of study data introduced by this social media interaction, many neuromuscular study protocols incorporated as an inclusion criterion the patient’s agreement not to share any of study experiences through social media with other patients during the study conduct.
The last step for any drug development program is the submission of all data for registration and approval by health authorities (HAs). Frequent interaction and communication with HA during the conduct of drug program is very important. Natural history of these diseases and awareness of the patients’ need to maintain their activities of daily living are valuable elements for developing treatments for these diseases. Therefore, it is advisable for the drug industry to gain alignment from the HA at an early stage of the program to facilitate approval process when reaching the final phase.
Having a safe and effective drug available to all patients is a common goal for both regulators and sponsors. With this in mind, adopting a collaborative approach between regulators and sponsors, would ensure smoother and faster interaction to achieve their common goal. Uncertainties in various aspects including biomarkers and PRO validations underscore the need for continuing dialogue between all the stakeholders in this arena to identify gaps in knowledge and unmet needs, and to address areas of ambiguity in development of diagnostics and therapies against hepatitis and other liver diseases.
Conclusion
The drug development process is becoming lengthier and more expensive. The rate of new drug approval fluctuates, depending on the research pipeline and targeted disease entities. The area of liver diseases and specially hepatitis is a complex one and is a challenging therapeutic area in drug development. Among the challenges encountered by liver disease studies are the use of placebo, lack of proper PRO in addition to longterm safety, and study duration. Hepatitis clinical trials have additional specific challenges that need to be considered. These challenges include various genotype of the HCV, identification of ideal drug regimen of short duration, in addition to population specific therapies (Cirrhosis and young population). With time, more efforts are directed to identify and validate PROs as well as relevant biomarkers. The concept of collaborative work between regulators and sponsors, should prevail and lead to achievement of their common goal that is having effective and safe treatment available for all patients.
References
- El Mouelhi M (2018) Drug Development and Challenges for liver diseases Clinical Trials. Egypt Liver J 8: 1-5.
- (2015) Kaiser Family Foundation Health Tracking Poll.
- (2019) Kaiser Family Foundation Health Tracking Poll.
- DiMasi JA, Grabowski HG, Hansen RW (2016) Innovation in the pharmaceutical industry: New estimates of R&D costs. J Health Econ 47: 20-33.
- Paul SM, Mytelka DS, Dunwiddie CT, Persinger CC, Munos BH, et al. (2010) How to improve R&D productivity: the pharmaceutical industry’s grand challenge. Nat Rev Drug Discov 9(3): 203-214.
- (2015) Centers for Medicare & Medicaid Services, Office of the Actuary, National Health Statistics Group.
- Elgharably A, Gomaa A, Crossey M, Norsworthy P, Waked I, et al. (2017) Hepatitis C in Egypt – past, present, and future. Int J Gen Med 10: 1-6.
- El-Zayadi A (2004) Curse of schistosomiasis on Egyptian liver. World J Gastroenterol 10(8): 1079-1081.
- Shepard CW, Finelli L, Alter MJ (2005) Global epidemiology of hepatitis C virus infection. Lancet Infect Dis 5(9): 558-567.
- Mohamoud YA, Mumtaz GR, Riome S, Miller D, Abu-Raddad LJ (2013) The epidemiology of hepatitis C virus in Egypt: a systematic review and data synthesis. BMC Infect Dis 13: 288.
- Morozov VA, Lagaye S (2018) Hepatitis C virus: Morphogenesis, infection and therapy. World J Hepatol 10(2): 186-212.
- Powell D, Alpert A, Pacula RL (2019) A Transitioning Epidemic: How the Opioid Crisis Is Driving the Rise in Hepatitis C. Health Affairs 38(2): 287-294.
- Macdonald A, Crowder K, Street A, McCormick C, Harris M (2004) The hepatitis C virus NS5A protein binds to members of the Src family of tyrosine kinases and regulates kinase activity. J Gen Virol 85(3): 721-729.
- Reyes GR (2002) The nonstructural NS5A protein of hepatitis C virus: an expanding, multifunctional role in enhancing hepatitis C virus pathogenesis. J Biomed Sci 9(3): 187-197.
- (2011) European Association for the Study of the Liver. EASL Clinical Practice Guidelines: management of hepatitis C virus infection. J Hepatol 55(2): 245-264.
- Mauss S, Berg T, Rockstroh J, Sarrazin C, Wedemeyer H (2017) Hepatology - Clinical textbook. (8th), Hamburg: Medizin Fokus Verlag.
- van de Ven N, Fortunak J, Simmons B, Ford N, Cooke GS, et al. (2015) Minimum target prices for production of direct-acting antivirals and associated diagnostics to combat hepatitis C virus. Hepatology 61(4): 1174-1182.
- Schneider MD, Sarrazin C (2014) Antiviral therapy of hepatitis C in 2014: do we need resistance testing? Antiviral Res 105: 64-71.
- Renau LP, Berenguer M (2018) Introduction to hepatitis C virus infection: Overview and history of hepatitis C virus therapies. Hemodial Int 22(1): S8-S21.
- (2018) Hepatitis C Guidance 2018 Update: AASLD-IDSA Recommendations for Testing, Managing, and Treating Hepatitis C Virus Infection. Clinical Infectious Diseases 67(10): 1477-1492.
- (2018) EASL Recommendations on Treatment of Hepatitis C 2018. J Hepatol 69(2): 461-511.
- Golabi P, Sayiner M, Bush H, Gerber LH, Younossi ZM (2017) Patient-Reported Outcomes and Fatigue in Patients with Chronic Hepatitis C Infection. Clin Liver Dis 21(3): 565-578.
- Obhrai J, Hall Y, Anand BS (2001) Assessment of fatigue and psychologic disturbances in patients with hepatitis C virus infection. J Clin Gastroenterol 32(5): 413-417.
- (2009) FDA Guidance for Industry –Patient-Reported Outcome Measures: Use in Medical Product Development to Support Labeling Claims. Silver Spring, MD: Food and Drug Administration.
- Chawla KS, Talwalkar JA, Keach JC, Malinchoc M, Lindor KD, et al. (2016) Reliability and validity of the Chronic Liver Disease Questionnaire (CLDQ) in adults with non-alcoholic steatohepatitis (NASH). BMJ Open Gastroenterol 3(1): e000069.
- Tada T, Kumada T, Toyoda H, Ito T, Sone Y, et al. (2015) Utility of real-time shear wave elastography for assessing liver fibrosis in patients with chronic hepatitis C infection without cirrhosis: Comparison of liver fibrosis indices. Hepatol Res 45(10): E122-E129.
- Suda T, Okawa O, Masaoka R, Gyotoku Y, Tokutomi N, et al. (2017) Shear wave elastography in hepatitis C patients before and after antiviral therapy. World J Hepatol 9(1): 64-68.
- Biasiolo A, Martini A, Pontisso P (2015) New biomarkers for clinical management of hepatitis C virus infected patients. World J Clin Infect Dis 5(4): 59-66.
- Mascia C, Vita S, Zuccalà P, Marocco R, Tieghi T, et al, (2017) Changes in inflammatory biomarkers in HCV infected patients undergoing direct acting antiviral-containing regimens with or without interferon. PLoS ONE 12(6): e0179400.
- Fujita K, Kuroda N, Morishita A, Oura K, Tadokoro T, et al. (2018) Fibrosis Staging Using Direct Serum Biomarkers is Influenced by Hepatitis Activity Grading in Hepatitis C Virus Infection. J Clin Med 7(9): 267.






























